Abstract
Sterol glycosyltransferases (SGTs) catalyse transfer of glycon moiety to sterols and their related compounds to produce diverse glyco-conjugates or steryl glycosides with different biological and pharmacological activities. Functional studies of SGTs from Withania somnifera indicated their role in abiotic stresses but details about role under biotic stress are still unknown. Here, we have elucidated the function of SGTs by silencing SGTL1, SGTL2 and SGTL4 in Withania somnifera. Down-regulation of SGTs by artificial miRNAs led to the enhanced accumulation of withanolide A, withaferin A, sitosterol, stigmasterol and decreased content of withanoside V in Virus Induced Gene Silencing (VIGS) lines. This was further correlated with increased expression of WsHMGR, WsDXR, WsFPPS, WsCYP710A1, WsSTE1 and WsDWF5 genes, involved in withanolide biosynthesis. These variations of withanolide concentrations in silenced lines resulted in pathogen susceptibility as compared to control plants. The infection of Alternaria alternata causes increased salicylic acid, callose deposition, superoxide dismutase and H2O2 in aMIR-VIGS lines. The expression of biotic stress related genes, namely, WsPR1, WsDFS, WsSPI and WsPR10 were also enhanced in aMIR-VIGS lines in time dependent manner. Taken together, our observations revealed that a positive feedback regulation of withanolide biosynthesis occurred by silencing of SGTLs which resulted in reduced biotic tolerance.
Similar content being viewed by others
Introduction
Withania somnifera, commonly known as ashwagandha or Indian ginseng is an important medicinal plant and a rich source of steroidal lactones and terpenoids1. Leaf and root extracts of the plant are widely utilized in preparations of various herbal drugs using its biological activities as antioxidative, anti-inflammatory, antidiabetic, antistress, antitumor, hepatoprotective, immunomodulatory, anticonvulsant, antiproliferative, cardioprotective, hypoglycemic, diuretic and hypocholesterolemic2,3. Withanolides present in the extract of different tissues are the important metabolites having various pharmacological properties. So far, many withanolides have been isolated from W. somnifera among which withaferin A and withanolide D were reported to inhibit angiogenesis4,5. Withanolide A, a major active constituent isolated from W. somnifera root predominantly induces axonal outgrowth in normal cortical neurons6.
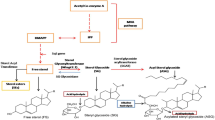
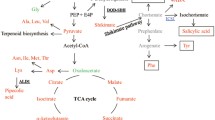
Present knowledge about the withanolide biosynthesis is very limited. Withanolides are C28 steroidal lactones which are biosynthesized by 5 carbon precursor isopentenyl diphophate (IPP) and its isomer dimethylallyl diphosphate (DMPP) via cytosolic mevalonate (MVA) pathway and plastid localised methyl-D-erythritol-4-phosphate (MEP) pathway leading to biosynthesis of 24-methylene cholesterol7. A series of desaturation, hydroxylation, epoxidation, cyclization, chain elongation and glycosylation steps are involved in withanolide biosynthesis8. Of these, glycosylation is one of the most widespread modifications in which simple or complex carbohydrate attaches to the biomolecules. The assembly of carbohydrates is made by a series of enzymatic process of specific glycosyltransferases (GTs), which sequentially transfer the monosaccharide moieties of their activated sugar donor (usually nucleotide donor) to the required acceptor such as lipids and proteins resulting in the formation of a glycosidic bond9,10.
Sterol glycosyltransferases (SGTs) in plants catalyze the transfer of glycon moieties to the sterols and their related compounds to generate steryl glycosides (SGs) or saponins. These glyco-conjugated sterols play very important role in defense of the plant against abiotic stresses and plant pathogen interactions11. The saponins produced by oats and tomato have been studied to their potential role in the defense of plants against phytopathogenic fungi12. SGs are membrane associated sterols and comprised of a sugar moiety attached to the hydroxyl (-OH) group at C-3 position of a sterol molecule13. The hydroxyl group at C-3 position is the most preferred one by SGTs for the enzymatic catalysis, followed by 27β-OH position or -OH group present at the side chain of a modified sterol backbone14,15. SGTs have been reported to occur as membrane associated as well as cytosolic. In addition, SGTs glycosylate steroidal hormones which function as growth and development regulator in plants16,17. In Solanum aculeatissimum, the accumulation of SaGT4A gene for the glycosylation of steroidal sapogenins in response to wounding stress indicates their role in plant defense18. WsSGTL1, one of the members of WsSGTs, has been examined to be involved in response to different abiotic stresses and insect resistance in heterologous expression systems2. However, extensive analysis of the role of SGTs in the glycosylation of terpenoids and their effect in the plant basal immunity is still lacking.
Virus-induced gene silencing (VIGS) is a quick method for functional analysis by knocking down the gene expression without the need of genetically transforms the plants19,20,21,22. Conventional VIGS assays initiate with a large fragment of gene which was converted and modified into small RNAs by the endogenous siRNA-based machinery causing off target gene silencing of the plant. Single artificial miRNA can provide better specificity by minimizing off-target effects23. We have developed artificial miRNA and VIGS (aMIR-VIGS) system for the functional characterization of WsSGTL genes under biotic stress. In the present study, an efficient protocol has been developed for the down-regulation of phytoene desaturase (PDS) gene by a combination of aMIR-VIGS system. We have applied this aMIR-VIGS system for the SGTLs members down-regulation of W. somnifera. Silenced lines led to significant accumulation of withanolides in leaves as compared to control plants. In addition, SGTLs down-regulation of W. somnifera affected the early gene transcript of the MVA and MEP pathway. Subsequently, the silneced lines lost their immunity and became susceptible to Alternaria alternata infection.
Results
Development of efficient aMIR-VIGS constructs
To develop aMIR-VIGS system for W. somnifera, we primarily targeted the PDS gene by designing amiRNA primer from the conserved region of the gene from closely related species of Solanaceae family (Supplementary Fig. 1a). The amipds construct was prepared by PCR based mutagenesis of miRNA159a of Arabidopsis thaliana and cloned into VIGS vector (Supplementary Fig. 1b) Syringe infiltration of this amipds-VIGS construct into W. somnifera plants developed bleaching (photobleaching) in the systemic leaves 15 to 20 days post inoculation (DPI) due to PDS gene silencing (Fig. 1a). First, the bleached regions were restricted to the veins of the leaves, later the symptoms extended to most of the leaf tissues24. The positive lines were also confirmed by coat protein specific primers (Fig. 1b). We have checked the level of PDS, by qRT-PCR analysis (Fig. 1c) which concludes that PDS mRNA expression was 75 to 90% less in the systemic tissue of amipds-VIGS lines than in control plants (empty vector).
(a) Phenotypic change in the leaves of silenced plant started from 15–20 DPI after the syringe infiltration of amipds-VIGS construct. (b) Positive lines of the plants checked by CP gene specific primer (amplicon size 351 bp). (c) PDS mRNA levels analysed by qRT-PCR. The expression levels of PDS transcripts were normalized to Actin gene specific primers. Data are means ± SE of three biological and two technical replicates.
Silencing of sterol glycosyltransferases
In order to investigate the role of SGTLs in W. somnifera, initially three different amiRNAs were designed from conserved sequences of WsSGTL1, WsSGTL2 and WsSGTL4, named as, 2misgt, 4misgt and 6misgt (Supplementary Fig. 2). To examine their silencing efficiency, amiRNAs were first cloned into pBI121 (Supplementary Fig. 3a) and then transformed into leaves of 3-weeks-old plants (Supplementary Fig. 3b). After 48 h and 72 h, qRT-PCR of 2misgt-pBI121 and 4misgt-pBI121 showed better silencing efficiency against members of SGTLs expression than 6misgt-pBI121 (Supplementary Fig. 3c,d). Further, 2misgt and 4misgt amiRNAs were utilized to clone into VIGS vector for the development of aMIR-VIGS system named as 2misgt-VIGS and 4misgt-VIGS (Supplementary Fig. 4a,b). The aMIR-VIGS construct of WsSGTLs members were transformed into leaves of 3-weeks-old plants. After 4 weeks of transformation, positive aMIR-VIGS lines were selected through PCR of CP specific primers (Supplementary Fig. 4c). The qRT-PCR were performed by using SGTL1, SGTL2 and SGTL4 gene specific primers and observed that their transcript level was decreased in the aMIR-VIGS plants. Interestingly, both aMIR-VIGS constructs showed almost same level of down-regulation of SGTLs members (Fig. 2a,b) as compared to control plants.
(a,b) The qRT-PCR showing the relative expression level of WsSGTLs members in 2misgt-VIGS and 4misgt-VIGS down-regulated lines as comparision to control (EV) with WsSGTL1, WsSGTL2 and WsSGTL4 gene specific primers. The expression levels of WsSGLTs transcript were normalized to Actin. (c,d) The relative WsSGTLs enzyme activity from the total protein of silenced lines by using stigmasterol and solenidine as accepter molecule respectively. Data are means ± SE of three biological and two technical replicates.
We have performed comparative nonradioactive sterol glycosyltransferase assay in the aMIR-VIGS lines of W. somnifera25 and control plants. Stigmasterol and solanidine were taken as acceptor molecule which have free 3β-OH moiety group. Accepter molecules were modified by donor substrate, uridine 5′-diphosphoglucose (UDP-glucose) with the help of SGT enzyme. This method takes advantage of a specific phosphatase to remove inorganic phosphate quantitatively from the leaving nucleotide diphosphate, such as UDP or GDP, of glycosyltransferase reactions. Total phosphate output/well in the protein reaction of 2misgts-VIGS and 4misgts-VIGS lines decreased significantly (upto 90%) as compared to control plants (Fig. 2c,d). This confirms that due to down-regulation of members of WsSGTs, silenced lines posses lesser SGT activity due to decreased expression of the genes.
Down-regulation of WsSGTLs modulate withanolides and phytosterols
To investigate the role of SGTL members in withanolide biosynthesis, we quantified withanolide A, withaferin A, withanoside V sitosterol and stigmasterol contents in leaves of each aMIR-VIGS plant after 40 DPI of transformation through high-performance liquid chromatography (HPLC) (Supplementary Fig. 6a–i). Quantitative estimation showed that withanolide A (upto 3.7 fold) and withaferin A (upto 1.7 fold) content significantly increased in 2misgt-VIGS and 4misgt-VIGS aMIR-VIGS lines as compared to control plants (Fig. 3a,b). In addition, concentration of sitosterol (upto 9.9 fold), stigmasterol (2.8 fold) also increased significantly in silenced lines (Fig. 3c,d). Analysis of withanoside V showed a significant reduction (approximately 45% to 77%) in 2misgt-VIGS and 4misgt-VIGS lines after down-regulation of SGTLs (Fig. 3e).
Relative quantification of HPLC analysis of withanolide A, withaferin A, sitosterol stigmasterol and withanoside V in the 2misgt-VIGS and 4misgt-VIGS down-regulated lines of W. somnifera as compare to EV. Data are means of three biological replicates plants. Asterisk indicate the significance level between control and down-regulated lines (*P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001) by Dunnett test.
Analysis was carried out to study whether the down-regulation of members of WsSGTL gene family affected the expression of genes involved in intermediate steps of the withanolide biosynthesis. To study this, transcript level of WsHMGR, WsDXR, WsFPPS, WsCYP710A1, WsDWF5 and WsSTE1 genes involved in MVA pathway were analysed by qRT-PCR in the aMIR-VIGS lines and control plants (Supplementary Fig. 7). There was a significant up-regulation (approx 3 fold) of WsCYP710A1 expression which might be responsible for the increase in the concentration of sitosterol and stigmasterol in silenced lines. Up-regulation of WsSTE1 (approx 3 fold) and WsDWF5 (approx 3.5 fold) was observed which might be responsible for increased withanolide A and withaferin A content in silenced lines. The expression of intermediate genes, WsHMGR, WsDXR, WsFPPS in aMIR-VIGS lines were also up-regulated upto 2.8, 10 and 2.9 fold, respectively. Our metabolite content and gene expression analysis conclude that down-regulation of SGTLs modulates the accumulation of withanolides by increasing expression of genes involved in MVA pathway (Fig. 4).
Solid one headed arrow indicates the single step irreversible while two headed for reversible reaction. Dashed purple arrow indicates several steps of reactions. All important enzyme of the pathway has been listed in blue box. Repression of SGTLs members by aMIR-VIGS system is represented by red dashed lines. Down-regulation of WsSGTLs members causes the increased in withanolides contents (black upward arrow) while decrease in the concentration of glycowithanolide (downward arrow). The up-regulation of important intermediate genes shown by upward red arrows indicates the increased withanolide biosynthesis in silenced lines.
Silencing of WsSGTLs affect growth and development of plant
The down-regulation of WsSGTLs resulted delay in the normal growth of aMIR-VIGS lines. After 3 weeks of transformation, plant height and leaf area of the silenced lines was measured. The height of the aMIR-VIGS lines was significantly shorter than control plants. This difference was maintained even after 10 weeks of growth (Fig. 5a,b), however, there was no change in the number of nodes and internodes. Total leaf area of the silenced lines was lesser than the control plants (Fig. 5c,d). These results suggest that the ratio of free sterol and its glycosylated forms may work as signalling molecules and regulate the growth of the plants. For the confirmation of this hypothesis, we have measured the ratio of free sitosterols and stigmasterol vs its glycosylated forms by acid hydrolysis26. The ratio of free phytosterol vs glycosylated phytosterols was higher in silenced lines than control plants (Fig. 5e,f). This experiment gave us indication of sterol imbalance in VIGS lines of W. somnifera and might be one of the reasons of shorting the height and leaf area of plants.
(a–d) Down-regulation of WsSGTL1, WsSGTL2 and WsSGTL4 of W. somnifera significantly affects the height and leaf area of the plants but no change in the number of nodes and internodes. (e,f) ratio of free phytosterol to their respective glycoside. Height data are means ± SE of three different plants and leaf area from three leaves of different plants at same node. HPLC data are the means ± SE of three technical replicate.
Suppression of WsSGTLs increases disease susceptibility
To determine the disease susceptibility of 2misgt-VIGS and 4misgt-VIGS line leaves against A. alternata, experiments were conducted under optimum growth conditions for the pathogen infection. First upper leaves of the silenced lines and control plants were inoculated with the fungal spore suspension. The severe necrotic symptoms were observed 7DPI of infection (Fig. 6a). After the infection of A. alternata, area of the lesion (Fig. 6b) and chlorosis in the leaves of 2misgt-VIGS and 4misgt-VIGS lines was much higher than control plants. The extracts isolated from the infected leaves were used for total colony forming unit count (cfu) which was significantly higher in silenced lines than control plants (Fig. 6c).
(a) Infection of W. somnifera with A. alternata strains, symptoms in the leaves in 7 days after inoculation. (b) Statistical data of lesion size 7 days after infection. For lesion size, the data are means ± SE of two independent experiments representing six biological replicates. Asterisk indicates a significant difference from the control (unpaired Student’s t-test; ***P < 0.05). (c) Total colony forming unit (cfu) count of the aMIR-VIGS plants leaves after 7 days infection of A. alternata. Data are the means of four biological replicates of each silenced lines. Asterisk indicates the significant level at *P ≤ 0.05, **P ≤ 0.01, ***P ≤ 0.001 by Dunnett test. (d) Aniline blue staining in the leaf of W. somnifera after the infection of A. alternata at 7 dpi at 20X magnifications on confocal microscope. Bars = 100 μm. (e,f) Hydrogen peroxide accumulation in leaves of W. somnifera were analysed after 48 h of infection by the microscopic (DAB staining, under light microscopy at 40 X magnification) and spectroscopic analysis. Data are the means ± SE of three different biological replicates. Asterisk indicates the significant difference from the control (unpaired Student’s t-test, **P ≤ 0.05). (g) Total SOD has been measured after 48 h infection of A. alternata in control and silenced lines of W. somnifera. Data are expressed in mean ± SE of three different biological replicates. Asterisk indicates the significant difference from the control (unpaired Student’s t-test, *P ≤ 0.05). (h) Stigmasterol/sitosterol ratio in control and silenced lines before and after 7 days A. alternata infection. Data are the means ± SE of three biological replicate samples. (*) asterisk is the indication of biotic stress condition.
Furthermore, callose deposition in the leaves of infected plants is another important parameter to check disease susceptibility against the biotic stress. After 7DPI of A. alternata infection, the leaf of the silenced lines and control plants was stained with aniline blue. Under confocal microscope, the higher amount of callose deposition was observed in the leaves of 2misgt-VIGS and 4misgt-VIGS lines as compared to the leaves of control plants (Fig. 6d). In addition to evaluate A. alternata induced disease symptoms, microscopic studies were conducted in down-regulated lines for cellular and intercellular reactive oxygen intermediates (ROIs). Hydrogen peroxide (H2O2) was detected by non-fluorescent (3, 3′-diaminobenzidine [DAB]) staining method. Microscopic analysis after 48 h infection revealed a strong dark brown precipitation in the leaves of 2misgt-VIGS and 4misgt-VIGS lines whereas no such precipitation was observed in the leaves of control plants (Fig. 6e). This was further accomplished by quantitative analysis of H2O2 where it increased (upto 1.8 fold) in the leaves of aMIR-VIGS lines after 48 h of fungal infection (Fig. 6f). We also quantified SOD level at 0 h and after 48 h post A. alternata infection in 2misgt-VIGS, 4misgt-VIGS lines and control plants. At 0 h, no significant differences were observed in the level of SOD, whereas, SOD level significantly increased upto 1.1, 2.7 and 2.1 for EV, 2misgt-VIGS and 4misgt-VIGS lines, respectively after 48 h of A. alternata infection, (Fig. 6g). The ratio of stigmasterol/sitosterol also maintains the innate immunity by increasing the disease susceptibility of the plant leaves. We have measured the stigmasterol/sitosterol ratio in plant before and after 7 days of A. alternata infection. After 7 days of stress, the concentration of sitosterol was decreased whereas, stigmasterol concentration increases in silenced lines and control plants. Although, stigmasterol/sitosterol ratio were higher in silenced lines than control plant (Fig. 6h). These results indicated that after down-regulation of members of WsSGTLs, the plants lose their basal immunity and became more prone to infection with A. alternata.
Salicylic acid level increases the expression of defence related genes in silenced lines
To investigate the contribution of defense related genes against A. alternata infection in silenced lines, real time expression analysis of some important genes, namely, WsPR1, WsDFS, WsSPI and WsPR10 was performed on the basis of earlier published transcriptome analysis27. The maximum enhancement in expression of WsPR1 was found upto 406 to 833 fold after 72 h of fungus infection in 2misgt-VIGS and 4misgt-VIGS respectively, whereas, upto 83 fold in EV as compared to mock plant (Fig. 7a[A] and 7b[A]). The increase in the expression level of WsDFS and WsSPI was also similar to the expression level of WsPR1 (Fig. 7a[B-C] and 7b[B-C]). Finally, it was concluded that after A. alternata infection, the expression of defense related genes in silenced lines of W. somnifera was highly up-regulated as compared to control plants.
(a,b) Expression level of different defense genes such as (A), WsPR1 (B), WsDFS (C), WsSPI have significantly increased while (D), WsPR10 showed delayed over-expression in silenced lines after 48 h and 72 h of fungal infection as compare to Mock, taken as control. (c) Salicylic acid quantification in the silenced plants leaves before and after 7 days infection of A. alternata from 40 DPI old plants. Data are expressed in the mean ± SE of three different biological replicates. Asterisk indicates the significance level as compare to control (unpaired Student’s t-test, at ***P ≤ 0.05).
The salicylic acid (SA) signalling cascades are the master regulator of defense process in plants which control the gene expression of large number of genes. Modulation of defense related gene expression encouraged us to study the possible role of SA. To know this, we measured SA levels before and after 7 days of A. alternata infection in aMIR-VIGS lines and control plants. Before the infection of A. alternata, no significant difference in the level of SA was observed in silenced lines and control plants. However after infection with A. alternata, SA level increased upto 3 fold in 2misgt-VIGS and 4misgt-VIGS lines as compared to control (Fig. 7c, Supplementary Fig. 10a–d). Altogether, these observations indicated that increased level of SA was responsible for modulation of defence related gene expressions which ultimately altered the basal disease resistance in aMIR-VIGS lines.
Discussion
W. somnifera is a medicinal plant rich in sterols, sterol glycosides and steroidal lactones which play a crucial role in adaptation of the plants to stress conditions28. In our previous studies, we reported that over-expression of WsSGTL1 in heterologous systems enhances germination, salt tolerance, heat tolerance, cold tolerance and insect resistance as compared to control plants2,28. In the present report, silencing of members of WsSGTs was done using the amiRNA technology29. However, Virus-induced gene silencing (VIGS) is also a quick method for knocking down the gene expression21. We designed artificial miRNA and developed efficient aMIR-VIGS system for the functional analysis of SGTLs members in W. somnifera.
The gene silencing by VIGS within a plant is highly dependent on the environmental factors and method of inoculation30,31,32. The marker gene used for the initiation of virus symptoms is GFP or PDS24,33. Gene silencing of PDS gene of W. somnifera through amipds-VIGS causes suppression of carotenoid biosynthesis where the affected plants became susceptible to photobleaching20,24,34,35. This protocol of aMIR-VIGS system has been used for the down-regulation of WsSGTs members to obtain a comprehensive understanding of their role in plants.
The members of WsSGTL are important modifying enzymes which participate in isoprenoid biosynthetic pathway, specifically in the glycosylation of phytosterols and withanolides. We have developed silenced lines of W. somnifera using aMIR-VIGS system. The down-regulation of members of WsSGTs was confirmed by comparative qRT-PCR analysis and SGT enzymatic assay of aMIR-VIGS lines and control plants. WsSGTLs down-regulated lines accumulated high amount of withanolide A, withaferin A, sitosterol and stigmasterol, whereas, as expected less amount of withanoside V in the leaves of silenced plants. It has been already reported that over-expression of glycosyltransferase genes leads to a significant increase in their respective glucosides36,. Recently, it was cleared that after over-expression of WsSGTL1 gene in homologous system increased the production of glycowithanolides37. To develop a better understanding of the effect of down-regulation of WsSGTLs, qRT-PCR of WsHMGR, WsDXR, WsFPPS, WsCYP710A1, WsSTE1 and WsDWF5 (genes of MVA pathway) was performed. From this analysis, it was concluded that WsSGTLs down-regulation positively affected the upstream genes of MVA pathway (Fig. 4, Supplementary Fig. 7). Recently, it was published that WsSQS leads to positive feedback and negative feed-forward regulation of sterol biosynthetic genes. Down-regulation of WsSQS significantly modulates the expression of MVA pathway genes located upstream and downstream of SQS5. From these results, we could conclude that the accumulation of withanolides and increased expression of biosynthetic pathway genes were positively affected by the down-regulation of members of WsSGTLs.
In addition, WsSGTLs down-regulated lines also showed phenotypic changes which suggested that WsSGTLs activity may be affecting the growth hormone signalling pathway in plants. In the silenced lines, height and leaf area were significantly shortened as compared to control plants (Fig. 5a–d). It was already proved that one members of WsSGTL gene family i.e., WsSGTL1 over-expression causes the alteration in phenotype of the plants such as enhanced growth and expansion of leaves37. Many reports suggested that glycosylation of important hormones, brassinosteroids and strigolactone was being carried out by the members of SGTs in plants13,38. The SDG8i glucosyltransferase and UGT74D1 were capable of influencing the biological activity of plant hormones such as strigolactone -oxindole-3-acetic acid (OxIAA) via glycosylation38. Gene mutants of UGT80B1and SMT1 have revealed roles of sterols modifications in embryo, vein, shoot and root patterning, cell expansion, polarity and proliferation, fertility, cellulose level maintenance, gravitropism, and hormone signalling39,40. After HPLC analysis of silenced lines it was concluded that the ratio of free phytosterol and their respective glycoside higher than control plants (Fig. 5e,f). It may be concluded that WsSGTLs down-regulation causes imbalance of glycosylated and nonglycosylated sterol which results in phenotypic changes of silenced lines. Besides this, the down-regulation might be affecting the signalling pathway of hormones which hinders the growth of plants.
Earlier reports showed the role of WsSGTL1 in response to abiotic stresses in heterologous system2,28 but no report is available regarding biotic stress. It was already reported that expression of SGTs were differentially modulated in different organs in response to external stimuli such as treatment of SA and methyl jasmonate in W. somnifera41, suggesting their role in defense. Here, we report that the down-regulation of WsSGTLs leads to increased susceptibility of plants against the infection of A. alternata.
Necrotrophic fungus, A. alternata, produces an AAL toxin (TA and TB) that causes necrosis in more than 200 species of Solanaceae family42,43. In biotic stress experiments, the leaves of silenced lines have 2–3 fold higher total cfu mL−1 count than control plant leaves. Callose also plays an important role in plant defense during interactions with pathogens. It rapidly synthesized and deposited just beneath the sites of attempted pathogen penetration44,45,46. The susceptibility was further evident by the massive callose deposition in the leaves of aMIR-VIGS lines as compared to control plants after one week of A. alternata infection. Further, we quantified H2O2 and SOD accumulation in silenced lines and control plants after the infection. It was reported earlier that SOD and H2O2 production plays a crucial role in restricting the growth of pathogen during hypersensitive response47,48,49,50,51. In the present study, WsSGTLs silenced lines accumulated high amount of H2O2 and SOD after the fungus infection. Several reports are available which emphasizes that exposure of stress resulted in increase in SOD activity due to increase in the ROS and H2O2 levels52,53. Since the high SOD level causes the high rate of H2O2 production in cell54. After pathogenic infection, plants induced array of metabolites change that potentially contributed in the susceptibility of the plants against pathogen infection. The conversion of sitosterol to stigmasterol is one of the significant marker of susceptibility against pathogenic infection and stigmasterol/sitosterol ratio become rise55,56. After the A. alternata infection, the conversion of sitosterol to stigmasterol was reported in the WsSGTLs silenced lines and control plants. This conversion was little high in the aMIR-VIGS lines than control plants. Resultant, the C22 desaturation of sterol was high in the silenced plant membrane after fungal infection and plants showed more disease susceptibility.
The level of SA was estimated before and after A. alternata infection in WsSGTLs silenced lines and control plants. SA plays important role in plant defense and involved in the activation of defense responses against biotrophic and hemi-biotrophic pathogens as well as in the establishment of systemic acquired resistance (SAR)57,58. It was also reported that programmed cell death was directly induced by level of SA58. This was one of the reasons of nercosis and callose deposition in the leaves of silenced lines in our study. Several previous reports demonstrated that the level of SA induced the expression level of PR proteins and other defense related genes59,60,61,62. To check this hypothesis, qRT-PCR of WsPR1, WsDFS, WsSPI and WsPR10 was performed in silenced and control plants and compared with the mock plants. Expression analysis showed up-regulation of these genes in time dependent manner63,64.
Finally, it may be concluded that after down-regulation of SGTL1, SGTL2 and SGTL4 genes of W. somnifera, withanolide contents were increased, whereas withanoside content was decreased. Also, this modulation was analysed with the increase in the expression of some important intermediate genes of MVA pathway. Silencing of WsSGTL members, modulation was observed in the free and glycosylated phytosterols which made the phenotypic variations in plants. Due to this modulation of withanolides,withanosides and ratio of nonglycosylated vs glycosylated phytosterols the plants might have lost their first line of defense. Further, the infection of A. alternata increased levels of SOD, H2O2 and SA significantly in down-regulated lines as compared to control plants. Higher level of SA also modulated the expression of WsPR1, WsDFS, WsSPI and WsPR10 genes of plant defense system. Altogether, we could demonstrate that the members of WsSGTLs play a significant role in maintaining the metabolic balance of the cell which facilitated the proper growth and defense system of the plants.
Methods
Preparation of artifitial miRNA
Expression of amiRNAs requires a mature miRNA backbone sequence, of which the stem region was replaced with desired sequences. A 545-bp fragment containing the entire sequence of the Arabidopsis miR159a was cloned into pTZ57R/T vector by PCR amplification using primers miR159-F1 (5′-ATATCTCCTTCATAGCTCTAATG-3′), miR159R1 (5′-AAATAACACGCTAAACATTGCTTCG-3′) and utilized for mutagenesis to yield amiRNA for WsPDS and members of WsSGTLs using oligonucleotide primers. For the preparation of these oligonucleotide primers, PDS sequences of closely related species of Solanaceae family and SGTLs sequences of W. somnifera were downloaded from National Centre for Biotechnology Information (NCBI) database. 21 bp conserve region for PDS and WsSGTLs were selected by multalin online tool (Supplementary Figs 1 and 2). Three conserve sequences of WsSGTLs 2misgt, 4misgt, 6misgt and one PDS named amipds (Supplementary Table 1) were selected on the basis of their specificity for the target. Three constructs have been prepared in pBI121 by using pTZ-miRNA159a as template named 2misgt-pBI121, 4misgt-pBI121 and 6misgt-pBI121 (Supplementary Fig. 3a) and silencing efficiency checked by syringe infiltration of these construct in leaves of the 3–4 week old plants after germination.
aMIR-VIGS construct preparation
Mutagenesis of pTZ57R/T-premiR159a construct was performed by PCR by using efficient 2misgt, 4misgt and amipds oligonucleotide primers. PCR product (247 bps) and pTRV vector were digested with Xba I and Sac I restriction enzymes (Fermentas, Fast Digest) and ligated with 1 μL of T4 DNA ligase (200 CEU μL−1 Fermentas). Ligated constructs were transformed into DH5α strain of E. coli and colonies were checked by amplification of 562 bp replicase, 351 bp coat protein and 247 bp amiRNA specific primers and further confirmation of positive construct was done with sequencing.
Plant growth and agroinfiltration
W. somnifera plants were grown at 28 °C under 16-h-light and 8-h-dark conditions in growth chamber. For VIGS assay, gene of interest (GOI)::TRV2 (2misgt-VIGS, 4misgt-VIGS and amipds::TRV2) constructs were transformed into GV3101 strain of Agrobacterium. Positive transformants were selected through colony PCR (Supplementary Fig. 4a,b) and grown on Luria-Bertani agar medium (50 mg mL−1 kanamycin, 50 mg mL−1 gentamycin and 25 mg mL−1 rifampicin). Two days before infiltration, 5-mL primary cultures of Agrobacterium strains were inoculated from single colonies on plates and grown for 16 h at 28 °C. 20 mL secondary culture was inoculated from the primary culture and grown for 5–6 h for obtaining OD600 0.4. The culture was harvested by centrifugation at 3000 × g at room temperature for 5 min. Cell pellet was resuspended in same volume of induction buffer, 10 mM MES (pH 5.5), 200 μM acetosyringone and incubated for 3–4 h at 28 °C at 80 rpm. Then pallet the culture again at 3000 × g and resuspended in the infiltration buffer containing 5 mM MES (pH 5.5) and OD600 was adjusted to 0.8–0.9. TRV1 + GOI: TRV2 cultures were mixed at 1:1 (v/v) ratio. A needleless syringe was used to inoculate the abaxial side of the lower leaves with TRV1 + GOI::TRV2 mixture of Agrobacterium culture. Infiltration of TRV1::TRV2 in the plants served as control (empty vector). The inoculated plants in the greenhouse were maintained at 21 ± 2 °C until the observation period was achieved.
RNA isolation and RT-PCR
Total RNA was extracted from upper leaves of 3-weeks-infiltered plants by using the RNA isolation kit (Sigma-Aldrich) and treated with RNase-free DNase I (Ambion). First-strand complementary DNA was synthesized using 5 μg of total RNA with oligo (dT) primer (Fermentas). The qRT-PCR was done in the StepOnePlus Real-Time PCR System (Applied Biosystems 7500). Primers used for qRT-PCR are listed in Supplementary Table S2. Three independent biological replicates and for each biological replicate, two technical replicates were analyzed by qRT-PCR analysis.
SGT assay
The SGT activity was measured by the Glycosyltransferase activity kit (R&D system/USA). The assay was carried out by detection of released inorganic phosphate using Malachite Green reagent25. Total protein was isolated by the ReadyPrep™ Protein Extraction Kit (Bio-Rad) and quantified by Bradford method65. For the SGT enzyme assay, phytosterols [stigmasterol (Sigma), solanidine (Sigma)] and UDP-glucose (Sigma) were used as accepter and donor substrates, respectively. The amount of phosphate removed from the acceptor molecule was calculated use via phosphate standard curve (Supplementary Fig. 5).
Biochemical Analysis by HPLC
The extraction procedure of withanolide (withanolide A and withaferin A) and glycowithanolide (withanoside V) has been followed by the protocol66. Qualitative and quantitative analysis of withanolides and withanoside were performed by HPLC-PDA with a Shimadzu (Japan) LC-10A system comprising LC-10AT dual-pump system, a SPD-10A PDA detector (operated at 227 nm), and Rheodyne injection valve with 20-μL sample loop. Compounds were separated on a RP-C18 column (Merck) (4.6 mm × 250 mm, 5-μm pore size) protected by a guard column containing the same packing. 1 mg mL−1 stock solutions of withaferin A, withanolide A and withanoside V (Sigma) were subsequently diluted to prepare solutions with concentrations in the range of 0.5 mg mL−1 to 50 mg mL−1 for preparation of standard curve. The sitosterol and stigmasterol estimation was carried out by following the protocol2. Ratio of these phytosterols of the plant leaves were checked by before and after acid hydrolysis by HCL26. For the quantification of SA, 200 mg sample was crushed into 1 mL of methanol and the volume was made to 2 mL. Mobile phase prepared from 20 mM phosphoric acid (A) (pH 2.0 adjusted) and acetonitrile (B) added in 50:50 ratio (A:B). Quantification was performed at 220 nm with PDA detector at 25 °C column temperature and 20 min RT with the flow rate of 1 mL min−1.
Biotic stress assay with A. alternata
Leaves of W. somnifera were surface sterilized with 1% NaOCl (v/v) and placed at blotting paper. The leaves were transferred into petriplates containing 0.8% agar. For pathogenic assay, 0.5% gelatine was freshly prepared to which 7–10 days old fungal spores were suspended to make the final fungal spore concentration of 106 cfu mL−1. Each leaf was treated with 5 μl of spore suspension at four different locations on the adaxial surface of the leaves. Control and pathogen infected leaves were kept at 25 °C under 70% relative humidity and 16 h of diurnal light. After 7 days of infection, leaves were sampled for measurement of lesion diameter and for total spore count.
Callose staining
For callose deposition assay, spore suspension sprayed on the leaves of control and transgenic plants with the help of an atomizer and pots were kept in moist chamber. Callose staining were performed after 5–7 DPI of incubation67. The infected leaves were incubated in 5 mL buffer [1 vol (water saturated phenol: glycerol: lactic acid: water in 1:1:1:1) +2 vol ethanol] at 65 °C temperature for 15–30 min. The leaves were transferred into fresh buffer for 25 h, rinsed 3 times each in ethanol and ddH2O. Stained in 150 mM K2HPO4 (pH 9.5) containing 0.01% aniline blue for 30 min, four washing was done in buffer (10 min each). The stained leaves were imaged under confocal microscope (Carl Zeiss’ LSM 510 META) with 20x Plan-apochromate lens. The excitation was done using diode laser 405 nm and emission was collected using band pass filter 470–500 nm.
DAB staining
For in situ detection of H2O2 DAB staining was carried out using leaves of transgenic and control plants68. The sample were collected after 48 h of fungal spray and infiltrated under gentle vacuum with 1 mg mL−1 DAB containing 0.05% v/v Tween 20 and 10 mM sodium phosphate buffer pH 7.0. The reaction was completed within 4–5 h post inoculation after visualizing the brown precipitate at the surface of the leaves. Leaves were fixed and then boiled for 15 min in ethanol: acetic acid: glycerol (3:1:1). Bleaching solution was replaced and leaves were incubated until the chlorophyll was completely removed. Leaves were observed by light microscopy under bright field at 4X magnification.
Measurement of H2O2
The H2O2 content of the leaves was measured spectrophotometrically after reaction with potassium iodide (KI)69 .The absorbance of the supernatant was measured at 390 nm from the spectrophotometer. The amount of hydrogen peroxide was calculated using a standard curve prepared with known concentrations of H2O2 (Supplementary Fig. 8a).
SOD assay
Superoxide dismutase (SOD, EC 1.15.1.1) activity was measured by nitrotetrazolium (NBT) photochemical assay70. The total protein of plants were measured by Bradford65 method using bovine serum albumin (BSA, Sigma Aldrich, USA) as a standard (Supplementary Fig. 8b).
Statistical analysis
Mean, standard error and number of replicates were used for statistical evaluation using GraphPad Prism 5. The statistical significance of differences between control and treated samples was tested by Dunnett’s test and unpaired Student’s t-test.
Additional Information
How to cite this article: Singh, G. et al. Silencing of sterol glycosyltransferases modulates the withanolide biosynthesis and leads to compromised basal immunity of Withania somnifera. Sci. Rep. 6, 25562; doi: 10.1038/srep25562 (2016).
References
Mirjalili, M. H., Moyano, E., Bonfill, M., Cusido, R. M. & Palazon, J. Steroidal lactones from Withania somnifera, an ancient plant for novel medicine. Molecules 14, 2373–2393 (2009).
Pandey, V. et al. WsSGTL1 gene from Withania somnifera, modulates glycosylation profile, antioxidant system and confers biotic and salt stress tolerance in transgenic tobacco. Planta 239, 1217–1231 (2014).
Bhattacharya, S. K., Bhattacharya, A., Sairam, K. & Ghosal, S. Anxiolytic-antidepressant activity of Withania somnifera glycowithanolides: an experimental study. Phytomedicine 7, 463–469 (2000).
Koduru, S., Kumar, R., Srinivasan, S., Evers, M. B. & Damodaran, C. Notch-1 inhibition by Withaferin-A: a therapeutic target against colon carcinogenesis. Mol. Cancer Ther. 9, 202–210 (2010).
Singh, A. K. et al. Virus-induced gene silencing of Withania somnifera squalene synthase negatively regulates sterol and defence-related genes resulting in reduced withanolides and biotic stress tolerance. Plant Biotechnol. J. 13, 12347, doi: 10.1111/pbi.12347 (2015).
Baitharu, I. et al. Withanolide A prevents neurodegeneration by modulating hippocampal glutathione biosynthesis during hypoxia. PLoS One 9, e105311 (2014).
Gupta, P. et al. De novo assembly, functional annotation and comparative analysis of Withania somnifera leaf and root transcriptomes to identify putative genes involved in the withanolides biosynthesis. PLoS One 8, e62714 (2013).
Senthil, K. et al. Transcriptome analysis reveals in vitro cultured Withania somnifera leaf and root tissues as a promising source for targeted withanolide biosynthesis. BMC Genomics 16, 14 (2015).
Ritsema, T. & Smeekens, S. C. Engineering fructan metabolism in plants. J. Plant Physiol. 160, 811–820 (2003).
Gloster, T. M. Advances in understanding glycosyltransferases from a structural perspective. Curr. Opin. Struct. Biol. 28, 131–141 (2014).
Wewer, V., Dombrink, I., vom Dorp, K. & Dormann, P. Quantification of sterol lipids in plants by quadrupole time-of-flight mass spectrometry. J. Lipid Res. 52, 1039–1054 (2011).
Osbourn, A. E. Preformed antimicrobial compounds and plant defense against fungal attack. Plant Cell 8, 1821–1831 (1996).
Chaturvedi, P., et al. Sterol glycosyltransferases-identification of members of gene family and their role in stress in Withania somnifera . Mol. Biol. Rep. 39, 9755–9764 (2012).
Ullmann, P., Ury, A., Rimmele, D., Benveniste, P. & Bouvier-Nave, P. UDP-glucose sterol beta-D-glucosyltransferase, a plasma membrane-bound enzyme of plants: enzymatic properties and lipid dependence. Biochimie. 75, 713–723 (1993).
Potocka, A. & Zimowski, J. Metabolism of conjugated sterols in eggplant. Part 2. Phospholipid : steryl glucoside acyltransferase. Acta Biochim. Pol. 55, 135–140 (2008).
Poppenberger, B. et al. The UGT73C5 of Arabidopsis thaliana glucosylates brassinosteroids. Proc. Natl. Acad. Sci. USA 102, 15253–15258 (2005).
Chaturvedi, P., Misra, P. & Tuli, R. Sterol glycosyltransferases–the enzymes that modify sterols. Appl. Biochem. Biotechnol. 165, 47–68 (2011).
Kohara, A. et al. A novel glucosyltransferase involved in steroid saponin biosynthesis in Solanum aculeatissimum . Plant Mol. Biol. 57, 225–239 (2005).
Ratcliff, F., Martin-Hernandez, A. M. & Baulcombe, D. C. Technical Advance. Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J. 25, 237–245 (2001).
Liu, Y., Schiff, M. & Dinesh-Kumar, S. P. Virus-induced gene silencing in tomato. Plant J. 31, 777–786 (2002).
Brigneti, G. et al. Virus-induced gene silencing in Solanum species. Plant J. 39, 264–272 (2004).
Senthil-Kumar, M. & Mysore, K. S. New dimensions for VIGS in plant functional genomics. Trends Plant Sci. 16, 656–665 (2011).
Tang, Y., Lai, Y. & Liu, Y. Virus-induced gene silencing using artificial miRNAs in Nicotiana benthamiana . Methods Mol. Biol. 975, 99–107 (2013).
Ruiz, M. T., Voinnet, O. & Baulcombe, D. C. Initiation and maintenance of virus-induced gene silencing. Plant Cell 10, 937–946 (1998).
Wu, Z. L., Ethen, C. M., Prather, B., Machacek, M. & Jiang, W. Universal phosphatase-coupled glycosyltransferase assay. Glycobiology 21, 727–733 (2011).
Misra, P. et al. Modulation of transcriptome and metabolome of tobacco by Arabidopsis transcription factor, AtMYB12, leads to insect resistance. Plant Physiol. 152, 2258–2268 (2010).
Ghosh Dasgupta, M., George, B. S., Bhatia, A. & Sidhu, O. P. Characterization of Withania somnifera leaf transcriptome and expression analysis of pathogenesis-related genes during salicylic acid signaling. PLoS One 9, e94803 (2014).
Mishra, M. K. et al. Overexpression of WsSGTL1 gene of Withania somnifera enhances salt tolerance, heat tolerance and cold acclimation ability in transgenic Arabidopsis plants. PLoS One 8, e63064 (2013).
Niu, Q. W. et al. Expression of artificial microRNAs in transgenic Arabidopsis thaliana confers virus resistance. Nat. Biotechnol. 24, 1420–1428 (2006).
Broderick, S. R. & Jones, M. L. An optimized protocol to increase virus-induced gene silencing efficiency and minimize viral symptoms in Petunia . Plant Mol. Biol. Report 32, 219–233 (2014).
Chen, J. C. et al. Chalcone synthase as a reporter in virus-induced gene silencing studies of flower senescence. Plant Mol. Biol. 55, 521–530 (2004).
Jiang, C. Z., Chen, J. C. & Reid, M. Virus-induced gene silencing in ornamental plants. Methods Mol. Biol. 744, 81–96 (2011).
Sha, A. et al. Virus-based microRNA silencing in plants. Plant Physiol. 164, 36–47 (2014).
Purkayastha, A. & Dasgupta, I. Virus-induced gene silencing: a versatile tool for discovery of gene functions in plants. Plant Physiol. Biochem. 47, 967–976 (2009).
Ma, X. et al. Different roles for RNA silencing and RNA processing components in virus recovery and virus-induced gene silencing in plants. J. Exp. Bot. 66, eru447v1, doi: 10.1093/jxb/eru447 (2014).
Lim, E. K. & Bowles, D. J. A class of plant glycosyltransferases involved in cellular homeostasis. EMBO J 23, 2915–2922 (2004).
Saema, S. et al. Ectopic overexpression of WsSGTL1, a sterol glucosyltransferase gene in Withania somnifera, promotes growth, enhances glycowithanolide and provides tolerance to abiotic and biotic stresses. Plant Cell Rep. in press, doi: 10.1007/s00299-015-1879-5 (2015).
Islam, S. et al. Increased biomass, seed yield and stress tolerance is conferred in Arabidopsis by a novel enzyme from the resurrection grass Sporobolus stapfianus that glycosylates the strigolactone analogue GR24. PLoS One 8, e80035 (2013).
DeBolt, S. et al. Mutations in UDP-Glucose:sterol glucosyltransferase in Arabidopsis cause transparent testa phenotype and suberization defect in seeds. Plant Physiol. 151, 78–87 (2009).
Carland, F., Fujioka, S. & Nelson, T. The sterol methyltransferases SMT1, SMT2, and SMT3 influence Arabidopsis development through nonbrassinosteroid products. Plant Physiol. 153, 741–756 (2010).
Sharma, L. K., Madina, B. R., Chaturvedi, P., Sangwan, R. S. & Tuli, R. Molecular cloning and characterization of one member of 3beta-hydroxy sterol glucosyltransferase gene family in Withania somnifera . Arch. Biochem. Biophys. 460, 48–55 (2007).
Chung, K. R. Stress response and pathogenicity of the necrotrophic fungal pathogen Alternaria alternata . Scientifica (Cairo) 2012, 635431 (2012).
Mesbaha, L., van der Weerdenb, G. M., Nijkampa, H. J. J. & Hillec, J. Sensitivity among species of Solanaceae to AAL toxins produced by Alternaria alternata f.sp. lycopersici. Plant Pathology 49, 734–741 (2000).
Voigt, C. A. Callose-mediated resistance to pathogenic intruders in plant defense-related papillae. Front. Plant Sci. 5, 168 (2014).
Luna, E. et al. Callose deposition: a multifaceted plant defense response. Mol Plant Microbe. Interact. 24, 183–193 (2011).
Hammond-Kosack, K. E. & Jones, J. D. Resistance gene-dependent plant defense responses. Plant Cell 8, 1773–1791 (1996).
Mur, L. A., Kenton, P., Lloyd, A. J., Ougham, H. & Prats, E. The hypersensitive response; the centenary is upon us but how much do we know? J. Exp. Bot. 59, 501–520 (2008).
Janknecht, R., Sander, C. & Pongs, O. (HX)n repeats: a pH-controlled protein-protein interaction motif of eukaryotic transcription factors? FEBS Lett. 295, 1–2 (1991).
Willekens, H. et al. Catalase is a sink for H2O2 and is indispensable for stress defence in C3 plants. EMBO J. 16, 4806–4816 (1997).
Pellinen, R. I., Korhonen, M. S., Tauriainen, A. A., Palva, E. T. & Kangasjarvi, J. Hydrogen peroxide activates cell death and defense gene expression in birch. Plant Physiol. 130, 549–560 (2002).
Alscher, R. G., Erturk, N. & Heath, L. S. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J. Exp. Bot. 53, 1331–1341 (2002).
Gill, S. S. & Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 48, 909–930 (2010).
Gomez, J. M., Hernandez, J. A., Jimenez, A., del Rio, L. A. & Sevilla, F. Differential response of antioxidative enzymes of chloroplasts and mitochondria to long-term NaCl stress of pea plants. Free Radic. Res. 31 Suppl, S11–18 (1999).
Muller, F. L. et al. High rates of superoxide production in skeletal-muscle mitochondria respiring on both complex I- and complex II-linked substrates. Biochem. J. 409, 491–499 (2008).
Wang, K., Senthil-Kumar, M., Ryu, C. M., Kang, L. & Mysore, K. S. Phytosterols play a key role in plant innate immunity against bacterial pathogens by regulating nutrient efflux into the apoplast. Plant Physiol. 158, 1789–1802 (2012).
Griebel, T. & Zeier, J. A role for beta-sitosterol to stigmasterol conversion in plant-pathogen interactions. Plant J. 63, 254–268 (2010).
War, A. R., Paulraj, M. G., War, M. Y. & Ignacimuthu, S. Role of salicylic acid in induction of plant defense system in chickpea (Cicer arietinum L.). Plant Signal. Behav. 6, 1787–1792 (2011).
Brodersen, P., Malinovsky, F. G., Hematy, K., Newman, M. A. & Mundy, J. The role of salicylic acid in the induction of cell death in Arabidopsis acd11 . Plant Physiol. 138, 1037–1045 (2005).
Jung, J. L., Fritig, B. & Hahne, G. Sunflower (Helianthus annuus L.) Pathogenesis-related proteins (induction by aspirin (acetylsalicylic acid) and characterization). Plant Physiol. 101, 873–880 (1993).
Gorlach, J. et al. Benzothiadiazole, a novel class of inducers of systemic acquired resistance, activates gene expression and disease resistance in wheat. Plant Cell 8, 629–643 (1996).
Zhang, J. et al. Expression of pathogenesis related genes in response to salicylic acid, methyl jasmonate and 1-aminocyclopropane-1-carboxylic acid in Malus hupehensis (Pamp.) Rehd. BMC Res . Notes 3, 208 (2010).
Bari, R. & Jones, J. D. Role of plant hormones in plant defence responses. Plant Mol. Biol. 69, 473–488 (2009).
Gorovits, R. et al. Expression of stress-response proteins upon whitefly-mediated inoculation of Tomato yellow leaf curl virus in susceptible and resistant tomato plants. Mol. Plant Microbe. Interact. 20, 1376–1383 (2007).
Roefaro, J. & Mukherjee, S. M. Olanzapine-lnduced hyperglycemic nonketonic coma. Ann. Pharmacother. 35, 300–302 (2001).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 72, 248–254 (1976).
Sangwan, R. S. et al. Withanolide A is inherently de novo biosynthesized in roots of the medicinal plant Ashwagandha (Withania somnifera). Physiol. Plant 133, 278–287 (2008).
Zhou, J., Spallek, T., Faulkner, C. & Robatzek, S. CalloseMeasurer: a novel software solution to measure callose deposition and recognise spreading callose patterns. Plant Methods 8, 49 (2012).
Bindschedler, L. V. et al. Peroxidase-dependent apoplastic oxidative burst in Arabidopsis required for pathogen resistance. Plant J. 47, 851–863 (2006).
Papadakis, A. K., Siminis, C. I. & Roubelakis-Angelakis, K. A. Reduced activity of antioxidant machinery is correlated with suppression of totipotency in plant protoplasts. Plant Physiol. 126, 434–444 (2001).
Beyer, W. F. Jr. & Fridovich, I. Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem. 161, 559–566 (1987).
Acknowledgements
The authors thank The Director, CSIR-National Botanical Research Institute, Lucknow, for the facilities provided. We are thankful to Dr. Abhishek Niranjan, CSIR-NBRI for helping in HPLC analysis. The authors are thankful to Department of Science and Technology (Project No. GAP 231425), Govt. of India, for financial support.
Author information
Authors and Affiliations
Contributions
P.M. and P.K.T. designed research experiments. G.S., M.T. and S.P.S. performed the experiments. G.S. and M.T. analysed data, prepared the figures and wrote the manuscript. P.M., P.K.T. and S.S. checked the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Singh, G., Tiwari, M., Singh, S. et al. Silencing of sterol glycosyltransferases modulates the withanolide biosynthesis and leads to compromised basal immunity of Withania somnifera. Sci Rep 6, 25562 (2016). https://doi.org/10.1038/srep25562
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep25562
- Springer Nature Limited
This article is cited by
-
Ectopic Expression of WsSGTL3.1 Gene in Arabidopsis thaliana Confers Enhanced Resistance to Pseudomonas syringae
Journal of Plant Growth Regulation (2022)
-
Overexpression of WssgtL3.1 gene from Withania somnifera confers salt stress tolerance in Arabidopsis
Plant Cell Reports (2021)
-
Fungal endophytes attune withanolide biosynthesis in Withania somnifera, prime to enhanced withanolide A content in leaves and roots
World Journal of Microbiology and Biotechnology (2019)
-
Compatibility of Inherent Fungal Endophytes of Withania somnifera with Trichoderma viride and its Impact on Plant Growth and Withanolide Content
Journal of Plant Growth Regulation (2019)
-
Elicitation Enhanced the Yield of Glycyrrhizin and Antioxidant Activities in Hairy Root Cultures of Glycyrrhiza glabra L.
Journal of Plant Growth Regulation (2019)