Abstract
Climate-induced forest die-off is widespread in multiple biomes, strongly affecting the species composition, function and primary production in forest ecosystems. Hydraulic failure and carbon starvation in xylem sapwood are major hypotheses to explain drought-induced tree mortality. Because it is difficult to obtain enough field observations on drought-induced mortality in adult trees, the current understanding of the physiological mechanisms for tree die-offs is still controversial. However, the simultaneous examination of water and carbon uses throughout dehydration and rehydration processes in adult trees will contribute to clarify the roles of hydraulic failure and carbon starvation in tree wilting. Here we show the processes of the percent loss of hydraulic conductivity (PLC) and the content of nonstructural carbohydrates (NSCs) of distal branches in woody plants with contrasting water use strategy. Starch was converted to soluble sugar during PLC progression under drought, and the hydraulic conductivity recovered following water supply. The conversion of NSCs is strongly associated with PLC variations during dehydration and rehydration processes, indicating that stored carbon contributes to tree survival under drought; further carbon starvation can advance hydraulic failure. We predict that even slow-progressing drought degrades forest ecosystems via carbon starvation, causing more frequent catastrophic forest die-offs than the present projection.
Similar content being viewed by others
Introduction
Recently, climate-induced forest die-offs have been found world-wide in response to prolonged drought and extreme heating1,2,3,4,5. Some of the most severe impacts of climate change will be the degradation of forest ecosystems, reducing the ecological benefits. Predictions of forest degradation under future climates are limited by our recent understanding of how mature trees respond to drought and how their survival or die-off is determined under prolonged drought. Currently, two non-pathogen-related hypotheses concerning drought-induced tree mortality have been proposed: hydraulic failure6 and carbon starvation7,8. The hydraulic failure hypothesis states that the physiological process of xylem embolism is caused by prolonged drought and is the major cause of tree and forest die-offs6,9,10,11. The carbon starvation hypothesis states that carbon shortages during drought cause physiological dysfunction and thereby lead to the death of forest trees7,12. Because each hypothesis may be too simple to explain tree mortality13,14, we sought to collect direct evidence relating to water and carbon use during survival or death processes.
Some studies have proposed that embolized xylem vessels can recover via refilling15,16,17,18,19 in a process promoted by injection of soluble sugar into the embolized vessels16,20,21,22,23,24,25. Experimental studies on seedlings have indicated that an increase in nonstructural carbohydrates (NSCs) in plant bodies enhances their drought tolerance12,16,25. However, the contribution of NSCs to drought tolerance in mature trees remains controversial26,27, because studies monitoring variations in NSCs and xylem embolism over time are lacking. Leaf photosynthetic and respiration rates and NSCs content in sapwood have been reported to decrease during dehydration processes28,29, whereas there are no reports of such changes during rehydration processes. Evaluation of carbon starvation and hydraulic failure hypotheses requires monitoring both of whole-plant carbon dynamics and hydraulic dynamics of xylem embolism and recovery during the dehydration and rehydration processes.
To evaluate the relationship between the hydraulic failure and carbon use processes, we simultaneously measured daily time courses of the PLC (percent loss of conductivity) and NSCs content in the xylem sapwood of distal branches, in addition to the leaf water potential, leaf gas exchange and stem respiration rates, in drought-tolerant woody plants. The measurements were periodically conducted during the gradual progression of drought and during the successive hydraulic recovery induced by irrigation or rainfall. To clarify the contribution of NSCs to hydraulic failure, we selected field-growing adult trees of two drought-tolerant woody plants, Hibiscus glaber Matsum. and Ligustrum micranthum Zucc. (Fig. 1). These species are endemic trees in the Bonin Islands in Japan and are usually found at dry sites with shallow soil30. Our data show that they have different mortality rates in distal branches under normal conditions; the longevity of 50% of the distal branches in the same age cohort is 2.4 years in Hibiscus and 4.1 years in Ligustrum, according to the lower wood density in Hibiscus (0.669 g cm−3) than Ligustrum (0.795 g cm−3). Furthermore, xylem cavitation tolerance against dehydration in Hibiscus branches is lower than that in Ligustrum branches. These interspecific differences should reflect different physiological performance with respect to hydraulic failure and carbon use. Our field study indicates that carbon starvation advances hydraulic failure during slow tree-dieback processes. We discuss the implications of our findings for global climate-induced forest die-offs and their influence on terrestrial ecosystems.
(A) The water-spending plant, Hibiscus glaber Matsum. (B) The water-conserving plant, Ligustrum micranthum Zucc. Hibiscus trees have thinner lamina with a shorter leaf lifespan and higher leaf gas exchange rates than do the Ligustrum trees. The lifespan of the distal branches in Hibiscus is also shorter than that of the branches in Ligustrum.
Results
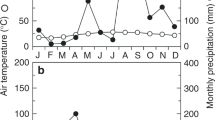
From the start of the experiment on 22 June 2014, the soil water content gradually decreased except in the case of one very small rainfall event on 11 July (the small shower did not show significant effects on the rehydration of plants). The water content increased rapidly after the areas received a plentiful amount of water (either by irrigation on 15–16 July or by rainfall on 21–22 July) (Fig. 2A). The predawn leaf water potential (ψpre) in Ligustrum decreased progressively with soil desiccation but increased rapidly following the increase in available water (Fig. 2B). Compared with that in Ligustrum, the range of decrease in ψpre in Hibiscus was much smaller. Midday net photosynthetic rates and daily stem respiration rates also slightly decreased with soil desiccation (Fig. 2C,D). After water had been provided, the leaf water potential, net photosynthetic rates and stem respiration rates increased in all individuals.
(A) Volumetric soil water content (VSWC) in control trees. (B) Leaf water potential before dawn. (C) Midday net photosynthetic rates. (D) Daily accumulated stem respiration rates. Red circles denote Hibiscus glaber, and blue squares denote Ligustrum micranthum. Open and closed symbols denote the control and irrigated trees, respectively. For the control trees, significant rainfall occurred on 21 July (open arrow); for the irrigated trees, we supplied water to the soil on 15 and 16 July in 2014 (purple arrow).
In both species, the PLC in distal branches increased with soil desiccation and decreased within two days of water supply (Fig. 3A,B). Although xylem embolism and hydraulic recovery involve different processes, hysteresis in PLC was not observed between the dehydration and rehydration processes (Fig. 3C,D). At a given ψpre, the PLC in Hibiscus was higher than that in Ligustrum, indicating that the mechanism for escaping severe xylem embolism functions well in Ligustrum.
(A) Daily time course of PLC in Hibiscus glaber. (B) Daily time course of PLC in Ligustrum micranthum. (C) The relationship between PLC and ψpre in Hibiscus glaber. (D) The relationship between PLC and ψpre in Ligustrum micranthum. In panels (A) and (B), open and closed symbols denote the control and irrigated trees, respectively. See the legend of Fig. 1 for the timing of the water supply. In panels (C) and (D), open and closed symbols show data during the dehydration process and during the rehydration process following the addition of water, respectively. The curves are fitted with a double-sigmoid function.
The amounts of soluble sugar and starch varied greatly, and the time-course data from both tree species exhibited a hysteresis loop during the desiccation and rehydration processes (Fig. 4). As PLC increased during soil desiccation, soluble sugar content also increased, and the starch content decreased. After water had been supplied, the soluble sugar content decreased, and the starch content increased. Time-course variations in the NSCs content in Hibiscus were greater than in Ligustrum. In Hibiscus, the change rates in xylem sugar contents were significantly correlated to PLC (R2 = 0.339, P = 0.047) and those in xylem starch contents were insignificantly but nearly correlated to PLC (R2 = 0.254, P = 0.095; Table S1); in Ligustrum, those in xylem sugar contents (R2 = 0.104, P = 0.306) and xylem starch contents (R2 = 0.145, P = 0.223) were not correlated to PLC. However, in Ligustrum, the amount of xylem sugar contents were insignificantly but nearly correlated to PLC (R2 = 0.233, P = 0.081) and the amount of xylem starch contents significantly decreased with PLC (R2 = 0.490, P = 0.005). Responses of NSCs dynamics in xylem sapwood to water condition were varied for species. Over all, the dynamics of NSCs within xylem sapwood seems to be associated with xylem cavitation (i.e. PLC) rather than water potential.
The daily variation of soluble sugar content in the control (A) and irrigation trees (B), and that of starch content in the control (C) and irrigation (D) control trees. Red and blue colors denote Hibiscus glaber and Ligustrum micranthum, respectively. The numbers in each panel show the order of the measurement days over the experimental period from 23 June (1) to 22 July (7) in 2014. Thin lines show the period of dehydration, and thick lines show the period of rehydration after being supplied with water. See the legend of Fig. 1 for the timing of the water supply.
Soluble sugar content was negatively correlated with starch content during all processes in Hibiscus and during the desiccation process in Ligustrum (Fig. 5). These relationships did not exhibit hysteresis during the dehydration and rehydration processes. Xylem cavitation triggers starch-to-sugar conversion, whereas the refilling of the embolized vessels promotes the reverse conversion. Hibiscus trees exhibited higher starch content and larger PLC variations than Ligustrum (Figs 4 and 5). The relatively small PLC variations in Ligustrum may explain the reason for non-significant correlation during the rehydration process in Ligustrum (Fig. 5B).
(A) The data show in Hibiscus glaber. (B) The data show in Ligustrum micranthum. Open and closed symbols show data during the dehydration process and the rehydration process after being supplied with water, respectively. A significant correlation was found in both processes in Hibiscus (P = 0.011), whereas a significant correlation was found only in the dehydration process in Ligustrum (P = 0.041).
Discussion
In the present study, we show that PLC can be a trigger of carbon dynamics between starch and soluble sugars within xylem sapwood in adult trees, and the amount of carbon dynamically transformed varies among tree species. The differences between the two species used in our study illustrate well that the carbon stored in xylem sapwood is an important determinant of the survival or drought-induced mortality of branches. During soil desiccation and watering, the distal branches of Hibiscus showed a greater change in PLC, with larger amounts of soluble sugar investment, than did the branches of Ligustrum (Fig. 4). These results are consistent with the physiology and anatomy of these trees; that is, healthy Hibiscus trees have a high NSCs (Fig. 5) and a larger fraction of parenchyma cells in their sapwood30. The short lifespan of Hibiscus twigs would be due to a high critical threshold of NSCs for shoot survival, and the frequent hydraulic segmentation of branches would function to suppress the transpirational water loss17,31. Their form and function can be thus consistent with their interspecific variations in resource-use strategy.
The recovery of embolized vessels is critical for tree survival under drought17,18,32. The hysteresis between the soluble sugar content and PLC (Fig. 4) and the negative relationship between soluble sugar and starch contents (Fig. 5) imply that the embolism process triggers a conversion from starch to sugar. A low starch content in xylem sapwood will result in a delay of the recovery of vessel dysfunction25. If the sapwood maintains a high sugar content until the next rainfall, sugar that is injected into embolized vessels may function in the recovery of dysfunctional vessels following rainfall23,24. A decrease in respiration rates with desiccation (Fig. 2D) is often observed, reducing ATP synthesis in mitochondria33. Such reduced respiration rates contribute to a positive carbon balance in the plant body beyond the critical threshold for metabolic homeostasis and survival during prolonged drought13,34. Energy produced by H+-ATPase on the plasma membrane is used for aquaporin-dependent hydraulic permeability35 and for the process of refilling embolized vessels36,37. Furthermore, treatment with an H+-ATPase inhibitor inhibits the refilling of embolized vessels16. Therefore, the increase in stem respiration rates following water-supply is associated with high energy requirement for the recovery of dysfunctional vessels. These facts indicate that a decrease in stored NSCs caused by drought can accelerate drought-induced tree mortality, because of the impaired hydraulic recovery of dysfunctional vessels due to carbon shortage. Thus, carbon starvation and hydraulic failure are not exclusive hypotheses in drought-induced tree mortality processes.
A theoretical perspective suspects the different processes of tree dieback according to drought conditions, i.e., severe drought will kill trees due to hydraulic failure, whereas mild drought will kill tree due to carbon starvation7. It is said that hydraulic failure was the main cause of forest die-off under prolonged drought (showing without the change in NSCs contents), according to simultaneous observation of NSCs and PLC in trees of an aspen forest in Colorado9 and of a rainforest in Amazon10. On the other hand, our notion that xylem embolism (i.e., an increase in PLC) seems to trigger starch-to-sugar conversion in sapwood (Figs 4 and 5 and Table S1) implies that drought-induced hydraulic failure and its recovery processes are closely linked with carbon dynamics within xylem sapwood. Although the physiological mechanisms for drought-induced tree die-offs are still controversial, their survival and death during drought events should include both processes of hydraulic failure and carbon use in xylem (Fig. 6).
When the amount of NSCs in the sapwood decreases due to drought, xylem hydraulic dysfunction does not recover following water supply, resulting in tree dieback induced by hydraulic failure. On the other hand, when the amount of NSCs is still remained at above a threshold level during drought, xylem dysfunction is recovered following water supply, resulting in survival after the drought.
The mechanism explored in this study implies that trees may become susceptible to drought after continuous environmental stress because of a gradual decrease in the resources stored in the plant body. Even if the stress is relatively mild, the amounts of NSCs reserves in individual trees will decrease during stressful periods. In such a condition, forests will be more easily destroyed by slightly more severe future droughts under global climate change. Conversely with sufficient resources, forest die-offs are mitigated even after the sudden severe drought. The stored carbon in sapwood would contribute to the homeostasis of forest ecosystems with reducing the hydraulic failure-induced forest die-offs. Such a mechanism is responsible for ongoing cases of forest die-offs for heating and drying in urban forests38. Under the chronic shortage in carbon resources induced by the progressing global changes, forest ecosystem may not be able to maintain the homeostasis. Therefore, we fear more extensive and much more frequent catastrophic forest die-offs via the weakening of present forests under the condition of low carbon assimilation (Fig. 6). An understanding of the carbon dynamics within the plant body will be essential for understanding the physiological mechanisms of forest growth and die-off, and their effects on terrestrial ecosystems39. This knowledge could show that the coordination between carbon use and water use plays an important role on resilience and vulnerability of forest dynamics to global climate change.
Methods
This study was conducted on the Ogasawara (Bonin) Islands in Japan (27°05′N, 142°13′E) in the northern Pacific Ocean. The mean air temperature was 23.2 °C, and the mean annual precipitation was 1,287 mm for the period from 2005 to 2014. The study year (2014) was a normal year with a relatively wet summer.
The study site is a dry dwarf forest, located on the elevated area of the island, where both Hibiscus glaber Matsum. and Ligustrum micranthum Zucc. grow sympatrically. We selected 2 healthy adult trees from each species. The heights of the canopy tops of the individuals were 1.1–1.6 m above the ground (Fig. 1). For each species, one tree was used for the artificial irrigation experiment, and the other (control) was used under natural conditions.
All measurements were performed for approximately one month, from 25 June to 22 July in 2014. The volumetric soil water content (VSWC) at a depth of 7.5 cm was monitored daily with an SM-150 (Delta-T, Cambridge, UK). For fully irrigation treatment, before sunset on 15 and 16 July, 0.6 m3 of water was supplied to 2.64 m2 of soil surface around the root systems of each individual. In natural conditions, on 21 and 22 July, increasing the VSWC in the control individuals.
Leaf gas exchange was periodically measured midday with an LI-6400 (LI-COR, Lincoln, NE, USA). Stem respiration rates at the main stem surface were measured daily by the closed-circuit method in the control individuals for each species with an infrared gas analyzer (IRGA; GMP 343, Vaisala, Helsinki, Finland). Leaf water potential before dawn was periodically measured with a pressure chamber (Model 1505-D-EXP, PMS, Corvallis, OR, USA). On the same days, 2 or 3 branches 30–50 cm in length were harvested before dawn, and twigs with 6-mm diameters, recut under water from the branches, were used for the measurements of PLC and NSCs contents. The measurement of twig hydraulic conductivity was conducted by adding gravimetric 5-kPa hydraulic pressure to the end of the examined twigs40. The NSCs content in the xylem sapwood was measured by quantifying the sugar content with the phenol-sulfuric acid method, and the starch content was assessed with the mutarotase-glucose oxidase method using a Glucose C-II test kit (Wako, Tokyo, Japan).
To evaluate the association between the dynamics of NSCs and water relations, the time-course changes in xylem sugar and starch contents and the amounts of xylem sugar and starch were plotted against PLC with trail of the changes. The change rates in sugar and starch contents (g g−1 day−1) were shown as:

To test the contribution of water relations on the dynamics of NSCs within xylem, the relationships between NSCs content (absolute contents and change rates) and water conditions (ψpre and PLC) were shown with the linear regression model (see Table S1).
Additional Information
How to cite this article: Yoshimura, K. et al. The dynamics of carbon stored in xylem sapwood to drought-induced hydraulic stress in mature trees. Sci. Rep. 6, 24513; doi: 10.1038/srep24513 (2016).
References
Adams, H. D. et al. Temperature sensitivity of drought-induced tree mortality portends increased regional die-off under grobal-change-type drought. Proc. Natl. Acad. Sci. USA 106, 7063–7066 (2009).
Allen, C. D. et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 259, 660–684 (2010).
Hoffmann, W. A., Marchin, R. M., Abit, P. & Lau, O. L. Hydraulic failure and tree dieback are associated with high wood density in a temoerate forest under extreme drought. Glob. Change Biol. 17, 2731–2742 (2011).
Nowacki, G. J. & Abrams, M. D. Is climate an important driver of post-European vegetation change in the Eastern Unted States? Glob. Change Biol. 21, 314–334 (2015).
Xu, C., McDowell, N. G., Sevanto, S. & Fisher, R. A. Our limited ability to predict vegetation dynamics under water stress. New Phytol. 200, 298–300 (2013).
Davis, S. D. et al. Shoot dieback during prolonged drought in Ceanothus (Rhamnaceae) chaparral of California: a possible case of hydraulic failure. Am. J. Bot. 89, 820–828 (2002).
McDowell, N. et al. Mechanism of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol, 178, 719–739 (2008).
McDowell, N. G. Mechanisms linking drought, hydraulics, carbon metabolism, and vegetation mortality. Plant Physiol. 155, 1051–1059 (2011).
Anderegg, W. R. et al. The roles of hydraulic and carbon stress in a widespread climate-induced forest die-off. Proc. Natl. Acad. Sci. USA 109, 233–237 (2012).
Rowland, L. et al. Death from drought in tropical forests is triggered by hydraulics not carbon starvation. Nature 528, 119–122 (2015).
Nardini, A., Battistuzzo, M. & Savi, T. Shoot desiccation and hydraulic failure in temperate woody angiosperms during an extreme summer drought. New Phytol. 200, 322–329 (2013).
O’Brien, M., Leuzinger, S., Philipson, C. D., Tay, J. & Hector, A. Drought survival of tropical tree seedlings enhanced by non-structural carbohydrate levels. Nature Clim. Change 4, 710–714 (2014).
Sala, A., Piper, F. & Hoch, G. Physiological mechanisms of drought-induced tree mortality are far from being resolved. New Phytol. 186, 274–281 (2010).
Zeppel, M. J. B., Anderegg, W. R. L. & Adams, H. D. Forest mortality due to drought: latest insights, evidence and unresolved questions on physiological pathways and consequences of tree death. New Phytol. 197, 372–374 (2013).
Hacke, U. G. & Sperry, J. S. Limits to xylem refilling under negative pressure in Laurus nobilis and Acer negundo . Plant Cell Environ. 26, 303–311 (2003).
Salleo, S., Lo Gullo, M. A. & de Paoli, D. Xylem recovery from cavitation-induced embolism in young plants of Laurus nobilis: a possible mechanism. New Phytol. 132, 47–56 (1996).
Yazaki, K., Sano, Y., Fujikawa, S., Nakano, T. & Ishida, A. Response to dehydration and irrigation in invasive and native sapling: osmotic adjustment versus leaf shedding. Tree Physiol. 30, 597–607 (2010).
Ogasa, M., Miki, N. H., Murakami, Y. & Yoshikawa, K. Recovery performance in xylem hydraulic conductivity is correlated with cavitation resistance for temperate deciduous tree species. Tree Physiol. 33, 335–344 (2013).
Knipfer, T., Eustis, A., Brodersen, C., Walker, A. M. & McElrone, A. J. Grapevine species from varied native habitats exhibit differences in embolism formation/repair associated with leaf gas exchange and root pressure. Plant Cell Environ. 38, 1503–1513 (2015).
Sauter, J. J., Wisniewski, M. & Witt, W. Interrelationships between ultrastructure, sugar levels, and frost hardiness of ray parenchyma cells during frost acclimation and deacclimation in poplar (Populus canadensis Moench) wood. J. Plant Physiol. 149, 451–461 (1996).
Bucci, S. J., Scholz, F. G., Goldstein, G., Meinzer, F. C. & Sternberg, L. D. S. L. Dynamic changes in hydraulic conductivity in petioles of two savanna tree species: factors and mechanisms contributing to the refilling of embolized vessels. Plant Cell Environ. 26, 1633–1645 (2003).
Salleo, S., Gullo, M. A. L., Trifilo, P. & Nardini, A. New evidence for a role of vessel-associated cells and phloem in the rapid xylem refilling of cavitated stems of Laurus nobilis L. Plant Cell Environ. 27, 1065–1076 (2004).
Zwieniecki, M. A. & Holbrook, N. M. Confronting Maxwell’s demoon: biophysics of xylem embolism repair. Trends Plant Sci. 31, 530–534 (2009).
Nardini, A., Lo Gullo, M. A. & Salleo, S. Refilling embolized xylem conduits: Is it a matter of phloem unloading? Plant Sci. 180, 604–611 (2011).
Secchi, F. & Zwieniecki, M. A. Sensing embolism in xylem vessels: the role of sucrose as a trigger for refilling. Plant Cell Environ. 34, 514–524 (2011).
Latt, C. R., Nair, P. K. R. & Kang, B. T. Reserve carbohydrate levels in the boles and structural roots of five multipurpose tree species in a seasonally dry tropical climate. For. Ecol. Manag. 146, 145–158 (2001).
Arndt, S. K., Livesley, S. J., Merchant, A., Bleby, T. M. & Grierson, P. F. Quercitol and osmotic adaptation of field-grown Eucalyptus under seasonal drought stress. Plant Cell Environ. 31, 915–924 (2008).
Mitchell, P. J. et al. Drought response strategies define the relative contributions of hydraulic dysfunction and carbohydrate depletion during tree mortality. New Phytol. 197, 862–872 (2013).
Sevanto, S., McDowell, N. G., Dickman, L. T., Pangle, R. & Pockman, W. T. How do trees die? A test of the hydraulic failure and carbon starvation hypotheses. Plant Cell Environ. 37, 153–161 (2014).
Ishida, A. et al. Coordination between leaf and stem traits related to leaf carbon gain and hydraulics across 32 drought-tolerant angiosperms. Oecologia 156, 193–202 (2008).
Pivovaroff, A. L., Sack, L. & Santiago, L. S. Coordination of stem and leaf hydraulic conductance in southern California shrubs: a test of the hydraulic segmentation hypothesis. New Phytol. 203, 842–850 (2014).
Meinzer, F. C. & McCulloh, K. A. Xylem recovery from drought-induced embolism: where is the hydraulic point of no return? Tree Physiol. 33, 331–334 (2013).
Atkin, O. K. & Macherel, D. The crucial role of plant mitochondria in orchestrating drought tolerance. Ann. Bot. 103, 584–597 (2009).
Sala, A. Lack of direct evidence for the carbon-starvation hypothesis to explain drought-induced mortality in trees. Proc. Natl Acad. Sci. USA 106, 68 (2009).
Kamaluddin, M. & Zwiazek, J. J. Naphthenic acids inhibit root water transport, gas ezchange and leaf growth in aspen (Populus tremuloides) seedlings. Tree Physiol. 22, 1265–1270 (2002).
Alves, G. et al. Plasma membrane H(+)-ATPase, succinate and isocitrate dehydrogenases activities of vessel-associated cells in walnut trees. J. Plant Physiol. 158, 1263–1271 (2001).
Secchi, F. & Zwieniecki, M. A. Patterns of PIP gene expression in Populus trichocarpa during recovery from xylem embolism suggest a major role for the PIP1 aquaporin subfamily as moderators of refilling process. Plant Cell Environ. 33, 1285–1297 (2010).
Osone, Y. et al. Responses of gas-exchange rates and water relations to annual fluctuations of weather in tree species of urban street trees. Tree Physiol. 34, 1056–1068 (2004).
Leuzinger, L. S. & Thomas, R. Q. How do we improve Earth system models? Integrating Earth system models, ecosystem models, experiments and long-term data. New Phytol. 191, 15–18 (2011).
Sperry, J. S., Donnelly, J. R. & Tyree, M. T. A method for measuring hydraulic conductivity and embolism in xylem. Plant Cell Environ. 11, 35–40 (1988).
Acknowledgements
This research was conducted in a national park of Japan and in a World Natural Heritage site recognized by UNESCO. Before the measurements, we received permission for examination from the Forest Agency of Japan and from the government of Tokyo. We wish to thank Ms. K. Otonari, Mr. N. Sugimura and Ms. T. Aihara for valuable help with the field and laboratory measurements. This study was supported by grants-in-aid from the Japan Society for the Promotion of Science (nos. 13J09663 and 26870833 to KY1; 18255011, 24370009, 15K00625 and 15K14746 to AI; 22370010, 26257405, 15H04420 and 22255004 to JY; 19780129 and 23780190 to KY3; 25850102, 14J08279 to MYO) and Sasakawa Scientific Research Grant (no. 27–510 to STS).
Author information
Authors and Affiliations
Contributions
All authors except for K.Yazaki, M.Y.O. and J.Y. conducted the field measurements. K.Yoshimura, A.I. and J.Y. mainly wrote the manuscript. K.Yoshimura, S.-T.S., K.Yazaki and M.Y.O. measured carbohydrate in our laboratory. All authors discussed and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yoshimura, K., Saiki, ST., Yazaki, K. et al. The dynamics of carbon stored in xylem sapwood to drought-induced hydraulic stress in mature trees. Sci Rep 6, 24513 (2016). https://doi.org/10.1038/srep24513
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep24513
- Springer Nature Limited
This article is cited by
-
Non-structural carbohydrate concentrations in contrasting dry and wet years in early- and late-successional boreal forest trees
Trees (2023)
-
Sapwood density underlies xylem hydraulics and stored carbohydrates across 13 deciduous tree species in a seasonally dry tropical forest in Thailand
Trees (2023)
-
Significance of current weather conditions for foliar traits of old-growth sessile oak (Quercus petraea Liebl) trees
Trees (2022)
-
Initial hydraulic failure followed by late-stage carbon starvation leads to drought-induced death in the tree Trema orientalis
Communications Biology (2019)
-
Xylem hydraulic safety and efficiency in relation to leaf and wood traits in three temperate Acer species differing in habitat preferences
Trees (2019)