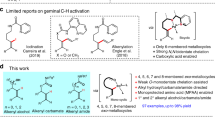
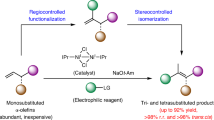
Abstract
The availability of structurally and stereochemically diverse chemical scaffolds is important to support continual development of drug discovery, functional materials and asymmetric synthesis. Axially chiral 1,3-dienes, particularly acyclic ones, are potentially valuable yet underutilized skeletons due to concerns about chiral stability and the absence of suitable synthetic conditions. Here we show an organocatalytic strategy for the direct functionalization of alkenyl C–H bonds, enabling the challenging atroposelective construction of a broad range of acyclic chiral 1,3-dienes in a modular manner. Intensive studies on the reaction mechanism show that the current strategy not only bypasses the traditional indirect process for olefinic C–H functionalization but also represents a departure from transition-metal-catalysed C(sp2)–H activation. These findings are expected to open avenues for research on olefin chemistry and to inspire investigation into the organocatalytic activation of other inert structures.








Similar content being viewed by others
Data availability
The X-ray crystallographic coordinates for the structure of 2w reported in this Article have been deposited at the CCDC under deposition number CCDC 2184813. These data can be obtained free of charge from the CCDC via http://www.ccdc.cam.ac.uk/data_request/cif. The data supporting the findings of this work are provided in the Supplementary Information, including experimental procedures, the characterization of new compounds and data of the DFT calculations, or are available from the authors upon reasonable request.
References
Yoon, T. P. & Jacobsen, E. N. Privileged chiral catalysts. Science 299, 1691–1693 (2003).
Carroll, M. P. & Guiry, P. J. P,N ligands in asymmetric catalysis. Chem. Soc. Rev. 43, 819–833 (2014).
Giacalone, F., Gruttadauria, M., Agrigento, P. & Noto, R. Low-loading asymmetric organocatalysis. Chem. Soc. Rev. 41, 2406–2447 (2012).
Akiyama, T. & Mori, K. Stronger Brønsted acids: recent progress. Chem. Rev. 115, 9277–9306 (2015).
Teichert, J. F. & Feringa, B. L. Phosphoramidites: privileged ligands in asymmetric catalysis. Angew. Chem. Int. Ed. 49, 2486–2528 (2010).
Noyori, R. & Takaya, H. BINAP: an efficient chiral element for asymmetric catalysis. Acc. Chem. Res. 23, 345–350 (1990).
Cheng, J. K., Xiang, S.-H., Li, S., Ye, L. & Tan, B. Recent advances in catalytic asymmetric construction of atropisomers. Chem. Rev. 121, 4805–4902 (2021).
Zask, A., Murphy, J. & Ellestad, G. A. Biological stereoselectivity of atropisomeric natural products and drugs. Chirality 25, 265–274 (2013).
Szlávik, Z. et al. Structure-guided discovery of a selective Mcl-1 inhibitor with cellular activity. J. Med. Chem. 62, 6913–6924 (2019).
Su, S. et al. Biphenyl acid derivatives as APJ receptor agonists. J. Med. Chem. 62, 10456–10465 (2019).
Zheng, Z. et al. Three-dimensional control of the helical axis of a chiral nematic liquid crystal by light. Nature 531, 352–356 (2016).
Bisoyi, H. K. & Li, Q. Light-driven liquid crystalline materials: from photo-induced phase transitions and property modulations to applications. Chem. Rev. 116, 15089–15166 (2016).
Pu, L. Fluorescence of organic molecules in chiral recognition. Chem. Rev. 104, 1687–1716 (2004).
Doherty, S. et al. Zirconium-mediated synthesis of a new class of 1,4-bis(diphenylphosphino)-1,3-butadiene-bridged diphosphine, NUPHOS: highly efficient catalysts for palladium-mediated cross couplings. J. Am. Chem. Soc. 123, 5110–5111 (2001).
Doherty, S. et al. Lewis acid platinum complexes of conformationally flexible NUPHOS diphosphines: highly efficient catalysts for the carbonyl−ene reaction. Organometallics 24, 5945–5955 (2005).
Doherty, S., Smyth, C. H., Harriman, A., Harrington, R. W. & Clegg, W. Can a butadiene-based architecture compete with its biaryl counterpart in asymmetric catalysis? Enantiopure Me-CATPHOS, a remarkably efficient ligand for asymmetric hydrogenation. Organometallics 28, 888–895 (2009).
Doherty, S., Knight, J. G. & Mehdi-Zodeh, H. Asymmetric carbonyl-ene and Friedel–Crafts reactions catalyzed by Lewis acid platinum group metal complexes of the enantiopure atropisomeric biaryl-like diphosphine (S)-Me2-CATPHOS: a comparison with BINAP. Tetrahedron Asymmetry 23, 209–216 (2012).
Ogasawara, M. et al. Atropisomeric chiral dienes in asymmetric catalysis: C2-symmetric (Z,Z)-2,3-bis[1-(diphenylphosphinyl)ethylidene]tetralin as a highly active Lewis base organocatalyst. Angew. Chem. Int. Ed. 52, 13798–13802 (2013).
Boer, F. P., Doorakian, G. A., Freedman, H. H. & McKinley, S. V. A study of the rotational process in sterically hindered dienes. J. Am. Chem. Soc. 92, 1225–1233 (1970).
Köbrich, G. et al. Chirale butadiene, 1H-NMR-spektroskopischer nachweis der behinderten rotation bei hexasubstituierten butadienen. Chem. Ber. 105, 3794–3806 (1972).
Rösner, M. & Köbrich, G. Enantiomerization of optically stable substituted butadienes. Angew. Chem. Int. Ed. 14, 708–709 (1975).
Pasto, D. J. & Scheidt, W. R. X-ray and dynamic nuclear magnetic resonance structural study of a 1,2-bis exocyclic diene. An example of a severely skewed diene. J. Org. Chem. 40, 1444–1447 (1975).
Yamada, M., Rivera-Fuentes, P., Schweizer, W. B. & Diederich, F. Optical stability of axially chiral push–pull-substituted buta-1,3-dienes: effect of a single methyl group on the C60 surface. Angew. Chem. Int. Ed. 49, 3532–3535 (2010).
Ervin, K. M. & DeTuri, V. F. Anchoring the gas-phase acidity scale. J. Phys. Chem. A 106, 9947–9956 (2002).
Beletskaya, I. P. & Cheprakov, A. V. The Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 100, 3009–3066 (2000).
Wei, Y. & Shi, M. Recent advances in organocatalytic asymmetric Morita–Baylis–Hillman/aza-Morita–Baylis–Hillman reactions. Chem. Rev. 113, 6659–6690 (2013).
Tang, S., Liu, K., Liu, C. & Lei, A. Olefinic C–H functionalization through radical alkenylation. Chem. Soc. Rev. 44, 1070–1082 (2015).
Mbofana, C. T. & Miller, S. J. Diastereo- and enantioselective addition of anilide-functionalized allenoates to N‑acylimines catalyzed by a pyridylalanine-based peptide. J. Am. Chem. Soc. 136, 3285–3292 (2014).
Zhang, J. et al. Recent advances in chelation-assisted site- and stereoselective alkenyl C–H functionalization. Chem. Soc. Rev. 50, 3263–3314 (2021).
Schreib, B. S. & Carreira, E. M. Palladium-catalyzed regioselective C–H iodination of unactivated alkenes. J. Am. Chem. Soc. 141, 8758–8763 (2019).
Jin, L., Zhang, P., Li, Y., Yu, X. & Shi, B.-F. Atroposelective synthesis of conjugated diene-based axially chiral styrenes via Pd (II)-catalyzed thioether-directed alkenyl C–H olefination. J. Am. Chem. Soc. 143, 12335–12344 (2021).
Mukherjee, S., Yang, J. W., Hoffmann, S. & List, B. Asymmetric enamine catalysis. Chem. Rev. 107, 5471–5569 (2007).
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008).
Metrano, A. J. & Miller, S. J. Peptide-based catalysts reach the outer sphere through remote desymmetrization and atroposelectivity. Acc. Chem. Res. 52, 199–215 (2019).
Silvi, M. & Melchiorre, P. Enhancing the potential of enantioselective organocatalysis with light. Nature 554, 41–49 (2018).
Chen, J. et al. Carbonyl catalysis enables a biomimetic asymmetric Mannich reaction. Science 360, 1438–1442 (2018).
Tsuji, N. et al. Activation of olefins via asymmetric Brønsted acid catalysis. Science 359, 1501–1505 (2018).
Beeson, T. D., Mastracchio, A., Hong, J.-B., Ashton, K. & MacMillan, D. W. C. Enantioselective organocatalysis using SOMO activation. Science 316, 582–585 (2007).
Mao, J.-H. et al. Organocatalyst-controlled site-selective arene C–H functionalization. Nat. Chem. 13, 982–991 (2021).
Gustafson, J. L., Lim, D. & Miller, S. J. Dynamic kinetic resolution of biaryl atropisomers via peptide-catalyzed asymmetric bromination. Science 328, 1251–1255 (2010).
Beleh, O. M., Miller, E., Toste, F. D. & Miller, S. J. Catalytic dynamic kinetic resolutions in tandem to construct two axis terphenyl atropisomers. J. Am. Chem. Soc. 142, 16461–16470 (2020).
Rauniyar, V., Lackner, A. D., Hamilton, G. L. & Toste, F. D. Asymmetric electrophilic fluorination using an anionic chiral phase-transfer catalyst. Science 334, 1681–1683 (2011).
Wang, Y.-M., Wu, J., Hoong, C., Rauniyar, V. & Toste, F. D. Enantioselective halocyclization using reagents tailored for chiral anion phase-transfer catalysis. J. Am. Chem. Soc. 134, 12928–12931 (2012).
Lu, T., Lu, Z., Ma, Z.-X., Zhang, Y. & Hsung, R. P. Allenamides: a powerful and versatile building block in organic synthesis. Chem. Rev. 113, 4862–4904 (2013).
Zhang, X. et al. An enantioconvergent halogenophilic nucleophilic substitution (SN2X) reaction. Science 363, 400–404 (2019).
Petrova, M. et al. Experimental and theoretical studies of bromination of diethyl 2,4,6-trimethyl-1,4-dihydropyridine-3,5-dicarboxylate. Heteroat. Chem. 25, 114–126 (2014).
Acknowledgements
We are grateful for financial support from the National Key R&D Program of China (2022YFA1503703 to B.T. and 2021YFF0701604 to B.T.), National Natural Science Foundation of China (22231004 to B.T., 22371113 to Y.-B.W., 22271135 to S.-H.X. and 21825105 to B.T.), Guangdong Innovative Program (2019BT02Y335 to B.T.) and Shenzhen Science and Technology Program (JCYJ20220818100604009 to Y.-B.W., JCYJ20210324105005015 to S.-H.X., JCYJ20210324104212035 to Y.-B.W. and KQTD20210811090112004 to B.T.). Computational work was supported by the resources from the Center of Computational Science and Engineering at SUSTech.
Author information
Authors and Affiliations
Contributions
B.T. and Y.-B.W. conceived and directed the project. Q.-H.W. and Y.-B.W. designed and performed the experiments. K.N.H. directed the DFT calculations and mechanism analysis. M.D. and Y.C. performed the DFT calculations and mechanism analysis. J.K.C. and S.-H.X. helped with the collection of some compounds and data analysis. B.T., K.N.H., Y.-B.W., Q.-H.W., P.Y., J.K.C. and S.-H.X. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Yan Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6, Tables 1–6, Methods and references.
Supplementary Data 1
Checkcif file of 2w.
Supplementary Data 2
Cif file of 2w.
Supplementary Data 3
Structure factors of 2w.
Supplementary Data 4
Structure factors of 2w.
Supplementary Data 5
Data of DFT Calculations Coordinates
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, QH., Duan, M., Chen, Y. et al. Organocatalytic olefin C–H functionalization for enantioselective synthesis of atropisomeric 1,3-dienes. Nat Catal 7, 185–194 (2024). https://doi.org/10.1038/s41929-023-01097-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-01097-x
- Springer Nature Limited