Abstract
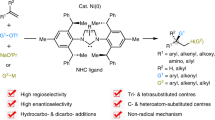
Despite tremendous efforts aimed at devising methods for stereoselective alkene synthesis, critical challenges are yet to be addressed. Direct access to a diverse range of 1-aryl(boryl)-1-methyl-functionalized tri- and tetrasubstituted trans alkenes, entities that are prevalent in many important molecules, through a catalytic manifold from readily available α-olefin substrates remains elusive. Here, we demonstrate that catalytic amounts of a non-precious N-heterocyclic carbene–Ni(I) complex in conjunction with a sterically bulky base promote site- and trans-selective union of monosubstituted olefins with a wide array of electrophilic reagents to deliver tri- and tetrasubstituted alkenes in up to 92% yield and >98% regio- and stereoselectivity. The protocol is amenable to the preparation of carbon- and heteroatom-substituted C=C bonds, providing distinct advantages over existing transformations. Utility is highlighted through concise stereoselective synthesis of biologically active compounds.





Similar content being viewed by others
Data availability
All data are available from the corresponding authors upon reasonable request. Crystallographic data for the structure reported in this article have been deposited at the Cambridge Crystallographic Data Centre, under deposition number CCDC 2018464 (7d). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Negishi, E., Wang, G., Rao, H. & Xu, Z. Alkyne elementometalation-Pd-catalyzed cross-coupling. Toward synthesis of all conceivable types of acyclic alkenes in high yields, efficiently, selectively, economically, and safely: ‘green’ way. J. Org. Chem. 75, 3151–3182 (2010).
Prunet, J. Progress in metathesis through natural product synthesis. Eur. J. Org. Chem. 2011, 3634–3647 (2011).
Negishi, E. et al. Recent advances in efficient and selective synthesis of di-, tri-, and tetrasubstituted alkenes via Pd-catalyzed alkenylation–carbonyl olefination synergy. Acc. Chem. Res. 41, 1474–1485 (2008).
Flynn, A. B. & Ogilvie, W. W. Stereocontrolled synthesis of tetrasubstituted olefins. Chem. Rev. 107, 4698–4745 (2007).
Eissen, M. & Lenoir, D. Mass efficiency of alkene syntheses with tri- and tetrasubstituted double bonds. ACS Sustain. Chem. Eng. 5, 10459–10473 (2017).
Maryanoff, B. E. & Reitz, A. B. The Wittig olefination reaction and modifications involving phosphoryl-stabilized carbanions. Stereochemistry, mechanism, and selected synthetic aspects. Chem. Rev. 89, 863–927 (1989).
Braun, M.-G., Quiclet-Sire, B. & Zard, S. Z. A highly stereoselective, modular route to (E)-vinylsulfones and to (Z)- and (E)-alkenes. J. Am. Chem. Soc. 133, 15954–15957 (2011).
Li, B. X. et al. Highly stereoselective synthesis of tetrasubstituted acyclic all-carbon olefins via enol tosylation and Suzuki–Miyaura coupling. J. Am. Chem. Soc. 139, 10777–10783 (2017).
Trost, B. M. & Ball, Z. T. Addition of metalloid hydrides to alkynes: hydrometallation with boron, silicon, and tin. Synthesis 2005, 853–887 (2005).
Itoh, T., Shimizu, Y. & Kanai, M. Ligand-enabled, copper-catalyzed regio- and stereoselective synthesis of trialkylsubstituted alkenylboronates from unactivated internal alkynes. J. Am. Chem. Soc. 138, 7528–7531 (2016).
Shimkin, K. W. & Montgomery, J. Synthesis of tetrasubstituted alkenes by tandem metallacycle formation/cross-electrophile coupling. J. Am. Chem. Soc. 140, 7074–7078 (2018).
Zhu, C. et al. A multicomponent synthesis of stereodefined olefins via nickel catalysis and single electron/triplet energy transfer. Nat. Catal. 2, 678–687 (2019).
Cuvigny, T., du Penhoat, H. & Julia, M. Isomérisation cis trans régiosélective de doubles liaison trisubstitutées. Tetrahedr. Lett. 21, 1331–1334 (1980).
Larionov, E., Li, H. & Mazet, C. Well-defined transition metal hydrides in catalytic isomerizations. Chem. Commun. 50, 9816–9826 (2014).
Kapat, A., Sperger, T., Guven, S. & Schoenebeck, F. E-olefins through intramolecular, radical relocation. Science 363, 391–396 (2019).
Zhang, S. et al. Cobalt(II)-catalyzed stereoselective olefin isomerization: facile access to acyclic trisubstituted alkenes. J. Am. Chem. Soc. 142, 8910–8917 (2020).
Yu, X., Zhao, H., Li, P. & Koh, M. J. Iron-catalyzed tunable and site-selective olefin transposition. J. Am. Chem. Soc. 142, 18223–18230 (2020).
Nguyen, T. T., Koh, M. J., Mann, T. J., Schrock, R. R. & Hoveyda, A. H. Synthesis of E- and Z-trisubstituted alkenes by catalytic cross-metathesis. Nature 552, 347–354 (2017).
Mu, Y., Nguyen, T. T., Koh, M. J., Schrock, R. R. & Hoveyda, A. H. E- and Z-, di- and tri-substituted alkenyl nitriles through catalytic cross-metathesis. Nat. Chem. 11, 478–487 (2019).
Mukherjee, N., Planer, S. & Grela, K. Formation of tetrasubstituted C–C double bonds via olefin metathesis: challenges, catalysts, and applications in natural product synthesis. Org. Chem. Front. 5, 494–516 (2018).
Qin, L., Ren, X., Lu, Y., Li, Y. & Zhou, J. Intermolecular Mizoroki–Heck reaction of aliphatic olefins with high selectivity for substitution at the internal position. Angew. Chem. Int. Ed. 51, 5915–5919 (2012).
Standley, E. & Jamison, T. F. Simplifying nickel (0) catalysis: an air-stable nickel precatalyst for the internally selective benzylation of terminal alkenes. J. Am. Chem. Soc. 135, 1585–1592 (2013).
Tasker, S., Gutierrez, A. & Jamison, T. F. Nickel-catalyzed Mizoroki–Heck reaction of aryl sulfonates and chlorides with electronically unbiased terminal olefins: high selectivity for branched products. Angew. Chem. Int. Ed. 53, 1858–1861 (2014).
Zheng, C., Wang, D. & Stahl, S. S. Catalyst-controlled regioselectivity in the synthesis of branched conjugated dienes via aerobic oxidative Heck reactions. J. Am. Chem. Soc. 134, 16496–16499 (2012).
Reid, W. B. & Watson, D. A. Synthesis of trisubstituted alkenyl boronic esters from alkenes using the boryl-Heck reaction. Org. Lett. 20, 6832–6835 (2018).
Diccianni, J. B., Heitmann, T. & Diao, T. Nickel-catalyzed reductive cycloisomerization of enynes with CO2. J. Org. Chem. 82, 6895–6903 (2017).
Dible, B. R., Sigman, M. S. & Arif, A. M. Oxygen-induced ligand dehydrogenation of a planar bis-µ-chloronickel(I) dimer featuring an NHC ligand. Inorg. Chem. 44, 3774–3776 (2005).
Green, S. A. et al. The high chemofidelity of metal-catalyzed hydrogen atom transfer. Acc. Chem. Res. 51, 2628–2640 (2018).
Sommer, H., Juliá-Hernández, F., Martin, R. & Marek, I. Walking metals for remote functionalization. ACS Cent. Sci. 4, 153–165 (2018).
Janssen-Müller, D., Sahoo, B., Sun, S.-Z. & Martin, R. Tackling remote sp3 C–H functionalization via Ni-catalyzed ‘chain-walking’ reactions. Isr. J. Chem. 60, 195–206 (2020).
Lee, W.-C., Wang, C.-H., Lin, Y.-H., Shih, W.-C. & Ong, T.-G. Tandem isomerization and C–H activation: regioselective hydroheteroarylation of allylarenes. Org. Lett. 15, 5358–5361 (2013).
Hazari, N., Nelvin, P. R. & Beromi, M. M. Well-defined nickel and palladium precatalysts for cross-coupling. Nat. Rev. Chem. 1, 0025 (2017).
Wang, S. C. et al. Mechanism of the Ni(0)-catalyzed vinylcyclopropane–cyclopentene rearrangement. J. Org. Chem. 74, 7822–7833 (2009).
Sakurada, J. & Satoh, T. Direct N- and C-alkenylation of nitrogen-containing heterocycles with magnesium alkylidene carbenoids. Tetrahedron 63, 3806–3817 (2007).
Takeda, T., Sato, K. & Tsubouchi, A. A new route to enol ethers. Synthesis 9, 1457–1465 (2004).
Yi, H. et al. Photocatalytic dehydrogenative cross-coupling of alkenes with alcohols or azoles without external oxidant. Angew. Chem. Int. Ed. 56, 1120–1124 (2017).
Miyazawa, M., Ishibashi, N., Ohnuma, S. & Miyashita, M. Stereospecific internal alkylation of terminal γ,δ-epoxy acrylates. Tetrahedron Lett. 38, 3419–3422 (1997).
Mu, Y., Nguyen, T. N., van der Mei, F. W., Schrock, R. S. & Hoveyda, A. H. Traceless protection for more broadly applicable olefin metathesis. Angew. Chem. Int. Ed. 58, 5365–5370 (2019).
Baumann, M. & Baxendale, I. R. An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles. Beilstein J. Org. Chem. 9, 2265–2319 (2013).
Brandi, A., Cicchi, S. & Cordero, F. M. Novel syntheses of azetidines and azetidinones. Chem. Rev. 108, 3988–4035 (2008).
Ray, S. & Sangita The potent triarylethylene pharmacophore. Drugs Future 29, 185–203 (2004).
Ephritikhine, M. A new look at the McMurry reaction. Chem. Commun. 2549–2554 (1998).
Ganiu, M. O., Cleveland, A. H., Paul, J. L. & Kartika, R. Triphosgene and DMAP as mild reagents for chemoselective dehydration of tertiary alcohols. Org. Lett. 21, 5611–5615 (2019).
Mannathan, S., Jeganmohan, M. & Cheng, C.-H. Nickel-catalyzed borylative coupling of alkynes, enones, and bis(pinacolato)diboron as a route to substituted alkenyl boronates. Angew. Chem. Int. Ed. 48, 2192–2195 (2009).
Mjalli, A. M. M. et al. Substituted imidazole derivatives, compositions, and methods of use as ptpase inhibitors. US patent WO2007/089857 A2 (2007).
Brown, H. C., Scouten, C. G. & Liotta, R. Hydroboration. 50. Hydroboration of representative alkynes with 9-borabicyclo[3.3.1]nonane—a simple synthesis of versatile vinyl bora and gem-dibora intermediates. J. Am. Chem. Soc. 101, 96–99 (1979).
McMurray, J., Mandal, P. K., Morlacchi, P., Knight, M. & Corry, D. B. STAT6 inhibitors. US patent WO2014/182928 A2 (2014).
Guerrero, P. G. Jr. et al. Synthesis of arotinoid acid and temarotene using mixed (Z)-1,2-bis(organylchalcogene)-1-alkene as precursor. Tetrahedron Lett. 53, 5302–5305 (2012).
Deloux, L. & Srebnik, M. Stereospecific synthesis of temarotene, its structural isomers, and mixed triaryl alkenes from gem-borazirconocene alkenes. J. Org. Chem. 60, 3276–3277 (1995).
Mitchell, P. & Teall, M. Sulfonyl piperidine derivatives and their use for treating prokineticin mediated gastrointestinal disorders. Patent WO2016/075457 A1 (2016).
Richalet, F., Weiler, S., El Shemerly, M. & Lane, H. Mitochondrial inhibitors for the treatment of proliferation disorders. Patent WO2019/072978 A1 (2019).
Sampson, P. B. et al. Kinase inhibitors and method of treating cancer with same. Patent WO2010/115279 A1 (2010).
Kalinin, D. V. et al. Novel potent proline-based metalloproteinase inhibitors: design, (radio)synthesis, and first in vivo evaluation as radiotracers for positron emission tomography. J. Med. Chem. 59, 9541–9559 (2016).
Diccianni, J., Lin, Q. & Diao, T. Mechanisms of nickel-catalyzed coupling reactions and applications in alkene functionalization. Acc. Chem. Res. 53, 906–919 (2020).
Shevick, S. L., Obradors, C. & Shenvi, R. A. Mechanistic interrogation of Co/Ni-dual catalyzed hydroarylation. J. Am. Chem. Soc. 140, 12056–12068 (2018).
Chen, F. et al. Remote migratory cross-electrophile coupling and olefin hydroarylation reactions enabled by in situ generation of NiH. J. Am. Chem. Soc. 139, 13929–13935 (2017).
Acknowledgements
This research was supported by the National University of Singapore Academic Research Fund Tier 1: R-143-000-A77-114 (M.J.K.) and by the National Institutes of Health grant no. R35GM137797 (O.G.). O.G. is grateful to the University of Maryland College Park for start-up funds and computational resources from UMD Deepthought2 and MARCC/BlueCrab HPC clusters and XSEDE (grant nos. CHE160082 and CHE160053). We thank G.K. Tan for X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
C.-F.L. and H.W. contributed equally to this work and are listed in alphabetical order in the author list. C.-F.L., H.W. and H.Z. developed the method and carried out the mechanistic studies. R.T.M. carried out the DFT calculations. O.G. directed the DFT studies. M.J.K. directed the investigations and wrote the manuscript with revisions provided by the other authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Catalysis thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Preliminary studies on the boron variant of the Ni-catalysed tandem reaction.
a, Control experiments revealed that borylation likely occurs, affording observed intermediate 11g′, prior to C=C bond migration. b, A tentative mechanistic pathway involving formation of a putative nickel-boryl species IX to promote boryl-Heck reaction. Under typical circumstances where catalytic amounts of LiOt-Bu were used, DMPU likely served as a hydride acceptor to afford the corresponding Ni-alkoxide VIII and turn over the catalytic cycle. In other instances, more base might be needed to regenerate the active Ni-tert-butoxide species (VIII, R = t-Bu). G, functional group; pin, pinacolato; DMPU, N,N′-dimethylpropyleneurea; IPr, 1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2, Figs. 1–4, Methods and References.
Supplementary Data 1
Crystallographic data for compound 7d.
Rights and permissions
About this article
Cite this article
Liu, CF., Wang, H., Martin, R.T. et al. Olefin functionalization/isomerization enables stereoselective alkene synthesis. Nat Catal 4, 674–683 (2021). https://doi.org/10.1038/s41929-021-00658-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-021-00658-2
- Springer Nature Limited
This article is cited by
-
Enantioselective synthesis of multifunctional alkylboronates via N-heterocyclic carbene–nickel-catalysed carboboration of alkenes
Nature Synthesis (2024)
-
Intermolecular trans-bis-silylation of terminal alkynes
Nature Synthesis (2023)