Abstract
Rice is a crop that requires high amount of water, and the drought is a major constraint in paddy cultivation. Water stress condition frequently prevails due to shortage of rain which results in significantly reduced plant growth and yield of rice. In the present study capability of Trichoderma spp. in imparting drought tolerance to rice, Oryza sativa was explored. Eleven local strains of Trichoderma spp. were applied to rice cv. Swarna Sub-1 through soil application (2 g/kg soil) and seed treatment (20 g/kg seed) under 0, 25, 50 and 75% less watering of the recommended amount. The soil application of T. harzianum AMUTHZ84 significantly promoted the shoot and root length (23.6 and 21.3%) followed by seed treatment (19.7 and 18.2%) under recommended level of irrigation condition (100% irrigation). Next in effectiveness was T. viride AMUTVR73 (21.5 and 18.1%) over untreated control. However, under 75% water availability, soil application with T. harzianum AMUTHZ82 was found superior over other isolates in enhancing shoot and root length (17.7 and 16.4%). The same isolate was also recorded to be superior under 50% (12.4 and 10.1%) and 25% water availability (9.3 and 8.1%) in enhancing the plant growth and biomass of rice cv. Swarna Sub-1. The isolate also significantly enhanced the leaf pigments, and photosynthesis in the rice plants grown under 25–75% water stress condition. In general, soil application of Trichoderma isolates was found more effective than seed treatment, and the T. harzianum AMUTHZ82 provided 8–17% enhancement in the plant growth, biomass, leaf pigments and photosynthesis of rice cv. Swarna Sub-1 grown under 25–75% water stress condition.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Rice is the primary and fundamental food source providing 20% of the global calories consumed by over half the world's population and fulfils the dietary requirements of a large proportion of the human population especially in Asia and Africa1. It is cultivated on 165.25 mh with an annual global production of around 787.29 mt 3. India has the largest land area under rice cultivation, but occupies second place in the tonnage globally after China. This is largely due to lower productivity rate of rice in India (4.21 tons/ha) over China (7.11 tons/ha) and the rest of the world 4.76 tons/ha2. In India, rice is grown in all states. West Bengal is the highest rice-producing state (16.72 mt), while Punjab has highest productivity rate (5.10 ton/ha). Uttar Pradesh is the second largest producer of rice, where around 5.60 mha of land is used to grow rice, with a total production of around 15.2 mt4.
Despite of being highest in rice acreage in the world, the annual gross productivity of rice in India is 40% less than China2. The significantly lower rice productivity rate in India is due to regular occurrence of biotic and abiotic stresses in rice cultivation5. Drought or lesser availability of water to paddy crop is a major and challenging abiotic stress to rice crop5. Rice plants require saturated or semi-saturated condition to maintain proper growth and development for better productivity. The crop yield is hampered severely when there is a limitation in the water availability5,6. The moderate drought can potentially reduce rice crop yields by up to 9–10% globally7,8,9.
Trichoderma spp. are a group of filamentous fungi that colonize the rhizosphere in all natural habitates, and may also form symbiotic relationships with plants7,11. They are renowned for their multifunctional capabilities, including suppression of plant pathogens2,12, promotion of plant growth, and modification of plant responses to abiotic stresses13. Several studies have demonstrated the positive impact of Trichoderma spp. in enhancing drought tolerance in rice14,15. Some species of Trichoderma, for example T. harzianum is an efficient mineral solubilizer and pathogen suppressor that can colonize the roots of crop plants and act as an efficient mycoparasite16,17,18. The biopriming with Trichoderma spp. can enhance nutrient absorption and improves water-holding capacity19, leading to gradual improvement in the plant growth and development under water stress conditions20,21,22. Besides the plant breeding for drought tolerance, no other reliable and effective technique has been developed to protect rice from water stress due to low rainfalls. In recent years potential of microorganisms in imparting tolerance against water stress has been evaluated23. Trichoderma spp. have demonstrated promising potential that can be exploited commercially to protect plants under moderate level of droughts24,25. Trichoderma spp. have been reported in reducing the impact of biotic and abiotic stress on plants26,27. The present study was carried out to examine and ascertain the role of Trichoderma spp. in protecting rice plants from the impact of water stress.
Results
Isolation and characterization of Trichoderma species
The DNA of Trichoderma species was sequenced by Macrogen® Incorporation, South Korea using universal primer (ex, 16S V3-V4, ITS, 18S) and Sanger sequencing method. The 16S and 18S rDNA gene sequences showed 99% similarity of the isolates with Trichoderma spp. (Fig. 1). The isolate showed 99 homologies with the ITS region of Trichoderma spp. Thus, the isolates were identified as Trichoderma harzianum, T. viride, T. virens, T. hamatum and T. koningii. The 16S and 18S rDNA gene sequences were deposited in NCBI gene bank under the accession number listed in Table 1. However, to avoid error in the grouping by ITS region, it is recommended to perform sequencing of the conserved region genes such as the TEF gene, Beta-tubulin gene, and Histone H3 gene for species-level identification in fungal isolates, especially, the TEF gene which shows a high level of sequence polymorphism among closely related species, even in comparison to the intron-rich portions of protein-coding genes such as the calmodulin gene, beta-tubulin gene, and histone H3 gene.
Plant growth
The rice cv. Swarna Sub-1 grown under water deficiency condition (75, 50 and 25% water availability of the recommended amount) showed the symptoms of water stress, exhibiting reduced plant growth, with shorter stems and smaller leaves in comparison to plants received the recommended level of irrigation. The symptoms varied with the level of water stress. In general, the rice cv. Swarna Sub-1 was found highly sensitive to drought (water stress). However, the soil application of T. harzianum AMUTHZ82 subsided the water stress symptoms and promoted shoot and root length by 19.7 and 16.8% with 75% watering, 17.4 and 14.1% with 50% watering and 12.3 and 11.2% with 25% watering over respective controls (Tables 2, 3). Next in effectiveness was T. viride AMUTVR73, other isolates did not produce the significant effect. The seed treatment also increased the length of shoot and root i.e., 16.1 and 13.4% (T. harzianum AMUTHZ82) and 10.6 and 8.2% (T. viride AMUTVR73) under 50% drought condition. Next in effectiveness were T. viride AMUTVR72, T. hamatum AMUTHAM_71, T. harzianum AMUTHZ81 and T. viride AMUTVR72 which significantly increased shoot and root length under drought conditions (P ≤ 0.05; Tables 2, 3). However, under normal irrigation (100% watering) highest promotion in the plant growth of rice was recorded, 21.3–23.6% (soil application) and 18–19.2% (seed treatment with T. harzianum AMUTHZ84. Next in effectiveness was T. viride AMUTVR73 (19.8 and 17.5%) over untreated control (Tables 2, 3).
Plant biomass production
The biomass production of rice plants grown under water stress conditions (75, 50 and 25% water availability of the recommended amount) was adversely affected. The fresh weight of shoot and root decreased by 21–26%, 36–45% and 52–62% at 75, 50, 25% watering, respectively over 100% watering (control). The soil application of T. harzianum AMUTHZ82 significantly promoted the plant biomass production under 75 and 50% water availability with a maximum increase 20.6 and 17.6% in shoot fresh weight and 18.5 and 15.3% in root fresh weight respectively, over control (P ≤ 0.05). Next in effectiveness was T. viride AMUTVR73, followed by T. harzianum AMUTHZ84 and T. viride AMUTVR71 which also significantly promoted the fresh weight of shoot and root (P ≤ 0.05; Tables 4, 5) over control. Under 25% water availability, T. harzianum AMUTHZ82 scored highest increase in fresh weight of shoot and root (13.8 and 11.3% respectively; Tables 4, 5), followed by T. viride AMUTVR73 (11.4 and 10.4%), and T. hamatum AMUTHAM_71 (8.2 and 8.1%) over control (P ≤ 0.05).
However, in 100% watered plants, maximum increase in biomass production was recorded with the soil application of T. harzianum AMUTHZ84 (22.6% and 21.4% respectively; Tables 4, 5) followed by seed treatment (22.1 and 20.7%). Next in effectiveness were T. viride AMUTVR73 (20.4 and 20.9%), T. harzianum AMUTHZ83 (19.3 and 19.3%) and T. viride AMUTVR71 (11.8 and 18.4%) over respective control. Other strains viz., T. koningii AMUTKNG_70 and T. virens AMUTVIRENS_71 did not significantly influence the biomass production of rice over respective control (P ≤ 0.05; Tables 4, 5). Overall, the soil application of Trichoderma AMUTHZ82 isolates induced 7–11% greater enhancement in the biomass production of rice in comparison to seed treatment.
Grain yield
The water stress caused significant decline (12–51%) in per plant grain yield production of rice cv. Swarna Sub-1 over normal irrigated control (Table 6). However, soil application of Trichoderma spp. improved the grain yield in both normal and water stress conditions. In normal irrigated condition, maximum increase in production of grain yield was recorded with the treatment of T. harzianum AMUTHZ84 (24.68%), followed by T. viride AMUTVR73 and T. harzianum AMUTHZ82 (22.08 and 20.13%, respectively; Table 6). Under 75 and 50% of water availability, T. harzianum AMUTHZ82 was found superior among all isolates in increasing grain yield by22.79 and 19.63%, respectively, over control (Table 6). Next in effectiveness were T. viride AMUTVR73, T. harzianum AMUTHZ84 and T. hamatum AMUTHM_71 which significantly increased grain yield of rice under drought conditions over control. Under 25% water availability of recommended amount, none of the isolates of Trichoderma were found effective in significantly enhancing the grain yield of rice plants over control. Overall, the soil application of Trichoderma AMUTHZ82 isolates induced 10–22% greater grain yield production of rice in comparison to seed treatment (Table 6).
Photosynthesis and transpiration rate
Rice plants grown under drought conditions experienced decline in the photosynthesis rate and transpiration rate (Figs. 2, 3). This resulted in reduced plant growth, and quality biomass production. The photosynthesis rate was found maximum in the rice plants grown under normal condition (100% water availability) with the soil application of T. harzianum AMUTHZ82 (7.12 µmol m−2 s−1), followed by T. viride AMUTVR72, T. harzianum AMUTHZ84, T. harzianum AMUTHZ83 and T. hamatum AMUTHAM_71. Lowest photosynthesis rate was recorded under 25% of water availability in comparison to 100% water availability. Soil application with T. harzianum AMUTHZ82 significantly enhanced the photosynthesis rate of rice plants growing under 75% (37% increase), 50% (19% increase) and 25% water availabilities (9% increase) over respective control. The seed treatment with Trichoderma spp. also minimized the negative effect of water stress on photosynthesis, but less than the soil application.
The transpiration rate was also influenced under low water availability with the treatment of Trichoderma spp. The plants treated with T. harzianum AMUTHZ82 showed minimum transpiration rate (2.36 µmol m−2 s−1), followed by T. viride AMUTVR72, T. hamatum AMUTHAM_71 and T. harzianum AMUTHZ84 under normal irrigated condition. The reduction in transpiration rate was observed lesser under 50% and 25% drought condition with the treatment of Trichoderma spp. over normal irrigated plants. Whereas, maximum transpiration rate was observed in untreated plant receiving recommended level of irrigation (Fig. 2, 3).
Leaf pigments
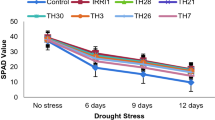
Total chlorophyll content (Chlorophyll A and B) in the leaves of rice plants under water stress conditions (75, 50 and 25% water availability of recommended amount) was recorded with a 20, 37 and 49% decline, respectively over control (Fig. 4). Under regular irrigation (100% watering), soil application of T. harzianum AMUTHZ84 resulted a maximum increase (21.3%) in chlorophyll content, followed by T. viride AMUTVR73 (17%) in the leaves of rice plants. Whereas, under 75 and 50% watering regime, T. harzianum AMUTHZ82 increased the total chlorophyll content (16.6 and 13.1%) over respective control (Figs. 4, 5). T. harzianum AMUTHZ83 and T. viride AMUTVR73 were found next in increasing chlorophyll content under 25% and 50% water stress conditions. The plants treated through seed treatment of Trichoderma isolates under water stress had significantly greater content of leaf pigment, but less than soil application. The plants receiving 25% water of the recommended amount recorded with 2–7% increase in total chlorophyll content with the soil application or seed treatment with Trichoderma isolates (Figs. 4, 5).
Discussion
The rice cv. Swarna Sub-1 was found sensitive to drought (water stress), and exhibited significant reduction in the plant growth, biomass production, leaf pigments, photosynthesis etc. grown under water stress conditions (75, 50 and 25% water of the recommended amount) in comparison to plants received normal irrigation (100% water availability of the recommended level). Adequate supply of water to a plant is essential for carrying out various physiological, biological and biochemical activities of the plant28,29. A decrease in the water supply influences the physiological activities especially the photosynthesis30,31, and subsequently the plant growth and development32, which directly affect the biomass production33,34 as observed in the present study.
The soil application of Trichoderma spp. especially T. harzianum AMUTHZ82 and T. viride AMUTVR73 subsided the negative effect of water stress on rice plants, and promoted the shoot and root growth, and biomass production of rice cv. Swarna Sub-1. Trichoderma spp. are universally occurring beneficial microbes in varied kind of soils13, and are generally known to suppress plant pathogenic fungi17,28, by parasitizing them or by producing metabolites, toxins, enzymes, etc. toxic to pathogens28,35. Trichoderma spp. also solubilize minerals present in soil, and produce hormones36,37, which may ultimately lead to plant growth promotion14,38,39. Some strains of Trichoderma spp. form endophytic relationship with roots, and develop symbiotic association with plants13,40. With such association, plants are able to efficiently use the water and nutrients in soil41, and are able to survive under various stress conditions such as nutrition deficiency, water stress, etc.42,43.
Khan et al.44 conducted a study in which the application of T. harzianum significantly enhanced plant height, root length, and shoot biomass of rice cv. Swarna Sub-1, under water-deficient conditions. Similarly, Haque et al.45 demonstrated that T. harzianum treated rice plants exhibited improved water-use efficiency, enhanced photosynthetic activity, and increased grain yield compared to non-treated plants during drought stress. Trichoderma spp. enhance plant health and stimulate the synthesis of plant growth-promoting substances such as auxins and cytokinins28, which play crucial roles in root development, nutrient uptake, and overall plant growth46,47. Additionally, T. harzianum triggers the expression of stress-responsive genes in rice, activating defence mechanisms and antioxidant systems that counteract the detrimental effects of water stress. The present study has demonstrated that soil application of T. harzianum AMUTHZ82 or T. viride AMUTVR73 may efficiently subside the impact of water stress at least up to 50% water availability levels and may enhance the plant growth and biomass production by 17–21%.
Conclusion
The rice cv. Swarna Sub-1 was found sensitive to drought (water stress), and exhibited significant reductions in the plant growth, biomass production, leaf pigments, photosynthesis etc. grown under water stress conditions (75, 50 and 25% water of the recommended amount) in comparison to plants received normal irrigation (100% water availability of the recommended level). The soil application of Trichoderma spp. especially T. harzianum AMUTHZ82 and T. viride AMUTVR73 subsided the negative effect of water stress on rice plants, and promoted the shoot and root growth and biomass production of rice. The present study has demonstrated that soil application of T. harzianum AMUTHZ82 or T. viride AMUTVR73 may efficiently reduce the impact of water stress at least up to 50% water availability levels and may enhance the plant growth and biomass production by 17–21%.
Materials and methods
Seeds of rice cv. Swarna Sub-1 were procured from Regional Centre, International Rice Research Institute, New Delhi, India.
Isolation and characterization of Trichoderma species
The local isolates of different Trichoderma spp. were isolated from rice fields using the serial dilution method on selective medium21. The corresponding GPS locations (latitude and longitude) of sampling sites were given in Table 7. The plates after inoculation were kept in an incubator at 25 ± 2 °C for 10 days. However, the fungus almost fully colonized the medium in 7 days. After incubation the Trichoderma colonies were pure cultured using the same culture medium and conditions. Thereafter, morphological characters of the Trichoderma isolates were studied to identify the species, followed by molecular characterization to confirm the identity48. The DNA of Trichoderma species was sequenced by Macrogen® Incorporation, South Korea, and the homology search was carried out using BLAST on the 18s rRNA-ITS region gene sequences to find and obtain the closest neighbour sequences from the NCBI database49. The MEGA-X software was used to access the Clustal-W alignment tool, and used to align all of the sequences. Simple comparison with the database of known species/isolates of Trichoderma harzianum, T. viride, T. virens, T. hamatum and T. konigii allowed the identification of the species. The UPGMA algorithm included in MEGA-X was used to create the phylogenetic tree (Fig. 1). The sequences have been deposited to the NCBI database and GenBank accession numbers listed in Table 1.
Mass multiplication of Trichoderma spp.
Eleven isolates of Trichoderma spp. as enlisted in Table 1 were mass cultured on sorghum grains in conical flasks. The seeds were soaked for 12 h. in distilled water containing sucrose (5%) and chloramphenicol (0.0003%). The seeds were kept in 500 ml capacity conical flasks and sterilized at 15 kg/cm2 steam pressure at 121 °C for 20 min. Next day the flasks were inoculated with the fungus, and incubated in an incubator for 10 days at 25 ± 2 °C. The sorghum seeds colonized by Trichoderma spp. were crushed with distilled water in an electrical grinder. The fungal suspension containing 10 g Trichoderma colonized seeds was applied to the soil (1 kg) in each pot a day before the planting of rice seedlings. For seed treatment, paste of sorghum seeds was applied to the rice seeds (@10 g/kg seeds), 3 h before their sowing in the nursery bed.
Treatments and plant cultures
The seedlings of rice cultivar Swarna Sub-1 were transplanted in the two sets of pots (1 seedling/pot) filled with sterilized soil and FYM (4:1). In one set of pots Trichoderma isolates were applied as soil application (@10 g/kg soil), whereas, in other set rice seedlings raised from Trichoderma treated seeds were planted. Four watering treatments viz., 100, 75, 50 and 25% of recommended irrigation were maintained for each isolate of Trichoderma spp. with five replicate/pots. Under 100% watering, the pots were watered regularly on alternate days. For 75% watering pots were watered every 4th day, and for 50% and 25% watering every 6th and 8th day, respectively. Similar treatments for drought have also been maintained in other researches50. Further, the soil moisture percentage is an important matrix for determining the drought level, but it is very difficult to use it as a treatment because of it changes with the progress of time. Since, there were 11 Trichoderma isolates, a total of 480 Pots were maintained, which were arranged in a complete randomized block design on the roof top receiving uniform sunlight. At maturity, 3 months after transplanting, the plants were harvested to measure plant length (shoot and root) and fresh weight per plant. The treatments for each Trichoderma isolate for soil application or seed treatment were as follows.
T1 = Plant + 100% watering (control)
T2 = Plant + 75% watering
T3 = Plant + 50% watering
T4 = Plant + 25% watering
T5 = Plant + 100% watering + Trichoderma isolate
T6 = Plant + 75% watering + Trichoderma isolate
T7 = Plant + 50% watering + Trichoderma isolate
T8 = Plant + 25% watering + Trichoderma isolate
Statistical analysis
The data obtained from the five replicates were used to calculate the means. However, replicate data were used to analyze least significant difference (LSD at P ≤ 0.05) with the help of Agricolae Package of R software51. Tukey HSD test was employed to mark significantly different treatments in the Tables. Two factor ANOVA was used to evaluate the data on plant growth variables.
Ethical statements
Both the authors have approved the content and authorship of the submitted manuscript, and all prevailing local, national, and international regulations and conventions, and scientific and ethical practices have been respected.
Plant compliance statement
The studies in this research article (plant field studies and experimental research, as well as the collection of plant material) were undertaken following laws and regulations pertinent to the institution.
Data availability
Both authors shall provide the datasets utilized and/or analyzed throughout the current work upon reasonable request.
References
Khan, M.R., Ahmad, I., Shah, M.H. and Ansari, M.S.A. Root-knot nematodes in cereal and pulse crops, and their management by novel biological and biotechnological approaches. In M. R. Khan (eds). Novel Biological and Biotechnological Applications in Plant Nematode Management, pp. 299–328 (Springer Nature, Singapore, 2023). ISBN: 978-981-99-2892-7.
Khan, M.R., Shah, M.H., & Ahamad, F. Relative effectiveness of eight Trichoderma species against white mould of pea caused by Sclerotinia sclerotiorum. Indian Phytopathol. 1–8 (2022).
FAOSTAT. https://www.fao.org/faostat/en/#compare (2022).
INDIASTAT. https://www.indiastat.com/table/agriculture/state-season-wise-area-production-productivity-ric/1440252 (2023).
Khan, M. R., Haque, Z. & Kausar, N. Management of the root-knot nematode Meloidogyne graminicola infesting rice in the nursery and crop field by integrating seed priming and soil application treatments of pesticides. Crop. Prot. 63, 15–25 (2014).
Al-Turki, A., Murali, M., Omar, A. F., Rehan, M. & Sayyed, R. Z. Exploring the recent advances in PGPR mediated resilience towards interactive effects of drought and salt stress in plants. Front. Microbiol. 14, 1214845. https://doi.org/10.3389/fmicb.2023.1214845 (2023).
Venuprasad, R., Lafitte, H. R. & Atlin, G. N. Response to direct selection for grain yield under drought stress in rice. Crop Sci. 47(1), 285–293 (2007).
Chepsergon, J., Mwamburi, L. & Kassim, M. K. Mechanism of drought tolerance in plants using Trichoderma spp. Int. J. Sci. Res. 3(11), 1592–1595 (2014).
Shah, S., Wang, D., Shah, F., Alharby, H., Bamagoos, A.A., Almjrashi, A., Alabdallah, A.M., Alzahrani, S.S., AbdElgawad, H., Adnan, M., Sayyed, R.Z., & Shah, H. Comprehensive impacts of climate change on rice production and adaptive strategies in China. Front Microbiol. 13. https://doi.org/10.3389/fmicb.2022.926059 (2022).
Lesk, C., Rowhani, P. & Ramankutty, N. Influence of extreme weather disasters on global crop production. Nature 529(7584), 84–87 (2016).
Khan, M. R., Majid, S., Mohidin, F. A. & Khan, N. A new bioprocess to produce low cost powder formulations of biocontrol bacteria and fungi to control fusarial wilt and root-knot nematode of pulses. Biol. Control 59(2), 130–140 (2011).
Mohiddin, F. A., Khan, M. R. & Khan, S. M. Why Trichoderma is considered super hero (super fungus) against the evil parasites?. Plant Pathol. J. 9(3), 92–102 (2010).
Shoresh, M. & Harman, G. E. The relationship between increased growth and resistance induced in plants by root colonizing microbes. Plant Signal Behav. 3(9), 737–739 (2008).
Harman, G. E., Howell, C. R., Viterbo, A., Chet, I. & Lorito, M. Trichoderma species—opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2(1), 43–56 (2004).
Brotman, Y. et al. Trichoderma-plant root colonization: Escaping early plant defense responses and activation of the antioxidant machinery for saline stress tolerance. PLOS Pathogens 9(4), e1003221. https://doi.org/10.1371/journal.ppat.1003221 (2013).
Schuster, A. & Schmoll, M. Biology and biotechnology of Trichoderma. Appl. Microbiol. Biotechnol. 87, 787–799 (2010).
Khan, M. R. Nematode biocontrol agents: Diversity and effectiveness against phytonematodes in sustainable crop protection. Indian Phytopathol. 69(4s), 453–463 (2016).
Afrouz, M. et al. Seed bio-priming with beneficial Trichoderma harzianum alleviates cold stress in maize. PeerJ 1, e15644. https://doi.org/10.7717/peerj.15644 (2023).
Doni, F., Isahak, A., Che Mohd Zain, C.R., Mohd Ariffin, S., Wan Mohamad, W.N.A., & Wan Yusoff, W.M. Formulation of Trichoderma sp. SL2 inoculants using different carriers for soil treatment in rice seedling growth. Springerplus 3, 1–5 (2014).
Bae, H. et al. The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J. Exp. Bot. 60(11), 3279–3295 (2009).
Mohammed, R. K. A. & Khan, M. R. Management of root-knot nematode in cucumber through seed treatment with multifarious beneficial microbes under protected cultivation. Indian Phytopathol. 74(4), 1035–1043 (2021).
Gowtham, H. G. et al. Insight into recent progress and perspectives in improvement of antioxidant machinery upon PGPR augmentation in plants under drought stress: A Review. Antioxidants 11(9), 1763. https://doi.org/10.3390/antiox11091763 (2022).
Marulanda, A., Barea, J. M. & Azcón, R. Stimulation of plant growth and drought tolerance by native microorganisms (AM fungi and bacteria) from dry environments: Mechanisms related to bacterial effectiveness. J. Plant Growth Regul. 28, 115–124 (2009).
Bernier, J., Atlin, G. N., Serraj, R., Kumar, A. & Spaner, D. Breeding upland rice for drought resistance. J. Sci. Food Agric. 88(6), 927–939 (2008).
Sudha, A. et al. Volatilome profiling and evaluation of plant growth stimulation and Anti-ergot activity of VOCs produced by Trichoderma asperelloides. Sydowia 76, 93–109. https://doi.org/10.12905/0380.sydowia76-2024-0093 (2023).
Contreras-Cornejo, H. A., Macías-Rodríguez, L., Beltrán-Peña, E., Herrera-Estrella, A. & López-Bucio, J. Trichoderma-induced plant immunity likely involves both hormonal-and camalexin-dependent mechanisms in Arabidopsis thaliana and confers resistance against necrotrophic fungi Botrytis cinerea. Plant Signal Behav. 6(10), 1554–1563 (2011).
Khan, M. R. & Mohiddin, F. A. Trichoderma: its multifarious utility in crop improvement. In New and Future Developments in Microbial Biotechnology and Bioengineering: Crop improvement through microbial biotechnology (eds Prasad, R. et al.) 263–291 (Elsevier publications, 2018).
Mir, M. I. et al. Multifarious indigenous diazotrophic rhizobacteria of rice (Oryza sativa L.) rhizosphere and their effect on plant growth promotion. Front. Nutri. 8, 781764. https://doi.org/10.3389/fnut.2021.781764 (2022).
Sagar, A., Sayyed, R.Z., Ramteke, P.W., Sharma, S., Marraiki, N., Elgorban, A.M., & Syed, A. ACC deaminase and antioxidant enzymes producing halophilic Enterobacter sp. PR14 promotes the growth of rice and millets under salinity stress. Physiol. Mol. Biol. Plants 26, 1847–54. https://doi.org/10.1007/s12298-020-00852-9 (2020).
Chaves, M. M., Flexas, J. & Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 103(4), 551–560 (2009).
Khan, M. R. & Rizvi, T. F. Effect of elevated levels of CO2 on powdery mildew development in five cucurbit species. Sci. Rep. 10, 4986 (2020).
Tardieu, F., & Simonneau, T. Variability among species of stomatal control under fluctuating soil water status and evaporative demand: Modelling isohydric and anisohydric behaviours. J. Exp. Bot. 419–432 (1998).
Farooq, M., Wahid, A., Kobayashi, N.S.M.A., Fujita, D.B.S.M.A., & Basra, S.M.A. Plant drought stress: effects, mechanisms and management. Sustain. Agric. 153–188. (2009).
Rizvi, T. F. & Khan, M. R. Biological and physiological responses of root-knot disease development on five cucurbits exposed to different concentrations of sulfur dioxide. Toxics 11, 334 (2023).
Rubio, M. B., Dominguez, S., Monte, E. & Hermosa, R. Comparative study of Trichoderma gene expression in interactions with tomato plants using high-density oligonucleotide microarrays. Microbiology 158(1), 119–128 (2012).
Suriani, N.L., Suprapta, D., Novizar, N., Parwanayoni, N., Darmadi, A., Dewi, D., Sudatri, N., Ahmad, F., Sayyed, R.Z., Syed, A., Elgorban, A.M., Bahkali, A.H., Enshasy, H.E., & Dalin, D.J. A mixture of piper leaves extracts and rhizobacteria for sustainable plant growth promotion and biocontrol of blast pathogen of organic bali rice. Sustainability. 12, 8490. https://www.mdpi.com/2071-1050/12/20/8490 (2020).
Khan, M.R., & Rizvi, T.F. Nanotechnology, a tool for reducing pesticide input in plant protection. In Bio-intensive approaches: application and effectiveness in plant diseases management, pp. 225–242 (Today and Tomorrow Publishes, New Delhi, 2018).
Basu, A., Prasad, P., Subha, N.D., Sadaf, K., Sayyed, R.Z., Reddy, M.S., & Hesham, E.E. Plant growth promoting rhizobacteria (PGPR) as green bioinoculants: recent developments, constraints, and prospects. Sustainability 13, 1140. https://www.mdpi.com/2071-1050/13/3/1140 (2021).
Khan, M.R., & Mohiddin, F.A. Biocontrol strategies for nematode management, an overview. In M. R. Khan (eds). Novel Biological and Biotechnological Applications in Plant Nematode Management, pp 116–141 (Springer Nature, Singapore, 2023). ISBN: 978-981-99-2892-7.
Enshasy, H.E.A., Ambehati, K.K. Ashraf, F., Ramchuran, S. Sayyed, R.Z. Amalin, D. Dailin, D.J., & Hanapi, S.Z. Trichoderma: Biocontrol agents for promoting plant growth and soil health. In Agriculturally important fungi for sustainable agriculture: Vol 2: Functional annotation for crop protection, pp. 239–259. (2020).
Mastouri, F., Björkman, T. & Harman, G. E. Seed treatment with Trichoderma harzianum alleviates biotic, abiotic, and physiological stresses in germinating seeds and seedlings. Phytopathol 100(11), 1213–1221 (2010).
Contreras-Cornejo, H. A., Macías-Rodríguez, L., Cortés-Penagos, C. & López-Bucio, J. Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol. 149(3), 1579–1592 (2009).
Contreras-Cornejo, H. A., Macías-Rodríguez, L. & Cortés-Penagos, C. Mechanism involved in Trichoderma harzianum-mediated maize drought tolerance. Physiol. Mol. Plant Pathol. 88, 41–53 (2014).
Khan, M. R. et al. Management of root-rot disease complex of mungbean caused by Macrophomina phaseolina and Rhizoctonia solani through soil application of Trichoderma spp. Crop. Prot. 119, 24–29 (2019).
Haque, M. Z., Anwar, M., Zaman, M. & Hasanuzzaman, M. Trichoderma harzianum enhances drought tolerance in rice by modulating physiological and biochemical attributes. J. Plant Growth Regul. 40, 764–778 (2021).
Vos, C. M., Yang, Y. & Harris, A. R. Exploring the impact of Trichoderma on nutrient acquisition by plants. Front. Microbiol. 12, 649403 (2021).
Khan, M.R., Ahamad, F., Rizvi, T. F., & Akram, M. Novel nano-materials and nano-formulations for nematode management. novel biological and biotechnological applications in plant nematode management. In M. R. Khan (eds). Springer Nature, Singapore. (2023). Pp. 256–279. ISBN: 978-981-99-2892-7
Vinale, F. et al. Trichoderma plant pathogen interactions. Soil Biol. Biochem. 40, 1–10 (2008).
El-Bondkly, A.M. Sequence analysis of industrially important genes from Trichoderma. In Biotechnology and biology of Trichoderma (pp. 377–392). Elsevier. (2014).
Yang, X., Wang, B., Chen, L., Li, P. & Cao, C. The different influences of drought stress at the flowering stage on rice physiological traits, grain yield, and quality. Sci. Rep. 9(1), 3742 (2019).
De Mendiburu, F., Simon, R. Agricolae-ten years of an open source statistical tool for experiments in breeding, agriculture and biology (No. e1748) Peer J. (2015).
Author information
Authors and Affiliations
Contributions
M.R.K. conceived the idea and critically checked the MS; M.H.S. executed the experiments, analyze the data and prepared the manuscript. Both authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Shah, M.H., Khan, M.R. Effectiveness of local isolates of Trichoderma spp. in imparting drought tolerance in rice, Oryza sativa. Sci Rep 14, 17672 (2024). https://doi.org/10.1038/s41598-024-67991-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-67991-x
- Springer Nature Limited