Abstract
Focal chondral defects of the knee are common and their management is challenging. This study investigated the efficacy and safety of Autologous Matrix-Induced Chondrogenesis (AMIC) for focal chondral defects of the knee. A systematic review and meta-analysis was conducted (according to the 2020 PRISMA statement) to investigate the efficacy of AMIC in improving symptoms and to compare AMIC versus microfracture (MFx). In January 2022, the following databases were accessed: Pubmed, Web of Science, Google Scholar, Embase. No time constrain was used for the search. All the clinical trials investigating AMIC and/or those comparing AMIC versus MFx for focal chondral defects of the knee were accessed. Only studies published in peer reviewed journals were considered. Studies which investigated other locations of the defects rather than knee were not eligible, nor those reporting data form mixed locations. Studies which reported data on revision settings, as well as those investigating efficacy on kissing lesions or multiple locations, were not suitable. The mean difference (MD) and odd ratio (OR) effect measure were used for continuous and binary data, respectively. Data from 18 studies (548 patients) were retrieved with a mean follow-up of 39.9 ± 26.5 months. The mean defect size was 3.2 ± 1.0 cm2. The visual analogue scale (VAS) decreased of − 3.9/10 (95% confidence interval (CI) − 4.0874 to -3.7126), the Tegner Activity Scale increased of + 0.8/10 (95% CI 0.6595 to 0.9405). The Lysholm Knee Scoring System increased of + 28.9/100 (95% CI 26.8716 to 29.1284), as did the International Knee Documentation Committee (IKDC) + 33.6/100 (95% CI 32.5800 to 34.6200). At last follow-up no patient showed signs of hypertrophy. 4.3% (9 of 210) of patients underwent revision procedures. The rate of failure was 3.8% (9 of 236). Compared to MFx, AMIC demonstrated lower VAS score (MD: − 1.01; 95% CI − 1.97 to 0.05), greater IKDC (MD: 11.80; 95% CI 6.65 to 16.94), and lower rate of revision (OR: 0.16; 95% CI 0.06 to 0.44). AMIC is effective for focal chondral defects of the knee. Furthermore, AMIC evidenced greater IKDC, along with a lower value of VAS and rate of revision compared to MFx.
Similar content being viewed by others
Introduction
Focal chondral defects of the knee are common1,2. Chondral defects impact negatively sport participation and the quality of life of affected patients3. If left untreated, chondral defects have limited chance to heal, and chronic pain may occur4,5,6. The management of chondral defects is challenging with unpredictable results7,8. For symptomatic defects smaller than 2 cm2, microfractures (MFx) have been proposed9,10,11,12. MFx is a bone marrow stimulating procedure of simple execution which can be conducted in a fully arthroscopic fashion13. During MFx, the cartilage is debrided to its viable border, and microfractures are performed to promote cell migration from the subchondral bone14,15. The bone marrow is the major hematopoietic and lymphoid organ, a niche to support self-renewal and differentiation of hematopoietic stem cells (HSC), multipotent progenitors (MPP), and lineage committed progenitors to produce blood cells16,17,18. Subchondral bone marrow cells are believed to enhance cartilage repair19,20,21. However, for bigger defects, the blood clot formed following MFx does not have enough mechanical resistance to remain in situ22. To overcome this limitation, in 2005, Behrens et al.23 firstly described an enhanced microfractures technique, which developed into Autologous Matrix-Induced Chondrogenesis (AMIC). In AMIC, a resorbable membrane is used to stabilize the clot and keep it stable in the joint cavity24,25. Different from other chondral procedures, AMIC does not necessitate to harvest any autologous tissue and is performed in a single session surgery26,27. These features make AMIC of special interest to both patients and surgeons28.
Several clinical studies evaluating the efficacy and safety of AMIC for focal chondral defects of the knee have been published24,25,29,30,31,32,33,34,35,36,37. However, in the past few years several studies have been published which have not yet been included in previous review22,27,38,39,40,41,42. Therefore, a systematic review and meta-analysis was conducted. The primary purpose of the present study was to investigate the efficacy and safety of AMIC for focal chondral defects of the knee. The secondary purpse was to investigate whether AMIC for focal chondral defects of the knee promotes a better outcome than MFx. We hypothesised that AMIC performed in the knee may be effective and safe to manage symptomatic chondral defects.
Methods
Eligibility criteria
All the clinical trials investigating AMIC and/or those comparing AMIC versus MFx for focal chondral defects of the knee were accessed. Only studies published in peer reviewed journals were considered. According to the author´ language capabilities, articles in English, German, Italian, French and Spanish were eligible. Only studies with level I to IV of evidence, according to Oxford Centre of Evidence-Based Medicine43, were considered. Reviews, opinions, letters, editorials were not considered. Studies which investigated other locations of the defects rather than knee were not eligible, nor were those reporting data from mixed locations. Studies which reported data on revision settings, and those investigating the efficacy of these techniques on kissing lesions or multiple locations, were not eligible. Animals, in vitro, biomechanics, computational, and cadaveric studies were not eligible. Missing quantitative data under the outcomes of interests warranted the exclusion of the study.
Search strategy
This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses: the 2020 PRISMA statement44. The PICOT algorithm was preliminary pointed out:
-
P (Problem): knee chondral defect;
-
I (Intervention): AMIC;
-
C (Comparison): MFx;
-
O (Outcomes): PROMs, rate of hypertrophy, failure, and revision surgery.
-
T (Timing): minimum 12 months follow-up.
In January 2022, the following databases were accessed: Pubmed, Web of Science, Google Scholar, Embase. No time constrain was set for the search. The following matrix of keywords were used in each database to accomplish the search: (knee) AND (chondral defects OR chondropathy OR cartilage defects) AND (Autologous Matrix-Induced Chondrogenesis OR AMIC OR surgery AND microfractures) AND (pain OR symptoms OR outcome AND patient reported outcome measures OR PROMs) OR (complications AND revision AND hypertrophy AND failure). No additional filters were used in the databases search.
Selection and data collection
Two authors (F. M. and H. S.) independently performed the database search. All the resulting titles were screened by hand and, if suitable, the abstract was accessed. The full-text of the abstracts which matched the topic were accessed. If the full-text was not accessible or available, the article was not considered for inclusion. A cross reference of the bibliography of the full-text articles was also performed for inclusion. Disagreements were debated and mutually solved by the authors. In case of further disagreements, a third senior author (N. M.) took the final decision.
Data items
Two authors (F. M. and H. S.) independently performed data extraction. The following data at baseline were extracted: author, year of publication and journal, length of the follow-up, number of patients with related mean age and BMI. Data concerning the following PROMs were collected at baseline and at last follow-up: Visual Analogue Scale (VAS), Tegner Activity Scale45, Lysholm Knee Scoring Scale46, and International Knee Documentation Committee (IKDC)47. The minimum clinically important difference (MCID) for the VAS was 2.7/10, 10/100 for the Lysholm score, 15/100 for the IKDC, 0.5/10 for the Tegner score48,49,50. Data from the following complications were also collected: hypertrophy, failures, and revision surgeries.
Assessment of the risk of bias and quality of the recommendations
The risk of bias were evaluated in accordance with the guidelines in the Cochrane Handbook for Systematic Reviews of Interventions51. Two reviewers (F. M. and H. S.) evaluated the risk of bias of the extracted studies independently. Disagreements were solved by a third senior author (N. M.). Randomised controlled trials (RCTs) were evaluated using the risk of bias of the software Review Manager 5.3 (The Nordic Cochrane Collaboration, Copenhagen). The following endpoints were evaluated: selection, detection, performance, attrition, reporting, and other bias. Non-RCTs were evaluated using the Risk of Bias in Nonrandomised Studies of Interventions (ROBINS-I) tool52. The quality of evidence of collective outcomes were evaluated using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) system was used53,54.
Synthesis methods
The statistical analyses were performed by the main author (F. M.) following the recommendations of the Cochrane Handbook for Systematic Reviews of Interventions55. For descriptive statistics, mean and standard deviation were used. To evaluate the improvement from baseline to last follow-up, the SPSS software was used. The mean difference (MD) was calculated, with 95% confidence interval (CI). The paired t-test was performed with values of P < 0.05 considered statistically significant. To compare AMIC versus MFx, a meta-analysis was conducted using the software Review Manager 5.3 (The Nordic Cochrane Collaboration, Copenhagen). For descriptive statistics, mean difference and standard deviation were used. The T-test was performed to assess baseline comparability, with values of P > 0.1 considered satisfactory. For continuous data, the inverse variance method with mean difference (MD) effect measure was used. For binary data, the Mantel–Haenszel method with odd ratio (OR) effect measure was used. The CI was set at 95% in all the comparison. Heterogeneity was assessed using \(\chi \) 2 and Higgins-I2 tests. If \(\chi \) 2 > 0.05, no statistically significant heterogeneity was found. A fixed model effect was used as default. If \(\chi \) 2 < 0.05 and Higgins-I2 > 60% high heterogeneity was found and a random model effect was used for analysis. Overall values of P < 0.05 were considered statistically significant.
Ethical approval
This study complies with ethical standards.
Registration and protocol
The present review was not registered.
Results
Study selection
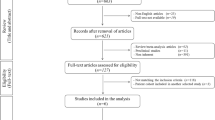
The literature search resulted in 1211 articles. Of them, 301 were excluded because of duplication. A further 890 studies were excluded as they did not match the eligibility criteria: not clinical studies (N = 177), language limitation (N = 5), not focusing on knee (N = 301), not focusing on AMIC (N = 407). Two studies were not included as they did not report quantitative data under the outcomes of interest. This left 18 studies for inclusion. The results literature search are shown in Fig. 1.
Risk of bias assessment
The Cochrane risk of bias tool was performed to investigate the risk of bias of RCTs. Given the number of retrospective studies included in the present investigation, the risk of selection bias was moderate. Few authors performed assessor blinding, leading to a moderate risk of detection bias. The risk of attrition and reporting biases was moderate, as was the risk of other bias. Concluding, the risk of bias graph evidenced a moderate quality of the methodological assessment of RCTs (Fig. 2).
Cochrane risk of bias tool. The risk of selection bias analysed the random sequence generation and the allocation concealment. The risk of detection bias in the blinding procedure during the outcome assessment were analysed. The risk of attrition bias refers to incomplete outcome data, such as missing outcome data from attrition during study enrollment or analysis. The risk of reporting bias refers to the selective publication of results based on their statistical or clinical relevance. If the authors identified additional risk of bias, these were considered as “other bias”. The risk of bias was evaluated in percentage as low, high, or unclear.
The ROBINS-I was applied to investigate the risk of bias of non-RCTs. No study evidenced critical risk of bias. Given the overall acceptable quality of the included studies, the overall risk of bias was moderate (Table 1).
Study characteristics and results of individual studies
Data from 548 patients were retrieved. 33% (180 of 548 patients) were female. The mean follow-up was 39.9 ± 26.5 months. The mean age was 27.0 ± 5.9 years and the mean BMI 27.1 ± 1.3 kg/m2. The mean defect size was 3.2 ± 1.0 cm2. The generalities and demographic of the included studies is shown in Table 2.
Efficacy of AMIC
The VAS decreased of – 3.9/10 (95% CI − 4.0874 to − 3.7126), the Tegner Activity Scale increased of + 0.8/10 (95% CI 0.6595 to 0.9405). The Lysholm Knee Scoring System increased of + 28.9/100 (95% CI 26.8716 to 29.1284), as did the IKDC + 33.6/100 (95% CI 32.5800 to 34.6200). These results are shown in greater detail in Table 3.
Complications
At last follow-up, no patient showed signs of hypertrophy. 4.3% (9 of 210) of patients underwent revision surgery. The rate of failure was 3.8% (9 of 236).
AMIC compared to MFx
Five studies were included in the meta-analyses29,31,37,39,40. At a mean follow-up of 40.3 months, the AMIC group demonstrated lower VAS score (MD: − 1.01; 95% CI − 1.97 to 0.05) and greater IKDC (MD: 11.80; 95% CI 6.65 to 16.94). At a mean follow-up of 43.6 months, the AMIC group demonstrated lower rate of revision (OR: 0.16; 95% CI 0.06 to 0.44). These results are shown in greater detail in Fig. 3.
Meta-analyses: forest plot of each comparison (IV inverse variance, OR odd ratio, MD mean difference, MH Mantel–Haenszel, CI confidence interval). The final effect and the relative confidence interval are represented respectively by the diamond and its lateral ends. The vertical line indicates the no effect threshold. The effect and the respective confidence interval of each study are represented by the square and the horizontal line, respectively.
Quality of the recommendations
The GRADE found limited effect in the estimated effect, and the true effect might be substantially different from the estimated effects. This relates to a low quality of the recommendations outcome rate of revision, and in IKDC and VAS scores (Fig. 4).
Discussion
General interpretation and clinical implication
The management of chondral defects of the knee is controversial, with unpredictable results. To date, no modality is considered definitive, and residual defect and symptoms recurrence is common. According to the main findings of the present study, AMIC seems to be effective to manage focal chondral defects of the knee. The increase in PROMs were greater than their MCID48,49,50. Furthermore, AMIC evidenced greater IKDC values, along with a lower value of VAS and lower rate of revision compared to MFx. Differently to other chondral procedures, AMIC does not necessitate to harvest or expand any autologous tissue, and is performed in a single session surgery. Therefore, AMIC should be considered in selected patients with symptomatic chondral defect of the knee.
Previous systematic reviews evaluated the efficacy of AMIC. Gao et al.56 evaluated the efficacy of AMIC in the knee including 12 studies. They found reduction in VAS and improvement of the Lysholm score within the first two years follow-up, but no improvement from two to five years follow-up56. Steinwachs et al.57 also performed a systematic review including 12 studies on AMIC. The Lysholm score, IKDC, and VAS were improved within the first two years follow-up alike, but they continued to improve after 3 years57. Previous systematic reviews also compared AMIC versus other common surgical strategies for chondral regeneration. Kim et al.58 compared AMIC (13 studies) versus MFx (18 studies). They evidenced greater values at IKDC evaluation compared to MFx, with no difference in Lysholm score, Tegner activity scale, and VAS for pain58. A recent systematic review of the same study group compared AMIC to matrix-induced autologous chondrocyte implantation (mACI) on the knee59. Although there were not statistical differences between the two interventions, given the single step procedure, avoidance of autologous cartilage harvest, and the need for chondrocyte expansion in a separate laboratory setting, AMIC may be preferable to mACI59. Another recent systematic review compared AMIC versus other chondral procedures including only RCTs60. Overall, AMIC demonstrated efficacy and safety in small- to medium-sized cartilage defects of the knee60.
Limitations of the included evidence
Between studies variability was evident. Most authors used a resorbable collagen I/III porcine derived membrane (Chondroguide®, Geistlich Biomaterials, Wolhusen, Switzerland)22,24,25,27,29,30,31,34,35,36,38,39,40. Enea et al.32 in 2013 published on the clinical application of AMIC using a polyglycolic acid and hyaluronic acid membrane enhanced with bone marrow concentrate. The same study group in 2015 published the results of AMIC using Biocollagen MeRG® collagen membrane (Bioteck, Vicenza, Italy) enhanced with bone marrow concentrate. Variability was also detected in the membrane fixation technique. Most authors fixed the membrane using fibrin glue22,24,25,27,31,32,33,34,35,36,38,39,40,41. In addition to its sealing, haemostatic, and adhesive proprieties, fibrin glue supports chondrocytes migration and proliferation61,62,63,64,65,66,67,68,69,70. Moreover, fibrin glue stimulates osteochondral scaffold fixation and cartilage regeneration71,72,73,74. Two authors compared AMIC fixed using fibrin glue versus suture29,37. Both authors reported better outcomes in the glued AMIC group. Membrane sutures produces fissures in the articular cartilage which may not heal, and may enlarge with time75,76. Suturing induces local cartilage impairment which may lead to pain, reduced healing, and premature osteoarthritis77. Most authors performed AMIC using a mini-arthrotomy or an arthrotomy22,27,29,31,34,35,36,37,38,39,40,41. Some authors24,25,32,33 used an arthroscopic technique to perform AMIC. These between studies variabilities may increase the risk of publication bias, and reduce the reliability of the present study.
Limitations of the review
The retrospective design of 55% (10 of 18) of the included studies represents another important limitation of the present investigation. Given the limited data available for inclusion, randomised and non randomised studies were not analysed separately. Most authors mixed patients who underwent chondral procedures on the femorotibial and patellofemoral joints, without reporting results separately. Moreover, most authors reported data from patients who underwent combined procedures. The description of the surgical approach, diagnosis, and rehabilitation protocols were often adequate, as were the criteria selection, outcome measures, and related timing of assessment. General health measures were seldom described, and the procedure to assess outcomes were often biased. To ensure the high quality of the included research and related validity of the findings, grey literature and not-peer reviewed articles were not considered. This may limit the number of investigations for inclusion and may limit the strength of the present study. The histopathology of the newly formed cartilage was not compared in the present meta-analysis. The characteristic of the new-formed cartilage at Magnetic Resonance Imaging (MRI) sequences were not investigated. Several studies analysed the magnetic resonance observation of cartilage repair tissue (MOCART) scoring system to evaluate the quality of the chondral regeneration. However, the MOCART score demonstrated no association with patient characteristics and with the surgical outcome in patients who underwent surgical management for knee and talus chondral defects78. The reviewers (F. M. and H. S.) who performed the literature search, data extraction, risk of bias assessment were the main authors of two of the included studies39,40. This may generate conflicts. Finally, a duplicate process in the literature search and data extraction was not conducted. These limitations impacted negatively on the reliability of the present study. Therefore, results from the present systematic review and meta-analysis should be considered carefully.
Conclusion
AMIC seems to be effective for the management of focal chondral defects of the knee. Furthermore, AMIC evidenced greater IKDC score, along with a lower value of VAS and rate of revision compared to MFx. The limited quantity and quality of the included studies limit the reliability of the present results and should be interpreted within the limitation of the present study.
Data availability
The datasets generated during and/or analysed during the current study are available throughout the manuscript.
References
Curl, W. W. et al. Cartilage injuries: A review of 31,516 knee arthroscopies. Arthroscopy 13(4), 456–460. https://doi.org/10.1016/s0749-8063(97)90124-9 (1997).
Figueroa, D. et al. Knee chondral lesions: Incidence and correlation between arthroscopic and magnetic resonance findings. Arthroscopy 23(3), 312–315. https://doi.org/10.1016/j.arthro.2006.11.015 (2007).
Heir, S. et al. Focal cartilage defects in the knee impair quality of life as much as severe osteoarthritis: A comparison of knee injury and osteoarthritis outcome score in 4 patient categories scheduled for knee surgery. Am. J. Sports Med. 38(2), 231–237. https://doi.org/10.1177/0363546509352157 (2010).
Kreuz, P. C. et al. Results after microfracture of full-thickness chondral defects in different compartments in the knee. Osteoarthr. Cartil. 14(11), 1119–1125. https://doi.org/10.1016/j.joca.2006.05.003 (2006).
Scillia, A. J. et al. Return to play after chondroplasty of the knee in National Football League athletes. Am. J. Sports Med. 43(3), 663–668. https://doi.org/10.1177/0363546514562752 (2015).
Davila Castrodad, I. M., Mease, S. J., Werheim, E., McInerney, V. K. & Scillia, A. J. Arthroscopic chondral defect repair with extracellular matrix scaffold and bone marrow aspirate concentrate. Arthrosc. Tech. 9(9), e1241–e1247. https://doi.org/10.1016/j.eats.2020.05.001 (2020).
Filardo, G. et al. Arthroscopic second generation autologous chondrocytes implantation associated with bone grafting for the treatment of knee osteochondritis dissecans: Results at 6 years. Knee 19(5), 658–663. https://doi.org/10.1016/j.knee.2011.08.007 (2012).
Bertho, P. et al. Treatment of large deep osteochondritis lesions of the knee by autologous matrix-induced chondrogenesis (AMIC): Preliminary results in 13 patients. Orthop. Traumatol. Surg. Res. 104(5), 695–700. https://doi.org/10.1016/j.otsr.2018.05.008 (2018).
Gudas, R. et al. A prospective randomized clinical study of mosaic osteochondral autologous transplantation versus microfracture for the treatment of osteochondral defects in the knee joint in young athletes. Arthroscopy 21(9), 1066–1075. https://doi.org/10.1016/j.arthro.2005.06.018 (2005).
Richter, D. L., Schenck, R. C. Jr., Wascher, D. C. & Treme, G. Knee articular cartilage repair and restoration techniques: A review of the literature. Sports Health 8(2), 153–160. https://doi.org/10.1177/1941738115611350 (2016).
Smith, G. D., Knutsen, G. & Richardson, J. B. A clinical review of cartilage repair techniques. J. Bone Jt. Surg. Br. 87(4), 445–449. https://doi.org/10.1302/0301-620X.87B4.15971 (2005).
Steinwachs, M. R., Guggi, T. & Kreuz, P. C. Marrow stimulation techniques. Injury 39(Suppl 1), S26-31. https://doi.org/10.1016/j.injury.2008.01.042 (2008).
Wen, H. J., Yuan, L. B., Tan, H. B. & Xu, Y. Q. Microfracture versus enhanced microfracture techniques in knee cartilage restoration: A systematic review and meta-analysis. J. Knee Surg. https://doi.org/10.1055/s-0040-1716552 (2020).
Orth, P., Gao, L. & Madry, H. Microfracture for cartilage repair in the knee: A systematic review of the contemporary literature. Knee Surg. Sports Traumatol. Arthrosc. 28(3), 670–706. https://doi.org/10.1007/s00167-019-05359-9 (2020).
Mithoefer, K., McAdams, T., Williams, R. J., Kreuz, P. C. & Mandelbaum, B. R. Clinical efficacy of the microfracture technique for articular cartilage repair in the knee: An evidence-based systematic analysis. Am. J. Sports Med. 37(10), 2053–2063. https://doi.org/10.1177/0363546508328414 (2009).
Travlos, G. S. Histopathology of bone marrow. Toxicol. Pathol. 34(5), 566–598. https://doi.org/10.1080/01926230600964706 (2006).
Travlos, G. S. Normal structure, function, and histology of the bone marrow. Toxicol. Pathol. 34(5), 548–565. https://doi.org/10.1080/01926230600939856 (2006).
Sebo, Z. L. et al. Bone marrow adiposity: Basic and clinical implications. Endocr. Rev. 40(5), 1187–1206. https://doi.org/10.1210/er.2018-00138 (2019).
Zhang, R., Ma, J., Han, J., Zhang, W. & Ma, J. Mesenchymal stem cell related therapies for cartilage lesions and osteoarthritis. Am. J. Transl. Res. 11(10), 6275–6289 (2019).
Berthelot, J. M., Le Goff, B. & Maugars, Y. Bone marrow mesenchymal stem cells in rheumatoid arthritis, spondyloarthritis, and ankylosing spondylitis: Problems rather than solutions?. Arthritis Res. Ther. 21(1), 239. https://doi.org/10.1186/s13075-019-2014-8 (2019).
McGonagle, D., Baboolal, T. G. & Jones, E. Native joint-resident mesenchymal stem cells for cartilage repair in osteoarthritis. Nat. Rev. Rheumatol. 13(12), 719–730. https://doi.org/10.1038/nrrheum.2017.182 (2017).
Miyahira, M. K. C. et al. Larger chondral lesions treated with collagen membrane—matrix-induced autologous chondrogenesis—show larger increase in clinical scores. Rev. Bras. Ortop. (Sao Paulo) 56(3), 333–339. https://doi.org/10.1055/s-0040-1712493 (2021).
Behrens, P. Matrixgekoppelte mikrofrakturierung. Arthroskopie 18, 193–197 (2005).
Schiavone Panni, A. et al. Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg. Sports Traumatol. Arthrosc. 26(4), 1130–1136. https://doi.org/10.1007/s00167-017-4503-0 (2018).
Schagemann, J. et al. Mid-term outcome of arthroscopic AMIC for the treatment of articular cartilage defects in the knee joint is equivalent to mini-open procedures. Arch. Orthop. Trauma Surg. 138(6), 819–825. https://doi.org/10.1007/s00402-018-2887-z (2018).
Gotze, C., Nieder, C., Felder, H. & Migliorini, F. AMIC for focal osteochondral defect of the talar shoulder. Life (Basel) 10, 12. https://doi.org/10.3390/life10120328 (2020).
de Girolamo, L. et al. Autologous matrix-induced chondrogenesis (AMIC) and AMIC Enhanced by autologous concentrated bone marrow aspirate (BMAC) allow for stable clinical and functional improvements at up to 9 years follow-up: Results from a randomized controlled study. J. Clin. Med. 8, 3. https://doi.org/10.3390/jcm8030392 (2019).
Bark, S. et al. Enhanced microfracture techniques in cartilage knee surgery: Fact or fiction?. World J. Orthop. 5(4), 444–449. https://doi.org/10.5312/wjo.v5.i4.444 (2014).
Anders, S., Volz, M., Frick, H. & Gellissen, J. A randomized, controlled trial comparing autologous matrix-induced chondrogenesis (AMIC(R)) to microfracture: Analysis of 1- and 2-year follow-up data of 2 centers. Open Orthop. J. 7, 133–143. https://doi.org/10.2174/1874325001307010133 (2013).
Astur, D. C. et al. Surgical treatment of chondral knee defects using a collagen membrane—autologus matrix-induced chondrogenesis. Rev. Bras. Ortop. 53(6), 733–739. https://doi.org/10.1016/j.rboe.2018.09.005 (2018).
Chung, J. Y. et al. Cartilage extra-cellular matrix biomembrane for the enhancement of microfractured defects. Knee Surg. Sports Traumatol. Arthrosc. 22(6), 1249–1259. https://doi.org/10.1007/s00167-013-2716-4 (2014).
Enea, D. et al. Single-stage cartilage repair in the knee with microfracture covered with a resorbable polymer-based matrix and autologous bone marrow concentrate. Knee 20(6), 562–569. https://doi.org/10.1016/j.knee.2013.04.003 (2013).
Enea, D. et al. One-step cartilage repair in the knee: Collagen-covered microfracture and autologous bone marrow concentrate. A pilot study. Knee 22(1), 30–35. https://doi.org/10.1016/j.knee.2014.10.003 (2015).
Gille, J. et al. Outcome of autologous matrix induced chondrogenesis (AMIC) in cartilage knee surgery: Data of the AMIC Registry. Arch. Orthop. Trauma Surg. 133(1), 87–93. https://doi.org/10.1007/s00402-012-1621-5 (2013).
Gudas, R., Maciulaitis, J., Staskunas, M. & Smailys, A. Clinical outcome after treatment of single and multiple cartilage defects by autologous matrix-induced chondrogenesis. J. Orthop. Surg. (Hong Kong) 27(2), 2309499019851011. https://doi.org/10.1177/2309499019851011 (2019).
Lahner, M. et al. Cartilage surgery in overweight patients: clinical and MRI results after the autologous matrix-induced chondrogenesis procedure. Biomed. Res. Int. 2018, 6363245. https://doi.org/10.1155/2018/6363245 (2018).
Volz, M., Schaumburger, J., Frick, H., Grifka, J. & Anders, S. A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years. Int. Orthop. 41(4), 797–804. https://doi.org/10.1007/s00264-016-3391-0 (2017).
Gille, J. et al. Autologous matrix-induced chondrogenesis for treatment of focal cartilage defects in the knee: A follow-up study. Orthop. J. Sports Med. 9(2), 2325967120981872. https://doi.org/10.1177/2325967120981872 (2021).
Migliorini, F. et al. Autologous matrix-induced chondrogenesis (AMIC) and microfractures for focal chondral defects of the knee: A medium-term comparative study. Life (Basel) 11, 3. https://doi.org/10.3390/life11030183 (2021).
Migliorini, F. et al. Management of patellar chondral defects with autologous matrix induced chondrogenesis (AMIC) compared to microfractures: A four years follow-up clinical trial. Life (Basel) 11, 2. https://doi.org/10.3390/life11020141 (2021).
Tradati, D. et al. AMIC-autologous matrix-induced chondrogenesis technique in patellar cartilage defects treatment: A retrospective study with a mid-term follow-up. J. Clin. Med. 9, 4. https://doi.org/10.3390/jcm9041184 (2020).
Waltenspul, M., Suter, C., Ackermann, J., Kuhne, N. & Fucentese, S. F. Autologous matrix-induced chondrogenesis (AMIC) for isolated retropatellar cartilage lesions: Outcome after a follow-up of minimum 2 years. Cartilage https://doi.org/10.1177/19476035211021908 (2021).
Howick J CI, Glasziou P, Greenhalgh T, Carl Heneghan, Liberati A, Moschetti I, Phillips B, Thornton H, Goddard O, Hodgkinson M (2011) The 2011 Oxford CEBM Levels of Evidence. Oxford Centre for Evidence-Based Medicine Available at https://www.cebm.net/indexaspx?o=5653
Page, M. J. et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 372, n71. https://doi.org/10.1136/bmj.n71 (2021).
Briggs, K. K. et al. The reliability, validity, and responsiveness of the Lysholm score and Tegner activity scale for anterior cruciate ligament injuries of the knee: 25 years later. Am. J. Sports Med. 37(5), 890–897. https://doi.org/10.1177/0363546508330143 (2009).
Lysholm, J. & Gillquist, J. Evaluation of knee ligament surgery results with special emphasis on use of a scoring scale. Am. J. Sports Med. 10(3), 150–154. https://doi.org/10.1177/036354658201000306 (1982).
Higgins LD, Taylor MK, Park D, Ghodadra N, Marchant M, Pietrobon R, Cook C, International Knee Documentation C. Reliability and validity of the International Knee Documentation Committee (IKDC) subjective knee form. Joint Bone Spine 74(6), 594–599. https://doi.org/10.1016/j.jbspin.2007.01.036 (2007).
Mostafaee, N. et al. Responsiveness of a Persian version of knee injury and osteoarthritis outcome score and tegner activity scale in athletes with anterior cruciate ligament reconstruction following physiotherapy treatment. Physiother. Theory Pract. 36(9), 1019–1026. https://doi.org/10.1080/09593985.2018.1548672 (2020).
Jones, K. J., Kelley, B. V., Arshi, A., McAllister, D. R. & Fabricant, P. D. Comparative effectiveness of cartilage repair with respect to the minimal clinically important difference. Am. J. Sports Med. 47(13), 3284–3293. https://doi.org/10.1177/0363546518824552 (2019).
Agarwalla, A. et al. Return to sport following isolated lateral opening wedge distal femoral osteotomy. Cartilage 13(1), 846S-852S. https://doi.org/10.1177/1947603520924775 (2021).
Cumpston, M. et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 10, 142. https://doi.org/10.1002/14651858.ED000142 (2019).
Sterne, J. A. et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355, i4919. https://doi.org/10.1136/bmj.i4919 (2016).
Brozek, J. L. et al. Grading quality of evidence and strength of recommendations in clinical practice guidelines. Part 1 of 3. An overview of the GRADE approach and grading quality of evidence about interventions. Allergy 64(5), 669–677. https://doi.org/10.1111/j.1398-9995.2009.01973.x (2009).
Atkins, D. et al. Grading quality of evidence and strength of recommendations. BMJ 328(7454), 1490. https://doi.org/10.1136/bmj.328.7454.1490 (2004).
Higgins JPT TJ, Chandler J, Cumpston M, Li T, Page MJ, Welch VA . Cochrane Handbook for Systematic Reviews of Interventions version 6.2. Cochrane 2021. Available from www.training.cochrane.org/handbook. Accessed on February 2022.
Gao, L., Orth, P., Cucchiarini, M. & Madry, H. Autologous matrix-induced chondrogenesis: a systematic review of the clinical evidence. Am. J. Sports Med. 47(1), 222–231. https://doi.org/10.1177/0363546517740575 (2019).
Steinwachs, M. R. et al. Systematic review and meta-analysis of the clinical evidence on the use of autologous matrix-induced chondrogenesis in the knee. Cartilage https://doi.org/10.1177/1947603519870846 (2019).
Kim, J. H., Heo, J. W. & Lee, D. H. Clinical and radiological outcomes after autologous matrix-induced chondrogenesis versus microfracture of the knee: A systematic review and meta-analysis with a minimum 2-year follow-up. Orthop. J. Sports Med. 8(11), 2325967120959280. https://doi.org/10.1177/2325967120959280 (2020).
Migliorini, F. et al. Matrix-induced autologous chondrocyte implantation versus autologous matrix-induced chondrogenesis for chondral defects of the talus: A systematic review. Br. Med. Bull. 138(1), 144–154. https://doi.org/10.1093/bmb/ldab008 (2021).
Karpinski, K., Haner, M., Bierke, S. & Petersen, W. Matrix-induced chondrogenesis is a valid and safe cartilage repair option for small- to medium-sized cartilage defects of the knee: A systematic review. Knee Surg. Sports Traumatol. Arthrosc. https://doi.org/10.1007/s00167-021-06513-y (2021).
Nair, M. A., Shaik, K. V., Kokkiligadda, A. & Gorrela, H. Tissue-engineered maxillofacial skeletal defect reconstruction by 3D printed beta-tricalcium phosphate scaffold tethered with growth factors and fibrin glue implanted autologous bone marrow-derived mesenchymal stem cells. J. Med. Life 13(3), 418–425. https://doi.org/10.25122/jml-2020-0044 (2020).
Sanghani-Kerai, A. et al. The development of a novel autologous blood glue aiming to improve osseointegration in the bone-implant interface. Bone Joint Res. 9(7), 402–411. https://doi.org/10.1302/2046-3758.97.BJR-2019-0073.R3 (2020).
Cassaro, C. V. et al. Fibrin biopolymer as scaffold candidate to treat bone defects in rats. J. Venom. Anim. Toxins Incl. Trop. Dis. 25, e20190027. https://doi.org/10.1590/1678-9199-JVATITD-2019-0027 (2019).
Morales-Conde, S., Balla, A., Alarcon, I. & Sanchez-Ramirez, M. Minimally invasive repair of ventral hernia with one third of tackers and fibrin glue: less pain and same recurrence rate. Minerva Chir. 75(5), 292–297. https://doi.org/10.23736/S0026-4733.20.08468-0 (2020).
Wong, A. I., McDonald, A., Jones, B. & Berkowitz, D. Patch-and-Glue: Novel technique in bronchoesophageal fistula repair and broncholith removal with stent and fibrin glue. J. Bronchol. Interv. Pulmonol. https://doi.org/10.1097/LBR.0000000000000732 (2020).
Karp, J. M. et al. Thrombin mediated migration of osteogenic cells. Bone 37(3), 337–348. https://doi.org/10.1016/j.bone.2005.04.022 (2005).
Brown, L. F. et al. Fibroblast migration in fibrin gel matrices. Am. J. Pathol. 142(1), 273–283 (1993).
Tani, K. et al. Thrombin enhances lung fibroblast proliferation in bleomycin-induced pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 5(1), 34–40. https://doi.org/10.1165/ajrcmb/5.1.34 (1991).
Chinni, C. et al. Thrombin, a survival factor for cultured myoblasts. J. Biol. Chem. 274(14), 9169–9174. https://doi.org/10.1074/jbc.274.14.9169 (1999).
Pagel, C. N. et al. Inhibition of osteoblast apoptosis by thrombin. Bone 33(4), 733–743. https://doi.org/10.1016/s8756-3282(03)00209-6 (2003).
Filardo, G. et al. Fibrin glue improves osteochondral scaffold fixation: Study on the human cadaveric knee exposed to continuous passive motion. Osteoarthr. Cartil. 22(4), 557–565 (2014).
Homminga, G. N., Buma, P., Koot, H. W., van der Kraan, P. M. & van den Berg, W. B. Chondrocyte behavior in fibrin glue in vitro. Acta Orthop. Scand. 64(4), 441–445. https://doi.org/10.3109/17453679308993663 (1993).
Singh, K., Moyer, H., Williams, J. K., Schwartz, Z. & Boyan, B. D. Fibrin glue: A scaffold for cellular-based therapy in a critical-sized defect. Ann. Plast. Surg. 66(3), 301–305. https://doi.org/10.1097/SAP.0b013e3181fc0507 (2011).
Zheng, M. H. et al. Matrix-induced autologous chondrocyte implantation (MACI): biological and histological assessment. Tissue Eng. 13(4), 737–746. https://doi.org/10.1089/ten.2006.0246 (2007).
Walker, E. A., Verner, A., Flannery, C. R. & Archer, C. W. Cellular responses of embryonic hyaline cartilage to experimental wounding in vitro. J. Orthop. Res. 18(1), 25–34. https://doi.org/10.1002/jor.1100180105 (2000).
Hunziker, E. B. & Quinn, T. M. Surgical removal of articular cartilage leads to loss of chondrocytes from cartilage bordering the wound edge. J. Bone Jt. Surg. Am. 85, 85–92. https://doi.org/10.2106/00004623-200300002-00011 (2003).
Hunziker, E. B. & Stahli, A. Surgical suturing of articular cartilage induces osteoarthritis-like changes. Osteoarthr. Cartil. 16(9), 1067–1073. https://doi.org/10.1016/j.joca.2008.01.009 (2008).
Migliorini, F. et al. Reliability of the MOCART score: A systematic review. J. Orthop. Traumatol. 22(1), 39. https://doi.org/10.1186/s10195-021-00603-w (2021).
Funding
Open Access funding enabled and organized by Projekt DEAL. The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
F.M.: literature search, data extraction, risk of bias assessment, conception and design, drafting, final approval; N.M.: supervision, revision, final approval; F.H.: writing, final approval; A.B.: literature search, data extraction, risk of bias assessment, final approval; A.B.: supervision, final approval; H.S.: drafting, final approval. All authors have agreed to the final version to be published and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Migliorini, F., Maffulli, N., Baroncini, A. et al. Autologous matrix-induced chondrogenesis is effective for focal chondral defects of the knee. Sci Rep 12, 9328 (2022). https://doi.org/10.1038/s41598-022-13591-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-13591-6
- Springer Nature Limited