Abstract
We examined whether 18F-fluorodeoxyglucose metabolism is associated with distant relapse-free survival (DRFS) and overall survival (OS) in women with estrogen receptor (ER)-positive, HER2-negative breast cancer. This was a cohort study examining the risk factors for survival that had occurred at the start of the study. A cohort from Asan Medical Center, Korea, recruited between November 2007 and December 2014, was included. Patients received anthracycline-based neoadjuvant chemotherapy. The maximum standardized uptake value (SUV) of 18F-fluorodeoxyglucose positron emission tomography/computed tomography (PET/CT) was measured. The analysis included 466 women. The median (interquartile range) follow-up period without distant metastasis or death was 6.2 (5.3–7.6) years. Multivariable analysis of hazard ratio (95% confidence interval [CI]) showed that the middle and high tertiles of SUV were prognostic for DRFS (2.93, 95% CI 1.62–5.30; P < 0.001) and OS (4.87, 95% CI 1.94–12.26; P < 0.001). The 8-year DRFS rates were 90.7% (95% CI 85.5–96.1%) for those in the low tertile of maximum SUV vs. 73.7% (95% CI 68.0–79.8%) for those in the middle and high tertiles of maximum SUV. 18F-fluorodeoxyglucose PET/CT may assess the risk of distant metastasis and death in ER-positive, HER2-negative patients.
Similar content being viewed by others
Introduction
Estrogen receptor (ER)-positive, HER2-negative breast cancer accounts for 60–70% of breast cancers1. Although patients are treated with curative intent, > 20% of patients experience relapse within 10–15 years and die from metastatic disease2. The recommended systemic treatment for patients with operable but high-risk tumors and those with locally advanced disease includes chemotherapy, followed by endocrine therapy. However, the diverse molecular characteristics of patients lead to diversity in the response to systemic therapies3,4. ER-positive, HER2-negative breast cancer does not benefit from systemic chemotherapy to the same extent as other subtypes5, and not all ER-positive breast cancers respond optimally to endocrine therapy. Identifying patients with newly diagnosed cancer who are at a high risk of relapse despite standard chemo- and endocrine therapy could help select patients who require more effective treatments or provide a basis for recommending participation in clinical trials6.
The diversity in transcriptional programs accounts for much of the biological heterogeneity of breast cancer7. The luminal epithelial-specific genes, including ER and proliferation genes, are the main gene clusters differentially expressed among intrinsic subtypes3. Likewise, multigene prognostic assays primarily rely on ER and proliferation-related gene expression8,9,10,11,12,13,14,15,16. A meta-analysis of gene expression profiles from large cohorts revealed that the capacity of prognostic signatures depends mainly on the detection of proliferation activity, and that ER expression status may contain only indirect information about prognosis16. The Warburg effect of aerobic glycolysis, a key metabolic hallmark of cancer, fuels cell growth and proliferation. Positron emission tomography/computed tomography (PET/CT) using 18F-fluorodeoxyglucose allows visualization of the increased glucose metabolism in malignant tumors. In ER-positive, HER2-negative breast cancers, the maximum standardized uptake value (SUV) of 18F-fluorodeoxyglucose is associated with poor prognostic factors, including progesterone receptor status17, histologic grade, Ki-67 expression18, and 21-gene recurrence score17. 18F-fluorodeoxyglucose PET/CT provides prognostic information in a non-invasive manner and with a low overall cost when previously performed for staging. However, no previous studies have determined the ability of 18F-fluorodeoxyglucose PET/CT to predict distant relapse-free survival (DRFS) and overall survival (OS)19,20,21.
The main objective of this study was to examine the association of tumor 18F-fluorodeoxyglucose metabolism with DRFS in patients with ER-positive, HER2-negative breast cancer treated with anthracycline-based neoadjuvant chemotherapy (NCT) followed by adjuvant endocrine therapy. Administration of chemotherapy before surgery has an advantage over adjuvant chemotherapy in that it allows the differentiation of predictive markers of the response to chemotherapy from prognostic factors. Therefore, we also examined whether the maximum SUV was associated with pathological complete response (pCR) to NCT.
Methods
Study design, setting, and patients
This retrospective cohort study analyzed women with breast cancer treated with NCT, followed by surgery and adjuvant endocrine therapy between November 2007 and December 2014 at Asan Medical Center (Asan cohort), located in Seoul, Republic of Korea. Follow-up ended on December 31, 2019. Risk factors were examined in relation to outcomes that had already occurred at the start of the study. The institutional review board of Asan Medical Center approved the study protocol and waived the requirement for informed consent (2019-1171). The study was performed following the Declaration of Helsinki and institutional guidelines.
The study analyzed a consecutive series of women with ER-positive, HER2-negative breast cancer who received anthracycline-based NCT. ER status was considered positive if Allred scores were 3 or higher. Immunostaining for HER2 was considered positive in the case of strong (3+) membranous staining in at least 10% of tumor cells. In situ hybridization methods determined HER2 positivity of tumors with 2+ immunostaining. Patients had American Joint Committee on Cancer clinical stage II or III tumors (tumor stage ≥ T2) by American Joint Committee on Cancer staging with a histological type of invasive ductal carcinoma. Patients underwent 18F-fluorodeoxyglucose PET/CT and received at least one cycle of NCT. Patients were excluded if they had a prior history of cancer or bilateral breast cancer. The number of patients enrolled during the study period determined the sample size of the Asan cohort.
18F-fluorodeoxyglucose PET/CT
18F-fluorodeoxyglucose PET/CT imaging was performed from the skull base to the upper thigh at 50–70 min after intravenous administration of 5.2–7.4 MBq/kg (0.14–0.2 mCi/kg) of 18F-fluorodeoxyglucose22. The median blood glucose level before 18F-fluorodeoxyglucose injection was 102 mg/dl (IQR 94–109). Two board-certified nuclear medicine physicians who were blinded to patient outcomes drew a volume of interest on the primary breast cancer or metastatic lymph nodes and assessed the maximum SUV of 18F-fluorodeoxyglucose uptake. The maximum standardized uptake values were harmonized across various PET/CT scanners (Biograph Sensation 16 and Biograph TruePoint 40, Siemens Healthineers, Knoxville, TN, USA; Discovery STE 8, Discovery PET/CT 690, and Discovery PET/CT 710, GE Healthcare, Milwaukee, WI, USA) without partial volume correction23. The recovery coefficient profiles of variable-sized hot cylinders of the American College of Radiology-approved PET phantoms (i.e., Esser phantom) were matched24,25. Annual 18F water cylinder phantom-based cross-calibration between PET and dose calibrator assured the uniform standardized uptake value of 1.0 between PET scanners25.
Variables
The primary outcome measure of this study was DRFS. The secondary outcome was OS. DRFS was defined as the interval from the date of NCT to the diagnosis of distant metastasis or death from breast cancer, non-breast cancer, or unknown causes26,27. OS was measured until the date of death from any cause. Pathologic complete response (pCR) was defined as the absence of residual invasive cancer on hematoxylin and eosin-stained samples of the complete resected breast specimen and all sampled regional lymph nodes26,28,29,30. Factors considered potential predictors of DRFS and OS that were prespecified in the study protocol were age, tumor stage, clinical lymph node stage, histologic grade, ER Allred score, progesterone receptor status, and Ki-67 expression31,32,33, as previously described26,28,29,34,35,36. All variables prior to NCT were retrieved from the electronic medical records of Asan Medical Center. Predictors that had continuous values or belonged to three or more categories were dichotomized based on commonly used cut-off values that are relevant for prognosis as follows: age (20–50 vs. > 50)31,32,33, tumor stage (T1–2 vs. T3–4)31,32,33, clinical N stage (N0 vs. N1–3)31,32,33, histologic grade (1 or 2 vs. 3)37,38, ER score (3–6 vs. 7–8)31,39, and Ki-67 expression (< 20% vs. ≥ 20%)31,40,41. The maximum SUV was analyzed as a continuous variable or grouped according to the median or tertile.
Statistical analysis
Data were expressed as the median and interquartile range (IQR) for continuous variables or numbers (%) for categorical variables. A two-sided P value of < 0.05 was considered significant. Categorical variables were compared using the χ2 test. For continuous variables, the Mann–Whitney U test was used. Associations were analyzed using linear regression analysis. Survival curves were estimated using the Kaplan–Meier method and compared with the log-rank test. Logistic regression analysis was used to determine the predictors of pCR, and Cox proportional hazard regression analysis was applied to identify independent predictors of DRFS and OS based on a predetermined statistical plan. Patients were censored to the date of the last disease assessment. For the multivariable analysis, a model was constructed including clinical and pathological predictors that were associated with the outcome at P ≤ 0.10. Post hoc analysis was performed to assess whether the overall association was consistent across all subgroups studied. For logistic and Cox regression, linearity, potential influential observations, and multicollinearity were assessed. The proportional hazard assumption was tested with Schoenfeld’s residual test and Schoenfeld’s partial residual plots. A restricted cubic spline plot was used for detecting nonlinearity and deviance residual for examining influential observations. Whether an overall association was consistent across all subgroups of patients categorized according to variables for survival analysis was examined. Estimates were presented with a 95% confidence interval (CI).
Missing clinical and pathological data were imputed using the multiple imputation method (m = 10). Ten imputed data sets were generated, and estimates from the multiple imputed data sets were combined according to Rubin’s rule42. The robustness of the results was demonstrated by performing a complete case analysis as a sensitivity analysis of the imputation model. Statistical tests were performed using R statistical software, version 3.6.1 (The R Foundation for Statistical Computing) and IBM SPSS Statistics for Windows, version 21 (IBM Corporation).
Results
Patient characteristics
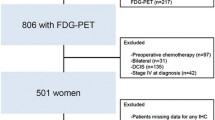
The study cohort comprised 466 women with breast cancer (Fig. 1). Patients’ demographics were typical of the neoadjuvant setting, including patients with regional lymph node disease or T3–4 tumors (Table 1). The median time between 18F-fluorodeoxyglucose PET/CT and the first date of NCT was 8 days (IQR 6–10). The median age of enrolled patients was 45 years (IQR 39–51). Patients received anthracycline (anthracycline and cyclophosphamide, n = 125, 26.8%) or taxane-based NCT (anthracycline, cyclophosphamide and taxane, n = 341, 73.2%) followed by breast-conserving surgery (n = 230, 49.4%) or mastectomy (n = 230, 49.4%). The clinical and pathological characteristics of enrolled patients are shown in Table 1. All patients except five who were lost to follow-up (n = 4) or had ER-negative cancer after surgery (n = 1) received adjuvant tamoxifen (n = 366, 78.5%) or aromatase inhibitor therapy (n = 95, 20.4%). The clinical and pathological characteristics of patients who did not undergo 18F-fluorodeoxyglucose PET/CT before NCT were not significantly different from those of patients who underwent 18F-fluorodeoxyglucose PET/CT (Supplementary Table S1). The maximum SUV ranged from 1.36 to 25.06, with a median value of 5.14 (Fig. 2), and was associated with clinical characteristics (Table 2).
Response to NCT
Of 460 patients who underwent surgery, 22 (4.8%) achieved a pCR. Univariable analysis showed that only progesterone receptor status (odds ratio 0.19; 95% CI 0.08–0.45, P < 0.001) was significantly associated with pCR (Supplementary Table S2). Multivariable analysis, including histologic grade and progesterone receptor, also showed that positivity for progesterone receptor was independently associated with pCR (odds ratio 0.21; 95% CI 0.08–0.51; P = 0.001).
Prognostic value
The median follow-up period for patients without distant metastasis or death was 6.2 (IQR 5.3–7.6) years. Distant metastasis or death did not occur in the 22 patients who achieved a pCR. Patients with pCR had significantly longer DRFS (P = 0.04, Supplementary Fig. S1) than those who did not achieve pCR, but there was no significant difference in OS (P = 0.15). Age, tumor stage, clinical N stage, Ki-67 expression, and maximum SUV (continuous and categorical) were associated with DRFS and OS (Table 3). Progesterone receptor status was also associated with OS. Multivariable analysis showed that maximum SUV was independently associated with DRFS and OS when analyzed as a categorical estimate by grouping the patients according to the median or tertile value (Table 4). The tumor stage and clinical N stage were also independent prognostic factors for DRFS and OS. Ki-67 expression was not independently associated with DRFS and OS regardless of whether we analyzed it as a continuous variable or several ordered categories. Kaplan–Meier estimates for patients who were free of distant metastasis or death were significantly different according to the maximum SUV (P < 0.001, Fig. 3). Post-hoc analysis of DRFS and OS showed that the overall association between maximum SUV and survival was consistent across most clinical and pathological subgroups large enough to provide sufficient outcome data (Supplementary Figs. S2, S3). When patients were stratified into subgroups according to independent prognostic factors, the increase in absolute risk of distant metastasis and death associated with high maximum SUV applied predominantly to patients with clinically node-positive disease (Supplementary Figs. S4, S5).
Kaplan–Meier curves of DRFS and OS by tertiles of the maximum standardized uptake value of 18F-fluorodeoxyglucose. The middle and high tertile categories were combined into one high-risk group because their outcome was significantly different from the low tertile. (a) The middle and high tertiles of SUV were prognostic for DRFS (hazard ratio 2.93, 95% confidence interval [CI] 1.62–5.30; P < 0.001). The 8-year DRFS rates were 90.7% (95% CI 85.5–96.1%) for those in the low tertile of maximum SUV vs. 73.7% (95% CI 68.0–79.8%) for those in the middle and high tertiles of maximum SUV. (b) The middle and high tertiles of SUV were prognostic for OS (hazard ratio 4.87, 95% CI 1.94–12.26; P < 0.001). The 8-year OS rates were 96.4% (95% CI 92.6–100%) for those in the low tertile of maximum SUV vs. 81.3% (95% CI 76.0–87.0%) for those in the middle and high tertiles of maximum SUV. DRFS distant relapse-free survival, OS overall survival, SUV standardized uptake value, Ter1 low tertile, Ter2–3 middle and high tertiles.
Discussion
The present study demonstrated that high tumor 18F-fluorodeoxyglucose metabolism was associated with reduced DRFS and OS after adjusting for standard prognostic factors in patients with ER-positive, HER2-negative breast cancer treated with anthracycline-based NCT, followed by adjuvant endocrine therapy. To the best of our knowledge, this study is the first to demonstrate the value of tumor 18F-fluorodeoxyglucose metabolism for the long-term prognosis of DRFS and OS in patients with ER-positive, HER2-negative breast cancer. 18F-fluorodeoxyglucose PET/CT may be helpful in selecting patients who need close monitoring or novel investigational treatment. In this study, maximum SUV was evaluated as a variable to assess tumor 18F-fluorodeoxyglucose metabolism because it is the most studied and most readily available routinely to assess tumor biology43. The harmonized SUV values from different PET scanners were obtained in patients with T2 or higher tumors. The strength of this study is that it studied a large and homogenous population with ER-positive, HER2-negative breast cancer patients.
Our results showed that negative progesterone receptor status was associated with a higher rate of pCR. However, the association with pCR appeared to be the opposite of its association with poor survival. Negative progesterone receptor status was associated with worse OS despite the higher chemotherapy sensitivity in these patients. The number of patients who achieved a pCR was small, and the rate of poor prognosis was mainly determined by those who did not achieve pCR. Previous studies investigating ER-positive breast cancer obtained the same results for multigene prognostic signatures26,44,45,46. This paradox is attributable to the relationship between the biological information provided by the predictor and the frequency and prognosis of false-positive predictions26. Less differentiated tumors are more likely to respond to chemotherapy and have a poor prognosis if they do not respond. These observations highlight that in ER-positive, HER2-negative breast cancer, survival is influenced not only by chemotherapy response but also by baseline biologic features and sensitivity to endocrine therapy45. Therefore, NCT that modestly increases the pCR rate is unlikely to improve prognosis47. In this study, the increased sensitivity to chemotherapy did not fully compensate for the poor baseline prognosis and low sensitivity to endocrine therapy. Meanwhile, 18F-fluorodeoxyglucose metabolism failed to show a significant association with pCR. Therefore, although 18F-fluorodeoxyglucose PET/CT was performed in the NCT setting, 18F-fluorodeoxyglucose metabolism in this study may reflect baseline prognostic features.
An important question is whether the results obtained in the neoadjuvant setting can be applied to the adjuvant setting. One limitation in this regard is that the prognostic information provided by 18F-fluorodeoxyglucose metabolism was obtained from a large number of patients with advanced clinical stages. Gene expression studies indicate that primary tumor samples and metastatic lymph node samples from the same individual are usually more similar to each other than to other samples, suggesting that the molecular program of primary breast cancer is retained in nodal metastases7. Furthermore, multigene assays have a prognostic value independent from the presence or absence of lymph node involvement48,49,50,51. These results suggest that 18F-fluorodeoxyglucose metabolism, a marker of cell growth and proliferation, can be used to identify a high-risk population in the adjuvant setting when prognostic gene expression data are not available.
The present study had several limitations. First, the outcome data collected was based on events that had already occurred at the start of the study. However, this study included all known predictors and potential confounders. The statistical methods were predetermined according to the primary objectives in the study protocol. Only a small number of patients did not undergo PET/CT, and there was no significant difference in characteristics between patients who did or did not undergo staging 18F-fluorodeoxyglucose PET/CT. Therefore, the possibility of information or selection bias was minimal. Second, we did not establish the cut-off value of the maximum SUV to define good and poor prognosis for patients. Prognostic characteristics of the cut-off value should be demonstrated on the independent data sets. The present results indicate that the harmonized maximum SUV of 4.1, which was the low tertile value, could be selected for a validation study based on the significance of the split in the survival curve.
In conclusion, high tumor 18F-fluorodeoxyglucose metabolism was associated with reduced DRFS and OS after adjusting for standard prognostic factors. 18F-fluorodeoxyglucose PET/CT may help classify ER-positive, HER2-negative patients into groups that would benefit from different therapeutic options.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Howlader, N. et al. US incidence of breast cancer subtypes defined by joint hormone receptor and HER2 status. J. Natl. Cancer Inst. 106, 055 (2014).
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: Patient-level meta-analysis of randomised trials. Lancet 378, 771–784 (2011).
Sørlie, T. et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl. Acad. Sci. U.S.A. 98, 10869–10874 (2001).
Goldhirsch, A. et al. Strategies for subtypes—Dealing with the diversity of breast cancer: Highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann. Oncol. 22, 1736–1747 (2011).
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomised trials. Lancet 365, 1687–1717 (2005).
Saura, C. et al. Neoadjuvant letrozole plus taselisib versus letrozole plus placebo in postmenopausal women with oestrogen receptor-positive, HER2-negative, early-stage breast cancer (LORELEI): A multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet Oncol. 20, 1226–1238 (2019).
Perou, C. M. et al. Molecular portraits of human breast tumours. Nature 406, 747–752 (2000).
Paik, S. et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N. Engl. J. Med. 351, 2817–2826 (2004).
van ‘t Veer, L. J. et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature 415, 530–536 (2002).
Wallden, B. et al. Development and verification of the PAM50-based Prosigna breast cancer gene signature assay. BMC Med. Genomics 8, 54 (2015).
Denkert, C. et al. Decentral gene expression analysis for ER+/Her2- breast cancer: Results of a proficiency testing program for the EndoPredict assay. Virchows Arch. 460, 251–259 (2012).
Sotiriou, C. et al. Gene expression profiling in breast cancer: Understanding the molecular basis of histologic grade to improve prognosis. J. Natl. Cancer Inst. 98, 262–272 (2006).
Carter, S. L., Eklund, A. C., Kohane, I. S., Harris, L. N. & Szallasi, Z. A signature of chromosomal instability inferred from gene expression profiles predicts clinical outcome in multiple human cancers. Nat. Genet. 38, 1043–1048 (2006).
Saal, L. H. et al. Poor prognosis in carcinoma is associated with a gene expression signature of aberrant PTEN tumor suppressor pathway activity. Proc. Natl. Acad. Sci. U.S.A. 104, 7564–7569 (2007).
Desmedt, C. et al. Biological processes associated with breast cancer clinical outcome depend on the molecular subtypes. Clin. Cancer Res. 14, 5158–5165 (2008).
Wirapati, P. et al. Meta-analysis of gene expression profiles in breast cancer: Toward a unified understanding of breast cancer subtyping and prognosis signatures. Breast Cancer Res. 10, R65 (2008).
Ahn, S. G. et al. Comparison of standardized uptake value of 18F-FDG-PET-CT with 21-gene recurrence score in estrogen receptor-positive, HER2-negative breast cancer. PLoS ONE 12, e0175048 (2017).
Higuchi, T. et al. Prognostic significance of preoperative 18F-FDG PET/CT for breast cancer subtypes. Breast 30, 5–12 (2016).
Groheux, D. et al. (18)FDG-PET/CT for predicting the outcome in ER+/HER2- breast cancer patients: Comparison of clinicopathological parameters and PET image-derived indices including tumor texture analysis. Breast Cancer Res. 19, 3 (2017).
Diao, W., Tian, F. & Jia, Z. The prognostic value of SUVmax measuring on primary lesion and ALN by (18)F-FDG PET or PET/CT in patients with breast cancer. Eur. J. Radiol. 105, 1–7 (2018).
Wen, W. et al. Prognostic value of maximum standard uptake value, metabolic tumor volume, and total lesion glycolysis of positron emission tomography/computed tomography in patients with breast cancer: A systematic review and meta-analysis. PLoS ONE 14, e0225959 (2019).
Chae, S. Y. et al. Comparison of diagnostic sensitivity of [(18)F]fluoroestradiol and [(18)F]fluorodeoxyglucose positron emission tomography/computed tomography for breast cancer recurrence in patients with a history of estrogen receptor-positive primary breast cancer. EJNMMI Res. 10, 54 (2020).
Ha, S. C. et al. Pretreatment tumor SUVmax predicts disease-specific and overall survival in patients with head and neck soft tissue sarcoma. Eur. J. Nucl. Med. Mol. Imaging 44, 33–40 (2017).
Lasnon, C. et al. Harmonizing SUVs in multicentre trials when using different generation PET systems: Prospective validation in non-small cell lung cancer patients. Eur. J. Nucl. Med. Mol. Imaging 40, 985–996 (2013).
Boellaard, R. et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: Version 2.0. Eur. J. Nucl. Med. Mol. Imaging 42, 328–354 (2015).
Hatzis, C. et al. A genomic predictor of response and survival following taxane-anthracycline chemotherapy for invasive breast cancer. JAMA 305, 1873–1881 (2011).
Hudis, C. A. et al. Proposal for standardized definitions for efficacy end points in adjuvant breast cancer trials: The STEEP system. J. Clin. Oncol. 25, 2127–2132 (2007).
Iwamoto, T. et al. Gene pathways associated with prognosis and chemotherapy sensitivity in molecular subtypes of breast cancer. J. Natl. Cancer Inst. 103, 264–272 (2011).
Miyake, T. et al. GSTP1 expression predicts poor pathological complete response to neoadjuvant chemotherapy in ER-negative breast cancer. Cancer Sci. 103, 913–920 (2012).
Jin, S. et al. 18F-fluorodeoxyglucose uptake predicts pathological complete response after neoadjuvant chemotherapy for breast cancer: A retrospective cohort study. J. Surg. Oncol. 107, 180–187 (2013).
Cardoso, F. et al. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 30, 1194–1220 (2019).
Andre, F. et al. Use of biomarkers to guide decisions on adjuvant systemic therapy for women with early-stage invasive breast cancer: ASCO clinical practice guideline update-integration of results from TAILORx. J. Clin. Oncol. 37, 1956–1964 (2019).
Henry, N. L. et al. Role of patient and disease factors in adjuvant systemic therapy decision making for early-stage, operable breast cancer: Update of the ASCO endorsement of the Cancer Care Ontario Guideline. J. Clin. Oncol. 37, 1965–1977 (2019).
Yoo, C. et al. Impact of immunohistochemistry-based molecular subtype on chemosensitivity and survival in patients with breast cancer following neoadjuvant chemotherapy. J. Breast Cancer 15, 203–210 (2012).
Jin, S. et al. 18 F-fluorodeoxyglucose uptake predicts pathological complete response after neoadjuvant chemotherapy for breast cancer: A retrospective cohort study. J. Surg. Oncol. 107, 180–187 (2013).
Kwon, Y. et al. Clinicopathological parameters and biological markers predicting non-sentinel node metastasis in sentinel node-positive breast cancer patients. Oncol. Rep. 25, 1063–1071 (2011).
Trudeau, M. E. et al. Prognostic factors affecting the natural history of node-negative breast cancer. Breast Cancer Res. Treat. 89, 35–45 (2005).
Rakha, E. A. et al. Prognostic significance of Nottingham histologic grade in invasive breast carcinoma. J. Clin. Oncol. 26, 3153–3158 (2008).
Dowsett, M. et al. Biomarker changes during neoadjuvant anastrozole, tamoxifen, or the combination: Influence of hormonal status and HER-2 in breast cancer—A study from the IMPACT trialists. J. Clin. Oncol. 23, 2477–2492 (2005).
Petrelli, F., Viale, G., Cabiddu, M. & Barni, S. Prognostic value of different cut-off levels of Ki-67 in breast cancer: A systematic review and meta-analysis of 64,196 patients. Breast Cancer Res. Treat. 153, 477–491 (2015).
Duffy, M. J. et al. Clinical use of biomarkers in breast cancer: Updated guidelines from the European Group on Tumor Markers (EGTM). Eur. J. Cancer 75, 284–298 (2017).
White, I. R., Royston, P. & Wood, A. M. Multiple imputation using chained equations: Issues and guidance for practice. Stat. Med. 30, 377–399 (2011).
Jadvar, H., Alavi, A. & Gambhir, S. S. 18F-FDG uptake in lung, breast, and colon cancers: Molecular biology correlates and disease characterization. J. Nucl. Med. 50, 1820–1827 (2009).
Parker, J. S. et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 27, 1160–1167 (2009).
Liedtke, C. et al. Genomic grade index is associated with response to chemotherapy in patients with breast cancer. J. Clin. Oncol. 27, 3185–3191 (2009).
Iwamoto, T. et al. First generation prognostic gene signatures for breast cancer predict both survival and chemotherapy sensitivity and identify overlapping patient populations. Breast Cancer Res. Treat. 130, 155–164 (2011).
Cortazar, P. et al. Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet 384, 164–172 (2014).
Albain, K. S. et al. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: A retrospective analysis of a randomised trial. Lancet Oncol. 11, 55–65 (2010).
Cardoso, F. et al. 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N. Engl. J. Med. 375, 717–729 (2016).
Lænkholm, A. V. et al. PAM50 risk of recurrence score predicts 10-year distant recurrence in a comprehensive Danish cohort of postmenopausal women allocated to 5 years of endocrine therapy for hormone receptor-positive early breast cancer. J. Clin. Oncol. 36, 735–740 (2018).
Sestak, I. et al. Comparison of the performance of 6 prognostic signatures for estrogen receptor-positive breast cancer: A secondary analysis of a randomized clinical trial. JAMA Oncol. 4, 545–553 (2018).
Acknowledgements
This work was supported by National Research Foundation Grants funded by the Korea Ministry of Science and ICT, Republic of Korea (Grant Numbers: NRF-2019R1A2C209022213), and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant Number HI18C2383).
Author information
Authors and Affiliations
Contributions
S.Y.C. and D.H.M. made a substantial contribution to the conception or design. S.Y.C., J.H.A., S.B.K., K.H.J., J.E.K., S.H.A., B.H.S., J.W.L., B.S.K., H.J.K., G.G., and J.O. made a substantial contribution to the acquisition of data. S.H.P. and S.Y.P. made a substantial contribution to the analysis of data. S.Y.C., S.H.P., H.S.L., J.H.A., S.Y.P., and D.H.M. made a substantial contribution to the interpretation of data. All authors drafted the manuscript or substantively revised it and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
Dr. SB Kim reports Grants from Novartis, Sanofi Genzyme, Kyowa Kirin Inc., Dongkook Pharma Co. Ltd, and Astra Zeneca, outside the submitted work. Dr. DH Moon reports Grants from the National Research Foundation of Korea, Life Molecular Imaging Gmbh (formerly Piramal Imaging GmbH), GE Healthcare, and Taiho Pharmaceutical outside the submitted work. The other authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chae, S.Y., Park, S.H., Lee, H.S. et al. Association between tumor 18F-fluorodeoxyglucose metabolism and survival in women with estrogen receptor-positive, HER2-negative breast cancer. Sci Rep 12, 7858 (2022). https://doi.org/10.1038/s41598-022-11603-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-11603-z
- Springer Nature Limited