Abstract
In Central Africa, the malaria vector Anopheles coluzzii is predominant in urban and coastal habitats. However, little is known about the environmental factors that may be involved in this process. Here, we performed an analysis of 28 physicochemical characteristics of 59 breeding sites across 5 urban and rural sites in coastal areas of Central Africa. We then modelled the relative frequency of An. coluzzii larvae to these physicochemical parameters in order to investigate environmental patterns. Then, we assessed the expression variation of 10 candidate genes in An. coluzzii, previously incriminated with insecticide resistance and osmoregulation in urban settings. Our results confirmed the ecological plasticity of An. coluzzii larvae to breed in a large range of aquatic conditions and its predominance in breeding sites rich in ions. Gene expression patterns were comparable between urban and rural habitats, suggesting a broad response to ions concentrations of whatever origin. Altogether, An. coluzzii exhibits a plastic response to occupy both coastal and urban habitats. This entails important consequences for malaria control in the context of the rapid urban expansion in Africa in the coming years.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
In the last century, urban development has transformed the landscape worldwide1,2. Animals and plants living in urban settings has been able to adapt to pollution, habitat fragmentation and/or strong alteration of their habitats3,4. For instance, urbanization had led to the reduction or extinction of different species such as moths, ivory-billet woodpecker or rodent’s populations5,6,7. Therefore, the ability to survive and/or to proliferate under urban settings entails ecological, physiological, behavioral, and/or genetic modifications that may influence biodiversity, conservation, and health policies8. Distribution of disease arthropods has been also affected by urban habitats. Mosquito vectors, such as Aedes aegypti, Ae. albopictus, Culex pipiens and Cx. quinquefasciatus, are examples of how mosquitoes closely associated to human can colonize and spread across cities9,10,11. For instance, the urban expansion of Aedes albopictus has been associated to chikungunya virus outbreaks across the world12,13.
In Africa, where the urbanization rate is among the fastest in the world14, malaria mosquitoes have been historically confined to rural areas because Anopheles larval development is strongly hindered in polluted water11,15. For this reason, previous entomological and epidemiological studies predicted that urban expansion in Africa would reduce malaria transmission11,15,16,17. Indeed, despite their favourable conditions in terms of blood meal opportunities and aquatic resources, African cities have been oases of reduced malaria transmission due to in part by the limited suitable vector breeding sites18,19. However, in the last decades, many works have recorded Anopheles presence in towns across the continent20,21,22,23,24,25,26,27. This caused alarm in public health community because Anopheles proliferation might lead to a risk of malaria transmission in a population with low or absent immunity to human malaria parasites18,28,29,30,31. The most abundant malaria mosquitoes in urban settings are members of the Anopheles gambiae complex32. Among them, Anopheles coluzzii is the predominant mosquito in cities of Central Africa20,22,33,34,35, whereas Anopheles arabiensis has been recorded in the cities of the West Africa36,37,38. Moreover, urban malaria expansion could also be promoted by the recent and troubling urban invasion by Anopheles stephensi in Sudan and Ethiopia39,40,41. In Central Africa, empirical studies revealed that An. coluzzii is more tolerant to ammonia concentrations than An. gambiae42,43. Ammonia is very abundant in urban settings, related to organic compounds 44,45. Therefore, the presence of this mosquito could be linked to its higher ability to breed in most polluted breeding sites of cities24,42,46,47,48,49.
Recently, Cassone et al., investigated the genetic basis of the presence of An. coluzzii in urban settings by transcriptomic analysis of larvae breeding in polluted sites of the city of Yaoundé (Cameroon)50. They found that genes related to oxidative stress and detoxification were upregulated in those larvae. Moreover, Antonio-Nkondjio and co-workers reported the association between An. coluzzii larvae presence in polluted sites and resistance to insecticides of adults in the cities of Yaoundé and Douala46, leading to the hypothesis that anthropogenic pressures have conditioned the evolution of this main malaria vector51. Interestingly, An. coluzzii is also more abundant than An. gambiae in coastal habitats due to its higher tolerance to salinity concentrations34,52. However, it is not known whether ammonia and salinity tolerance share a common physiological mechanism origin in this malaria mosquito. Although, the ability of this mosquito to colonize breeding sites with higher concentrations of salts and ammonia lead to hypothesize that similar ecological and genetic factors are incriminated.
To better understand the ecological and genetic drivers involved in the colonization of breeding sites by An. coluzzii, we performed a physicochemical analysis of the breeding across five coastal sites (rural and urban) in Cameroon and Gabon. Our study revealed the complexity and diversity of the breeding sites colonized by An. coluzzii, highlighting the role of some biotic and abiotic factors in the species distribution. Nonetheless, transcriptomic analysis of ten candidate’s genes involved in detoxification and osmoregulation revealed comparable levels across breeding sites analyzed, suggesting a common genetic response against different ions presence. These results show the ability of An. coluzzii to colonize a broad number of aquatic conditions, with direct consequences for malaria control in urban settings.
Results
Breeding site diversity
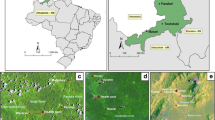
In this study, 59 breeding sites and 1351 larvae were analyzed at five localities in Cameroon and Gabon (Fig. 1, Tables 1, and S1) that display great diversity in terms of the values of physicochemical variables and types of settings (Table 2, Fig. S1, Table S1). The Principal Components Analysis (PCA) selected the first two principal components (PCs) using the broken stick model test 53 (Fig. 2A). These two PCs accounted for 47.17% of the total variance. PC1 (35.93%) can be interpret as a salinity gradient of breeding sites, characterized by sodium (Na+) and chlorine (Cl−) concentrations (Fig. 2B). PC2 (11.24%) was correlated with physicochemical variables associated to organic pollution, characterized by concentrations of ammonia (NH4+) and phosphates (PO4−) and pH (Fig. 2B). When the different breeding sites were plotted per locality, the PCA showed a partial overlapping, with Douala as the most differentiated locality (Fig. 2C). On the other hand, the PCA analysis allowed discriminating between urban and rural settings (Fig. 2D). The segregation between these settings was higher along PC2 (organic pollutant gradient) than PC1 (salinity gradient) (Fig. 2D). The analysis of the 22 parameters measured in this study revealed that the concentration of alkalinity, calcium (Ca2+), NH4+, carbonate (HCO3−), significantly varied between rural and urban habitats (Table 2). Analysis of these parameters among sites showed significant variations in concentrations, for instance, of F−, Ca2+, alkalinity, Cl- and HCO3− (Table 2). Other variables, such as NH4+ and nitrates (NO3−), were comparable among sites54,55,56. Finally, analysis of the differences among sites colonized by Culex species, a biomarker of organic pollution11,57,58,59, revealed that only the presence vegetation significantly varied between sites colonized or not by Culex species (Table 2).
Breeding sites characterization according to the physico-chemical variables. (A) Principal components selected with the Broken Stick model. (B) Canonical weight of each variable on the principal components; the arrow size indicates the importance of the parameter. (C) and (D) PCA diagrams showing the correlations between rural and urban breeding sites and physicochemical characteristics.
Spatial distribution and ecological modelling of An. coluzzii in coastal sites
Molecular species identification of 1351 Anopheles larvae from the 59 breeding sites (Table 1, Table S1) showed that An. coluzzii was the predominant species (n = 1181), compared with An. gambiae (n = 166) (Fig. 1). Only four hybrids were found and removed for further analysis (Table S1). Anopheles coluzzii frequency varied among localities, decreasing from urban (Libreville: 95.78%; Kribi: 95.33%; Douala: 92.13%) to rural areas (Cocobeach: 76.30%; Grand Batanga: 69.70%) (Table 1, Fig. 1). The differences in An. coluzzii frequency were not significant among locality (ANOVA, F = 1.179, P-value > 0.05), and among habitats (ANOVA, F = 0.045, P-value > 0.05). Our ecological modelling approach retained the first two PCs, habitat (urban or rural), altitude, and presence of Culex spp as significant predictors to explain the relative An. coluzzii frequency variation across localities and breeding sites (Table 3). Moreover, the best model (with ΔAIC = 0, Akaike Weight = 0.36) showed that the interactions between PC1/PC2 and habitat and Culex were significant (Table 3). Our results revealed that in urban settings, An. coluzzii frequency increased with high concentrations of mineral salts and organic matter, thus confirming its higher tolerance to salts and ammonia42,52. Moreover, altitude, which was correlated to distance to the sea level and salinity gradient (PC1) (r = 0.39, P-value < 0.05), greatly influenced An. coluzzii frequency compared with An. gambiae in rural habitats, but not in urban settings (Fig. 3).
Genetic response to habitats and environmental stressors
Finally, we assessed the expression of eight genes implicated in insecticide resistance (six genes of the CYP6 subfamily62,63, CYP4G1664,65, and acetylcholinesterase 1(Ace-1)62) and of two genes involved in osmoregulation (Na/K transporting ATPase and V-type H + -transporting ATPase subunit 166,67,68,69) (Table S2) in mosquitoes collected at 15 breeding sites across the urban (Douala; n = 3; Libreville n = 6) and rural settings (Grand Batanga; n = 2; Cocobeach n = 4). After the “Bonferroni” correction, the comparison of their expression profile did not show any significant difference between habitats (Wilcoxon test, P-value > 0.05) (Fig. 4A), despite the large gene expression variations across sites (Figs. S2, S3). Although, all genes under study tended to be strongly expressed, only CYP6Z1 and Ace1 showed significantly higher fold changes than the other genes (t test, P-value < 0.05, after Bonferroni correction) (Figs. S2, S3). As most of these genes, particularly those belonging to the CYP6 subfamily, have been repeatedly involved in insecticide resistance in An. gambiae in different parts of Africa62,63,70,71,72,73,74, and in other Anopheles species, such as Anopheles funestus75, Anopheles minimus76 and Anopheles sinensis76. The insecticide resistance status (i.e. presence of the kdr-West mutation) was investigated in a subsample of 80 mosquitoes from the five localities. Overall, 89% of these mosquitoes were homozygotes for the resistant alleles (Table S3). This indicates that the kdr mutation is presumably almost fixed across localities, thus possibly explaining the absence of expression differences between habitats (Fig. 4A), and among sites (Figs. S2 and S3)63. In order to investigate the environmental patterns of genes expression, we performed a coinertia analysis between the set of environmental predictors and the genes expressions changes of the studied genes. Our results identified co-relationships between most of the genes involved in insecticide resistance and the ions of anthropogenic origin (Fig. 4B). Only, the NaKATPase_β2 gene involved in osmoregulation processes, was highly correlated with the salinity (Fig. 4B). These results were confirmed by a correlation analysis among genes. All the expression patterns of gene involved in insecticide resistance were highly correlated (Fig. 4C), with the exception of the NaKATPase_β2 gene.
Correlation between gene expression profiles, habitat (rural and urban) and environmental predictors. (A) Association between habitat type and expression for each gene. (B) Coinertia diagram between gene expression on the left, and environmental predictors on the right. (C) Correlogram between genes (“number” = correlation coefficients; significance code 0’***’, 0.001'**', 0.01'*', 0.05'.' and 0.1' '; each blue cross (*) corresponds to a site).
Discussion
During the last decades, an increasing number of studies reported the presence of Anopheles in the African cities20,21,22,23,24,25,26,27. The present study investigated a large number of the ecological characteristics of the breeding sites exploited by An. coluzzii in rural and urban sites across coastal habitats of Central Africa, where this species is predominant compared with its sister species An. gambiae34,42,52. Three results emerge from our study. First, An. coluzzii shows an ability to colonize a large variety of water habitats, from natural ones to more anthropogenic areas. Second, An. coluzzii is more prone to breed in sites with higher concentrations of ions than An. gambiae, whatever the urbanization degree77,78. Third, gene expression patterns of the ten candidate’s genes in An. coluzzii are comparable between urban and rural habitats, suggesting that An. coluzzii exhibits a general response to the ions presents in each breeding site rather than specific responses to urban pollutants and to environmental stress. Altogether, our study suggests that An. coluzzii adaptation to urban settings is rooted in its ability to exploit more ions rich aquatic habitats.
Anopheles coluzzii ecological success is illustrated by its phenotypic plasticity34,77,78. At the larval stage, this mosquito exhibits an extraordinary adaptability to develop in a great variety of breeding sites22,79,80,81,82, with a preference for more permanent ones83. Our study confirmed the wide range of aquatic conditions where this species can lay and develop, in coastal and urban settings of Central Africa. Concerning the physicochemical characteristics of the breeding sites, our results were similar to those of previous studies in Cameroon84 and Kenya85, except for conductivity, because they concern inland sites (Table 2). Compared with published findings from other urban coastal sites in West Africa, we found lower concentrations of pollutants, such as NH4+ and NO3–86, revealing that An. coluzzii can still colonize breeding sites with a higher concentration of these ions (Table 2). We recorded the presence of Culex. The most abundant Culex species in the cities, Cx. quinquefasciatus, exhibits a high tolerance for organic matter but very low tolerance to salinity11,52,57,58,59, therefore, being a good biomarker of high ammonia and low salts concentrations. In this sense, Kudom et al., 2015 observed higher NH4+ concentration and lower conductivity at breeding sites colonized by Culex quinquefasciatus80. We did not observe any significant difference of NH4+ concentration between breeding sites with/without Culex. Moreover, we found a large variation of physicochemical parameters among and within sites. Cities are not uniform habitats because they combine multiple and heterogeneous ecological settings3,87. From their origins, African towns have displayed different degrees of urbanization88. This feature may help mosquitoes that are less tolerant to water pollutants to survive and breed within cities. For instance, we recorded the presence of An. gambiae in our three urban settings (Libreville, Douala and Kribi). Anopheles gambiae has already been recorded in other cities of Central and West Africa. Its relative frequency varies from absence in Yaoundé and Douala (Cameroon)22,84 to predominant in Bangui (Central Africa Republic)89, and intermediate in Ghana90. Therefore, it would be interesting to perform phenotypic experiments across cities, including larval transplantation, in order to investigate survival, physiological changes or behavioral response to predation, for instance, in both species91,92. Unfortunately, the temporal physico-chemical characteristics were not considered in the present study. Important variations could occur across the year, which may provide key information at a very fine scale to better understand frequency changes between An. gambiae and An. coluzzii and therefore, urban local adaptation drivers93.
We then analyzed the distribution of An. coluzzii relative to its sister species An. gambiae. We observed a positive and significant association of An. coluzzii relative frequency with ion concentrations, represented by PC1 and PC2 (Fig. 2B, Table 3). PC1 may be interpreted as a salinity gradient (i.e. conductivity), while PC2 can be interpreted as a human pollution gradient because correlation with concentrations of NH4+ and PO4−. Our results are in agreement with previous phenotypic experiments that revealed An. coluzzii higher tolerance to ammonia and salinity42,43,52, in congruence with, previous studies in Burkina Faso that showed higher An. coluzzii frequency in more anthropogenic habitats83. However, it is not clear if tolerance to different ions has the same physiological origin. In aquatic arthropods, including Anopheles species, Na+ and K+ ions are actively transported via the Na/K pump94. This physiologic mechanism could also be involved in NH4+ transport through interaction with the potassium binding site of the pump, as observed in other organisms95,96,97. For instance, future studies will study how the inhibition of the Na/K pump could affect the tolerance to salinity and ammonium in Anopheles. The habitat type (urban vs. rural) also had a significant effect on the relative frequency of An. coluzzii, showing that there is an ecological segregation between An. coluzzii and An. gambiae. In coastal and urban habitats of Central Africa, An. coluzzii remains the predominant species, while An. gambiae dominates the rural areas22,34. This distribution pattern has been also observed through the cities of West-Central Africa, such as Accra90, Cotonou98, Abidjan99 or Luanda100, while in inland cities, other species are more prone to proliferate, such as An. arabiensis in Bobo-Dioulasso101. It leads to think that distance to coast is associated to the predominance of Anopheles coluzzii, with the exception of Dakar, where An. arabiensis is the predominant species102. In addition, our modelling approach revealed the influence of lower altitude on An. coluzzii frequency in rural, but not in urban habitats. In coastal areas, altitude is related to the distance to the sea, and therefore it is negatively correlated with conductivity (r = − 0.16), Na+ (r = -0.15), and Cl− (r = − 0.15). The observation of this pattern in urban areas suggests that other factors, such as organic pollution, play a more important role. Finally, the model reveals the co-occurrence of Culex species and An. coluzzii (Table 3). The fact that An. coluzzii is significantly found in the same breeding sites as Culex, probably Cx. quinquefasciatus20,103,104,105 that is known to breed in larval habitats rich in decomposing organic matter22,106,107, suggests that this mosquito may be developing mechanisms to tolerate organic stress. However, the quantification of other parameters such as oxygen demand (COD) or biochemical oxygen demand (BOD) should be consider in order to improve the characterization of organic pollution in the breeding sites108,109,110. In addition, nitrites (or nitrates)46,48 and heavy metals that reveal pollution related to human activities such as agriculture, energy conversion and industry could potential provide clues of the urban pollution nature111.
To better understand the proliferation process of Anopheles in urban settings, we then compared the gene expression of ten candidate genes involved in detoxification and osmoregulation62,63,70,112 (Table S2) from specimens collected in urban and rural coastal sites. Our results did not find any significant difference between habitats, although all genes tended to be overexpressed with regard to the house-keeping gene used in this study. Associated with this result, the expression of each gene is highly correlated with that of the other nine genes. These results are consistent with a common response to high ion concentrations, of whatever origin. The function of these genes could be influenced by the physicochemical parameters of each breeding site. In the town of Accra (Ghana), King et al., studied the correlation of the expression of five genes with the different physicochemical variables of the breeding sites86, and found a significant positive correlation of all genes with Ca2+ levels. In the same sense, our co-inertia analysis showed a strong correlation between most of the genes involved in insecticide resistance and ions (Ca2+ and F−). In larvae of Bombyx mori (the silkworm), exposure to F- pollution is strongly correlated with overexpression of cytochrome oxidases belonging to the CYP6 (CYP6AB4, CYP6AE and CYP6B29) and CYP4 (CYP4M5, CYP4M9) subfamilies113. Thus, gene expression could be the result of a specific ionic signal or the interaction between several genes. Also, this observation confirms the fact that there are interactions between tolerance to environmental chemicals present in the breeding sites and insecticide resistance in Anopheles51,114,115. Indeed, several studies mention the involvement of the cytochrome oxidase P450 family in the detoxification of insecticide molecules and pollutants present in the environment63,86,116,117,118. Also, the NaKATPase_β2 gene, related to the Na/K pump, was found to be particularly strongly correlated with salinity. This pump has been already incriminated in the response to salinity in mosquitoes of the genus Aedes119 and the genus Anopheles, particularly An. merus112. Finally, the correlogram between the different genes showed a significant correlation between genes involved in insecticide resistance and those involved in osmoregulation, mainly VtHi2. This result may suggest a possible overlap or interaction between osmoregulation and detoxification in the immature forms of An. coluzzii. Overall, these results reveal a global pattern of enzyme activities in response to the ions studied at the breeding sites86, particularly of genes involved in detoxification, such as the P450 family.
Conclusion
During its history, An. coluzzii has strengthened its deadly relationship with humans by taking advantage of anthropogenic activities, such as the agriculture83,120, up to the colonization of urban environments22,48,121. In Central Africa, our study confirmed the ecological plasticity of this species that can colonize a large spectrum of ion concentrations. Compared with its sister species An. gambiae, An. coluzzii has become predominant in more ion rich habitats, such as coastal and urbans areas. These results raise important questions about the origin and evolution of its osmoregulatory capacities52. Africa is undergoing a large transformation through a fast urbanization14. The colonization of this habitat by Anopheles could have important health consequences. For this reason, it is important to determine the fitness cost of this adaptation, how the vector competence will evolve, and what genes are involved in the underlying ecological and behavioral mechanisms. Finally, our study highlighted a similar gene expression profile of specimens coming from rural and urban habitats, which are characterized by exposure to different ions. Future studies should focus on predicting the risk of malaria transmission according to the specific socio-economic and ecological characteristics of the urban settings. This is a priority to achieve the goal of malaria control in Africa in the coming decades.
Material and methods
Study sites and larval collections
Mosquito larvae were collected in September 2014 and January 2015 in Cameroon and Gabon, respectively. Coastal sites were selected in urban and rural habitats (Table 1). In Cameroon, sampling was carried in Douala, Kribi and Grand Batanga. Douala (urban), the economic capital of Cameroon, presents the highest urbanization level in the country. The locality has a flat topography, making difficult the discharge of wastewater. Kribi (urban) is a port town in the southern region of the country, with a rapid demographic and urban growth due to economic development. Grand Batanga (rural) is a coastal village, south of Kribi where the main activities are fishing and plantations. In Gabon, sampling was carried out in Libreville and Cocobeach. Libreville (urban) is located along the northwest coast of the country. It is characterized by rapid population growth, associated with haphazard urban development and poor management of waste produced by human activities, particularly at its periphery. Cocobeach (rural) is a locality with moderate population growth and low human activities (mainly fishing).
Potential breeding sites were systematically inspected to determine the presence of Anopheles larvae122. To standardize the genetic analyses, larvae were collected from 7.30 am to 11.30 am using the dipping technique50,122. At each breeding site, four pools of ten Anopheles larvae at stage L3-L4 were collected. The first pool was stored in 1 ml of 100% ethanol for molecular analysis to species identification. The other pools were stored in 2 ml of RNAlater (Qiagen), for subsequent expression analysis. All samples were stored at -20 °C.
Physicochemical characteristics of the breeding sites
The physical and chemical characteristics of the larval habitats were investigated using in situ and ex situ measurements. In situ measures characterized the parameters related to the physicochemical characteristics of the breeding sites, such as hydrogen potential (pH), conductivity (μS/cm), dissolved oxygen (%), temperature (°C) and salinity (mg/l), using a WTW 3110 instruments. Ordinal variables, such as turbidity (nothing, low, way and high), percentage of vegetation, mosquito larval density (low, way and high), and categorical variables, such as the presence/absence of mosquito larvae of the Culex genus, and breeding site physiognomy (puddle, drainage channel, canoe, etc.), were also investigated (Table S1 and Fig. S1). Ex situ measures were carried out at the Geochemical Water Analysis Laboratory (LAGE/CRH-IRGM) of Yaoundé, Cameroon. To this aim, 200 mL of water from each breeding site were filtered using 0.22 μm cellulose acetate filter (SIGMA-ALDRICH). Then, the alkalinity and the concentration of sodium (Na+), ammonia (NH4+), nitrates (NO3−), fluoride (F−), magnesium (Mg2+), chlorine (Cl−), potassium (K+), calcium (Ca2+), sulphates (SO42−), hydrogen carbonate (HCO3−), and phosphates (PO43−) were determined using a standardized ion chromatography spectrometry procedure.
Molecular Anopheles species identification, insecticide resistance status, and transcriptomic analysis
To determine the presence and proportion of An. coluzzii at each breeding site, total genomic DNA was extracted from the first pool of ten larvae stored in 100% ethanol using the DNeasy Extraction Kit (Qiagen, Manchester, UK), according to the manufacturer’s instruction. Species identification of the An. gambiae complex was determined based on a ribosomal DNA-based PCR–RFLP assay60 The presence of the target site L1014F (kdr-w) mutations was determined using the TaqMan assay, as described in Bass et al.123.
Breeding sites with higher An. coluzzii frequency were selected for gene expression analysis. After collection, the specimens were immediately stored in an RNAlater reagent to preserve RNA. Total RNA was individually isolated from the remaining three pools using the RNeasy Kit (Qiagen) following the manufacturer's instructions. To minimize the variable level of gene expression from one larva to another at the same site, three RNA pools were constructed using the Mini-Elute kit (Qiagen) following the manufacturer's instructions. After purification of the RNA from each pool with the Turbo DNA-free Kit (Qiagen), RNA quality and quantity were assessed using a Nanodrop spectrophotometer (Nanodrop Technologies UK) and Qubit 2.0 Fluorometer (Invitrogen), respectively. RNA was amplified and converted into double-stranded cDNA using the SuperScript III First-Strand Synthesis System (Life Technologies). The PCR protocol developed by Santolomazza124 was used to discriminate between An. coluzzii and An. gambiae, the only two members of the An. gambiae complex present at our breeding sites.
Previous studies identified genes with divergent expression profiles in larvae developing in urban breeding sites50,125. Ten candidate genes involved in insecticide resistance, detoxification mechanisms and osmoregulation62,73,119,125 were selected, and when needed, specific primers were designed using Primer v3 (bioinfo.ut.ee/primer3-0.4.0/) (Table S2). Expression levels were estimated following the formula: (Ct candidate gene – Ct housekeeping gene)/ Ct housekeeping gene, where Ct is the threshold cycle. Moreover, we normalized our analysis using the RPL19 housekeeping gene126. PCR reactions were carried out in 96-well plates (Applied Biosystems). Each well contained 10 µl of Master mix SYBR Green (10X) (Applied Biosystems), 0.2 µl of sense and antisense primers (10 µM), 7.6 µl of distilled H2O, and 2 µl of cDNA. Gene expression was quantified using the 7500 Fast Real Time PCR System (Applied Biosystems), and the following amplification conditions: 2 min at 50 °C, 10 min of pre-incubation at 95 °C followed by 40 cycles for 15 s at 95 °C and one minute at 60 °C.
Statistical analysis
First, a principal components analysis (PCA) was performed with the quantitative variables to ordinate the sites and the urban vs. rural habitats using the package ade4127. The PCA also allows integrating the significant PCs after selection with the Broken Stick Model as independent variables, thus reducing the risk of multicollinearity and the number of variables53,61.
Then, An. coluzzii frequency variation (in relation to An. gambiae) according to the different physicochemical parameters of the breeding sites and habitats was investigated by building a binomial generalized linear mixed model using the package lme4128. Response variable was the frequency of An. coluzzii (in relation to An. gambiae) in larval habitats and explanatory variables were the different physicochemical parameters of the breeding sites. It should be noted that in order to normalize the physicochemical variables, they first underwent a Boxcox transformation using the ‘’boxcoxfr’’ function of the AID package129. The significant interaction between variables was estimated with the Akaike Information Criterion (AIC) for model selection130. AIC changes were evaluated when model terms were added or removed using the dredge function in the MuMIn package34. The model with the lowest AIC was considered the best model. However, the models with a ΔAICC < 2131 were retained. Accordingly, multi-model parameter estimates were calculated with the MuMIn package. The ''Predict'' function of the lme4 package and the plot3D package were used to determine and describe how the relative An. coluzzii frequency (in relation to An. gambiae) would evolve according to the habitat type, altitude and PCs. The average expression levels of each gene were compared between rural and urban areas using the Wilcoxon test. In order to investigate the correlation between the general pattern of gene expression and water quality in larval habitats, a coinertia analysis was carried out using the "coinertia" function of the ade4 package. And the correlogram between genes was done using the "matrix Plot" function of the GGally package132. All statistical analyses were carried out using the R 3.5.2 software (https://cran.r-project.org).
References
Chin, A. Urban transformation of river landscapes in a global context. Geomorphology 79, 460–487 (2006).
Thomas, W. L. Man's role in changing the face of the earth. (The University of Chicago, 1956).
Johnson, M. T. & Munshi-South, J. Evolution of life in urban environments. Science 358, eaam8327 (2017).
Dubois, J. & Cheptou, P.-O. Effects of fragmentation on plant adaptation to urban environments. Philos. Trans. R. Soc. B Biol. Sci. 372, 20160038 (2017).
Cavia, R., Cueto, G. R. & Suárez, O. V. Changes in rodent communities according to the landscape structure in an urban ecosystem. Landsc. Urban Plan. 90, 11–19 (2009).
Jackson, J. A. Ivory-billed Woodpecker (Campephilus principalis): Hope, and the interfaces of science, conservation, and politics. Auk 123, 1–15 (2006).
McIntyre, N. E. Ecology of urban arthropods: a review and a call to action. Ann. Entomol. Soc. Am. 93, 825–835 (2000).
Crispo, E., Moore, J. S., Lee-Yaw, J. A., Gray, S. M. & Haller, B. C. Broken barriers: Human-induced changes to gene flow and introgression in animals: An examination of the ways in which humans increase genetic exchange among populations and species and the consequences for biodiversity. BioEssays 33, 508–518 (2011).
Triteeraprapab, S. et al. Transmission of the nocturnal periodic strain of Wuchereria bancrofti by Culex quinquefasciatus: establishing the potential for urban filariasis in Thailand. Epidemiol. Infect. 125, 207–212 (2000).
Carrieri, M., Bacchi, M., Bellini, R. & Maini, S. On the competition occurring between Aedes albopictus and Culex pipiens (Diptera: Culicidae) in Italy. Environ. Entomol. 32, 1313–1321 (2003).
Doby, J. & Mouchet, J. Écologie larvaire de quelques espèces de Culicidés dans la région de Yaoundé (Sud-Cameroun). Bulletin de la Société de Pathologie Exotique 50, 945–957 (1957).
Delatte, H. et al. Aedes albopictus, vecteur des virus du chikungunya et de la dengue à la Réunion: biologie et contrôle. Parasite 15, 3–13 (2008).
Vazeille, M., Moutailler, S., Pages, F., Jarjaval, F. & Failloux, A. B. Introduction of Aedes albopictus in Gabon: what consequences for dengue and chikungunya transmission?. Tropical Med. Int. Health 13, 1176–1179 (2008).
United Nations, Department of Economic and Social Affairs, Population Division. World Urbanization Prospects: The 2018 Revision (ST/ESA/SER.A/420). (New York, United Nations, 2019).
Trape, J. F. L'impact de l'urbanisation sur le paludisme en Afrique centrale. Doctoral dissertation, Université Paris 11 (1986).
Robert, V. et al. Malaria transmission in urban sub-Saharan Africa. Am. J. Trop. Med. Hyg. 68, 169–176 (2003).
Hay, S. I., Guerra, C. A., Tatem, A. J., Noor, A. M. & Snow, R. W. The global distribution and population at risk of malaria: past, present, and future. Lancet. Infect. Dis 4, 327–336 (2004).
Keiser, J. et al. Urbanization in sub-saharan Africa and implication for malaria control. Am. J. Trop. Med. Hyg. 71, 118–127 (2004).
Hay, S. I., Guerra, C. A., Tatem, A. J., Atkinson, P. M. & Snow, R. W. Urbanization, malaria transmission and disease burden in Africa. Nat. Rev. Microbiol. 3, 81 (2005).
Mourou, J.-R. et al. Malaria transmission and insecticide resistance of Anopheles gambiae in Libreville and Port-Gentil, Gabon. Malaria J. 9, 1475–2875 (2010).
Ndo, C., Menze-Djantio, B. & Antonio-Nkondjio, C. Awareness, attitudes and prevention of malaria in the cities of Douala and Yaoundé (Cameroon). Parasit. Vectors 4, 181 (2011).
Kamdem, C. et al. Anthropogenic habitat disturbance and ecological divergence between incipient species of the malaria mosquito Anopheles gambiae. PloS One 7, e39453 (2012).
Akogbeto, M., Chippaux, J.-P. & Coluzzi, M. . Le. paludisme urbain côtier à Cotonou (République du Bénin). Étude entomologique. Revue d’Epidémiologie Santé Publique 40, 233–239 (1992).
Awolola, T., Oduola, A., Obansa, J., Chukwurar, N. & Unyimadu, J. Anopheles gambiae ss breeding in polluted water bodies in urban Lagos, southwestern Nigeria. J. Vect. Borne Diseas. 44, 241 (2007).
Ravoahangimalala, R., Randrianambinintsoa, E., Tchuinkam, T. & Robert, V. Malaria in the urban highland area of Antananarivo, Madagascar: bioecology of Anopheles arabiensis. Bull. Soc. Pathol. Exot. 101, 348 (2008).
Labbo, R. et al. Ecology of urban malaria vectors in Niamey, Republic of Niger. Malaria J. 15, 314 (2016).
Klinkenberg, E. et al. Malaria and irrigated crops, Accra, Ghana. Emerg. Infect. Dis. 11, 1290 (2005).
Baudon, D. & Spiegel, A. Paludisme urbain, paludisme de demain pour l’Afrique sub-saharienne. Bull. Soc. Pathol. Exot. 96, 3–155 (2003).
Carme, B. Reducing the risk of malaria acquisition by urban dwellers of sub-Saharan Africa during travel in malaria-endemic areas. J. Infect. Dis. 170, 257–258 (1994).
Silva, P. M. & Marshall, J. M. Factors contributing to urban malaria transmission in sub-Saharan Africa: a systematic review. J. Trop. Med. https://doi.org/10.1155/2012/819563 (2012).
Bouyou-Akotet, M. K. et al. Falciparum malaria as an emerging cause of fever in adults living in Gabon, Central Africa. BioMed. Res. Internat. (2014).
Coetzee, M. et al. Anopheles coluzzii and Anopheles amharicus, new members of the Anopheles gambiae complex. Zootaxa 3619, 246–274 (2013).
Fontaine, M. C. et al. Extensive introgression in a malaria vector species complex revealed by phylogenomics. Science 347, 1258524 (2015).
Tene, F. B. et al. Habitat segregation and ecological character displacement in cryptic African malaria mosquitoes. Evolut. Appl. 7 (2015).
Mourou, J.-R. et al. Malaria transmission in Libreville: results of a one year survey. Malar. J. 11, 40 (2012).
Trape, J.-F. et al. Malaria morbidity among children exposed to low seasonal transmission in Dakar, Senegal and its implications for malaria control in tropical Africa. Am. J. Trop. Med. Hyg. 48, 748–756 (1993).
Dukeen, M. Y. & Omer, S. Ecology of the malaria vector Anopheles arabiensis Patton (Diptera: Culicidae) by the Nile in northern Sudan. Bull. Entomol. Res. 76, 451–467 (1986).
Robert, V., Awono-Ambene, H. & Thioulouse, J. Ecology of larval mosquitoes, with special reference to Anopheles arabiensis (Diptera: Culcidae) in market-garden wells in urban Dakar, Senegal. J. Med. Entomol. 35, 948–955 (1998).
Takken, W. & Lindsay, S. Increased threat of urban malaria from Anopheles stephensi mosquitoes, Africa. Emerg. Infect. Dis. 25, 1431 (2019).
Seyfarth, M., Khaireh, B. A., Abdi, A. A., Bouh, S. M. & Faulde, M. K. Five years following first detection of Anopheles stephensi (Diptera: Culicidae) in Djibouti, Horn of Africa: populations established—malaria emerging. Parasitol. Res. 118, 725–732 (2019).
Sinka, M. et al. A new malaria vector in Africa: predicting the expansion range of Anopheles stephensi and identifying the urban populations at risk. Proceedi. Nat. Acad. Sci. 117 (2020).
Tene, B. et al. Physiological correlates of ecological divergence along an urbanization gradient: differential tolerance to ammonia among molecular forms of the malaria mosquito Anopheles gambiae. BMC Ecol. 13, 1 (2013).
Akpodiete, N. O. & Tripet, F. Laboratory and microcosm experiments reveal contrasted adaptive responses to ammonia and water mineralisation in aquatic stages of the sibling species Anopheles gambiae (sensu stricto) and Anopheles coluzzii. Parasit. Vectors 14, 1–19 (2021).
Udo , W., In Su, C. & Eva-Maria, D. (ed Environmental Protection Agency) (2009).
Huff, L., Delos, C., Gallagher, K. & Beaman, J. Aquatic life ambient water quality criteria for ammonia-freshwater. Washington DC: US Environmental Protection Agency (2013).
Antonio-Nkondjio, C. et al. Anopheles gambiae distribution and insecticide resistance in the cities of Douala and Yaounde(Cameroon): influence of urban agriculture and pollution. Malar. J. 10, 154–154 (2011).
Djouaka, R. F. et al. Does the spillage of petroleum products in Anopheles breeding sites have an impact on the pyrethroid resistance?. Malar. J. 6, 159 (2007).
Tene Fossog, B. et al. Water quality and Anopheles gambiae larval tolerance to pyrethroids in the cities of Douala and Yaounde (Cameroon). J. Trop. Med. 2012 (2012).
Ossè, R., Bangana, S., Aïkpon, R., Kintonou, J. & Sagbohan, H. Adaptation of Anopheles coluzzii Larvae to polluted breeding Sites in Cotonou: a strengthening in Urban Malaria transmission in Benin. Vector Biology Journal 6, 2 (2019).
Cassone, B. J. et al. Gene expression divergence between malaria vector sibling species Anopheles gambiae and Anopheles coluzzii from rural and urban Yaounde Cameroon. Mol. Ecol. 23, 2242–2259 (2014).
Kamdem, C., Fouet, C., Gamez, S. & White, B. J. Pollutants and insecticides drive local adaptation in African malaria mosquitoes. Mol. Biol. Evol. 34, 1261–1275 (2017).
Kengne, P., Charmantier, G., Blondeau-Bidet, E., Costantini, C. & Ayala, D. Tolerance of disease-vector mosquitoes to brackish water and their osmoregulatory ability. Ecosphere 10, e02783. https://doi.org/10.1002/ecs2.2783 (2019).
Peres-Neto, P. R., Jackson, D. A. & Somers, K. M. How many principal components? Stopping rules for determining the number of non-trivial axes revisited. Comput. Stat. Data Anal. 49, 974–997 (2005).
Panagopoulos, G., Lambrakis, N., Tsolis-Katagas, P. & Papoulis, D. Cation exchange processes and human activities in unconfined aquifers. Environ. Geol. 46, 542–552 (2004).
Elhatip, H., Afşin, M., Dirik, K., Kurmaç, Y. & Kavurmacı, M. Influences of human activities and agriculture on groundwater quality of Kayseri-Incesu-Dokuzpınar springs, central Anatolian part of Turkey. Environ. Geol. 44, 490–494 (2003).
Azedine, H., Lynda, C. & Younes, S. Wastewater discharge impact on groundwater quality of Béchar city, southwestern Algeria: an anthropogenic activities mapping approach. Procedia Eng. 33, 242–247 (2012).
Hamon, J., Burnett, G., Adam, J.-P., Rickenbach, A. & Grjébine, A. Culex pipiens fatigans Wiedemann, Wuchereria bancrofti Cobbold, et le développement économique de l’Afrique tropicale. Bull. World Health Organ. 37, 217 (1967).
Subra, R. Biology and control of Culex pipiens quinquefasciatus* Say, 1823 (Diptera, Culicidae) with special reference to Africa. Int. J. Trop. Insect Sci. 1, 319–338 (1981).
Brengues, J. Culex pipiens fatigans Wiedemann, en Afrique tropicale: son importance et son contrôle. Med. Trop. 38, 691–694 (1978).
Fanello, C., Santolamazza, F. & Della, T. A. Simultaneous identification of species and molecular forms of the Anopheles gambiae complex by PCR-RFLP. Med. Vet. Entomol. 16, 461–464 (2002).
Cangelosi, R. & Goriely, A. Component retention in principal component analysis with application to cDNA microarray data. Biol. Direct 2, 2 (2007).
Edi, C. V. et al. CYP6 P450 enzymes and ACE-1 duplication produce extreme and multiple insecticide resistance in the malaria mosquito Anopheles gambiae. PloS Genet. 10, e1004236 (2014).
Djouaka, R. F. et al. Expression of the cytochrome P450s, CYP6P3 and CYP6M2 are significantly elevated in multiple pyrethroid resistant populations of Anopheles gambiae ss from Southern Benin and Nigeria. BMC Genom. 9, 538 (2008).
Balabanidou, V. et al. Cytochrome P450 associated with insecticide resistance catalyzes cuticular hydrocarbon production in Anopheles gambiae. Proc. Natl. Acad. Sci. 113, 9268–9273 (2016).
Mueller, P. et al. Pyrethroid tolerance is associated with elevated expression of antioxidants and agricultural practice in Anopheles arabiensis sampled from an area of cotton fields in Northern Cameroon. Mol. Ecol. 17, 1145–1155 (2008).
Larsen, E. H. et al. Osmoregulation and excretion. Compr. Physiol. 4, 405–573 (2011).
Lin, L.-Y., Horng, J.-L., Kunkel, J. G. & Hwang, P.-P. Proton pump-rich cell secretes acid in skin of zebrafish larvae. Am. J. Physiol. Cell Physiol. 290, C371–C378 (2006).
Mantel, L. H. & Farmer, L. L. Osmotic and ionic regulation. Internal Anatomy Physiol. Regul. 5, 53–161 (1983).
Furriel, R., McNamara, J. & Leone, F. Characterization of (Na+, K+)-ATPase in gill microsomes of the freshwater shrimp Macrobrachium olfersii. Comput. Biochem. Physiol. B: Biochem. Mol. Biol. 126, 303–315 (2000).
Chiu, T.-L., Wen, Z., Rupasinghe, S. G. & Schuler, M. A. Comparative molecular modeling of Anopheles gambiae CYP6Z1, a mosquito P450 capable of metabolizing DDT. Proc. Natl. Acad. Sci. 105, 8855–8860 (2008).
Antonio-Nkondjio, C. et al. Investigation of mechanisms of bendiocarb resistance in Anopheles gambiae populations from the city of Yaoundé, Cameroon. Malaria J. 15, 424 (2016).
David, J.-P., Ismail, H. M., Chandor-Proust, A. & Paine, M. J. I. Role of cytochrome P450s in insecticide resistance: impact on the control of mosquito-borne diseases and use of insecticides on Earth. Phil. Trans. R. Soc. B 368, 20120429 (2013).
Müller, P. et al. Field-caught permethrin-resistant Anopheles gambiae overexpress CYP6P3, a P450 that metabolises pyrethroids. PloS Genet. 4, e1000286 (2008).
Mitchell, S. N. et al. Identification and validation of a gene causing cross-resistance between insecticide classes in Anopheles gambiae from Ghana. Proc. Natl. Acad. Sci. 109, 6147–6152 (2012).
Irving, H., Riveron, J., Ibrahim, S. S., Lobo, N. & Wondji, C. Positional cloning of rp2 QTL associates the P450 genes CYP6Z1, CYP6Z3 and CYP6M7 with pyrethroid resistance in the malaria vector Anopheles funestus. Heredity 109, 383 (2012).
Rongnoparut, P., Boonsuepsakul, S., Chareonviriyaphap, T. & Thanomsing, N. Cloning of cytochrome P450, CYP6P5, and CYP6AA2 from Anopheles minimus resistant to deltamethrin. J. Vector Ecol. 28, 150–158 (2003).
Costantini, C. et al. Living at the edge: biogeographic patterns of habitat segregation conform to speciation by niche expansion in Anopheles gambiae. BMC Ecol. 9, 16 (2009).
Simard, F. et al. Ecological niche partitioning between Anopheles gambiae molecular forms in Cameroon: the ecological side of speciation. BMC Ecol. 9, 17 (2009).
Sattler, M. A. et al. Habitat characterization and spatial distribution of Anopheles sp. mosquito larvae in Dar es Salaam (Tanzania) during an extended dry period. Malaria J. 4, 4 (2005).
Kudom, A. A. Larval ecology of Anopheles coluzzii in Cape Coast, Ghana: water quality, nature of habitat and implication for larval control. Malar. J. 14, 447 (2015).
Etang, J. et al. Anopheles coluzzii larval habitat and insecticide resistance in the island area of Manoka, Cameroon. BMC Infect. Dis. 16, 217 (2016).
Mbida, A. M. et al. Nouvel aperçu sur l’écologie larvaire d’Anopheles coluzzii Coetzee et Wilkerson, 2013 dans l’estuaire du Wouri, Littoral-Cameroun. Bulletin de la Société de pathologie exotique 110, 92–101 (2017).
Gimonneau, G. et al. Larval habitat segregation between the molecular forms of the mosquito Anopheles gambiae in a rice field area of Burkina Faso, West Africa. Med. Vet. Entomol. 26, 9–17 (2012).
Djamouko-Djonkam, L. et al. Spatial distribution of Anopheles gambiae sensu lato larvae in the urban environment of Yaoundé, Cameroon. Infect. Dis. Poverty 8, 84 (2019).
Dida, G. O. et al. Spatial distribution and habitat characterization of mosquito species during the dry season along the Mara River and its tributaries, in Kenya and Tanzania. Infect. Dis. Poverty 7, 2 (2018).
King, S. A. et al. The Role of Detoxification Enzymes in the Adaptation of the Major Malaria Vector Anopheles gambiae (Giles; Diptera: Culicidae) to Polluted Water. J. Med. Entomol. 54, 1674–1683 (2017).
Donihue, C. M. & Lambert, M. R. Adaptive evolution in urban ecosystems. Ambio 44, 194–203 (2015).
Coret, C., Zaugg, R. & Chouin, G. Les villes en Afrique avant 1900. Bilan historiographique et perspectives de recherche. Afriques. Débats, méthodes et terrains d’histoire (2020).
Ndiath, M. O. et al. Composition and genetics of malaria vector populations in the Central African Republic. Malar. J. 15, 1–10 (2016).
Mattah, P. A. D. et al. Diversity in breeding sites and distribution of Anopheles mosquitoes in selected urban areas of southern Ghana. Parasit. Vectors 10, 1–15 (2017).
Diabaté, A. et al. Larval development of the molecular forms of Anopheles gambiae (Diptera: Culicidae) in different habitats: a transplantation experiment. J. Med. Entomol. 42, 548–553 (2005).
Briones, M. J. I., Ineson, P. & Piearce, T. G. Effects of climate change on soil fauna; responses of enchytraeids, Diptera larvae and tardigrades in a transplant experiment. Appl. Soil. Ecol. 6, 117–134 (1997).
Szulkin, M., Munshi-South, J. & Charmantier, A. Urban evolutionary biology (Oxford University Press, 2020).
Bradley, T. Physiology of osmoregulation in mosquitoes. Annu. Rev. Entomol. 32, 439–462 (1987).
Raabe, W. & Lin, S. Pathophysiology of ammonia intoxication. Exp. Neurol. 87, 519–532 (1985).
Ip, Y., Chew, S. & Randall, D. Ammonia toxicity, tolerance, and excretion. Fish Physiol. 20, 109–148 (2001).
Randall, D. J. & Tsui, T. Ammonia toxicity in fish. Mar. Pollut. Bull. 45, 17–23 (2002).
Yadouléton, A. et al. The impact of the expansion of urban vegetable farming on malaria transmission in major cities of Benin. Parasit. Vectors 3, 118 (2010).
Girod, R., Orlandi-Pradines, E., Rogier, C. & Pages, F. Malaria transmission and insecticide resistance of Anopheles gambiae (Diptera: Culicidae) in the French military camp of Port-Bouet, Abidjan (Cote d’Ivoire): implications for vector control. J. Med. Entomol. 43, 1082–1087 (2006).
Cuamba, N., Choi, K. S. & Townson, H. Malaria vectors in Angola: distribution of species and molecular forms of the Anopheles gambiae complex, their pyrethroid insecticide knockdown resistance (kdr) status and Plasmodium falciparum sporozoite rates. Malar. J. 5, 2 (2006).
Jones, C. M. et al. Insecticide resistance in Culex quinquefasciatus from Zanzibar: implications for vector control programmes. Parasit. Vectors 5, 1–9 (2012).
Pagès, F. et al. Malaria transmission in Dakar: a two-year survey. Malar. J. 7, 178 (2008).
Corbel, V. et al. Multiple insecticide resistance mechanisms in Anopheles gambiae and Culex quinquefasciatus from Benin, West Africa. Acta Tropica 101, 207–216 (2007).
Nchoutpouen, E. et al. Culex species diversity, susceptibility to insecticides and role as potential vector of lymphatic filariasis in the city of Yaoundé, Cameroon. PLoS Negl. Trop. Dis. 13, e0007229 (2019).
Antonio-Nkondjio, C. et al. High mosquito burden and malaria transmission in a district of the city of Douala, Cameroon. BMC Infect. Dis. 12, 275 (2012).
Calhoun, L. M. et al. Combined sewage overflows (CSO) are major urban breeding sites for Culex quinquefasciatus in Atlanta, Georgia. Am. J. Trop. Med. Hyg. 77, 478–484 (2007).
Lines, J., Harpham, T., Leake, C. & Schofield, C. Trends, priorities and policy directions in the control of vector-borne diseases in urban environments. Health Policy Plan. 9, 113–129 (1994).
Adje, D. D. et al. Étude de la pollution organique de la rivière Okedama dans la Commune de Parakou. Afrique Sci. 15, 299–305 (2019).
Mpakam, H. et al. Etude des facteurs de pollution des ressources en eau en milieu urbain: cas de Bafoussam (Ouest-Cameroun). Actes du colloque international sur le thème" changements climatiques et évaluation environnementale", de Niamey (Niger) (2009).
Adams, N. & Bealing, D. Organic pollution: biochemical oxygen demand and ammonia. Handbook of Ecotoxicology, 728–749 (1997).
Mireji, P. O. et al. Heavy metals in mosquito larval habitats in urban Kisumu and Malindi, Kenya, and their impact. Ecotoxicol. Environ. Saf. 70, 147–153 (2008).
White, B. J. et al. Dose and developmental responses of Anopheles merus larvae to salinity. J. Exp. Biol. 216, 3433–3441 (2013).
Zhao, G.-D. et al. Transcription profiling of eight cytochrome P450s potentially involved in xenobiotic metabolism in the silkworm, Bombyx mori. Pesticide Biochem. Physiol. 100, 251–255 (2011).
Oliver, S. V. & Brooke, B. D. The effect of metal pollution on the life history and insecticide resistance phenotype of the major malaria vector Anopheles arabiensis (Diptera: Culicidae). PloS One 13, e0192551 (2018).
Nkya, T. E. et al. Impact of agriculture on the selection of insecticide resistance in the malaria vector Anopheles gambiae: a multigenerational study in controlled conditions. Parasit. Vectors 7, 1–12 (2014).
David, J.-P., Ismail, H. M., Chandor-Proust, A. & Paine, M. J. I. Role of cytochrome P450s in insecticide resistance: impact on the control of mosquito-borne diseases and use of insecticides on Earth. Philos. Trans. R. Soc. B Biol. Sci. 368, 20120429 (2013).
Wondji, C. S. et al. Two duplicated P450 genes are associated with pyrethroid resistance in Anopheles funestus, a major malaria vector. Genome Res. 19, 452–459 (2009).
Vontas, J., Katsavou, E. & Mavridis, K. Cytochrome P450-based metabolic insecticide resistance in Anopheles and Aedes mosquito vectors: Muddying the waters. Pesticide Biochem. Physiol. 69, 104666 (2020).
Patrick, M. L., Aimanova, K., Sanders, H. R. & Gill, S. S. P-type Na+/K+-ATPase and V-type H+-ATPase expression patterns in the osmoregulatory organs of larval and adult mosquito Aedes aegypti. J. Exp. Biol. 209, 4638–4651 (2006).
White, B. J., Collins, F. H. & Besansky, N. J. Evolution of Anopheles gambiae in Relation to Humans and Malaria. Annual Review of Ecology, Evolution, and Systematics 42 (2011).
Dabiré, K. et al. Occurrence of natural Anopheles arabiensis swarms in an urban area of Bobo-Dioulasso city, Burkina Faso, West Africa. Acta Tropica 132, S35–S41 (2014).
Service, M. W. Mosquito ecology field sampling methods. 2nd edn, (Elsevier Applied Science, 1993).
Bass, C. et al. Detection of knockdown resistance (kdr) mutations in Anopheles gambiae: a comparison of two new high-throughput assays with existing methods. Malar. J. 6, 1–14 (2007).
Santolamazza, F. et al. Insertion polymorphisms of SINE200 retrotransposons within speciation islands of Anopheles gambiae molecular forms. Malar. J. 7, 163 (2008).
Tene F. B. et al. Resistance to DDT in an urban setting: common mechanisms implicated in both M and S forms of Anopheles gambiae in the city of Yaoundé Cameroon. PloS one 8 (2013).
Nsango, S. E. et al. AP-1/Fos-TGase2 axis mediates wounding-induced Plasmodium falciparum killing in Anopheles gambiae. J. Biolog. Chem. 288 (2013).
Dray, S. & Dufour, A.-B. The ade4 package: implementing the duality diagram for ecologists. J. Statis. Software 22 (2007).
Bates, D., Maechler, M., Bolker, B. & Walker, S. (2015).
Bartlett, M. S. Properties of sufficiency and statistical tests. Proc. R. Soc. London. Ser. A Math. Phys. Sci. 160, 268–282 (1937).
Akaike, H. A new look at the statistical model identification. IEEE Trans. Autom. Control 19, 716–723 (1974).
Burnham, K. P. & Anderson, D. R. Multimodel inference: understanding AIC and BIC in model selection. Sociol. Methods Res. 33, 261–304 (2004).
Schloerke, B., Crowley, J., Cook, D., Briatte, F., Marbach, M., Thoen, E., Elberg, A., & Larmarange, J. GGally: extension to ‘ggplot2’. R package version 1.4. 0. R Foundation for Statistical Computing. (2018).
Acknowledgements
We specially thank the members of the ESV-GAB (CIRMF, Franceville, Gabon), Medical Entomology Laboratory (OCEAC, Yaoundé, Cameroon) and Laboratoire d'Analyse Géochimiques des Eaux (LAGE, Yaoundé, Cameroon), who participated in the fieldwork in Gabon and Cameroon. Funds were provided by the Agence Universitaire de la Francophonie (AUF, ‘Salt-Adapt’ Project), the Institut de Recherche pour le Développement (IRD, ‘URBANO’ Project), and the Centre International de Recherches Médicales de Franceville (CIRMF). N. L-P was funded by AUF and CIRMF scholarships. Finally, we specially thank Ray Gaab for insights and comments on the manuscript.
Author information
Authors and Affiliations
Contributions
NMLP, BTF and DA designed the research. NMLP, BTF and RET did the fieldwork. RET and JJB did the water analysis. NMLP, OAE BTF, RAE, CT, PK and NB performed the molecular analysis for species identification. NMLP and NB carried out the gene expression analysis. NMLP, CC and DA performed the statistical analysis. NMLP, NR, CC and DA wrote the paper. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Longo-Pendy, N.M., Tene-Fossog, B., Tawedi, R.E. et al. Ecological plasticity to ions concentration determines genetic response and dominance of Anopheles coluzzii larvae in urban coastal habitats of Central Africa. Sci Rep 11, 15781 (2021). https://doi.org/10.1038/s41598-021-94258-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-94258-6
- Springer Nature Limited
This article is cited by
-
Utility of plasma anti-gSG6-P1 IgG levels in determining changes in Anopheles gambiae bite rates in a rural area of Cameroon
Scientific Reports (2024)
-
Assessment of environmental and spatial factors influencing the establishment of Anopheles gambiae larval habitats in the malaria endemic province of Woleu-Ntem, northern Gabon
Malaria Journal (2024)
-
Rehydration of the sleeping chironomid, Polypedilum vanderplanki Hinton, 1951 larvae from cryptobiotic state up to full physiological hydration (Diptera: Chironomidae)
Scientific Reports (2022)