Abstract
During the 1999–2002 water years, a hydrogeological research project was carried out on the unconfined aquifer of Trifilia in the Peloponnese. Seawater intrusion due to overpumping, and intensive use of fertilizers caused the groundwater quality degradation that is a typical case for the coastal aquifers in Greece. Isopiezometric maps along with ion distribution balances, ion distribution maps and factor analysis indicate the existence of three zones of groundwater quality. In the first zone of saline water, a cation exchange process between the Ca2+ of sediments and the Na+ of groundwater contribute to the formation of the water type Na+-Ca2+-Cl−. In the second zone, which is considered as a transition zone, dominate the Ca2+-Na+-HCO3 −-Cl− water type. In the third zone of Ca2+-HCO3 −-SO4 2− water type, relationships among Ca2+, SO4 2−, NO3 − and NH4 can be attributed to the dissociation of ammonium nitrate and sulfate fertilizers on one hand, and Ca2+ derivation from cation exchange processes between water, rocks and clay minerals, such as smectite and illite, on the other.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
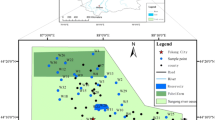
The study area lies in the Trifilia province of southwestern Peloponnesus and covers a total area of 120 km2 (Fig. 1). The mean annual air temperature is 16.3 °C, with a minimum of 11.1 °C in February and a maximum of 25.7 °C in August. The mean annual precipitation at Gargalianoi meteorological station is 784 mm (Panagopoulos and Lambrakis 2001). The dominant land use in the area is cultivation of olives (83%), vegetables (12%, mainly tomatoes and watermelons), and greenhouse crops (5%).
Geological map of western Trifilia (Perrier 1980, Filiatra sheet)
Irrigation demands within the coastal regions are covered by exploiting the unconfined aquifer of Pleistocene sediments through a large number of shallow wells and deeper boreholes.
A widespread agricultural practice in the area is the use of inorganic fertilizers, which pollute the groundwater. According to Sampatakakis and others (1994), the mean annual amount of fertilizers applied in the Trifilia area is 1,850 Kg/ha. Compositions of these fertilizers have changed during the last decades. In 1960, 70–80% of the fertilizers were ammonium sulfate [(NH4)2SO4], 10–15% was ammonium nitrate (NH4NO3), 10–15% was mixed nitrogen (N), phosphorous (P) and potassium (K), and the remaining 5–10% were trace elements. In 1970, these values changed to 50–60, 25–30, 20–30 and 5–10% respectively, while in 1980 the percentage contribution was 20–30, 30–40, 30–40 and 10%, respectively.
Nitrate contamination of groundwater is of environmental concern. One of the main sources of contamination is the high input of nitrogen fertilizers in intensive agriculture (Roth and Fox 1990; Johnson and others 1991). Yadav (1997) showed that 15% of the annually applied nitrogen fertilizer, and even greater percentages of residual nitrogen, were leached into groundwater. This load contributes to chemical, physical and biological processes.
When applying factor analysis to the hydrochemical data of a coastal aquifer, Giménez (1994) found a factor with a high correlation coefficient between Ca2+ and NO3 −. He explained this relationship as a result of processes developed after the application of nitrogenated fertilizers. These processes add Ca2+ to the water as a consequence of a likely ion exchange between NH4 + and Ca2+. Comber and others (1967) refer that NH4 + derived from nitrogenous fertilizer dissociation, can replace an equivalent quantity of Ca2+ in the interlayers of clay minerals and then reaches the aquifer. A number of similar observations for Ca2+, SO4 2−, NO3 − and NH4 + interrelations have been referred in the literature (Ruiz and others 1990; Covelli and others 1998; Stigter and others 1998; Jeong 2001). In the hydrochemical study of a coastal karst aquifer located in Malia, Greece (Lambrakis and Papatheodorou 2001), a similar factor exhibited high positive loadings on Ca2+, Mg2+, NH4 +, NO3 − and SO4 2−, suggesting a nitrate contribution of anthropogenic origin. Daskalaki (2002) studying the hydrochemical character and quality of groundwater in Greece, found a clear relationship between Ca2+ and NO3 − in almost all the alluvial aquifers of Greece, and many sedimentary and metamorphic carbonate aquifers of Central and North Greece. Ca2+ was attributed to the mineralogical composition of the aquifer, and NO3 to agricultural activities.
The aim of this article is to examine the impact of intensive land use and fertilizer application in western Trifilia of Peloponnesus, Greece, and contribute to the interpretation of the hydrochemical processes taking place in such unconfined coastal aquifers.
Materials and methods
The impact of fertilizer application in western Trifilia was investigated by studying the hydrogeology and hydrochemistry of the unconfined coastal aquifer. The inventory of the water points indicated that more than 300 shallow wells and deep boreholes exploit the aquifer. A series of 75 wells was used for piezometric measurements. Among these, 35 sample points were defined for chemical analyses (Fig. 2). Eight boreholes of the aquifer were tested for hydraulic parameter determination, and three core samples were collected to determine mineralogical composition. Statistical processing of the groundwater chemical analyses data was made using R-mode factor analysis.
Sampling and hydrochemical measurements
Sampling runs were undertaken biannually from May 1999 to October 2001. The samples were collected at the well-pump outflow in two polyethylene bottles. All samples were filtered on-site through 0.45 μm pore size Millipore® filters. Unstable parameters such as pH, electric conductivity (EC), dissolved oxygen (DO) and redox potential (Eh), were determined on-site by using an ion/EC meter with combined electrodes. The other analyses were performed soon after collection in the Laboratory of Hydrogeology, University of Patras. The first sample of 0.5 L volume was acidified with 2 ml of 65% HNO3 for cation analysis. The second non-acidified aliquot (1 L) was used to determine bicarbonates (HCO3 −) and chloride (Cl−) using Hach® titration kits. Sulfates (SO4 2−), nitrates (NO3 −) and ammonium (NH4 +) were determined using a spectrophotometer. Finally, the major cations Ca2+, Mg2+, Na+ and K+ were determined by atomic absorption spectroscopy. The overall precision of the analyses is within ±5%. Processing of the data was carried out using a software package developed in the Laboratory of Hydrogeology, University of Patras (Lambrakis 1991). A statistical overview of the measured parameters is presented in Table 1 (May 2001).
Mineralogical composition
Three core samples (S1, S2, S3) were collected from the Pleistocene sediments of the study area (Fig. 1). The mineral compositions of the clay fraction (<2 μm) extracted by sedimentation were determined by X-ray diffraction (XRD) using a Philips PW1050/25 diffractometer with Ni-filtered CuKa radiation. Oriented clay powders were prepared by the dropper method and were scanned at 1° 2θ/min from 3 to 30°2θ. For each <2 μm specimen, clay minerals were identified from three XRD patterns (air-dried at 25 °C, ethylene-glycol solvated, and heated at 490 °C for 2h). X-ray diffractograms indicate that smectite and illite minerals dominate the clay fraction of the studied samples (Figs. 3, and 4). Calcite, quartz and traces of plagioclase were also present.
Factor Analysis
R-mode factor analysis offers a comparison of relationships among variables in terms of samples, and requires a homogeneous population in order to be meaningful without any important loss of information. Factor analysis also allows us to determine the geographical distribution of the resulting factors. Geological interpretation of the factors gives insight into the main processes that may control the distribution of hydrochemical parameters. Following Davis (1986), R-mode factor analysis was applied to study the interrelations between the 13 variables examined in the western Trifilia aquifer data-set for the wet season (May) of the hydrological year 2001 (n=35 samples; Table 1). There are many recommendations for selecting the optimum number of factors (“amount” of cumulative variance, Eigenvalues greater than 1, “scree plot”, etc.; Davis 1986; Reyment and Jőreskop 1993), but the most straightforward solution to this problem is to extract as many factors as the ruling theory demands. According to this criterion, three factors have been chosen for this study, which together explain more than 76% of the total variance (Table 2). Apart from Eh, all the other parameters have high communality (0.65–0.98), which shows that the 3-Factor model describes them well. The correlation coefficients between variables and the factors (loadings) are listed in Table 2.
Geological and hydrogeological setting
The bedrock of the research area belongs to the Tripolis Series and is composed of a thick sequence of Eocene and dolomitic limestones overlain by Oligocene flysch (Perrier 1980). Pleistocene and Holocene deposits unconformably overlie all of these sediments (Fig. 1). The Pleistocene sediments host an extensive unconfined aquifer and are comprised mainly of calcitic sandstone and marly limestone, with a thickness ranging from 30 to 100 m, which were deposited in a shallow marine environment.
The piezometric map (Fig. 5) provides a good description of the aquifer and shows that the general groundwater flow is SW towards the coast of the Ionian Sea. The aquifer presents low hydraulic loads, close to zero or negative levels, in its coastal sector. Towards the inland and to the east, the water table has positive values that reach 93 m. The hydraulic gradients are smaller in the west than the east, and in the wider area of Filiatra and Marathopolis vary between 1.2 and 2.5%. Eastwards, in the area of Chochlasti, gradients are greater and vary between 5 and 7%. This is due to the smaller transmissivity in the eastern part of the aquifer compared to the western part, as proved by pumping tests. These tests show that transmissivity varies between 4×10−3 m2/s and 4.9×10−2 m2/s in the western part.
The water balance parameters have been computed based on the procedures described by Thornthwaite and Mather (1955). The potential evapotranspiration has been computed based on temperature data using Thornthwaite’s formula (1948). The average annual volume of actual evapotranspiration is 23.5×106 m3 and corresponds to 48.1% of the annual precipitation. The mean annual volume of the area’s surface runoff is 14.7×106 m3 and corresponds to 30.2% of the mean annual precipitation, while the remaining 10.6×106 m3 (21.7% of the mean annual precipitation) is the recharge groundwater.
Results and discussion
Groundwater chemistry
Based on the Piper diagram (Fig. 6), three different water types can be discerned in the study area. These are the following: Na+-Ca2+-Cl− water type (Group 1), Ca2+-Na+-HCO3 −-Cl− water type (Group 2) and Ca2+-HCO3 −-SO4 2− water type (Group 3). Their distribution is shown in Fig. 7.
The Na+-Ca2+-Cl− water type occupies the coastal zone (zone 1) and is dominated by Na+. The water samples of this zone are characterized by higher electrical conductivity and Na+, K+, Mg2+ and Cl− concentrations compared with samples from the two other types (Table 3). The chloride distribution map (Fig. 8) shows three regions in the coastal zone with high chloride concentrations (Filiatra, Agia Kyriaki, Marathopolis), while the highest concentration (844 mg/l) is found in the Marathopolis region. The increased salinity of the groundwater in this zone could be attributed to seawater intrusion due to the low hydraulic load of the aquifer here. This low hydraulic load (Fig. 5) is caused by the overexploitation of the aquifer to meet increased irrigation demands and to the lack of rational water management.
Seawater intrusion causes ion exchange phenomena between brackish water and the aquifer’s matrix. Presuming that Ca2+ is the dominant ion, as is the case for the sediments of the study area, the following equations can describe the above-mentioned process (Appelo and Postma 1993):
where X is the soil exchanger.
By calculating a composition based on conservative mixing between seawater and freshwater of the area (Appelo and Postma 1993), we obtained the concentration of the different ions as deficits (negative numbers) and excesses (positive numbers, Table 4), expressed as Δion=Cm – Cex, where Cm is the measured concentration and Cex is the concentration expected by freshwater – seawater conservative mixing. Table 4 shows that all groups have excess in Ca2+ concentration, while Na+ is obviously in deficit in the first group and in excess in the other groups.
Similar observations can be seen in Fig. 9a and b where the straight line is the mixing line between the area’s sea and fresh waters. Samples from the first zone belonging to the first group show ion exchange phenomena as described above, due to seawater intrusion.
The chemical composition of group 2 with water type (Ca2+-Na+-HCO3 −-Cl−), is characterized mainly by the dominance of Ca2+ and lower salt content compared to the first water type. Groundwater becomes enriched with ions such as Na+ and Cl− (Table 3) from dissolving minerals that move along a flow line towards the sea.
The third group, water type Ca2+-HCO3 −-SO4 2−, has samples with a chemical composition dominated by Ca2+ and HCO3 − ions that characterizes fresher waters. Thus, zone three can be considered as the recharge zone, while the second zone can be considered as a transitional zone between fresh and saline water. The SO4 2− ion participation in this group’s water type can be attributed to the contamination of groundwater by fertilizers. Table 3 shows that the maximum values of NO3 − concentrations also belong to the samples of this group. The excess in Ca2+ concentrations can be attributed to ion exchange phenomena due not to seawater intrusion, because the hydraulic load is high and Na+ concentrations are also in excess, but to NH4 + ions due to extensive application of fertilizers (Comber and others 1967; Ruiz and others 1990; Gimenez 1994; Covelli and others 1998; Jeong 2001).
R-mode Factor Analysis
Following the application of R-mode Factor Analysis, a three-factor model was produced that accounted for more than 75% of the total variance (Table 2). Factor 1 accounts for nearly 40% of the total variance and has high positive (0.89–0.97) loadings for EC, Mg2+, Na+, K+ and Cl−, and moderate negative loading for HCO3 − (Table 2). The spatial distribution of factor scores for factor 1 (Fig. 10a) has a discernible pattern; the higher scores occur in the coastal zone (zone 1 of Fig. 7), moderate scores are present in the transitional zone (zone 2 of Fig. 7), and lower scores occur in the aquifer’s recharge area (zone 3 of Fig. 7). This particular pattern in conjunction with high positive loadings of EC, Mg2+, Na+, K+ and Cl−, reflects the intrusion of seawater in the coastal zone of the unconfined aquifer.
Factor 2 accounts for 23.4% of the data’s total variance and exhibits high positive loadings (0.77–0.89) on Ca2+, SO4 2−, NO3 − and NH4 + (Table 2). The spatial distribution of this factor shows that the area with higher factor scores has an elliptic shape and is located in Chochlasti region (Fig. 10b). Together with the loading profile, this geographical pattern suggests that the second factor is a contamination factor of anthropogenic origin (fertilizers).
Factor 3 accounts for 13.8% of the total variance. The interrelationship between the variables associated with this factor, dissolved oxygen (DO), Eh and pH, is well known (Freeze and Cherry 1979).
Nitrate contamination
Due to extensive use of fertilizers in the area, the groundwater contains an increased concentration of NO3 − and NH4 + (Fig. 11a, b), especially in the third zone (zone 3 of Fig. 7) of the aquifer (Table 3), with maximum values seen in Chochlasti region (290.4 and 3.73 mg/l respectively). Ca2+ and SO4 2− display a similar distribution pattern to NO3 − and NH4 + in the region of Chochlasti with values of 340.8 and 460 mg/l respectively (Fig. 12a, b; Table 3), suggesting close relationships between these chemical species.
These characteristic chemical distribution patterns of the groundwater are similar to the results of the R-mode factor analysis shown in Fig. 10b. Nitrate, ammonium, calcium and sulfate distribution maps (Figs. 11a, b; and 12a, b respectively) also show higher concentrations of these ions in the same region. The cross-plots in Fig. 13 support the existence of strong relationships among these parameters in zone 3 of the aquifer, as the correlation coefficient between Ca2+ and total nitrogen (NO3 −+NH4 +) is extremely high (0.958), while the correlation coefficient between Ca2+ and SO4 2− is 0.757.
The relatively higher concentrations of nitrates and ammonium in zone 3 could be attributed to the intense application of nitrogen fertilizers to the soil, mostly NH4NO3 and (NH4)2SO4. Thus, the applied fertilizers move through the unsaturated zone and infiltrate into the aquifer. Nitrate and sulfate salts are very soluble in water (over 10 g/l at 20 °C; Hatziioannou 1985) and thus dissociate according to the following reactions:
causing an increase of nitrate and sulfate concentrations, which is facilitated by the oxidizing environment of the aquifer. NH4OH is a weak base and dissociates according to the following reaction (Hatziioannou 1985):
that causes an increase of ammonium concentration in the aquifer.
The relative high concentration of Ca2+ in zone 3 and its relationship with NH4 + could be attributed to ion exchange reactions between water, rock and the clay minerals smectite and illite.
Due to the dominance of calcite in the Pleistocene sediments of the study area, it is reasonable to assume that most of the interlayer sites of smectite were occupied by Ca2+. In such an environment, Ca2+ could replace the cations which were originally present in the interlayer of smectite. The high NH4 + concentration in the water samples of zone 3, could promote the saturation of Ca-smectite with NH4 + because the replacing power of NH4 + is greater than that of Ca2+ (Grim 1968), according to the ion exchange reaction:
Thus, the interlayer Ca2+ cations of smectite are “exchanged out” and liberated in the groundwater. This exchange of cations could explain the enrichment of water samples of zone 3 in Ca2+ and the observed positive correlation between Ca2+ and NH4 +. Illite may also contribute to the enrichment of Ca2+ in the aquifer, though to a lesser extent, due to its lower cation exchange capacity.
Conclusions
The hydrogeological and hydrochemical study of the western Trifilia region reveals marked seawater intrusion in the unconfined aquifer of Pleistocene sediments in the coastal zone, due to the reduction of the water level below sea level caused by intensive pumping. As a result, Na+, Ca2+ and Cl− ions dominate the groundwater composition in this zone. Two further zones, the second and third, were defined in the region. These two zones lack direct sea influences on groundwater chemistry. Ion balances showed that an excess of Ca2+ concentrations in the water samples of the first zone can more or less be equilibrated with Na+ and K+ concentrations that are in deficit due to ion exchange phenomena caused by the seawater intrusion. Ca2+ concentrations are still in excess in water samples of the other two zones and present a high correlation with nitrogen. The intensive land use and the nitrogen-based fertilizers applied in the study area have resulted in high nitrate, sulfate and ammonium concentrations (290.4, 460 and 3.73 mg/l respectively) in the groundwater of the third zone. It is suggested that the hydrochemical composition of the groundwater is influenced by cation exchange processes, which are facilitated by the presence of the clay minerals smectite and illite in the Pleistocene sediments of the unconfined aquifer.
The high amount of NH4 + in the water of zone 3, coupled with its high replacing power, promotes the saturation of smectite with NH4 +. Cations, e.g. Ca2+, which were in the interlayers of smectite and, to a lesser degree, illite, and that were subsequently substituted by NH4 +, were liberated into the groundwater. This cation exchange process leads to the high correlation between Ca2+ and NH4 + in samples contaminated by nitrates and sulfates.
References
Appelo CAJ, Postma D (1993) Geochemistry, groundwater and pollution, pp 315–318
Comber NM, Willcox JS, Jones HT, Townsend WN (1967) Introduction to the agricultural chemistry. WNM Giourdas, Athens (translation in Greek)
Covelli S, Cucchi F, Mosca R (1998) Monitoring of percolation water to discriminate surficial inputs in a karst aquifer. Environ Geol 36(3–4):296–304
Daskalaki P (2002) Spatiotemporal changes of groundwater quality in Greece. Ph.D. Thesis, University of Patras, Greece (in Greek)
Davis JC (1986) Statistics and data analysis in geology. Wiley, New York
Freeze RA, Cherry JA (1979) Groundwater. Prentice-Hall, Englewood Cliffs, New Jersey
Giménez E (1994) Caracterización hidrogeoquímica de los procesos de salinización del aquífero detrítico costero de la Plana de Castellon. Ph.D. Thesis, Universidad de Granada, Spain (in Spanish)
Grim RE (1968) International series in the Earth and Planetary Sciences. McGraw-Hill Book Company, New York
Hatziioannou TP (1985) Assay and chemical equilibrium. O.P.D.B., Athens (in Greek)
Jeong CH (2001) Effect of land use and urbanization on hydrochemistry and contamination of groundwater from Taejon area, Korea. J Hydrol 253:194–210
Johnson SL, Adams RM, Perry GM (1991) The on-farm costs of reducing groundwater pollution. AM J Agric Econ 73:1063–1073
Lambrakis N (1991) Elaboration of chemical analysis data. Mineral Wealth 74:53–60 (in Greek)
Lambrakis N, Papatheodorou GS (2001) Factor analysis and interpretation of the hydrochemistry of a coastal aquifer, Crete, Greece. Proc Int Conf “Ecological Protection of the Planet Earth I”, Xanthi, Greece, vol. 1, pp 59–67
Panagopoulos GP, Lambrakis NJ (2001) Investigation of the quality of groundwater of SW Trifilia karstic aquifer in respect with the hydrogeological conditions of the area. Bulletin of Geological Society of Greece, vol. XXXIV/5, Proc 9th Int Congr, Athens, Greece, pp 1885–1892
Perrier R (1980) Geological map of Greece, Filiatra sheet, I.G.M.E., Athens
Reyment R, Jőreskog KG (1993) Applied factor analysis in the natural sciences. Cambridge University Press, Cambridge
Roth GW, Fox RH (1990) Soil nitrate accumulations following nitrogen fertilized corn in Pennsylvania. J Environ Qual 19:243–248
Ruiz F, Gomis V, Blasco P (1990) Application of factor analysis to the hydrogeochemical study of a coastal aquifer. J Hydrol 11:169–177
Sampatakakis P, Makris A, Gidonis E, Tzoulis H (1994) Phenomena of undergroundwater pollution in Trifilia coastal area from the use of agricultural fertilizers. Bulletin of the Geological Society of Greece, vol. XXX/4, Proc 7th Int Congr, Thessaloniki, Greece, pp 115–125 (in Greek)
Stigter TY, van Ooijen SPJ, Post VEA, Appelo CAJ, Carvalho Dill AMM (1998) A hydrogeological and hydrochemical explanation of the groundwater composition under irrigated land in a Mediterranean environment, Algarve, Portugal. J Hydrol 208:262–279
Tellam JM, Lloyd JW (1986) Problems in the recognition of seawater intrusion by chemical means: an example of apparent chemical equivalence. J Engin Geol 19:389–398
Thornthwaite CW (1948) An approach toward a relational classification of climate. Geogr Rev 38(1):55–94
Thornthwaite CW, Mather JR (1955) The water budget and its use in irrigation water in the year book of agriculture. US Dept Agric, pp 346–358
Yadav SN (1997) Formulation and estimation of nitrate-nitrogen leaching from corn cultivation. J Environ Qual 26:808–814
Acknowledgements
The current research was conducted on the frames of “K. Karatheodori” project, financed by the Research Committee of Patras University. The first author would like to thank the Greek State Scholarships Foundation for the economical support of his research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panagopoulos, G., Lambrakis, N., Tsolis-Katagas, P. et al. Cation exchange processes and human activities in unconfined aquifers. Env Geol 46, 542–552 (2004). https://doi.org/10.1007/s00254-004-1064-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00254-004-1064-6