Abstract
Temperature affects the development, population dynamics, reproduction and population size of insects. Thaumastocoris peregrinus Carpintero et Dellape (Heteroptera: Thaumastocoridae) is a eucalyptus pest. The objective of this study was to determine biological and life table parameters of T. peregrinus on Eucalyptus benthamii at five temperatures (18 °C; 22 °C; 25 °C; 27 °C and 30 °C) with a relative humidity (RH) of 70 ± 10% and photoperiod of 12 hours. The duration of each instar and the longevity of this insect were inversely proportional to the temperature, regardless of sex. The nymph stage of T. peregrinus was 36.4 days at 18 °C and 16.1 days at 30 °C. The pre-oviposition period was 5.1 days at 30 °C and 13.1 days at 18 °C and that of oviposition was 7.6 days at 30 °C and 51.2 days at 22 °C. The generation time (T) of T. peregrinus was 27.11 days at 22 °C and 8.22 days at 30 °C. Lower temperatures reduced the development and increased the life stage duration of T. peregrinus. Optimum temperatures for T. peregrinus development and reproduction were 18 and 25 °C, respectively.
Similar content being viewed by others
Introduction
The frequent introduction and establishment of exotic insect pests on eucalyptus plantations in Brazil are impacting and reducing productivity. The bronze bug Thaumastocoris peregrinus Carpintero & Dellapé (Hemiptera: Thaumastocoridae), an Australian eucalyptus pest was first recorded in Brazil in 20091. At high infestations, this insect decreases the photosynthetic rate, leading to partial or total plant defoliation, and in some cases, plant death2,3.
Studies have focused on the biology4,5,6, chemical control7, chemical ecology8,9, morfology10, remote sensing for monitoring11,12 and biological control13,14,15,16 of this pest, aiming to minimize losses. However, the effect of temperature on the biological parameters of this species is not yet well known.
Ambient temperature is one of the most important abiotic factors affecting the survival, development rate, abundance, behavior and fitness of insects17,18,19,20. In fact, each insect species has an optimum temperature at which they thrive, with lower and upper limits for development21,22. High temperatures can decrease fecundity, hatching and survival of these organisms23, while low temperatures can affect the sex ratio (reduce male proportion), behavior, and population distribution of insects24.
The study of temperature in life-history variables, such as nymph development period, adult longevity and fecundity is crucial to the development of pest-management strategies25. The temperature decrease Parapoynx crisonalis (Lepidoptera: Pyralidae) life tables26 and Brachmia macroscopa (Lepidoptera: Gelechiidae) development and fecundity27. Thus, the objective of this study was to evaluate the effect of different temperatures on biological parameters of T. peregrinus.
Results
Nymph development
The nymph development period of T. peregrinus differed across temperatures (Kruskal-Wallis on ranks; df = 4, H = 168.42, P < 0.001) (Table 1). Furthermore, this parameter affected the duration of each instar (first-instar, Kruskal-Wallis on ranks; df = 4, H = 219.31, P < 0.001; second instar, Kruskal-Wallis on ranks; df = 4, H = 198.67, P < 0.001; third instar, Kruskal-Wallis on ranks; df = 4, H = 172.49, P < 0.001; fourth instar, Kruskal-Wallis on ranks; df = 4, H = 134.77, P < 0.001; and fifth instar, Kruskal-Wallis on ranks; df = 4, H = 126.4, P < 0.001) of this insect.
Adult reproduction and longevity
The pre-oviposition period of T. peregrinus decreased linearly with increased temperature, ranging from 13 (18 °C) to 5 (30 °C) days (Table 2). The fertility of this insect was similar at 22 °C (64 eggs), 18 °C (45.9 eggs), 25 °C (58.1) and 27 °C (49.1), while it was lower at 30 °C (22 eggs) (Table 2).
Female longevity of T. peregrinus was longest at 22 °C (53 days) and that of males at 18 to 22 °C (57 and 54 days, respectively) (Table 2). Temperature did not affect the sex ratio of this insect (GLM-binomial: χ2190 = 1.96, p = 0.74) (Table 2).
Survival analysis
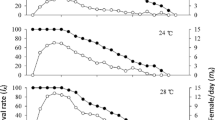
Temperature affected the survival rates of T. peregrinus nymphs (Mantel-Haenzel Test; χ2 = 53.6, P < 0·0001) (Fig. 1A), females (Mantel-Haenzel Test; χ2 = 60.9, P < 0·0001) (Fig. 1B), and males (Mantel-Haenzel Test; χ2 = 103, P < 0.0001) (Fig. 1C).
Survival analysis using the Cox’s Proportional Hazards model showed a higher death risk (hazard ratio; HR) for nymphs and adults (females and males) of T. peregrinus as temperature increased (Table 3) and (Fig. 2).
Threshold development and thermal constants
The linear regression estimative for the temperature limit of T. peregrinus first, second, third, fourth and fifth instars was 7.70, 9.85, 10.33, 10.24 and 10.45 °C, respectively (Fig. 1). The T. peregrinus thermal constant (K) per instar was 79.34 degree-day (DD) (first), 58.58 DD (second), 55.57 DD (third), 62.24 DD (fourth) and 88.91 DD (fifth). The accumulated temperature for the nymph-to-adult period of this insect was 338.50 DD, with a temperature limit of 9.93 °C (Fig. 1).
Life table
The net reproductive rate (R0) of T. peregrinus was higher at 25 °C (6.39) and 18 °C (4.45), the latter being similar to that at 22 °C (4.00). The net reproduction rate was lower at 30 °C (0.13). The generation time (T) of T. peregrinus varied between 27.11 days at 22 °C to 8.22 at 30 °C, and the intrinsic growth rate (rm) and finite increase (λ) of T. peregrinus were higher at 25 °C (0.046 and 1.047, respectively) and lower at 30 °C (0.084 and 0.919, respectively) (Table 4).
Discussion
Temperature strongly influences insect development in both single generation progeny and in organisms that are established and successfully continued for multiple generations28. Thaumastocoris peregrinus development and reproduction reinforces the temperature effect on insects29, with the duration of its juvenile stage decreasing as temperature increases, as found for Corythucha ciliate (Say) (Hemiptera: Tingidae) and Loxostege sticticalis (L.) (Lepidoptera: Crambidae)30.
The shorter duration of each instar and of the adult period of T. peregrinus at higher temperatures is due to increased metabolism, food intake and energy, allowing the insect to reach the next stage31,32. Other factors, such as poor food quality9,33,34, decreased the survival and/or insect growth rate35. The ladybird Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae)36,37, the dragonfly Ischnura verticalis (Odonata: Coenagrionidae)38, and the locust Romalea microptera (Orthoptera: Romaleidae)39 had shorter juvenile stages at increased temperatures.
Thaumastocoris peregrinus had a shorter pre-oviposition period with increased temperature, indicating the effect of this parameter on this organism. This is also reflected in the mating and egg laying of T. peregrinus as reported for Phenacoccus madeirensis Green (Hemiptera: Pseudococcidae)40 and Leptocoris achinensis (Dallas) (Hemiptera: Alydidae)41 and food/temperature and bioecology interactions42 as reported for Cimex lectularius (Linnaeus 1758; Hemiptera: Cimicidae)43. The number of eggs per T. peregrinus female at 26 °C on Eucalyptus urophylla x Eucalyptus camaldulensis5 and Eucalyptus scoparia at different temperatures4 and with E. tereticornis at 25 °C44 varied within certain limits45. The longer pre-oviposition period, at least for some T. peregrinus females at lower temperatures could be due to the longer time required for this predator to develop its ovary46.
Thaumastocoris peregrinus female and male longevity was increased at temperatures between 18 to 22 °C, which could be due to reduced metabolic processes at lower temperatures, affecting development and life history47. At low metabolic rates, certain physiological processes are suppressed, for example reproduction48, in order to maintain more crucial processes for survival. The effect of low temperatures on longevity have been reported for Monosteira unicostata Mulsant & Rey 1852 (Hemiptera: Tingidae) and Cleruchoides noackae Lin & Huber, 2007 (Hymenoptera: Mymaridae)49,50.
The optimal temperature range for T. peregrinus development and reproduction between 25 and 30 °C was similar to those reported for egg, nymph and egg-adult periods, respectively, for this bug44,51, as well as for Nezara viridula (L.) (Hemiptera: Pentatomidae) collected in soybean fields at climatically different locations28. The linear increase in the ratio between instars and of the adult stage duration of T. peregrinus (1/D) confirms the energy gain for its physiological processes52.
The low survival at high temperatures indicates a phenotypic plasticity for T. peregrinus in different environments53.
The higher thermal constant of T. peregrinus nymph development, 338.50 DD with a minimum of 9 °C shows the impact of low temperatures on this insect51,54. This result was also observed for Axinoscymnus cardilobus (Ren and Pang) (Coleoptera: Coccinellidae), with 204 DD; it took 67 days at a minimum of 9.07 °C to complete one generation, while this was 120 days55 at 17 °C. However, the accumulated temperature for the nymph-to-adult period of T. peregrinus, with 395.43 DD with a temperature limit of 9.93 °C shows its high adaptive potential. This species needed 905.65 DD in Canberra, Australia, to complete a generation and survived at temperatures below 1.5 °C, with adults recovering at higher temperatures51.
The e R0, rm, T and λ of T. peregrinus showed shorter development periods and higher growth rates with increased temperature, similar to that reported for Megacopta cribraria (F.) (Hemiptera: Plastaspidae)56 and Jakowleffia setulosa (Jakovley, 1874) (Hemiptera: Lygaeidae)57. These characteristics are important to understand the impact of temperature on insect growth, survival, reproduction and population increase58,59. This is necessary because the energy generated by the anabolism and catabolism metabolic processes for insect growth and reproduction depends on the environmental temperature60.
The environmental temperature affected the development, fertility, longevity and mortality of T. peregrinus. Thus, the definition of thermal requirements for T. peregrinus can assist traditional techniques in managing this pest. As well, this important data can be used in simulating population dynamics, monitoring, population peaks, occurrence, ecological zoning and modeling in order to manage this pest.
Material and Methods
Insect rearing and temperature conditions
The experiments were conducted at the Forest Entomology Laboratory of Embrapa Florestas in Colombo, Paraná state, Brazil. Thaumastocoris peregrinus was reared in the laboratory at 24 ± 2 °C, 60 ± 10% RH, and a photoperiod of 12:12 h L:D on bouquets of Eucalyptus benthamii Maiden & Cambage (Myrtales: Myrtaceae) branches. The branches were fixed in a piece of foam to prevent drowning the insects in a 500-mL glass flask filled with water61,62. The effect of temperature on various biological parameters of T. peregrinus was evaluated at five constant temperatures (18, 22, 25, 27 and 30 ± 2 °C) with a photoperiod of 12:12 L: D and RH 70 ± 10% in climatic chambers (BOD Specification: Type B.O.D M.S.Mistura; model MSM 011/G; SERIES 1002.0157, Volts 220, W700).
Nymph development
Newly hatched T. peregrinus nymphs were individually placed in acrylic plates (2.8 cm diameter × 1.5 cm) with a Eucalyptus benthamii fresh leaf disk (2.1 cm diameter) with its petiole introduced in a hydrogel layer (hydroplan-EB/HyC, SNF SA Floger) to maintain the leaf turgor. The eucalyptus leaf discs were replaced every two days. The duration and viability of T. peregrinus instars were assessed daily. Instar changes were evaluated based on the exuvia presence. Survival was evaluated in relation to the number of live individuals beginning each instar.
Adult reproduction and longevity
Thaumastocoris peregrinus adults (<24 h old) were sexed based on its morphological characteristics6. A couple of this insect was placed per Petri dish (5.0 cm in diameter) with a fresh E. benthamii leaf disc (4.9 cm diameter). The pre-oviposition (female emergence to the first egg laying) and oviposition period, fecundity (number of eggs per female per day), longevity and mortality of T. peregrinus were evaluated. The males were not replaced. Mortality data were used to calculate longevity. Females were maintained until death, and egg numbers were use in the analysis.
Development thresholds and thermal constants
The temperature development threshold (Tt) and thermal constant (K) of T. peregrinus were estimated using the hyperbole method63, based on the duration of the different instars, the nymph stage and the egg-adult period at 18, 22, 25, 27 and 30 ± 2 °C. The T. peregrinus instar development rate and nymph-to-adult period was regressed against temperature using a linear equation given by the formula: 1/D = a + bT, where, 1/D is the insect development time (D) in days, and T is the temperature (°C). The intercept ratio over the slope of the regression line corresponds to the threshold temperature (Tt) and the thermal constant (K) was estimated by taking the inverse of the slope (1/b)64.
Life table analysis
The T. peregrinus fertility life table at each temperature was built with specific survival at age x (lx), specific fertility (mx) and number of offspring reaching the age x in the next generation (lxmx). These data were used to calculate the net reproductive rate (R0), time between generations (T), innate ability to increase (rm) and finite rate of increase (λ) of this insect65.
Biological parameter analysis
All data were first analyzed using the Shapiro-Wilk and Bartlett tests to determine data normality and homogeneity. The data related to each instar duration and of the nymph-to-adult period did not conform to normality, even after log transformation. Therefore, the comparisons were made using the non-parametric Kruskal-Wallis test. Pre-oviposition, oviposition, fecundity, oviposition rate and female and male longevity data were normally distributed, and thus they were analyzed using a linear model followed by a post hoc pairwise comparisons performed using Tukey HSD test (function glht, package multcomp)66. Sex ratio was analyzed using a generalized linear model (GLM) assuming binomial distribution67,68. The analyses were performed with the software R, version 3.3.2. The fertility life table was analyzed by Jackknife and the averages compared by Student’s t-test using the software SAS version 9.169.
Survival analysis
Survival curves were fitted and analysed using Kaplan-Meier survival probabilities (R version 3.3.2, “survival”, “survminer” packages)70,71 followed by a pairwise comparisons Mantel-Haenszel Test (Log-Rank test) and Cox Proportional-Hazard Model (PH Model). The data evaluated at 30 °C was used as the reference for the other treatments (temperatures) on Multivariate Cox regression. Individuals who did not die by the end of the nymph period were censored (0 = death event did not occur; 1 = death event occurred). The adult individuals were not censored, because the experiment finished with the death of all insects (females and males).
Change history
16 January 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Wilcken, C. F. et al. Bronze bug Thaumastocoris peregrinus Carpintero & Dellapé (Hemiptera: Thaumastocoridae) on Eucalyptus in Brazil and its distribution. J. Plant Prot. Res. 50(02), 184–188 (2010).
Jacobs, D. H. & Neser, S. Thaumastocoris australicus Kirkaldy (Heteroptera: Thaumastocoridae): a new insect arrival in South Africa, damaging to Eucalyptus trees: research in action. S. Afr. J. Sci. 101(5), 233–236 (2005).
Nadel, R. L. et al. DNA bar-coding reveals source and patterns of Thaumastocoris peregrinus invasions in South Africa and South America. Biol. Invasions. 12, 1067–1077 (2010).
Noack, A. & Rose, H. Life-history of Thaumastocoris peregrinus and Thaumastocoris sp. in the laboratory with some observations on behaviour. Gen. Appl. Entomol. 36, 27–33 (2007).
Soliman, E. P. et al. Biology of Thaumastocoris peregrinus in different eucalyptus species and hybrids. Phytoparasitica 40(3), 223–230 (2012).
Nadel, R. L. & Noack, A. E. Current understanding of the biology of Thaumastocoris peregrinus in the quest for a management strategy. Int. J. Pest Manage. 58(3), 257–266 (2012).
Noack, A. E. et al. Efficacy of Imidacloprid in the control of Thaumastocoris peregrinus on Eucalyptus scoparia in Sydney, Australia. Arboric. Urb. Forest. 35, 192–196 (2009).
Martins, C. B. C. et al. Volatile chemicals of adults and nymphs of the Eucalyptus pest, Thaumastocoris peregrinus (Heteroptera: Thaumastocoridae). Psyche. 2012, 1–6 (2012).
Martins, C. B. & Zarbin, P. H. Volatile organic compounds of conspecific-damaged Eucalyptus benthamii influence responses of mated females of Thaumastocoris peregrinus. J. Chem. Ecol. 39, 602–611 (2013).
Noack, E. A., Cassis, G. & Rose, H. A. Systematic revision of Thaumastocoris Kikaldy (Hemiptera: Heteroptera: Thaumastocoridae). Zootaxa 3121, 1–60 (2011).
Oumar, Z. & Mutanga, O. The potential of remote sensing technology for the detection and mapping of Thaumastocoris peregrinus in plantation forests. Southern Forests: A J. Forest Sci. 73, 23–31 (2011).
Oumar, Z., Mutanga, O. & Ismail, R. Predicting Thaumastocoris peregrinus damage using narrow band normalized indices and hyperspectral indices using field spectra resampled to the Hyperion sensor. Int. J. Appl. Earth Obs. 21, 113–121 (2013).
Mascarin, G. M., Duarte, V. S., Brandão, M. M. & Delalíbera, I. Jr. Natural occurrence of Zoophthora radicans (Entomophthorales: Entomophthoraceae) on Thaumastocoris peregrinus (Heteroptera: Thaumastocoridae), an invasive pest recently found in Brazil. J. Invertebr. Pathol. 110, 401–404 (2012).
Souza, G. K. et al. First record of a native heteropteran preying on the introduced eucalyptus pest, Thaumastocoris peregrinus (Hemiptera: Thaumastocoridae), in Brazil. Fla. Entomol. 95, 517–520 (2012).
Mutitu, E. K. et al. Biology and rearing of Cleruchoides noackae (Hymenoptera: Mymaridae), an egg parasitoid for the biological control of Thaumastocoris peregrinus (Hemiptera: Thaumastocoridae). J. Econ. Entomol. 106, 1979–1985 (2013).
Dias, T. K. R. et al. Predation of Thaumastocoris peregrinus (Hemiptera: Thaumastocoridae) by Atopozelus opsimus (Hemiptera: Reduviidae) in Brazil. Invert. Surviv. J. 11, 224–227 (2014).
Harrington, R., Fleming, R. & Woiwood, I. P. Climate change impacts on insect management and conservation in temperate regions: can they be predicted? Agri. Forest Entomol. 3, 233–240 (2001).
Zheng, F. S., Du, Y. Z., Wang, Z. J. & Xu, J. J. Effect of temperature on the demography of Galerucella birmanica (Coleoptera: Chrysomelidae). Insect Sci. 15, 375–380 (2008).
Thomson, L. J., Macfadyen, S. & Hoffmann, A. A. Predicting the effects of climate change on natural enemies of agricultural pests. Biol. Control 52, 296–306 (2010).
Ju, R. T., Wang, F. & Li, B. Effects of temperature on the development and population growth of the sycamore lace bug, Corythucha ciliata. J. Insect Sci. 11, 1–16 (2011).
Briere, J. F., Pracos, P., Le Roux, A. Y. & Pierre, J. S. A novel rate model of temperature-dependent development for arthropods. Environ. Entomol. 28, 22–29 (1999).
Trudgill, D. L., Honek, A., Li, D. & Van Straalen, N. M. Thermal time– concepts and utility. Ann. Appl. Biol. 146, 1–14 (2005).
Cui, X., Wan, F., Xie, M. & Liu, T. Effects of heat shock on survival and reproduction of two white fly species, Trialeurodes vaporariorum and Bemisia tabaci biotype B. J. Insect Sci. 8, 1–10 (2008).
Nigro, R. G., Campos, M. C. C. & Perondini, A. L. P. Temperature and the progeny sex-ratio in Sciara ocellaris (Diptera, Sciaridae). Genet. Mol. Biol. 30, 152–158 (2007).
Kang, L., Chen, B., Wei, J. N. & Liu, T. X. Roles of thermal adaptation and chemical ecology in Liriomyza distribution and control. Ann. Rev. Entomol. 54, 127–145 (2009).
Chen, Q. et al. Age-stage, two-sex life table of Parapoynx crisonalis (Lepidoptera: Pyralidae) at different temperatures. PLoS ONE 12, 3 (2017).
Ma, L., Wang, X., Liu, Y., Su, M. Z. & Huang, G. H. Temperature effects on development and fecundity of Brachmia macroscopa (Lepidoptera: Gelechiidae). PLoS ONE 12(3), e0173065 (2017).
Chanthy, P., Martin, R. J., Gunning, R. V. & Andrew, N. R. Influence of temperature and humidity regimes on the developmental stages of green vegetable bug, Nezara viridula (L.) (Hemiptera: Pentatomidae) from inland and coastal populations in Australia. Gen. Appl. Entomol. 43, 37–55 (2015).
Davidson, J. On the relationship between temperature and rate of development of insects at constant temperatures. J. Anim. Ecol. 13, 26–38 (1994).
Cheng, Y. et al. Response of reproductive traits and longevity of Beet Webworm to temperature, and implications for migration. J. Insect Sci. 15, e154 (2015).
Brown, J. H. et al. Toward a metabolic theory of ecology. Ecology 85, 1771–1789 (2004).
Poncio, S. et al. Effect of temperature on immature of Stiretrus decastigmus (Hemiptera: Pentatomidae). Braz. J. Biol. 76, 495–499 (2016).
Larsson, S., Bjorkman, C. & Gref, R. Responses of Neodiprion sertifer (Hym., Diprionidae) larvae to variation in needle resin acid concentration in Scots pine. Oecologia 70, 77–84 (1986).
Lacerda, M. C. et al. Development and reproduction of Podisus distincus (Heteroptera: Pentatomidae) fed on larva of Bombyx mori (Lepidoptera: Bombycidae). Braz. J. Biol. 64, 237–242 (2004).
Kollberg, I. et al. Temperature affects insect outbreak risk through tritrophic interactions mediated by plant secondary compounds. Ecosphere 6, 1–17 (2015).
Dmitriew, C. & Rowe, L. Effects of early resource limitation and compensatory growth on lifetime fitness in the ladybird beetle (Harmonia axyridis). J. Evol. Biol. 20, 1298–1310 (2007).
Dmitriew, C. M. The evolution of growth trajectories: what limits growth rate? Biol. Rev. 86, 97–116 (2011).
Dmitriew, C., Cooray, M. & Rowe, L. Effects of early resource-limiting conditions on patterns of growth, growth efficiency and immune function at emergence in a damselfly (Odonata: Coenagrionidae). Can. J. Zool. 85, 310–318 (2007).
Flanagin, V. L., Haase, S. P. & Julian, S. A. Effects of growth rates on development to metamorphosis in the lubber grasshopper, Romalea microptera. Oecologia 125, 162–169 (2000).
Chong, J. H., Oetting, R. D. & Van Iersel, M. W. Temperature effects on the development, survival, and reproduction of the Madeira mealybug, Phenacoccus madeirensis Green (Hemiptera: Pseudococcidae), on chrysanthemum. Ann. Entomol. Soc. Am. 96, 539–543 (2003).
Yamashita, K. I., Sudo, K. I., Adachi, T. & Miura, K. Estimation of number of annual generations using effective heat unit of development for the rice bug, Leptocorisa chinensis (Dallas) (Hemiptera: Alydidae). Appl. Entomol. Zool. 40, 621–624 (2005).
Jamieson, M. A., Trowbridge, A. M., Raffa, K. F. & Lin Droth, R. L. Consequences of climate warming and altered precipitation patterns for plant-insect and multitrophic interactions. Plant Physiol. 160, 1719–1727 (2012).
Weeks, E. N. et al. Tracking bed bugs (Cimex lectularius): a study of the effect of physiological and extrinsic factors on the response to bed bug-derived volatiles. J. Exp. Biol. 216, 460–469 (2013).
Martinez, G. et al. Life-history analysis of Thaumastocoris peregrinus in a newly designed mass rearing strategy. B. Insectol. 67, 199–205 (2014).
Lysyk, T. J. Influence of temperature on oviposition by Dermacentor andersoni (Acari: Ixodidae). J. Med. Entomol. 51, 932–940 (2014).
Park, H. H., Park, C. G. & Ahn, J. J. Oviposition model of Cnaphalocrocis medinalis Guenée (Lepidoptera: Pyralidae). J. Asia. Pac. Entomol. 17, 781–786 (2014).
Keil, G., Cummings, E. & de Magalhães, J. P. Being cool: how body temperature influences ageing and longevity. Biogerontology 16, 383–397 (2015).
Boivin, G. Phenotypic plasticity and fitness in egg parasitoids. Neotrop. Entomol. 39, 457–463 (2010).
Sánchez-Ramos, I. et al. Effect of temperature on the survival and development of the immature stages of Monosteira unicostata (Hemiptera: Tingidae). Eur. J. Entomol. 112, e664 (2015).
Souza, A. R. D. et al. Longevity of Cleruchoides noackae (Hymenoptera: Mymaridae), an egg parasitoid of Thaumastocoris peregrinus (Hemiptera: Thaumastocoridae), with various honey concentrations and at several temperatures. Fla. Entomol. 99, 33–37 (2016).
Saavedra, M. C., Avila, G. A., Withers, T. M. & Holwell, G. I. The potential global distribution of the Bronze bug Thaumastocoris peregrinus Carpintero and Dellapé (Hemiptera: Thaumastocoridae). Agr. Forest Entomol. 17, 375–388 (2015).
Clissold, F. J. & Simpson, S. J. Temperature, food quality and life history traits of herbivorous insects. Curr. Opin. Insect Sci. 11, 63–70 (2015).
García-Robledo, C. et al. Limited tolerance by insects to high temperatures across tropical elevational gradients and the implications of global warming for extinction. PNAS 113, 680–685 (2016).
Nondillo, A. et al. Exigências térmicas e estimativa do número de gerações anuais de Frankliniella occidentalis (Pergande) (Thysanoptera: Thripidae) em morangueiro. Neotrop. Entomol. 37, 646–650 (2008).
Huang, Z., Ren, S. & Musa, P. D. Effects of temperature on development, survival, longevity, and fecundity of Bemisia tabaci Gennadius (Homoptera: Aleyrodidae) predator, Axinoscymnus cardilobus (Coleoptera: Coccinellidae). Biol. Control 46, 209–215 (2008).
Shi, S. S., Cui, J. & Zang, L. S. Development, survival, and reproduction of Megacopta cribraria (Heteroptera: Plataspidae) at different constant temperatures. J. Econ. Entomol. 107, 2061–2066 (2014).
Jia, H. et al. Effects of temperature on the growth, development and reproduction of Jakowleffia setulosa (Hemiptera: Lygaeidae). Acta. Entomol. Sin. 57, 935–942 (2014).
Ramalho, F. S. et al. Temperature-dependent fecundity and life table of the fennel aphid Hyadaphis foeniculi (Passerini) (Hemiptera: Aphididae). PLoS ONE 10, e0122490 (2015).
San Choi, K. & Kim, D. S. Effect of temperature on the fecundity and longevity of Ascotis selenaria (Lepidoptera: Geometridae): Developing an oviposition model. J. Econ. Entomol. 109, 1267–1272 (2016).
Angilletta, M. J. & Dunham, J. R. The temperature–size rule in ectotherms: simple evolutionary explanations may not be general. Am. Nat. 162, 332–342 (2003).
Barbosa, L. R. et al. Criação massal do percevejo bronzeado, Thaumastocoris peregrinus Carpinteiro & Dellapé, 2006 (Hemiptera: Thaumastocoridae). Embrapa, Brasília, Brasil, https://doi.org/10.13140/RG.2.2.36424.11528 [In Portuguese] (2016).
Carpintero, D. L. & Dellapé, P. M. A new species of Thaumastocoris Kirkaldy from Argentina (Heteroptera: Thaumastocoridae: Thaumastocorinae). Zootaxa 1228, 61–68 (2006).
Haddad, M. L., Parra, J. R. P. & Moraes, R. C. B. Métodos para estimar os limites térmicos inferior e superior de desenvolvimento de insetos. Piracicaba, FEALQ. 29p (1999).
Campbell, A. et al. Temperature requirements of some aphids and their parasites. J. Appl. Ecol. 11, 431–438 (1974).
Southwood, T. R. E. 1978. Ecological methods: with particular reference to the study of insect populations (2nd ed. Chapman & Hall, New York, NY) 524 (2014).
Hothorn, T., Bretz, F. & Westfall, P. Simultaneous inference in general parametric models. Biom. J. 50, 346–363 (2008).
Wilson K., Hardy I. C. W. (In press) Analysis of sex ratios and other proportional data. In: HardyICW (ed.) Sex ratio handbook. Cambridge University Press, Cambridge, UK (2002).
Girondot, M. How to work correctly statistically about sex ratio (Version of 12th April 2014) 27 (2014).
SAS, The SAS System for Windows, Release 9.0. SAS Institute, Cary, N. C. (2000).
Therneau T. A package for survival analysis in S. R package version 2.38, http://CRAN.R-project.org/package=survival (2015).
Kassambara A., Kosinski M. & Biecek P. R package version 0.3. survminer: drawing survival curves using ggplot2; p. 1 (2017).
Acknowledgements
To Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG) and“ Programa Cooperativo sobre Proteção Florestal/PROTEF do Instituto de Pesquisas e Estudos Florestais/IPEF” for financial support.
Author information
Authors and Affiliations
Contributions
L.R.B., F.S., A.P.R. and E.P.S. designed the research; L.R.B., F.S., E.P.S., A.P.R. and J.M.C. performed the experiments; A.J.V.Z., C.F.W., F.S., J.C.Z. and L.R.B., analyzed the data, A.J.V.Z., C.F.W., F.S., J.C.Z., J.M.C. and L.R.B., wrote and approved the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Barbosa, L.R., Santos, F., Soliman, E.P. et al. Biological parameters, life table and thermal requirements of Thaumastocoris peregrinus (Heteroptera: Thaumastocoridae) at different temperatures. Sci Rep 9, 10174 (2019). https://doi.org/10.1038/s41598-019-45663-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45663-5
- Springer Nature Limited
This article is cited by
-
Hydrogel as an alternative to agar for laboratory rearing of the green peach aphid Myzus persicae
CABI Agriculture and Bioscience (2023)
-
Biology and predation potential of the hemipteran predator, Rhynocoris marginatus (Fab., 1794) on the fall armyworm, Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae)
Egyptian Journal of Biological Pest Control (2022)
-
Thermal requirements and estimates of the annual number of generations of Heliothrips haemorrhoidalis in strawberry-producing regions of Brazil
Phytoparasitica (2022)
-
Development and fertility parameters of a predatory bug, Dortus primarius Distant (Miridae: Deraeocorinae) at different temperatures
International Journal of Tropical Insect Science (2022)
-
Demographic parameters of the reduviid predator, Rhynocoris marginatus (Reduviidae: Hemiptera) fed on two lepidopterous insect pests
BioControl (2021)