Abstract
The neurotransmitter dopamine has central roles in mood, appetite, arousal and movement1. Despite its importance in brain physiology and function, and as a target for illicit and therapeutic drugs, the human dopamine transporter (hDAT) and mechanisms by which it is inhibited by small molecules and Zn2+ are without a high-resolution structural context. Here we determine the structure of hDAT in a tripartite complex with the competitive inhibitor and cocaine analogue, (–)-2-β-carbomethoxy-3-β-(4-fluorophenyl)tropane2 (β-CFT), the non-competitive inhibitor MRS72923 and Zn2+ (ref. 4). We show how β-CFT occupies the central site, approximately halfway across the membrane, stabilizing the transporter in an outward-open conformation. MRS7292 binds to a structurally uncharacterized allosteric site, adjacent to the extracellular vestibule, sequestered underneath the extracellular loop 4 (EL4) and adjacent to transmembrane helix 1b (TM1b), acting as a wedge, precluding movement of TM1b and closure of the extracellular gate. A Zn2+ ion further stabilizes the outward-facing conformation by coupling EL4 to EL2, TM7 and TM8, thus providing specific insights into how Zn2+ restrains the movement of EL4 relative to EL2 and inhibits transport activity.
Similar content being viewed by others
Main
Dopamine and the dopaminergic circuits in the brain are intimately involved in mood, reward, motivation and movement5. Outside the brain, dopamine participates in signalling in the eye, cardiovascular system and pancreas6. Within the central nervous system, dopamine is produced by a small number of neurons located in the midbrain that project throughout the brain, acting as vehicles of dopamine release to diverse regions, including the striatum, limbic system and neocortex7, thus explaining the profound effect of dopaminergic signalling on brain function. Dysfunction of dopaminergic signalling underpins Parkinson’s disease8 and multiple psychological disorders9, and illicit and therapeutic drugs, including medications used to treat attention deficit hyperactivity disorder, modulate dopaminergic signal transduction10. Widely used therapeutic or illicit drugs, such as methylphenidate, amphetamines or cocaine, target the human dopamine transporter (hDAT), perturbing or inhibiting dopamine transport and thus disrupting dopaminergic signalling11.
The hDAT is a member of the neurotransmitter sodium symporter (NSS) family of transporters, which in turn belong to the larger family of SLC6 transporters12, integral membrane proteins that harness ion gradients to achieve concentrative reuptake of small molecules by way of an alternating access mechanism13. The hDAT uses Na+ and Cl− gradients to enable substrate uptake, with K+ promoting the return of the transporter to the extracellular-facing conformation, following unbinding of substrate and ions within the cytoplasm14. The activity of hDAT is distinct from its biogenic amine transporter relatives, however, in that transport activity is inhibited by physiologically related levels4 of Zn2+, which is co-released with neurotransmitters15, as well as by synthetic small molecules, such as KM822 and MRS7292, which target largely uncharacterized, allosteric site(s)3,16,17. Although studies on a transport-inactive Drosophila dopamine transporter (dDAT) illuminated its overall structure and the mechanism by which substrates and inhibitors bind to the central site18,19,20, the molecular structure of functionally active hDAT and the mechanisms of small molecules and ions acting on allosteric sites, and at the central site, remain unresolved. Here we define the binding site and non-competitive inhibition mechanism of MRS72923, elaborate a structure-based mechanism for Zn2+ modulation of transport, and map the binding site of β-CFT, a high-affinity cocaine analogue.
Overall architecture of ∆-hDAT complex
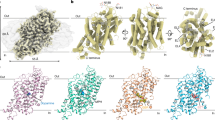
To facilitate expression and purification, we removed 56 residues from the N terminus that are predicted to be unstructured, and used the point mutant I248Y, which provided modest thermostability21, together yielding the Δ-hDAT construct. Δ-hDAT exhibits dopamine transport (Fig. 1a) and [3H]WIN35428 binding (Fig. 1b) activities similar to the full-length, wild-type transporter22,23. Following expression in mammalian cells, detergent solubilization and purification in the presence of MRS7292 and β-CFT, we obtained monodisperse and homogenous Δ-hDAT (Extended Data Fig. 1a,b) for cryo-electron microscopy (cryo-EM) grid preparation. Inclusion of both MRS7292 and β-CFT yields a highly stable complex, facilitating transporter isolation and single-particle cryo-EM studies21. Collection of a large single-particle cryo-EM dataset and extensive image processing (Extended Data Fig. 2a), which included ab initio-based 3D classification followed by non-uniform refinement, ultimately yielded a cryo-EM reconstruction of Δ-hDAT at 3.19 Å (Extended Data Fig. 2b–e). The resulting density map allowed for fitting of nearly the entire polypeptide chain, the placement of most side chains (Extended Data Fig. 3), the positioning of bound ligands, a Zn2+ and a Na+ ion, as well as the definition of multiple detergent or lipid molecules. Both in the single-particle classifications and in the biochemical analysis of the transporter, we observe detergent-solubilized Δ-hDAT as a monomer, although many previous studies find that dopamine transporters and related NSSs exist as dimers or higher-ordered oligomers24. Further experiments are required to understand how to retain oligomeric species upon membrane solubilization.
a, Saturation uptake of [3H]dopamine in HEK293 GnTI− cells expressing ∆-hDAT (black) and full-length hDAT (blue). Uptake in the presence of 10 µM MRS7292 is shown in orange and pink for ∆-hDAT and full-length hDAT, respectively. The Michaelis constant (Km) values for [3H]dopamine uptake by ∆-hDAT and full-length hDAT were 0.55 ± 0.07 and 0.56 ± 0.13 μM, and reaction rate at infinite substrate concentration (Vmax) values were 342.8 ± 11.7 and 190.9 ± 10.7 fmol min−1 per well, respectively. Data were analysed using a Michaelis–Menten kinetics model. The uptake assay was performed in n = 3 biological replicates, with each in technical triplicate. Data are mean ± s.d. b, Scintillation proximity assay (SPA) using [3H]WIN35428 and purified His-tagged ∆-hDAT. The dissociation constant (Kd) for [3H]WIN35428 binding by ∆-hDAT was 6.5 ± 0.91 nM. Data are mean ± s.d. Assays were done in n = 3 independent replicates, each with technical triplicates. c, Structure of ∆-hDAT showing β-CFT in the central site and MRS7292 in the allosteric site. NAG represents an N-acetylglucosamine modification at N188. d, Slab view of ∆-hDAT in a surface representation showing how the transporter adopts an outward-open conformation. e, Chemical structure of β-CFT (prepared using ChemDraw 18.2). f, Density associated with β-CFT, contoured at 10σ, within 2 Å of the ligand atoms. g, Close-up representation of β-CFT bound to the central site. Hydrogen-bonding interactions are shown as black, dashed lines.
The overall structure of Δ-hDAT adheres to the canonical LeuT fold25, a conserved architecture among the SLC6 transporters, with transmembrane helices TM1–5 related to TM6–10 by a pseudo two-fold axis of symmetry, aligned approximately parallel to the membrane (Fig. 1c). The Δ-hDAT structure resolved here, bound with multiple inhibitory small molecules and ions, adopts an outward-open conformation (Fig. 1d) where the central ligand binding site, also known as the S1 site, is accessible to bulk solvent via the extracellular vestibule. In accord with the outward-open conformation, the distance between two conserved residues of the extracellular gate, F320 on TM6b and Y156 on TM3, is approximately 13 Å. The cytoplasmic gate is closed, consistent with an outward-open state, with TM1a residing within the protein core (Fig. 1c), interacting extensively with TM5, TM6b and TM7. The C-terminal ‘latch’, which caps the cytoplasmic face of the transporter, is—to our knowledge—the most extensive cytoplasmic motif observed to date in an NSS (Extended Data Fig. 4a), and includes three short C-terminal helices (CT1, CT2 and CT3) that cover the cytoplasmic termini of TM3, TM10 and TM12 (Extended Data Fig. 4b), further stabilizing the closed conformation of the cytoplasmic gate.
The activity of hDAT and related NSSs is modulated by lipids and cholesterol26,27,28 and, accordingly, we find multiple lipid or lipid-like density features that we have modelled as either linear alkyl chains or cholesteryl hemisuccinate (CHS) molecules (Extended Data Fig. 5a). We observe densities consistent with either CHS or cholesterol in the Δ-hDAT structure that are equivalent to sites in dDAT18,19 (Extended Data Fig. 5b) and near to a cholesterol site in the human serotonin transporter29 (hSERT) (Extended Data Fig. 5c). We also find density for CHS at an additional site, in a groove formed by TM4, TM5 and TM8 (Extended Data Fig. 5d). Covalent modification of extracellular-exposed surfaces, by way of N-linked glycosylation at N181, N188 and N205, all on EL2, confers maximal transport activity upon hDAT and, when ablated, alters the potency of cocaine-like drugs30. For the Δ-hDAT expressed in GnTI− cells, a line that yields core N-linked carbohydrate similar to HEK293 cells31, we observe prominent density for glycosylation at N188, whereas density for modification at N181 and N205 is too weak to model.
Central site pharmacology
There is a high degree of amino acid sequence conservation between the monoamine transporters (MATs)—hDAT, human noradrenaline transporter (hNET) and hSERT. Nevertheless, decades of pharmacological studies have led to the development of transporter-selective small molecule inhibitors that bind to the central site. β-CFT (Fig. 1e,f), which is used in the present structure determination, has modest selectivity for binding to hDAT over hNET and hSERT32, whereas reboxetine prefers hNET over hDAT and hSERT33, a selectivity that is conferred by residues both within and outside of the central site34. The classic selective serotonin reuptake inhibitor S-citalopram shows a strong preference for binding to hSERT over hDAT and hNET35. Inspection of the complex of β-CFT with Δ-hDAT enables us to visualize key interactions between the transporter and ligand and to define the overall transporter conformation, thus providing information on how residues within the binding site may sculpt transporter selectivity. The role(s) of residues outside of the central binding site in modulating ligand selectivity will require further investigation.
β-CFT occupies the central site of Δ-hDAT (Fig. 1g), consistent with its action as a competitive inhibitor of dopamine uptake36. Using the ‘A, B and C’ representation of the central site37, the tropane moiety of β-CFT is positioned toward subsite A, facing D79 and A81 on TM1b, F76 on TM1a and G323 on TM6. The fluorophenyl moiety of β-CFT is in subsite B, near residues on TM3, TM6 and TM8. V152, S422 and Y156 participate in van der Waals contacts, and F326 on the TM6a–TM6b linker forms an edge-to-face contact with the phenyl ring of the fluorophenyl group (Fig. 1g). Akin to the dDAT–β-CFT complex19, the carbomethoxy group protrudes toward the base of the extracellular gate25,38, yet does not disturb the critical hydrogen-bonding interaction between Y156 and D7939,40. Subsite B residues M427 and G153, when introduced into the corresponding positions in dDAT, enhance β-CFT binding19 and, in hSERT, these same residues are leucine and alanine, respectively29,41. These differences in subsite B residue composition may contribute to the selectivity of Δ-hDAT for β-CFT.
Subsite C residues in MATs are involved in accommodating chemical moieties of bulky inhibitor molecules, as seen in the S-citalopram–hSERT complex29, in which T497 and V501 provide a mixed polar and non-polar surface for accommodating the cyano group of S-citalopram (Extended Data Fig. 6a). T497 is an alanine in both hDAT and hNET; thus, differential residue composition in subsite C can explain in part the selectivity of S-citalopram for hSERT. The superposition of β-CFT-bound ∆-hDAT with the cocaine-bound, native, porcine SERT (pSERT) (Protein Data Bank (PDB): 8DE342) and β-CFT-bound dDAT (PDB: 4XPG) structures shows overall α-carbon root mean-square deviation (r.m.s.d.) values of 1.05 and 1.08 Å, respectively, with the central site being well superimposed (Extended Data Fig. 6b). The central site in Δ-hDAT is solvent-accessible via the extracellular vestibule, as a consequence of the swung-out position of F320 (Extended Data Fig. 6c), similar to the dDAT complex with β-CFT19. Of note, in the complex of pSERT with the β-CFT analogue cocaine42 (PDB: 8DE3), the side chain of the equivalent phenylalanine residue is swung-in, covering the central site and occluding the ligand from the extracellular solution (Extended Data Fig. 6c).
Molecular dynamics simulations and analysis further support the binding pose of β-CFT at the central site in the ∆-hDAT cryo-EM structure (Supplementary Fig. 2). Analysis of the fluctuation of β-CFT moieties revealed more stable tropane and fluorophenyl groups compared to the greater fluctuations and solvent exposure for the carbomethoxy group (Supplementary Fig. 3). The per-residue contact analysis (Supplementary Fig. 4) revealed that residues F76, A77, D79, A81, F320 and G323 from subsite A frequently interact with the tropane moiety, and residues V152, G153, Y156, F326, S422, A423 and G426 interact with the fluorophenyl moiety, which probably aids in stabilizing β-CFT. Additionally, consistent hydrogen bonding (Supplementary Fig. 5a) between the acidic side chain of D79 and the tropane moiety (via the tropane nitrogen atom) and comparison of each simulated binding mode with the cryo-EM model (Supplementary Fig. 6), as well as the measurement of the in silico density of spatial sampling of β-CFT (Supplementary Fig. 7) support stable binding across all simulated replicas (Extended Data Fig. 7d).
A density feature adjacent to β-CFT, and indicative of a bound ion, parallels the Na2 site in LeuT43, dDAT18, and hSERT29, suggesting the presence of a similar Na+-coordinating site in ∆-hDAT (Extended Data Fig. 7e). The Na+ ion is coordinated with the main chain oxygens of V78 and L418 on TM1 and TM8, respectively, and side chains of D421 and S422 on TM8, with a mean coordination distance of 2.3 Å.
MRS7292 sculpts an allosteric site
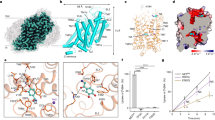
The N-methanocarba nucleoside analogue MRS7292 slows the unbinding of the central site ligand β-CFT21, acts as a non-competitive inhibitor of dopamine transport, and has a chemical structure unlike previously characterized hDAT ligands3,16. To understand how MRS7292 inhibits dopamine transport and slows the dissociation of ligands from the central site, we determined the structure of Δ-hDAT in complex with MRS7292 and β-CFT (Fig. 2a). The density is located underneath EL4, adjacent to TM1b, and about 13 Å above the central binding site, where β-CFT is found (Fig. 2b,c). In accord with the largely non-polar character of MRS7292, the associated binding pocket is primarily hydrophobic and lined by aromatic and aliphatic amino acid residues (Fig. 2a). The MRS7292-binding site is spatially distinct from the hSERT allosteric site (Extended Data Fig. 7a) defined by the binding of citalopram29, vilazodone44 or the allosterically bound serotonin molecule41. The allosteric site ligands in hSERT and Δ-hDAT are at least 13 Å distant from the central site, consistent with the conclusion that the allosteric ligands do not directly contact the ligand bound to the central site.
a, MRS7292 binds underneath EL4 and adjacent to TM1b, in a hydrophobic pocket additionally defined by TM7. Key residues in the MRS site are indicated in stick representation with the carbon atoms of MRS7292 in orange. Subsites I, II and III are shown. b, Chemical structure of MRS7292 (prepared using ChemDraw 18.2). c, Density associated with MRS7292, contoured at 12σ within 2 Å of the ligand atoms. d, Effect of MRS7292 on [3H]dopamine uptake in ∆-hDAT and ∆-hDAT mutants. Data were analysed using nonlinear regression (Methods). IC50 measurements were performed in n = 3 biological replicates (each in technical triplicate). Data are mean ± s.d. e, Superposition of the MRS site in ∆-hDAT and the equivalent site in hSERT (PDB: 7LIA) using α-carbon atoms of TM3 and TM8. f, Close-up view of the MRS site showing the cryo-EM model and the final conformations of the simulation replicas.
The MRS7292 compound is inserted deeply into Δ-hDAT with only a small amount of surface area exposed to the solution (Fig. 2a). Buried underneath EL4 and sandwiched between TM1b and a short helix of EL4a, the MRS7292-binding site (MRS site) can be divided into three subsites: the ring clasp (I), the adenosine sandwich (II) and the thienyl anchor (III) (Fig. 2a). The hydroxyl-decorated, rigid methanocarba ring is in subsite I, clasped underneath the turn in EL4, making extensive van der Waals interactions. The terminal alkyl group of the methyl ester is in close proximity to a hydrophobic groove formed by I159 and W162 on TM3, F391 on EL4b and F472 on TM10 (Extended Data Fig. 7b). The carbonyl oxygen of the methyl ester may interact with D476 via a water-mediated hydrogen bond, leaving the secondary hydroxyl groups as the major solvent-exposed portions of the ligand.
Gripping the MRS7292 ligand is a sandwich-like interaction between the adenine group and the indole moiety of W84 on one side, and the polypeptide main chain of residues 388–389 on the other side (Fig. 2a). Testament to the crucial role of W84 in subsite II, mutation to alanine or cysteine severely compromises the potency of MRS7292 (Extended Data Fig. 7c,d). By contrast, the W84C mutant increases the potency of KM822, a small molecule inhibitor of hDAT that is structurally distinct from MRS729217, thus suggesting distinction in binding site(s) between MRS7292 and KM822. Near the indole NH group of W84, and possibly interacting with the N1 nitrogen of MRS7292 via a water molecule, is D385. Consistent with the importance of the aspartate, mutation to an alanine leads to almost 50% reduction in MRS7292-mediated inhibition of dopamine transport (Fig. 2d). Substitution of D385 by an asparagine retained potency of MRS7292 as evident by a similar inhibition curve to ∆-hDAT (Fig. 2d). This suggests that asparagine is able to form a water-mediated hydrogen bond with the N1 of MRS7292 like its aspartate counterpart in the parental ∆-hDAT. Two hydrogen bonds augment the sandwich of subsite II, one involving the exocyclic NH group of the adenine moiety and the main chain carbonyl oxygen of K384 (EL4) and the second between the hydroxyl of Y394 and the N5 atom on the adenine ring. The latter interaction is important because substitution of the N5 with a CH yields a compound that no longer inhibits hDAT transport activity16. The alkyne group links the adenine and thienyl moieties, in an axle-like fashion, and is surrounded by L80, V364, I390 and Y394, which together act like a bushing for the linear, carbon-carbon triple bond. In accord with the relevance of Y394, we find that substitution of Y to F diminishes the potency of MRS7292, thus substantiating the importance of the hydrogen bond between the hydroxyl group of Y394 and the N5 atom of the MRS7292 adenine ring (Extended Data Fig. 7c,d).
The 5-chlorothien-2-yl entity is deeply buried in subsite III, surrounded by a constellation of non-polar amino acids that include L280, V364, Y394, F411 and M414 (Extended Data Fig. 7e). The aromatic residues participate in edge-to-face interactions with the five-membered thienyl ring, while the aliphatic amino acids supply van der Waals contacts. M414 has a relatively flexible yet non-polar side chain, and to probe its role in the interactions with the thienyl moiety, we prepared the ∆-hDAT(M414L) variant. Notably, the M414L mutant showed an enhanced potency for MRS7292, as evidenced by an approximately threefold decrease in the half-maximal inhibitory concentration (IC50) value compared with ∆-hDAT (Fig. 2d and Extended Data Fig. 7d). We speculate that the smaller leucine side chain creates a larger pocket to accommodate the thienyl moiety, thereby reducing steric hindrance and enhancing MRS7292-mediated inhibition of dopamine transport. Most of the residues interacting with MRS7292 are conserved in related NSSs yet the hDAT orthologues, including hNET and hSERT, are less sensitive to inhibition of uptake activity by MRS729219. Of the non-conserved residues, V364 is an isoleucine in hNET and hSERT (Fig. 2e). Surprisingly, mutation of V364 to isoleucine enhanced apparent MRS7292 affinity, resulting in a nearly fourfold decrease in the IC50 value compared with the wild-type-like parent (Fig. 2d and Extended Data Fig. 7d). To mimic a more hSERT-like MRS7292-binding pocket in Δ-hDAT, we generated a double mutant with an additional substitution of I390 to leucine in the V364I background. The double mutant showed enhanced potency towards MRS7292 with a corresponding approximately threefold decrease in IC50 value, similar to the V364I mutant. However, I390L alone resulted in a modest increase in the IC50 value, indicated by the marginal shift of the curve compared with ∆-hDAT. Taken together, and reminiscent of the determinants of central site ligand selectivity, the mechanism by which MRS7292 exhibits selectivity for ∆-hDAT over hNET and hSERT is partially dependent upon residues outside of the immediate binding site.
Molecular dynamics-based r.m.s.d. analysis showed stable positioning of MRS7292 across five simulation replicas (Supplementary Fig. 2), with marginal atomic fluctuations (Fig. 2f). The hydroxyl-decorated methanocarba (pseudo-ribose) moiety exhibited slightly higher fluctuations compared to the adenine and thienyl rings (Supplementary Fig. 3). The radial distribution function shows solvent exposure for the pseudo-ribose moiety of MRS7292 (Supplementary Fig. 3). Furthermore, analysis of the per-residue contact probability (Supplementary Fig. 4) showed that the 15 highest coordinating residues from Δ-hDAT stabilized MRS7292 within its cryo-EM pose. Moreover, the adenine ring of MRS7292 and the indole ring of W84 maintained a stacking-like interaction more than 99% of the time (Supplementary Table 1 and Supplementary Figs. 4 and 8). Hydrogen bond analysis (Supplementary Figs. 4 and 5) indicated stable interactions with key groups, such as the exocyclic NH group (N2 atom) of the adenine ring and the backbone carbonyl oxygen (O) of K384 (in EL4), as well as the hydroxyl group of Y394 and the N5 atom of the adenine ring. Additionally, the five-membered thienyl ring exhibited stabilization through hydrophobic interactions with L280, V363, V364, F411 and M414. Overlay of the MRS7292-binding site from all the simulation replicas with the cryo-EM pose is shown in Supplementary Fig. 9.
MRS7292 binds to Δ-hDAT by way of an induced-fit mechanism. Although we do not yet have a structure of Δ-hDAT in the absence of MRS7292, and thus cannot visualize its binding site in an apo state, by comparing the MRS site of Δ-hDAT to the equivalent region of a closely related hSERT structure (PDB: 7LIA), we speculate that the binding of MRS7292 to Δ-hDAT results in substantial conformational changes. Compared with hSERT, we estimate that MRS7292 binding displaces TM1b and TM6a by 1.9 and 2.1 Å and leads to their reorientations by 3.1 and 3.8°, respectively (Extended Data Fig. 7f). Compared with dDAT, TM1b and TM6a are displaced by 2.6 and 2.8 Å and reoriented by 9.0 and 3.2°, respectively (Extended Data Fig. 7g). We suggest that MRS7292 binding also readjusts the conformation of EL4a, as well as the turn between EL4a and EL4b, to sculpt the polypeptide chain for optimal interactions with the N-methanocarba and adenine rings of MRS7292.
In the context of MATs, the allosteric site of hSERT has been the most well characterized, beginning from when it was first suggested by ligand-unbinding studies to more recently, when it has been structurally defined in complexes of hSERT with inhibitors, including S-citalopram29 and vilazodone44, and with the substrate serotonin41. Comparison of the location of the allosteric site in hSERT to the allosteric ‘MRS site’ in Δ-hDAT shows that they are entirely distinct locations on extracellular-facing regions of the transporters (Extended Data Fig. 7a). Whereas the allosteric site in hSERT is largely formed by residues from TM10, TM11 and TM12, including the di-proline motif (P560-P561) in EL6 and P499 on TM10, the allosteric site in Δ-hDAT largely involves EL4 and TM1b, together with residues from TM5, TM7 and TM8. Further inspection of the region of Δ-hDAT that is equivalent to the allosteric site in hSERT provides a structural explanation for why Δ-hDAT is not sensitive to the same allosteric ligand as hSERT. As examples, structural superposition shows that P561 in hSERT is substituted by an arginine in hDAT, and the equivalent proline residue in the di-proline motif (P546) in Δ-hDAT is situated at a Cα–Cα distance of 5.9 Å from hSERT P561 (Extended Data Fig. 8a). Furthermore, P499 in hSERT is not conserved in Δ-hDAT and is substituted by T482. Similarly, the non-polar pocket formed by TM6a, TM10 and TM11 that accommodates the fluorophenyl moiety of S-citalopram in hSERT is distinct in comparison to Δ-hDAT (Extended Data Fig. 8b). The equivalent region in Δ-hDAT is more polar in nature with T316, T482, S539 in place of A331, P499 and F556, respectively, in Δ-hDAT and hSERT. Taken together, although Δ-hDAT retains the overall structural motif of the hSERT allosteric site, differences in amino acid composition mean that hSERT allosteric ligands probably do not bind to Δ-hDAT. Nevertheless, the site may still be a target for suitably tailored small molecules.
Zinc restrains extracellular loops
Zn2+ is packaged in vesicles and released upon vesicle fusion with the presynaptic membrane45, modulating the activities of synaptic neurotransmitter receptors and transporters. Since the discovery of Zn2+ inhibition of dopamine transport decades ago, several key residues involved in Zn2+ binding have been identified4. In the absence of a high-resolution structure, proposed mechanisms of inhibition have been developed through analysis of hDAT topology, mutagenesis studies and computational modelling4,46. A previous study showed that Zn2+ coordination by H193, H375 and E396 inhibits translocation of dopamine while potentiating WIN35428 binding at the central site by restraining EL2 and EL447. In agreement with these findings, examination of the Δ-hDAT cryo-EM density map shows the presence of nearly continuous density between a cluster of histidine residues, a glutamate, and the aspartate D191 on EL2 and EL4, suggestive of the presence of a bound ion. Although we have not supplemented the buffers with Zn2+ salts or ions during Δ-hDAT purification, elemental analysis of the purified Δ-hDAT protein revealed the presence of around 3.9 μM of Zn2+, probably from the lysed cells or from the cell growth medium, the latter of which contains about 4.1 µM Zn2+. Fitting of a Zn2+ ion to the density yielded reasonable coordination geometry (Fig. 3a and Extended Data Fig. 9a). The Zn2+ ion is coordinated by H193 on EL2 and H375 at the juncture of EL4a and TM7, with Zn2+-to-nitrogen interaction distances of 2.3 Å (Fig. 3a). E396 on EL4b defines a third ligand to the Zn2+ ion with an interaction distance of 2.1 Å (Fig. 3a). D191 was previously proposed to stabilize Zn2+ coordination through hydrogen bonding with H193, and mutation of this residue to an asparagine resulted in an apparent threefold decrease in Zn2+ affinity47; however, our cryo-EM density map shows that D191 is in close proximity to the Zn2+ site, with a carboxylate oxygen to Zn2+ distance of 2.3 Å (Fig. 3a). Analysis of the binding site geometry is further consistent with a bound Zn2+ ion. A computational modelling study proposed D206 on EL2 as a fourth Zn2+-coordinating residue46. However, in the current structure of Δ-hDAT, D206 is nearly 15 Å from the Zn2+ site.
a, Location and close-up view of the Zn2+ site showing the multivalent coordination of the divalent ion. The Zn–O and Zn–N distances are expressed in Å. b, Superposition of the Zn2+ site in ∆-hDAT and a predicted model of ∆-hDAT in an inward-open state showing the displacement of H375 on EL4 by 4.2 Å between the outward-open to inward-open conformations. TM3 and TM8 were aligned using α-carbon atoms. c, Characterization of [3H]dopamine uptake in HEK293 GnTI− cells expressing ∆-hDAT and the mutants T211E and T211H in the presence of Zn2+. Data were analysed using a nonlinear regression model (Methods). The experiments were performed in n = 3 biological replicates with each in technical triplicate. Data are mean ± s.d.
Mutation of the residues involved in Zn2+ coordination or the introduction of the same residues at equivalent sites in related NSSs (Extended Data Fig. 9b) ablates or introduces Zn2+ sensitivity, respectively4. Thus, when H193 is mutated to lysine, the capacity for Zn2+ to inhibit transport is compromised4. Similarly, when a histidine is introduced at the equivalent site in the Zn2+-insensitive hNET, the resulting K189H variant of hNET becomes sensitive to Zn2+ inhibition of noradrenaline transport4. hSERT is not sensitive to Zn2+, probably because the residues equivalent to H193 and H375 are phenylalanine and arginine, respectively, in hSERT (Extended Data Fig. 9b).
Zn2+ coordination in Δ-hDAT restrains EL2 in a distinct conformation compared with members of the related NSS transporters hSERT, GlyT1 and GAT1 (Extended Data Fig. 9c–e). Structures of hSERT through different states of its transport cycle have revealed that movement of EL4 relative to EL2 accompanies the transition from outward-open to inward-facing conformations42. By analysing the structure of Δ-hDAT in the context of the transport mechanism of hSERT, we speculate that coordination of Zn2+ inhibits transport by restricting movement of EL4, thus preventing the conformational change from the outward-open to the inward-facing state (Fig. 3b).
Further inspection of the Zn2+ site revealed a nearby residue, T211 (Fig. 3a), that itself is not within coordination distance of the Zn2+ but, we hypothesized, when mutated to either a glutamate or histidine, the respective carboxyl or imidazole groups would be close enough to interact favourably with the ion (Extended Data Fig. 9f,g). To increase Zn2+ potency in uptake experiments, we conducted assays at pH 8.5, thus favouring the deprotonated state of the coordinating histidine residues. Indeed, we found that the IC50 values for Zn2+ in the T211E and T211H mutants were around 100-fold and 20-fold lower, respectively, than that of ∆-hDAT (Fig. 3c). The high sensitivity of T211E to Zn2+ made it difficult to obtain a full inhibition curve under the same conditions used for ∆-hDAT and T211H, and for this reason, a second set of IC50 measurements was taken at pH 7.5 for the T211E mutant (Extended Data Fig. 9h). We suggest that the two mutants bind Zn2+ with higher affinity than Δ-hDAT, thus bolstering the identification of the Zn2+ site.
Conclusion
Despite the overarching role of dopamine and dopaminergic signals in brain development, function and disease, and the importance of drugs in modulating the activity of hDAT, a structural understanding of transporter mechanism and allosteric inhibition has proved elusive. By elucidating the structure of Δ-hDAT bound with a trifecta of antagonistic agents, we show how β-CFT occupies the central binding site, arresting the transporter in an outward-open conformation, adjacent to a sodium ion bound at the Na2 site. The allosteric inhibitor, MRS7292, binds above the central site and underneath EL4, immediately adjacent to TM1b, via an induced-fit mechanism, occupying a binding pocket that is not present in the closely related hSERT protein. Binding of MRS7292 displaces TM1b toward TM6a, and we speculate that the allosteric ligand locks these key helices in place, together with EL4, thus preventing isomerization of the transporter to an inward-facing state. Although mutants of residues in contact with MRS7292 reduce potency of MRS7292, swapping of non-conserved residues between hSERT and hDAT suggests that amino acids outside of immediate contact with MRS7292 also confer selective binding of MRS7292 to hDAT. A Zn2+ ion occupies a binding site immediately above the MRS7292 ligand, coordinated by residues on both EL2 and EL4. By tethering EL4 to EL2, the bound Zn2+ ion may restrict movement of EL4 upon transporter rearrangement to inward-facing conformations, thus providing insight into how Zn2+ inhibits transport activity (Fig. 4). All together, these bound agents restrict the conformational mobility of Δ-hDAT, preventing isomerization to occluded or inward-facing states, and more generally, they provide fresh insights into how small molecules and ions can modulate structure and activity of MATs.
β-CFT binds at the central or S1 site, stabilizing the outward-open conformation of ∆-hDAT. Binding of MRS7292 at the allosteric binding pocket near TM1b and below EL4 leads to a conformational change in TM1b, TM6 and EL4 that precludes their movement, further stabilizing the outward-open conformation and enhancing β-CFT binding at the central site. Binding of Zn2+ restrains the EL4 loop, precluding its movement upon transition to the inward-open conformation, thus inhibiting transport.
Methods
Plasmid and constructs
The hDAT cDNA (UniProt ID Q01959) was cloned into the pEG-BacMam vector48 with an N-terminal His-StrepII–eGFP tag and a 3C protease site (LEVLFQGP) between the eGFP tag and the start of the hDAT protein-coding sequence. This construct also harboured an N-terminal deletion of the proteolytically labile 56 amino acids and included a thermostabilizing mutation (I248Y) as previously described21 and will be referred to as Δ-hDAT throughout. Point mutations were introduced using site-directed mutagenesis verified by DNA sequencing.
Protein expression and purification
Baculovirus-mediated expression of Δ-hDAT was performed following standard protocol48, as previously described21, with minor modifications. In brief, HEK293 GnTI− (Ric-15) cells31 at a density of 3.5 to 4.0 × 106 cells per ml were transduced with Δ-hDAT P2 virus at a multiplicity of infection of 2.5 and cultured in Erlenmeyer flasks at 37 °C with 8% CO2 for 12 h, followed by the addition of 10 mM sodium butyrate48. Subsequently, the transduced cultures were shifted to 30 °C and incubated for a total of 48 h. The cells were collected by centrifugation at 4,000 rpm for 15 min (TX 1000 rotor, Thermo Scientific), washed in ice-cold phosphate buffered saline, flash frozen in liquid nitrogen, and stored at −80 °C until further use. Cell pellets were thawed on ice and resuspended in a resuspension buffer composed of 50 mM HEPES pH 7.8, 200 mM NaCl, and 20% glycerol supplemented with 0.8 μM aprotonin, 2 μg ml−1 leupeptin, 2 μM pepstatin A and 1 mM phenylmethylsulfonyl fluoride (PMSF). The cells were lysed by sonication and centrifuged at 40,000 rpm for 60 min (Type 45 Ti rotor) to pellet the membrane fraction. Membrane pellets were resuspended using a Dounce homogenizer in resuspension buffer in which the glycerol concentration was raised to 30%, flash frozen, and stored at −80 °C. All centrifugation steps were carried out at 4 °C, unless otherwise stated.
Frozen membranes from 4.8 l of culture were thawed on ice and solubilized in a solution of 10 mM lauryl maltose neopentyl glycol (LMNG), 2 mM CHS, 50 mM HEPES pH 7.8, 200 mM NaCl, 10 μM MRS7292, 2 μM β-CFT, 0.8 μM aprotonin, 2 μg ml−1 leupeptin, 2 μM pepstatin A, and 1 mM PMSF by constant stirring for about 3 h at 4 °C. The resulting solution was clarified by ultracentrifugation at 40,000 rpm for 60 min (Type 45 Ti rotor). Meanwhile, green fluorescent protein-nanobody (GNB) resin49, prepared by coupling the GFP-nanobody protein to CNBr Sepharose resin at a concentration of 1 mg ml−1, was equilibrated in 0.1 mM LMNG, 0.02 mM CHS, 50 mM HEPES pH 7.8, 200 mM NaCl, 25 μM palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC) and 10% glycerol. The pre-equilibrated GNB resin was added to the solubilized membrane supernatant for binding in batch mode on a 3D shaker at 4 °C for 3 h. The protein-bound GNB resin was then packed into a gravity column and washed with a total of 12 column volumes of wash buffer consisting of 0.06% digitonin, 0.006% CHS, 50 mM HEPES pH 7.8, 200 mM NaCl, 25 μM POPC, 10% glycerol, 10 μM MRS7292, 2 μM β-CFT. The tag-free Δ-hDAT protein was eluted overnight in wash buffer containing 3C protease, concentrated, and further purified by size-exclusion chromatography (SEC) in 0.02% digitonin, 0.002% CHS, 50 mM HEPES pH 7.8, 200 mM NaCl, 4 μM MRS7292, 500 nM β-CFT and 25 μM POPC.
Cryo-EM sample, grid preparation and data collection
SEC-purified Δ-hDAT protein was concentrated to about 7 mg ml−1 using a 100 kDa cutoff filter and used immediately for preparation of cryo-EM grids. Holey grids (Quantifoil R 1.2/1.3 Au 200 mesh) were rendered hydrophilic by glow-discharge at 15 mA for 30 s and were used immediately. A solution of 3 μl of concentrated Δ-hDAT was applied to the grid and blotted for 3 s with no wait time, single blotting in 100% humidity at 15 °C, followed by plunge freezing in liquid ethane using a Vitrobot Mark IV vitrification system (Thermo Scientific). Cryo-EM data were collected using a Titan Krios (300 keV) microscope fitted with a Falcon4i direct electron detector and a Selectris X Energy filter (Thermo Scientific) and SerialEM v4.1.0 beta24 software. The images were recorded in electron event representation (EER) format using a defocus range of −1.0 to −2.5 µM, a total dose of 50 e− Å−2, a physical pixel size of 0.743 Å, and an energy filter slit width of 6 eV.
Cryo-EM image processing
A total of 14,460 cryo-EM images in EER50 format were imported into CryoSparc, versions 4.2.1 and 4.4.051 and motion corrected using patch motion correction followed by contrast transfer function (CTF) estimation and curation of the micrographs. Micrographs with poor CTF fits were discarded, leaving a total of 14,277 micrographs for further image processing. Particles were picked by reference-free blob picking using elliptical blobs with 80 Å and 150 Å minor and major axes, respectively. A total of 7,117,202 particles were picked initially. Particles were then extracted with a box size of 256 pixels, Fourier cropped to 64 pixels, and subjected to multiple rounds of 2D classification. The 2D classes showing promising transmembrane helix features were selected in three rounds of 2D classification, followed by ab initio-based 3D reconstruction and classification52. In brief, four ab initio classes were generated in duplicate jobs with the following parameters: initial batch size: 300; final batch size: 1,000; number of final iteration: 500; max alignment resolution: 8 Å; and initial alignment resolution: 20 Å. Particles from the best class with distinct transmembrane helices were pooled from both replicates, and duplicates were removed. Next, particles were re-extracted with the same box size as above with Fourier cropping by a factor of two. Subsequent 2D classification followed by ab initio-based 3D reconstruction–classification was repeated as described above, but with maximum and minimum alignment resolutions of 6 Å and 12 Å, respectively. The best class was then selected, and particles were pooled as previously described. Finally, the particles were re-extracted without any Fourier cropping with a box size of 384 pixels, and ab initio-based classification was carried out as previously described with initial and maximum alignment resolutions of 8 Å and 4.5 Å, respectively, and final iteration set at 350. Classes with the most well-defined Δ-hDAT features were selected for pooling particles. After removal of duplicates, the particles were used for non-uniform refinement53, with initial low-pass resolution of 12 Å, followed by four additional passes of refinement with the minimize over per-particle scale parameter on. Non-uniform refinement resulted in a 3D reconstruction of a Δ-hDAT map at a resolution of 3.19 Å, based on a Fourier shell correlation (FSC) cutoff of 0.143 with 177,494 particles.
Model building and refinement
The final cryo-EM map of Δ-hDAT was interpreted by fitting an AlphaFold-derived model54 (AF-Q01959-F1) of hDAT in ChimeraX55 using rigid body fitting. The N-terminal 56 residues were truncated in the AlphaFold model of Δ-hDAT. The fitted model and map were then manually adjusted using COOT (v0.9.8.6)56 and then further refined in Phenix v1.20.1-448757 using real space refinement58 in an iterative manner. The restraints for the MRS7292 compound were generated using the elbow program in Phenix and used in subsequent refinement steps. MolProbity59 was used to assess the quality of the refined model with respect to geometric restraints, all atom clash score, and Ramachandran statistics, and Check My Metal was used to assess Zn2+ and Na+ site stereochemistry60. The comprehensive validation program in Phenix was used to obtain the final refinement statistics (Extended Data Table 1). In order to assess overfitting during refinement, the FSCwork and FSCfree curves were compared61,62. ∆-hDAT coordinates were shaken using PDB tools in Phenix with random shifts of 0.5 r.m.s.d. The resultant model was superposed, using α-carbon atoms, with the input model to confirm the change in r.m.s.d. This shaken model was refined against one of the two half-maps and the resultant model-versus-map FSC curve was termed as FSCwork. A map-versus-model curve with this shaken-refined model and the other half-map, which was not used in any refinement, was obtained using the comprehensive cryo-EM validation tool in Phenix. This FSC curve was termed as FSCfree. The FSCwork and FSCfree curves were plotted and analysed for overfitting. Structural figures and illustrations were prepared using PyMOL (The PyMOL Molecular Graphics System, version 2.5.5, Schrödinger) and ChimeraX v1.6.155.
Model of inward-open ∆-hDAT
The model of an inward-open conformation was generated using the SWISS model server63 and the inward-open structure of hSERT (PDB:7LI6) as a template.
Dopamine uptake assay
HEK293 GnTI− cells were transduced with Δ-hDAT and full-length hDAT P2 viruses, propagated in SF9 cells using standard methods as described in ‘Protein expression and purification’, at a cell density of 2.5 – 3.0 × 106 cells per ml, followed by incubation at 37 °C for 6 h. After 6 h, sodium butyrate was added to a final concentration of 10 mM, and the cells were transferred to 30 °C with 8% CO2 for 6 h. The transduced cells were seeded into 96-well poly-d-lysine coated Isoplates (Perkin Elmer) at a density of 100,000 cells per well. The plates were then incubated at 30 °C for 12–16 h before the uptake assay was initiated. The cells were initially washed with 37 °C uptake assay buffer composed of 25 mM HEPES pH 7.4, 120 mM NaCl, 5 mM KCl, 1.2 mM CaCl2, 1.2 mM MgSO4, 5 mM d-glucose, 1 mM ascorbic acid, and 1 µM Ro 41-0960, followed by incubation in 50 µl of the same buffer for 10 min. Replicates with 10 µM of GBR12909 in the uptake assay buffer were used to measure background. For assessing the effect of MRS7292 on uptake, 10 µM of MRS7292 was added to the uptake buffer. Cells were then incubated with 50 µl of [3H]dopamine with a specific activity of 45.6 Ci mmol−1 (hot:cold ratio of 1:100) in uptake assay buffer at a concentration range from 30 to 0.0137 µM for 10 min. The uptake reaction was stopped by adding 100 µl of chilled inhibition buffer, uptake assay buffer supplemented with 2.5 µM GBR12909. Two consecutive washes with 100 µl of inhibition buffer were carried out, followed by resuspension of cells in 100 µl of 1% Triton X-100. Finally, 100 µl of liquid scintillation cocktail was added to each well and [3H] counts were measured using a MicroBeta2 (Perkin Elmer). Background counts from three replicates were averaged and subtracted from total counts. Data were fit to the Michaelis–Menten equation to determine the kinetic parameters of dopamine uptake from three independent experiments (n = 3 biological replicates starting from transduction), each with triplicate measurements.
For [3H]dopamine uptake experiments on ∆-hDAT mutants, HEK293 GnTI− cells were transduced and incubated as described above. For IC50 measurements of the MRS7292 compound, cells expressing ∆-hDAT and ∆-hDAT with W84A, W84C, D385A, D385N, V364I, Y394F, M414L, I390L and V364I/I390L mutations were washed with uptake buffer and then incubated with 25 µl of uptake buffer containing MRS7292 at concentrations ranging from 30 to 0.0411 µM (10 to 0.0137 µM for the M414L mutant) for 20 min. Replicates containing 10 µM of GBR12909 were used to measure background. To initiate uptake, 25 µl of 50 nM [3H] dopamine (35.5 Ci mmol−1) was added, and the cells were incubated for 10 minutes. Uptake was quenched by adding 50 µl of ice-cold inhibition buffer. Subsequent washing steps and radioactivity measurements were carried out as previously described.
For IC50 measurements of Zn2+, uptake assays were carried out as described for MRS7292 with the following alterations. The uptake buffer contained EPPS at pH 8.5 in place of HEPES. Cells were washed once with uptake buffer containing 1 mM ethylenediaminetetraacetic acid (EDTA) to chelate ambient Zn2+ from the cell growth medium, followed by second wash without EDTA. Cells were then incubated for 15 min in uptake buffers containing 0.3–300 µM added Zn2+ for ∆-hDAT, 0.1–100 µM added Zn2+ for T211H, and 0.03–30 µM added Zn2+ for T211E. Measurements of [3H]dopamine uptake in the absence of added Zn2+ were obtained using buffer with 1 mM EDTA to chelate ambient Zn2+. The IC50 of Zn2+ for the T211E mutant was also measured at pH 7.5 to obtain a more complete inhibition curve. Elemental analysis of the uptake buffer revealed ambient Zn2+ present at about 100 nM. The estimated ‘free’ Zn2+ concentrations are used in the IC50 plots, where following the 1 mM EDTA wash we assume that there is ‘zero’ Zn2+ and in the subsequent Zn2+ concentrations we estimate that there is about 100 nM ambient Zn2+, from the uptake buffer, in addition to the added Zn2+ concentrations.
Specific counts were obtained by subtracting background counts (averaged from technical triplicates) from total counts. The specific uptake activity as percentages of the control was plotted against either MRS7292 or Zn2+ concentrations using GraphPad Prism v7.05. Specific uptake activity in 1 pM MRS7292 and 1 mM EDTA was set to 100% for the MRS7292 and Zn2+ IC50 measurements, respectively. The data points were fitted using nonlinear regression models in GraphPad Prism v7.05: [inhibitor] versus normalized response with variable slope: \(y=100/(1+({x}^{{\rm{H}}{\rm{i}}{\rm{l}}{\rm{l}}{\rm{s}}{\rm{l}}{\rm{o}}{\rm{p}}{\rm{e}}}/{{\rm{I}}{\rm{C}}}_{50}^{{\rm{H}}{\rm{i}}{\rm{l}}{\rm{l}}\,{\rm{s}}{\rm{l}}{\rm{o}}{\rm{p}}{\rm{e}}}))\) for analysis of inhibition by MRS7292 and [inhibitor] versus response with variable slope: \(y={\rm{b}}{\rm{o}}{\rm{t}}{\rm{t}}{\rm{o}}{\rm{m}}+({\rm{t}}{\rm{o}}{\rm{p}}-{\rm{b}}{\rm{o}}{\rm{t}}{\rm{t}}{\rm{o}}{\rm{m}})/(1+({x}^{{\rm{H}}{\rm{i}}{\rm{l}}{\rm{l}}{\rm{s}}{\rm{l}}{\rm{o}}{\rm{p}}{\rm{e}}}/{{\rm{I}}{\rm{C}}}_{50}^{{\rm{H}}{\rm{i}}{\rm{l}}{\rm{l}}\,{\rm{s}}{\rm{l}}{\rm{o}}{\rm{p}}{\rm{e}}}))\) for analysis of inhibition by Zn2+. Data were collected from three independent experiments (n = 3 biological replicates starting from transduction), each performed with three technical replicates.
Scintillation proximity assay
For SPA64, His-tagged ∆-hDAT protein was purified as described in ‘Protein expression and purification’ but with Strep-tactin resin, utilizing the Twin strep affinity tag, and without β-CFT (WIN35428). The various buffer systems were unchanged. YSi-Cu SPA beads at 1 mg ml−1 were added to ∆-hDAT (30 nM) in SEC buffer (0.02% digitonin, 0.002% CHS, 50 mM HEPES pH 7.8, 200 mM NaCl, 25 µM POPC and 4 µM MRS7292). [3H]-WIN35428 (82.8 Ci mmol−1) in 20 mM HEPES pH 7.8 and 100 mM NaCl was used at concentration points ranging from 0 to 150 nM. For background measurement, 100 µM of GBR12909 was added to the assay buffer. Reactants were added to a 96-well isoplate, briefly mixed on a shaker at room temperature, and [3H] counts were recorded using a MicroBeta2. Data were collected from three independent experiments (n = 3), each performed in technical triplicate, using the same purified protein sample. Background subtracted counts were plotted and analysed by a single-site binding model via nonlinear regression analysis in GraphPad Prism v7.05.
Molecular dynamics simulation
Simulation setup
The Δ-hDAT cryo-EM structure was used to prepare the simulation systems, after removing all unwanted molecular species except for the ligands (β-CFT and MRS7292) and the Zn2+ ion. A missing disordered region (EL2) was modelled using the Schrödinger Prime module65 (Schrödinger release 2023-2: Prime, Schrödinger), and the protein-prepared wizard66 was used to assign the protonation states of titratable residues. All histidine residues were assigned as neutral (HID) except for His129, which was protonated (HIP). A disulfide bond was introduced between Cys180 and Cys189. The protein was internally hydrated using the DOWSER plugin67,68 of VMD69. The CHARMM-GUI Membrane-Builder70 was then used to construct the initial lipid bilayer for embedding the protein. The protein’s orientation in the bilayer was derived from the Orientations of Proteins in Membranes (OPM) database71. Subsequently, the structure was inserted into a heterogeneous lipid bilayer, and sterically clashing lipid molecules were removed. The bilayer consisted of POPC and cholesterol (CHL) at a percentage ratio of 3:2. Slabs of 40-Å TIP3P water molecules were placed above and below the bilayer. Na+ and Cl– counterions were added to neutralize the systems to a total salt concentration of 0.15 M, resulting in the entire simulation unit cell (102 Å × 102 Å × 134 Å) containing approximately 122,000 atoms. LEaP was utilized to assign force field parameters for all the molecular species in the system. The Δ-hDAT protein, lipids, Na+ and Cl– ions, and TIP3P waters were described using AMBER ff19SB72, Lipid2173, and monovalent ion parameters for TIP3P water74, respectively. The Zn2+ ion was described using the Li–Merz parameters75 for highly charged metal ions. CUFIX corrections76 were applied to nonbonded interactions between specific pairs of charged chemical moieties. LigPrep with the OPLS4 force field77 was used to minimize the β-CFT and MRS7292 structures. In addition, the ionized state of these ligands was realized by Epik78 at a pH value of 7.0 ± 2.0. AMBER force field 2 (GAFF2)79,80 parameter sets were used for the ligands (β-CFT and MRS7292). A typical system setup is depicted in Supplementary Fig. 2.
Simulation conditions
All simulations were performed using the Amber2081 suite and pmemd.cuda module. To eliminate bad contacts between solute and solvent water molecules in the system, energy minimization and equilibration simulations were conducted in three stages prior to the production runs. Firstly, energy minimization was performed while applying harmonic restraints on the lipid and solute heavy atoms (k = 10 kcal mol–1 Å–2). The entire system was then minimized for 10,000 steps, followed by an additional 5,000 steps of energy minimization using the Steepest Descent algorithm and the conjugate gradient method. Secondly, a two-step equilibration simulation was carried out. The system was first heated from 0 K to approximately 100 K, and then gradually to 310 K with the protein and lipid restrained over 100 ps in the NVT ensemble. Subsequently, all simulated complexes underwent 10 repeats of unconstrained NPT dynamics (5 ns, each) at 310 K and 1 atm. Finally, a 1.0 µs production simulation was conducted for each complex within the NPT ensemble at 310 K and 1 atm, using periodic boundary conditions. The temperature and pressure were maintained using a Langevin thermostat82 and a Monte Carlo barostat83, respectively. Electrostatic interactions were calculated with a distance cutoff of 10 Å, using the particle mesh Ewald (PME)84 method. The SHAKE algorithm85 was used to maintain all constraints for bonds involving hydrogens, and the time step was set to 2.0 fs. In total, we conducted five independent replicas, leading to a cumulative sampling of 5 µs (5 runs × 1 µs each) and storing 500,000 frames.
Trajectory analysis
For visualization and analysis, we used VMD69 and AmberTools2286,87 along with in-house scripts. To quantify the stacking of hDAT:W84 and MRS7292, we established a threshold for when their two rings form a stacking interaction as follows. The threshold is based on the distance between the heavy atom centres of masses (COMs) of the indole ring of W84 (atoms: CG, CD1, NE1, CE2, CZ2, CH2, CZ3, CE3 and CD2) and the adenine ring of MRS7292 (atoms: N3, C4, N1, C3, C5, N4, C6, N5 and C2). We considered MRS7292 and W84 molecules to be stacked if their COM distance was ≤5 Å and the angle between their ring normals was ≤45°. The per-residue contact profile was calculated using an in-house tcl script in VMD69. For each snapshot within each run, the distance between every heavy atom pair, from the ligands and the protein, respectively, was computed, and distances ≤4 Å were considered a contact. Across the entire trajectory, if a residue exhibited contacts with ligands for more than 40% of the total time, it was designated as having a stable contact.
Cell line statement
Sf9 cells for generation of baculovirus and expression of recombinant antibody fragment are from Thermo Fisher (12659017, lot 421973). The cells were not authenticated experimentally for these studies. The cells were tested negative for Mycoplasma contamination using the CELLshipper Mycoplasma Detection Kit M-100 from Bionique.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The cryo-EM maps and coordinates for the ∆-hDAT structure have been deposited in the Electron Microscopy Data Bank (EMDB) under accession number EMD-43128 and in the Protein Data Bank (PDB) under accession code 8VBY. All molecular dynamics trajectories generated for this study and simulation input files have been deposited in a Zenodo repository and are freely available at https://doi.org/10.5281/zenodo.11391489 (ref. 88). Source data are provided with this paper.
References
Bjorklund, A. & Dunnett, S. B. Dopamine neuron systems in the brain: an update. Trends Neurosci. 30, 194–202 (2007).
Clarke, R. L. et al. Compounds affecting the central nervous system. 4. 3 Beta-phenyltropane-2-carboxylic esters and analogs. J. Med. Chem. 16, 1260–1267 (1973).
Janowsky, A., Tosh, D. K., Eshleman, A. J. & Jacobson, K. A. Rigid adenine nucleoside derivatives as novel modulators of the human sodium symporters for dopamine and norepinephrine. J. Pharm. Expt. Therapeutics 357, 24–35 (2016).
Norregaard, L., Fredericksen, D., Nielsen, E. O. & Gether, U. Delineation of an endogenous zinc-binding site in the human dopamine transporter. EMBO J. 17, 4266–4273 (1998).
Speranza, L., di Porzio, U., Viggiano, D., de Donato, A. & Volpicelli, F. Dopamine: the neuromodulator of long-term synaptic plasticity, reward and movement control. Cells 10, 735 (2021).
Bucolo, C., Leggio, G. M., Drago, F. & Salomone, S. Dopamine outside the brain: the eye, cardiovascular system and endocrine pancreas. Pharmacol. Ther. 203, 107392 (2019).
Kandel, E. R., Schwartz, J. H.and Jessel, T. M. Principles of Neural Science (Appleton and Lange, 1991).
Goetz, C. G. The history of Parkinson’s disease: early clinical descriptions and neurological therapies. Cold Spring Harb. Perspect. Med. 1, a008862 (2011).
Klein, M. O. et al. Dopamine: functions, signaling, and association with neurological diseases. Cell. Mol. Neurobiol. 39, 31–59 (2019).
Groom, M. J. & Cortese, S. Current pharmacological treatments for ADHD. Curr. Top. Behav. Neurosci. 57, 19–50 (2022).
Newman, A. H., Ku, T., Jordan, C. J., Bonifazi, A. & Xi, Z. X. New drugs, old targets: tweaking the dopamine system to treat psychostimulant use disorders. Annu. Rev. Pharmacol. Toxicol. 61, 609–628 (2021).
Rudnick, G., Krämer, R., Blakely, R. D., Murphy, D. L. & Verrey, F. The SLC6 transporters: perspectives on structure, functions, regulation, and models for transporter dysfunction. Pflugers Arch. 466, 25–42 (2014).
Mitchell, P. A general theory of membrane transport from studies of bacteria. Nature 180, 134–136 (1957).
Schmidt, S. G. et al. The dopamine transporter antiports potassium to increase the uptake of dopamine. Nat. Commun. 13, 2446 (2022).
Frederickson, C. J. et al. Synaptic release of zinc from brain slices: factors governing release, imaging, and accurate calculation of concentration. J. Neurosci. Methods 154, 19–29 (2006).
Tosh, D. K. et al. Scaffold repurposing of nucleosides (adenosine receptor agonists): enhanced activity at the human dopamine and norepinephrine sodium symporters. J. Med. Chem. 60, 3109–3123 (2017).
Aggarwal, S. et al. Identification of a novel allosteric modulator of the human dopamine transporter. ACS Chem. Neurosci. 10, 3718–3730 (2019).
Penmatsa, A., Wang, K. H. & Gouaux, E. X-ray structure of dopamine transporter elucidates antidepressant mechanism. Nature 503, 85–90 (2013).
Wang, K. H., Penmatsa, A. & Gouaux, E. Neurotransmitter and psychostimulant recognition by the dopamine transporter. Nature 521, 322–327 (2015).
Penmatsa, A., Wang, K. H. & Gouaux, E. X-ray structures of Drosophila dopamine transporter in complex with nisoxetine and reboxetine. Nat. Struct. Mol. Biol. 22, 506–508 (2015).
Navratna, V., Tosh, D. K., Jacobson, K. A. & Gouaux, E. Thermostabilization and purification of the human dopamine transporter (hDAT) in an inhibitor and allosteric ligand bound conformation. PLoS ONE 13, e0200085 (2018).
Kristensen, A. S. et al. SLC6 neurotransmitter transporters: structure, function and regulation. Pharmacol. Rev. 63, 585–640 (2011).
Aggarwal, S. & Mortensen, O. V. Discovery and development of monoamine transporter ligands. Adv. Neurobiol. 30, 101–129 (2023).
Jayaraman, K. et al. SLC6 transporter oligomerization. J. Neurochem. 157, 919–929 (2021).
Yamashita, A., Singh, S. K., Kawate, T., Jin, Y. & Gouaux, E. Crystal structure of a bacterial homologue of Na+/Cl− dependent neurotransmitter transporters. Nature 437, 215–223 (2005).
Hong, W. C. & Amara, S. G. Membrane cholesterol modulates the outward facing conformation of the dopamine transporter and alters cocaine binding. J. Biol. Chem. 285, 32616–32626 (2010).
Jones, K. T., Zhen, J. & Reith, M. E. Importance of cholesterol in dopamine transporter function. J. Neurochem. 123, 700–715 (2012).
Zeppelin, T., Ladefoged, L. K., Sinning, S., Periole, X. & Schiott, B. A direct interaction of cholesterol with the dopamine transporter prevents its out-to-inward transition. PLoS Comput. Biol. 14, e1005907 (2018).
Coleman, J. A., Green, E. M. & Gouaux, E. X-ray structures and mechanism of the human serotonin transporter. Nature 532, 334–339 (2016).
Li, L. B. et al. The role of N-glycosylation in function and surface trafficking of the human dopamine transporter. J. Biol. Chem. 279, 21012–21020 (2004).
Reeves, P. J., Callewaert, N., Contreras, R. & Khorana, H. G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl Acad. Sci. USA 99, 13419–13424 (2002).
Kuhar, M. J. et al. Studies of selected phenyltropanes at monoamine transporters. Drug Alcohol Depend. 56, 9–15 (1999).
Wong, E. H. et al. Reboxetine: a pharmacologically potent, selective, and specific norepinephrine reuptake inhibitor. Biol. Psychiatry 47, 818–829 (2000).
Andersen, J., Ringsted, K. B., Bang-Andersen, B., Stromgaard, K. & Kristensen, A. S. Binding site residues control inhibitor selectivity in the human norepinephrine transporter but not in the human dopamine transporter. Sci. Rep. 5, 15650 (2015).
Owens, M. J., Knight, D. L. & Nemeroff, C. B. Second-generation SSRIs: human monoamine transporter binding profile of escitalopram and R-fluoxetine. Biol. Psychiatry 50, 345–350 (2001).
Gu, H., Wall, S. C. & Rudnick, G. Stable expression of biogenic amine transporters reveals differences in inhibitor sensitivity, kinetics, and ion dependence. J. Biol. Chem. 269, 7124–7130 (1994).
Wang, H. et al. Structural basis for action by diverse antidepressants on biogenic amine transporters. Nature 503, 141–145 (2013).
Krishnamurthy, H., Piscitelli, C. L. & Gouaux, E. Unlocking the molecular secrets of sodium-coupled transporters. Nature 459, 347–355 (2009).
Kitayama, S. et al. Dopamine transporter site-directed mutations differentially alter substrate transport and cocaine binding. Proc. Natl Acad. Sci. USA 89, 7782–7785 (1992).
Bismuth, Y., Kavanaugh, M. P. & Kanner, B. I. Tyrosine 140 of the gamma-aminobutyric acid transporter GAT-1 plays a critical role in neurotransmitter recognition. J. Biol. Chem. 272, 16096–16102 (1997).
Yang, D. & Gouaux, E. Illumination of serotonin transporter mechanism and role of the allosteric site. Sci. Adv. 7, eabl3857 (2021).
Yang, D., Zhao, Z., Tajkhorshid, E. & Gouaux, E. Structures and membrane interactions of native serotonin transporter in complexes with psychostimulants. Proc. Natl Acad. Sci. USA 120, e2304602120 (2023).
Yamashita, A., Singh, S. K., Kawate, T., Jin, Y. & Gouaux, E. Crystal structure of a bacterial homologue of Na+/Cl−-dependent neurotransmitter transporters. Nature 437, 215–223 (2005).
Plenge, P. et al. The antidepressant drug vilazodone is an allosteric inhibitor of the serotonin transporter. Nat. Commun. 12, 5063 (2021).
Aniksztejn, L., Charton, G. & Ben-Ari, Y. Selective release of endogenous zinc from the hippocampal mossy fibers in situ. Brain Res. 404, 58–64 (1987).
Stockner, T. et al. Mutational analysis of the high-affinity zinc binding site validates a refined human dopamine transporter homology model. PLoS Comput. Biol. 9, e1002909 (2013).
Loland, C. J., Norregaard, L. & Gether, U. Defining proximity relationships in the tertiary structure of the dopamine transporter. Identification of a conserved glutamic acid as a third coordinate in the endogenous Zn2+-binding site. J. Biol. Chem. 274, 36928–36934 (1999).
Goehring, A. et al. Screening and large-scale expression of membrane proteins in mammalian cells for structural studies. Nat. Protoc. 9, 2574–2585 (2014).
Kubala, M. H., Kovtun, O., Alexandrov, K. & Collins, B. M. Structural and thermodynamic analysis of the GFP:GFP-nanobody complex. Protein Sci. 19, 2389–2401 (2010).
Guo, H. et al. Electron-event representation data enable efficient cryoEM file storage with full preservation of spatial and temporal resolution. IUCrJ 7, 860–869 (2020).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Wright, N. J. et al. Methotrexate recognition by the human reduced folate carrier SLC19A1. Nature 609, 1056–1062 (2022).
Punjani, A., Zhang, H. & Fleet, D. J. Non-uniform refinement: adaptive regularization improves single-particle cryo-EM reconstruction. Nat. Methods 17, 1214–1221 (2020).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Pettersen, E. F. et al. UCSF ChimeraX: structure visualization for researchers, educators, and developers. Protein Sci. 30, 70–82 (2021).
Casanal, A., Lohkamp, B. & Emsley, P. Current developments in Coot for macromolecular model building of electron cryo-microscopy and crystallographic data. Protein Sci. 29, 1069–1078 (2020).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. D 75, 861–877 (2019).
Afonine, P. V. et al. Real-space refinement in PHENIX for cryo-EM and crystallography. Acta Crystallogr. D 74, 531–544 (2018).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
Gucwa, M. et al. CMM-An enhanced platform for interactive validation of metal binding sites. Protein Sci. 32, e4525 (2023).
Amunts, A. et al. Structure of the yeast mitochondrial large ribosomal subunit. Science 343, 1485–1489 (2014).
Alvadia, C. et al. Cryo-EM structures and functional characterization of the murine lipid scramblase TMEM16F. eLife 8, e44365 (2019).
Waterhouse, A. et al. SWISS-MODEL: homology modelling of protein structures and complexes. Nucleic Acids Res. 46, W296–W303 (2018).
Quick, M. & Javitch, J. A. Monitoring the function of membrane transport proteins in detergent-solubilized form. Proc. Natl Acad. Sci. USA 104, 3603–3608 (2007).
Jacobson, M. P. et al. A hierarchical approach to all-atom protein loop prediction. Proteins 55, 351–367 (2004).
Sastry, G. M., Adzhigirey, M., Day, T., Annabhimoju, R. & Sherman, W. Protein and ligand preparation: parameters, protocols, and influence on virtual screening enrichments. J. Comput. Aided Mol. Des. 27, 221–234 (2013).
Gumbart, J., Trabuco, L. G., Schreiner, E., Villa, E. & Schulten, K. Regulation of the protein-conducting channel by a bound ribosome. Structure 17, 1453–1464 (2009).
Zhang, L. & Hermans, J. Hydrophilicity of cavities in proteins. Proteins 24, 433–438 (1996).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Jo, S., Kim, T., Iyer, V. G. & Im, W. CHARMM-GUI: a web-based graphical user interface for CHARMM. J. Comput. Chem. 29, 1859–1865 (2008).
Lomize, M. A., Pogozheva, I. D., Joo, H., Mosberg, H. I. & Lomize, A. L. OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res. 40, D370–D376 (2012). (Database issue).
Tian, C. et al. ff19SB: amino-acid-specific protein backbone parameters trained against quantum mechanics energy surfaces in solution. J. Chem. Theory Comput. 16, 528–552 (2020).
Dickson, C. J., Walker, R. C. & Gould, I. R. Lipid21: complex lipid membrane simulations with AMBER. J. Chem. Theory Comput. 18, 1726–1736 (2022).
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926–935 (1983).
Li, P., Song, L. F. & Merz, K. M. Jr Parameterization of highly charged metal ions using the 12-6-4 LJ-type nonbonded model in explicit water. J. Phys. Chem. B 119, 883–895 (2015).
Yoo, J. & Aksimentiev, A. New tricks for old dogs: improving the accuracy of biomolecular force fields by pair-specific corrections to non-bonded interactions. Phys. Chem. Chem. Phys. 20, 8432–8449 (2018).
Lu, C. et al. OPLS4: improving force field accuracy on challenging regimes of chemical space. J. Chem. Theory Comput. 17, 4291–4300 (2021).
Shelley, J. C. et al. Epik: a software program for pKa prediction and protonation state generation for drug-like molecules. J. Comput. Aided Mol. Des. 21, 681–691 (2007).
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
He, X., Man, V. H., Yang, W., Lee, T. S. & Wang, J. A fast and high-quality charge model for the next generation general AMBER force field. J. Chem. Phys. 153, 114502 (2020).
Case, D. A. et al. AMBER 2020 (University of California, San Francisco, 2020).
Pastor, R. W., Brooks, B. R. & Szabo, A. An analysis of the accuracy of Langevin and molecular dynamics algorithms. Mol. Phys. 65, 1409–1419 (1988).
Aqvist, J., Wennerstrom, P., Nervall, M., Bjelic, S. & Brandsdal, B. O. Molecular dynamics simulations of water and biomolecules with a Monte Carlo constant pressure algorithm. Chem. Phys. Lett. 384, 288–294 (2004).
Darden, T., York, D. & Pedersen, L. Particle mesh Ewald: an N.log(N) method for Ewald sums in large systems. J. Chem. Phys. 98, 10089–10092 (1993).
Ryckaert, J.-P., Ciccotti, G. & Berendsen, H. J. C. Numerical integration of the cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanes. J. Comput. Physics 23, 327–341 (1977).
Case, D. A. et al. AmberTools. J. Chem. Inf. Model. 63, 6183–6191 (2023).
Roe, D. R. & Cheatham, T. E. 3rd PTRAJ and CPPTRAJ: software for processing and analysis of molecular dynamics trajectory data. J. Chem. Theory Comput. 9, 3084–3095 (2013).
Sk, M. F. & Tajkhorshid, E. Raw data for: Structure of the human dopamine transporter and mechanisms of allosteric inhibition. Zenodo https://doi.org/10.5281/zenodo.11391488 (2024).
Acknowledgements
We thank members of the Gouaux and Baconguis laboratories for input, R. Courtney for manuscript preparation, C. Sun for advice on cryo-EM data processing, M. Ralle and S. Miller for ICPMS measurements, which were performed in the OHSU Elemental Analysis Core with partial support from NIH (S10OD028492), J. Coleman (University of Pittsburgh) for his suggestions on biochemical experiments and on the manuscript, A. Janowsky for discussions related to hDAT inhibitors, and T. Provitola for assistance with figures. We acknowledge use of the OHSU Multiscale Microscopy Core (MMC), the Pacific Northwest Cryo-EM Center (PNCC) and the cryo-EM facility at Janelia research campus. PNCC is supported by NIH grant U24GM129547 and accessed through EMSL (grid.436923.9), a DOE Office of Science User Facility sponsored by the Office of Biological and Environmental Research. The computational component of the project was supported by the National Institutes of Health (Grants R24-GM145965 and P41-GM104601 to E.T.). Molecular dynamics simulations were performed using ACCESS allocations (Grant MCA06N060 to E.T.) through the support of National Science Foundation grant number ACI-1548562. The experimental work was supported by an NIH grant to E.G. (R01 MH070039) and NIDDK Intramural grant to K.A.J. (ZIADK031127). E.G. is an investigator of the Howard Hughes Medical Institute and thanks Bernard and Jennifer LaCroute for generous support.
Author information
Authors and Affiliations
Contributions
D.K.S., V.N. and E.G. designed the project. D.T. and K.A.J. prepared the MRS compound. V.N. established constructs and protein expression conditions, D.K.S. prepared all cryo-EM samples and carried out cryo-EM analysis and structure determination. D.K.S. and A.C. prepared and expressed mutant constructs and performed ligand binding experiments and uptake assays. M.F.S. and E.T. carried out the molecular dynamics studies. D.K.S. and E.G. wrote the initial draft of the manuscript, and all authors edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Zhenhua Shao, Harald Sitte and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Biochemical characterization of ∆-hDAT.
(a) Fluorescence-detection size-exclusion chromatography (FSEC) of purified ∆-hDAT protein. (b) SDS-PAGE analysis of the FSEC sample was done once and visualized by silver staining. See Supplementary Fig. 1 for gel.
Extended Data Fig. 2 Cryo-EM image processing and analysis.
(a) Cryo-EM workflow. A detailed description of the image processing steps and parameters is included in the Methods section. Scale bar = 50 nm. (b) Local resolution map along with (c) the gold standard Fourier shell correlation (GSFSC) curve and (d) angular sampling of the cryo-EM reconstruction. (e) FSCwork/FSCfree and map versus model curve.
Extended Data Fig. 3 Density associated with transmembrane helices, EL4, and the C-terminal latch.
Isomesh map features are contoured at 8 σ and within 2 Å of the atoms associated with each feature.
Extended Data Fig. 4 C-terminal latch of ∆-hDAT and structural comparison with other NSS transporters.
(a) Comparison of the C-terminal latch of ∆-hDAT with the human serotonin transporter (hSERT; PDB: 7LIA), human GABA transporter (GAT1; PDB: 7SK2), and human glycine transporter (GlyT1, PDB: 6ZBV). The structures were aligned using α-carbon atoms. (b) Close up view of the C-terminal latch of ∆-hDAT showing the proximity of TM12, TM3, and TM10.
Extended Data Fig. 5 Lipid and lipid-like features in the ∆-hDAT reconstruction.
(a) Overall distribution of lipid-like features in the cryo-EM reconstruction of ∆-hDAT. CHS and alkyl chains of possible lipid molecules are represented as orange and magenta sticks, respectively. The associated cryo-EM density features are shown in isomesh map representation contoured at 8 σ within 2 Å of the atoms of putative CHS and alkyl chain molecules. (b) Structural alignment of ∆-hDAT with dDAT (PDB: 4XPG) and (c) with hSERT (PDB: 5I73) showing the positions of CHS or cholesterol molecules in the proximity of lipid molecules in ∆-hDAT. Structural superposition was done using α-carbon atoms of the whole structures. (d) Additional putative CHS molecule in a groove formed by TMs 4, 5 and 8.
Extended Data Fig. 6 Central binding site of ∆-hDAT and comparison with related transporters.
(a) Overlay of the central binding pockets of S-citalopram-bound hSERT (5I73) and β-CFT-bound ∆-hDAT. The structural alignment was done using α-carbon atoms of TM3 and TM8 of ∆-hDAT. (b) Superposition of the central sites occupied by β-CFT in dDAT (PDB: 4XPG), cocaine in pSERT (PDB: 8DE3) and β-CFT in ∆-hDAT. (c) Key phenylalanines ‘above’ the central sites in pSERT (PDB: 8DE3) and dDAT (PDB: 4XPG) that participate in defining the occluded or outward open states. The gray sphere represents the center of mass of β-CFT in ∆-hDAT. (d) (d) Superposition of the central site showing the relative pose of β-CFT in the cryo-EM reconstruction of ∆-hDAT and five replicas of MD simulation derived poses. All structural superpositions in (b)-(d) were done using α-carbon atoms of whole structures. (e) Coordination of a Na+ ion at the Na2 site. The distances between the Na+ ion and the coordinating oxygen atoms of the likely interacting residues are shown in yellow dashed lines and represented in Å.
Extended Data Fig. 7 The MRS7292 site in ∆-hDAT and structural comparison with hSERT and dDAT.
(a) The locations of allosteric sites for the respective transporter complexes. The allosteric ligands are shown in sphere representation, and the central site molecules are shown as sticks. (b) Illustration of the MRS site showing the position of the methyl ester moiety (dashed circle). (c) Effect of MRS7292 on [3H]dopamine uptake in ∆-hDAT mutants. Data was plotted and fitted using a non-linear regression model as described in ‘Methods’. Data from n = 3 biological replicates, each performed in technical triplicate, are represented as mean values +/− standard deviation. (d) Total, specific [3H]dopamine uptake for ∆-hDAT and ∆-hDAT mutant controls from the IC50 experiments. Data from n = 3 biological replicates, each performed in technical triplicate, are represented as mean values +/− standard deviation. (e) Accommodation of the thienyl moiety (dashed circle) into the MRS subsite. (f) Alignment of the MRS7292 (shown as teal sticks) binding pocket with the binding pocket of hSERT in complex with serotonin (7LIA) and (g) dDAT in complex with β-CFT (4XPG). Superposition of structures was performed using α-carbon atoms of TM3 and TM8 of ∆-hDAT. The superposition shows how TMs 1b and 6a are displaced and reoriented whereas TMs 1a and 6b superimpose relatively well.
Extended Data Fig. 8 Allosteric site in hSERT and comparison to ∆-hDAT.
(a) Structural superposition of the allosteric binding pocket for serotonin (5-HT) (PDB: 7LIA) with the equivalent region in ∆-hDAT. The α-carbon to α-carbon distance (yellow dashed line) is indicated in Å. (b) Superposition of the S-citalopram (CIT) (PDB: 5I73) binding pocket with the equivalent region in ∆-hDAT. Superpositions were done using α-carbon atoms of TM3 and TM8. Selected residues and the allosteric molecules are represented as sticks.
Extended Data Fig. 9 Zinc site analysis and alignments with other NSSs.
(a) Isomesh map representation of coulomb density associated with the Zn2+ site in the ∆-hDAT cryo-EM reconstruction, contoured at 8 σ within 2.0 Å of the atoms associated with the structural feature. (b) Alignment of the amino acid sequence encompassing the Zn2+ binding region of ∆-hDAT with the equivalent sequences of related transporters. Residues that directly coordinate Zn2+ in ∆-hDAT are outlined in red. (c)-(e) Alignment of the zinc binding site of ∆-hDAT with the equivalent regions in hSERT (PDB:7LIA), the human glycine transporter (GlyT1; PDB:6ZBV), and the human GABA transporter (GAT1; PDB:7SK2) demonstrates the unique position of EL2 in Zn2+-bound ∆-hDAT compared to related transporters. All structural alignments used α-carbon atoms of TM3 and TM8 of ∆-hDAT. (f,g) Simple models of T211E and T211H were created by substituting the mutated residue for T211 and selecting rotamers with favorable chi1 and chi2 angles to estimate distances to the zinc ion (Å). (h) Inhibition curve for T211E at pH 7.5. Data was analyzed using a nonlinear regression model as described in ‘Methods’. Data from n = 3 biological replicates, each performed in technical triplicate, are represented as mean values +/− standard deviation.
Supplementary information
Supplementary Information
This file contains Supplementary Table 1 and Supplementary Figs. 1–9.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Srivastava, D.K., Navratna, V., Tosh, D.K. et al. Structure of the human dopamine transporter and mechanisms of inhibition. Nature 632, 672–677 (2024). https://doi.org/10.1038/s41586-024-07739-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-024-07739-9
- Springer Nature Limited