Abstract
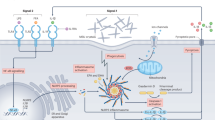
A central aspect of the pathogenesis of gout is elevated urate concentrations, which lead to the formation of monosodium urate crystals. The clinical features of gout result from an individual's immune response to these deposited crystals. Genome-wide association studies (GWAS) have confirmed the importance of urate excretion in the control of serum urate levels and the risk of gout and have identified the kidneys, the gut and the liver as sites of urate regulation. The genetic contribution to the progression from hyperuricaemia to gout remains relatively poorly understood, although genes encoding proteins that are involved in the NLRP3 (NOD-, LRR- and pyrin domain-containing 3) inflammasome pathway play a part. Genome-wide and targeted sequencing is beginning to identify uncommon population-specific variants that are associated with urate levels and gout. Mendelian randomization studies using urate-associated genetic variants as unconfounded surrogates for lifelong urate exposure have not supported claims that urate is causal for metabolic conditions that are comorbidities of hyperuricaemia and gout. Genetic studies have also identified genetic variants that predict responsiveness to therapies (for example, urate-lowering drugs) for treatment of hyperuricaemia. Future research should focus on large GWAS (that include asymptomatic hyperuricaemic individuals) and on increasing the use of whole-genome sequencing data to identify uncommon genetic variants with increased penetrance that might provide opportunities for clinical translation.
Key points
-
The progression to clinically evident gout is thought to occur via several phases — from hyperuricaemia to the deposition of monosodium urate crystals to clinical gout that results from an innate immune response to these crystals.
-
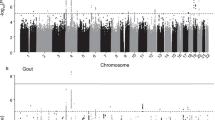
Genome-wide association studies reveal that the genetic basis of hyperuricaemia is dominated by loci containing urate transporters involved in the excretion of urate by the kidneys and the gut.
-
The genetic basis of progression to clinical gout is not well understood, although candidate gene studies have detected association of genes encoding proteins that influence the activation and activity of the NLRP3 (NOD-, LRR- and pyrin domain-containing 3) inflammasome with gout; future research should focus on large genome-wide association studies in gout, including using control asymptomatic individuals with hyperuricaemia.
-
Genome-wide and targeted sequencing data reveal uncommon, population-specific and penetrant genetic variants that provide relatively immediate insights into the pathogenesis of hyperuricaemia and gout and opportunities for clinical translation.
-
Genetic variants that predict the response of individuals with hyperuricaemia to urate-lowering drugs are being identified.




Similar content being viewed by others
References
Dalbeth, N., Merriman, T. R. & Stamp, L. K. Gout. Lancet 388, 2039–2052 (2016).
Zhu, Y., Pandya, B. J. & Choi, H. K. Prevalence of gout and hyperuricemia in the US general population: the national health and nutrition examination survey 2007–2008. Arthritis Rheum. 63, 3136–3141 (2011).
Richette, P., Clerson, P., Perissin, L., Flipo, R. M. & Bardin, T. Revisiting comorbidities in gout: a cluster analysis. Ann. Rheum. Dis. 74, 142–147 (2015).
Robinson, P. C. & Hosburgh, S. Gout: joints and beyond, epidemiology, clinical features, treatment and co-morbidities. Maturitas 78, 245–251 (2014).
Merriman, T. R. An update on the genetic architecture of hyperuricemia and gout. Arthritis Res. Ther. 17, 98 (2015).
Campion, E. W., Glynn, R. J. & DeLabry, L. O. Asymptomatic hyperuricemia. risks and consequences normative aging study. Am. J. Med. 82, 421–426 (1987).
Krishnan, E., Lessov-Schlaggar, C. N., Krasnow, R. E. & Swan, G. E. Nature versus nurture in gout: a twin study. Am. J. Med. 125, 499–504 (2012).
Kalousdian, S., Fabsitz, R., Havlik, R., Christian, J. & Rosenman, R. Heritability of clinical chemistries in an older twin cohort: the NHLBI Twin Study. Genet. Epidemiol. 4, 1–11 (1987).
Whitfield, J. & Martin, N. Inheritance and alcohol as factors influencing plasma uric acid levels. Acta Genet. Med. Gemello 32, 117–126 (1983).
Emmerson, B. T., Nagel, S. L., Duffy, D. L. & Martin, N. G. Genetic control of the renal clearance of urate: a study of twins. Ann. Rheum. Dis. 51, 375–377 (1992).
Monga, M., Macias, B., Groppo, E. & Hargens, A. Genetic heritability of urinary stone risk in identical twins. J. Urol. 175, 2125–2128 (2006).
Nilsson, S. E., Read, S., Berg, S. & Johansson, B. Heritabilities for fifteen routine biochemical values: findings in 215 Swedish twin pairs 82 years of age or older. Scan. J. Clin. Lab. Invest. 69, 562–569 (2009).
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2010).
Kottgen, A. et al. Genome-wide association analyses identify 18 new loci associated with serum urate concentrations. Nat. Genet. 45, 145–154 (2013).
Cadzow, M., Merriman, T. R. & Dalbeth, N. Performance of gout definitions for genetic epidemiological studies: analysis of UK Biobank. Arthritis Res. Ther. 19, 181 (2017).
Perez-Ruiz, F., Calabozo, M., Erauskin, G. G., Ruibal, A. & Herrero-Beites, A. M. Renal underexcretion of uric acid is present in patients with apparent high urinary uric acid output. Arthritis Rheum. 47, 610–613 (2002).
Ichida, K. et al. Decreased extra-renal urate excretion is a common cause of hyperuricemia. Nat. Comm. 3, 764 (2012).
Khanna, D. et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: systematic non-pharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res. 64, 1431–1436 (2012).
Richette, P. et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 76, 29–42 (2017).
Stamp, L. K. et al. Impaired response or insufficient dosage? Examining the potential causes of “inadequate response” to allopurinol in the treatment of gout. Semin. Arthritis Rheum. 44, 170–174 (2014).
Dalbeth, N. et al. Urate crystal deposition in asymptomatic hyperuricaemia and symptomatic gout: a dual energy CT study. Ann. Rheum. Dis. 74, 908–911 (2015).
Roddy, E., Zhang, W. & Doherty, M. Are joints affected by gout also affected by osteoarthritis? Ann. Rheum. Dis. 66, 1374–1377 (2007).
Dalbeth, N. et al. Tendon involvement in the feet of patients with gout: a dual-energy CT study. Ann. Rheum. Dis. 72, 1545–1548 (2013).
Chhana, A., Lee, G. & Dalbeth, N. Factors influencing the crystallization of monosodium urate: a systematic literature review. BMC Musculoskelet. Disord. 16, 296 (2015).
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006).
Dalbeth, N. et al. Cellular characterization of the gouty tophus: a quantitative analysis. Arthritis Rheum. 62, 1549–1556 (2010).
Gibson, T., Waterworth, R., Hatfield, P., Robinson, G. & Bremner, K. Hyperuricaemia, gout and kidney function in New Zealand Maori men. Br. J. Rheumatol. 23, 276–282 (1984).
Okada, Y. et al. Meta-analysis identifies multiple loci associated with kidney function-related traits in east Asian populations. Nat. Genet. 44, 904–909 (2012).
Tin, A. et al. Genome-wide association study for serum urate concentrations and gout among African Americans identifies genomic risk loci and a novel URAT1 loss-of-function allele. Hum. Mol. Genet. 20, 4056–4068 (2011).
Dehghan, A. et al. Association of three genetic loci with uric acid concentration and risk of gout: a genome-wide association study. Lancet 372, 1953–1961 (2008).
Li, C. et al. Genome-wide association analysis identifies three new risk loci for gout arthritis in Han Chinese. Nat. Comm. 6, 7041 (2015).
Li, Z. et al. Replication of gout/urate concentrations GWAS susceptibility loci associated with gout in a Han Chinese population. Sci. Rep. 7, 4094 (2017).
Matsuo, H. et al. Genome-wide association study of clinically defined gout identifies multiple risk loci and its association with clinical subtypes. Ann. Rheum. Dis. 75, 652–659 (2016).
Phipps-Green, A. J. et al. Twenty-eight loci that influence serum urate levels: analysis of association with gout. Ann. Rheum. Dis. 75, 124–130 (2016).
Urano, W. et al. Effect of genetic polymorphisms on development of gout. J. Rheumatol. 40, 1374–1378 (2013).
Batt, C. et al. Sugar-sweetened beverage consumption: a risk factor for prevalent gout with SLC2A9 genotype-specific effects on serum urate and risk of gout. Ann. Rheum. Dis. 73, 2101–2106 (2013).
McKinney, C. et al. Multiplicative interaction of functional inflammasome genetic variants in determining the risk of gout. Arthritis Res. Ther. 17, 288 (2015).
Rasheed, H., Stamp, L. K., Dalbeth, N. & Merriman, T. R. Interaction of the GCKR and A1CF loci with alcohol consumption to influence the risk of gout. Arthritis Res. Ther. 19, 161 (2017).
Rasheed, H. et al. Association of the lipoprotein receptor-related protein 2 gene with gout and non-additive interaction with alcohol consumption. Arthritis Res. Ther. 15, R177 (2013).
Scharpf, R. B. et al. Copy number polymorphisms near SLC2A9 are associated with serum uric acid concentrations. BMC Genet. 15, 81 (2014).
Higashino, T. et al. Multiple common and rare variants of ABCG2 cause gout. RMD Open 3, e000464 (2017).
Stiburkova, B. et al. Functional non-synonymous variants of ABCG2 and gout risk. Rheumatology 56, 1982–1992 (2017).
Boocock, J. et al. Trans-ancestral meta-analysis identifies nine new loci associated with serum uric acid concentrations [abstract]. Arthritis Rheumatol. 68 (Suppl. 10), 2273 (2016).
Merriman, T. R., Choi, H. K. & Dalbeth, N. The genetic basis of gout. Rheum. Dis. Clin. North Am. 40, 279–290 (2014).
Boyle, E. A., Li, Y. I. & Pritchard, J. K. An expanded view of complex traits: from polygenic to omnigenic. Cell 169, 1177–1186 (2017).
Doring, A. et al. SLC2A9 influences uric acid concentrations with pronounced sex-specific effects. Nat. Genet. 40, 430–436 (2008).
Vitart, V. et al. SLC2A9 is a newly identified urate transporter influencing serum urate concentration, urate excretion and gout. Nat. Genet. 40, 437–442 (2008).
Cleophas, M. et al. ABCG2 polymorphisms in gout: insights into disease susceptibility and treatment approaches. Pharmacogenomics Pers. Med. 10, 129–142 (2017).
Merriman, T. R. et al. Non-coding genetic variant maximally associated with serum urate levels is functionally linked to HNF4A-dependent PDZK1 expression [abstract]. Arthritis Rheumatol. 69, S10 (2017).
Beer, N. L. et al. The P446L variant in GCKR associated with fasting plasma glucose and triglyceride levels exerts its effect through increased glucokinase activity in liver. Hum. Mol. Genet. 18, 4081–4088 (2009).
Rees, M. et al. Cellular characterisation of the GCKR P446L variant associated with type 2 diabetes risk. Diabetologia 55, 114–122 (2012).
Sakiyama, M. et al. Identification of rs671, a common variant of ALDH2, as a gout susceptibility locus. Sci. Rep. 6, 25360 (2016).
Sulem, P. et al. Identification of low-frequency variants associated with gout and serum uric acid levels. Nat. Genet. 43, 1127–1130 (2011).
Shin, J., Kim, Y., Kong, M. & Lee, C. Genetic architecture for susceptibility to gout in the KARE cohort study. J. Hum. Genet. 57, 379–384 (2012).
Kuo, J. Z. et al. Trans-ethnic fine mapping identifies a novel independent locus at the 3' end of CDKAL1 and novel variants of several susceptibility loci for type 2 diabetes in a Han Chinese population. Diabetologia 56, 2619–2628 (2013).
Nakayama, A. et al. GWAS of clinically defined gout and subtypes identifies multiple susceptibility loci that include urate transporter genes. Ann. Rheum. Dis. 76, 869–877 (2017).
Li, C. et al. Genetic variants associated with tophi occurrence by a genome wide association study of 1888 patients. Gout and Hyperuricemia 4, 12–20 (2017).
Merriman, T. R. et al. Genome-wide association study of gout in people of European ancestry [abstract]. Arthritis Rheumatol. 69, S10 (2017).
Vasiliou, V. et al. ALDH16A1 is a novel non-catalytic enzyme that may be involved in the etiology of gout via protein–protein interactions with HPRT1. Chem. Biol. Interact. 202, 22–31 (2013).
Charkoftaki, G. et al. Transcriptomic analysis and plasma metabolomics in Aldh16a1-null mice reveals a potential role of ALDH16A1 in renal function. Chem. Biol. Interact. 276, 15–22 (2017).
Hollis-Moffatt, J. E. et al. The renal urate transporter SLC17A1 locus: confirmation of association with gout. Arthritis Res. Ther. 14, R92 (2012).
Tanner, C. et al. Population-specific resequencing associates the ATP-binding cassette subfamily C member 4 gene with gout in New Zealand Māori and Pacific men. Arthritis Rheumatol. 69, 1461–1469 (2017).
Farrés, J. et al. Effects of changing glutamate 487 to lysine in rat and human liver mitochondrial aldehyde dehydrogenase. A model to study human (Oriental type) class 2 aldehyde dehydrogenase. J. Biol. Chem. 269, 13854–13860 (1994).
Yamanaka, H. et al. Analysis of the genotypes for aldehyde dehydrogenase 2 in Japanese patients with primary gout. Adv. Exp. Med. Biol. 370, 53–56 (1994).
Qing, Y. F. et al. Association of TLR4 gene rs2149356 polymorphism with primary gouty arthritis in a case-control study. PLoS ONE 8, e64845 (2013).
Rasheed, H. et al. The Toll-like receptor 4 (TLR4) variant rs2149356 and risk of gout in European and Polynesian sample sets. PLoS ONE 11, e0147939 (2016).
Chen, Y. et al. CARD 8 rs2043211 polymorphism is associated with gout in a Chinese male population. Cell. Physiol. Biochem. 35, 1394–1400 (2015).
Landvik, N. E. et al. A specific interleukin-1B haplotype correlates with high levels of IL1B mRNA in the lung and increased risk of non-small cell lung cancer. Carcinogenesis 30, 1186–1192 (2009).
Delgado-Lista, J. et al. Interleukin 1B variant-1473G/C (rs1143623) influences triglyceride and interleukin 6 metabolism. J. Clin. Endocrinol. Metab. 96, E816–E820 (2011).
Chang, W. C. et al. Genetic variants of PPAR-gamma coactivator 1B augment NLRP3-mediated inflammation in gouty arthritis. Rheumatology 56, 457–466 (2017).
Shaukat, A. et al. Replication of genetic association of peroxisome proliferator-activated receptor gamma-1B with gout in a New Zealand Polynesian sample set [abstract]. Arthritis Rheumatol. 69 (Suppl. 10), 1127 (2017).
Gosling, A. L. et al. Mitochondrial genetic variation and susceptibility to gout in Maori and Pacific people living in Aotearoa New Zealand. Ann. Rheum. Dis. 77, 571–578 (2017).
Merriman, T. & Terkeltaub, R. PPARGC1B: insight into the expression of the gouty inflammation phenotype: PPARGC1B and gouty inflammation. Rheumatology 56, 323–325 (2017).
Cardona, F. et al. Contribution of polymorphisms in the apolipoprotein AI-CIII-AIV cluster to hyperlipidaemia in patients with gout. Ann. Rheum. Dis. 64, 85–88 (2005).
Rasheed, H. et al. Replication of association of the apolipoprotein A1-C3-A4 gene cluster with the risk of gout. Rheumatology 55, 1421–1430 (2016).
Hyka, N. et al. Apolipoprotein AI inhibits the production of interleukin-1β and tumor necrosis factor-α by blocking contact-mediated activation of monocytes by T lymphocytes. Blood 97, 2381–2389 (2001).
Chiang, S. et al. Increased level of MSU crystal-bound protein apolipoprotein AI in acute gouty arthritis. Scand. J. Rheumatol. 43, 498–502 (2014).
CriȘan, T. O. et al. Soluble uric acid primes TLR-induced proinflammatory cytokine production by human primary cells via inhibition of IL-1Ra. Ann. Rheum. Dis. 75, 755–762 (2016).
Dong, Z. et al. Effects of multiple genetic loci on the pathogenesis from serum urate to gout. Sci. Rep. 7, 43614 (2017).
Merriman, T. R. et al. Pleiotropic effect of ABCG2 in gout [abstract]. Arthritis Rheumatol. 68 (Suppl. 10), 2276 (2016).
Basseville, A. et al. Histone deacetylase inhibitors influence chemotherapy transport by modulating expression and trafficking of a common polymorphic variant of the ABCG2 efflux transporter. Canc. Res. 72, 3642–3651 (2012).
Wang, J. et al. Association between gout and polymorphisms in GCKR in male Han Chinese. Hum. Genet. 131, 1261–1265 (2012).
Kanbay, M. et al. Uric acid in metabolic syndrome: from an innocent bystander to a central player. Eur. J. Intern. Med. 29, 3–8 (2016).
Robinson, P. C., Choi, H. K., Do, R. & Merriman, T. R. Insight into rheumatological cause and effect through the use of Mendelian randomization. Nat. Rev. Rheumatol. 12, 486–496 (2016).
Hughes, K., Flynn, T., de Zoysa, J., Dalbeth, N. & Merriman, T. R. Mendelian randomization analysis associates increased serum urate, due to genetic variation in uric acid transporters, with improved renal function. Kidney Int. 85, 344–351 (2014).
Lyngdoh, T. et al. Serum uric acid and adiposity: deciphering causality using a bidirectional Mendelian randomization approach. PLoS ONE 7, e39321 (2012).
Rasheed, H., Hughes, K., Flynn, T. J. & Merriman, T. R. Mendelian randomization provides no evidence for a causal role of serum urate in increasing serum triglyceride levels. Circ. Cardiovasc. Genet. 7, 830–837 (2014).
Palmer, T. M. et al. Association of plasma uric acid with ischaemic heart disease and blood pressure: Mendelian randomisation analysis of two large cohorts. Br. Med. J. 347, f4262 (2013).
Keenan, T. et al. Causal assessment of serum urate levels in cardiometabolic diseases through a Mendelian randomization study. J. Am. Coll. Cardiol. 67, 407–416 (2016).
Sluijs, I. et al. A Mendelian randomization study of circulating uric acid and type 2 diabetes. Diabetes 64, 3028–3036 (2015).
White, J. et al. Plasma urate concentration and risk of coronary heart disease: a Mendelian randomisation analysis. Lancet Diabetes Endocrinol. 4, 327–336 (2016).
Dalbeth, N., Stamp, L. K. & Merriman, T. R. The genetics of gout: towards personalised medicine? BMC Med. 15, 108 (2017).
Ko, T. M. et al. Use of HLA-B*58:01 genotyping to prevent allopurinol induced severe cutaneous adverse reactions in Taiwan: national prospective cohort study. BMJ 351, h4848 (2015).
He, W., Phipps-Green, A., Stamp, L. K., Merriman, T. R. & Dalbeth, N. Population-specific association between ABCG2 variants and tophaceous disease in people with gout. Arthritis Res. Ther. 19, 43 (2017).
Spiliopoulou, A. et al. Genomic prediction of complex human traits: relatedness, trait architecture and predictive meta-models. Hum. Mol. Genet. 24, 4167–4182 (2015).
Wen, C. C. et al. Genome-wide association study identifies ABCG2 (BCRP) as an allopurinol transporter and a determinant of drug response. Clin. Pharmacol. Ther. 97, 518–525 (2015).
Roberts, R. L. et al. ABCG2 loss-of-function polymorphism predicts poor response to allopurinol in patients with gout. Pharmacogenomics J. 17, 201–203 (2017).
Wallace, M. C. et al. Association between ABCG2 rs2231142 and poor response to allopurinol: replication and meta-analysis. Rheumatology 57, 656–660 (2018).
Ichida, K. et al. Clinical and molecular analysis of patients with renal hypouricemia in Japan-influence of URAT1 gene on urinary urate excretion. J. Am. Soc. Nephrol. 15, 164–173 (2004).
Charles, B. A. et al. A genome-wide association study of serum uric acid in African Americans. BMC Med. Genomics 4, 17 (2011).
Chittoor, G. et al. Genetic variation underlying renal uric acid excretion in Hispanic children: the Viva La Familia Study. BMC Med. Genet. 18, 6 (2017).
Giri, A. K. et al. Genome wide association study of uric acid in Indian population and interaction of identified variants with type 2 diabetes. Sci. Rep. 6, 21440 (2016).
Huffman, J. E. et al. Modulation of genetic associations with serum urate levels by body-mass-index in humans. PLoS ONE 10, e0119752 (2015).
Kamatani, Y. et al. Genome-wide association study of hematological and biochemical traits in a Japanese population. Nat. Genet. 42, 210–215 (2010).
Karns, R. et al. Genome-wide association of serum uric acid concentration: replication of sequence variants in an island population of the Adriatic Coast of Croatia. Ann. Hum. Genet. 76, 121–127 (2012).
Kenny, E. E. et al. Increased power of mixed models facilitates association mapping of 10 loci for metabolic traits in an isolated population. Hum. Mol. Genet. 20, 827–839 (2010).
Kolz, M. et al. Meta-analysis of 28,141 individuals identifies common variants within five new loci that influence uric acid concentrations. PLoS Genet. 5, 1000504 (2009).
Korostishevsky, M. et al. Genomics and metabolomics of muscular mass in a community-based sample of UK females. Eur. J. Hum. Genet. 24, 277–283 (2016).
Li, S. et al. The GLUT9 gene is associated with serum uric acid levels in Sardinia and Chianti cohorts. PLoS Genet. 3, e194 (2007).
Li, W. D. et al. A genome wide association study of plasma uric acid levels in obese cases and never-overweight controls. Obesity 21, E490–E494 (2013).
McArdle, P. F. et al. Association of a common nonsynonymous variant in GLUT9 with serum uric acid levels in Old Order Amish. Arthritis Rheum. 58, 2874–2881 (2008).
Middelberg, R. P. et al. Genetic variants in LPL, OASL and TOMM40/APOE-C1-C2-C4 genes are associated with multiple cardiovascular-related traits. BMC Med. Genet. 12, 123 (2011).
Nagy, R. et al. Exploration of haplotype research consortium imputation for genome-wide association studies in 20,032 Generation Scotland participants. Genome Med. 9, 23 (2017).
Merriman, T. R. et al. Trans-ancestral meta-analysis identifies 13 new loci associated with serum urate levels [abstract OP0263]. Ann. Rheum. Dis. 76 (Suppl. 2) 165 (2017).
Son, C.-N. et al. ABCG2 polymorphism is associated with hyperuricemia in a study of a community-based Korean cohort. J. Korean Med. Sci. 32, 1451–1459 (2017).
Suhre, K. et al. Human metabolic individuality in biomedical and pharmaceutical research. Nature 477, 54–60 (2011).
Voruganti, V. S. et al. Genome-wide association analysis confirms and extends the association of SLC2A9 with serum uric acid levels to Mexican Americans. Front. Genet. 4, 279 (2013).
Wallace, C. et al. Genome-wide association study identifies genes for biomarkers of cardiovascular disease: serum urate and dyslipidemia. Am. J. Hum. Genet. 82, 139–149 (2008).
Yang, Q. O. et al. Multiple genetic loci influence serum urate levels and their relationship with gout and cardiovascular disease risk factors. Circ. Cardiovasc. Genet. 3, 523–530 (2010).
Zemunik, T. et al. Genome-wide association study of biochemical traits in Korčula Island, Croatia. Croat. Med. J. 50, 23–33 (2009).
Yang, B. et al. A genome-wide association study identifies common variants influencing serum uric acid concentrations in a Chinese population. BMC Med. Genomics 7, 10 (2014).
Reynolds, R. J. et al. Serum urate gene associations with incident gout, measured in the Framingham heart study, are modified by renal disease and not by body mass index. Rheumatol. Int. 36, 263–270 (2016).
Stark, K. et al. Common polymorphisms influencing serum uric acid levels contribute to susceptibility to gout, but not to coronary artery disease. PLoS ONE 4, e7729 (2009).
Zhang, Y. et al. Associations of gout with polymorphisms in SLC2A9, WDR1, CLNK, PKD2, and ABCG2 Chinese Han and Tibetan populations. Int. J. Clin. Exp. Pathol. 9, 7503–7517 (2016).
Zhou, Z. W. et al. Polymorphisms in GCKR, SLC17A1 and SLC22A12 were associated with phenotype gout in Han Chinese males: a case-control study. BMC Med. Genet. 16, 66 (2015).
Zheng, C., Yang, H., Wang, Q., Rao, H. & Diao, Y. Association analysis of five SNP variants with gout in the Minnan population in China. Turk. J. Med. Sci. 46, 361–367 (2016).
Kim, Y. S. et al. Genetic analysis of ABCG2 and SLC2A9 gene polymorphisms in gouty arthritis in a Korean population. Korean J. Intern. Med. 30, 913–920 (2015).
Wan, W., Xu, X., Zhao, D., Pang, Y. & Wang, Y. Polymorphisms of uric transporter proteins in the pathogenesis of gout in a Chinese Han population. Genet. Mol. Res. 14, 2546–2550 (2015).
Matsuo, H. et al. Common defects of ABCG2, a high-capacity urate exporter, cause gout: a function-based genetic analysis in a Japanese population. Sci. Transl. Med. 1, 5ra11 (2009).
Li, R. et al. A meta-analysis of the associations between the Q141K and Q126X gene variants and gout risk. Int. J. Clin. Exp. Pathol. 8, 9812–9823 (2015).
Sakiyama, M. et al. The effects of URAT1/SLC22A12 nonfunctional variants, R90H and W258X, on serum uric acid levels and gout/hyperuricemia progression. Sci. Rep. 6, 20148 (2016).
Schunkert, H. et al. Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat. Genet. 43, 333–338 (2011).
Acknowledgements
The authors thank the Health Research Council of New Zealand and the University of Otago, Dunedin, New Zealand, for salary funding for T.J.M. and T.R.M.
Reviewer information
Nature Reviews Rheumatology thanks K. Pavelka and the other, anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
T.J.M., N.D. and T.R.M. contributed to researching data for the article; N.D., E.A.S. and T.R.M. contributed to discussion of content; N.D. and T.R.M. contributed to writing the article; and T.J.M., N.D. and E.A.S. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
N.D. declares that she has received consulting fees, speaker fees or grants from Ardea/AstraZeneca, Cymabay, Crealta, Horizon and Takeda, which have developed or marketed urate-lowering therapies for management of gout. T.R.M. declares that he has received consulting fees, speaker fees or grants from Ardea/AstraZeneca and Horizon, which have developed or marketed urate-lowering therapies for management of gout. The other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
1000 Genomes: www.internationalgenome.org
Genome Aggregation Database: gnomAD.broadinstitute.org
Trans-Omics for Precision Medicine (TOPMed): www.nhlbiwgs.org
Supplementary Information
41584_2018_4_MOESM1_ESM.pdf
Supplementary Figure S1 Genetic epidemiological (Mendelian randomization) evidence that serum urate is not causal for comorbidities associated with gout
Rights and permissions
About this article
Cite this article
Major, T.J., Dalbeth, N., Stahl, E.A. et al. An update on the genetics of hyperuricaemia and gout. Nat Rev Rheumatol 14, 341–353 (2018). https://doi.org/10.1038/s41584-018-0004-x
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-018-0004-x
- Springer Nature Limited
This article is cited by
-
Atavistic strategy for the treatment of hyperuricemia via ionizable liposomal mRNA
Nature Communications (2024)
-
Hyperuricemia and its related diseases: mechanisms and advances in therapy
Signal Transduction and Targeted Therapy (2024)
-
Social Listening in Gout: Impact of Proactive vs. Reactive Management on Self-Reported Emotional States
Rheumatology and Therapy (2024)
-
Recent developments in selective laser processes for wearable devices
Bio-Design and Manufacturing (2024)
-
Lactobacillus rhamnosus GG ameliorates hyperuricemia in a novel model
npj Biofilms and Microbiomes (2024)