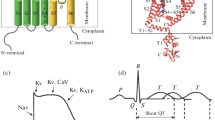
Abstract
Voltage-gated ion channels (VGICs), including those for Na+, Ca2+ and K+, selectively permeate ions across the cell membrane in response to changes in membrane potential, thus participating in physiological processes involving electrical signalling, such as neurotransmission, muscle contraction and hormone secretion. Aberrant function or dysregulation of VGICs is associated with a diversity of neurological, psychiatric, cardiovascular and muscular disorders, and approximately 10% of FDA-approved drugs directly target VGICs. Understanding the structure–function relationship of VGICs is crucial for our comprehension of their working mechanisms and role in diseases. In this Review, we discuss how advances in single-particle cryo-electron microscopy have afforded unprecedented structural insights into VGICs, especially on their interactions with clinical and investigational drugs. We present a comprehensive overview of the recent advances in the structural biology of VGICs, with a focus on how prototypical drugs and toxins modulate VGIC activities. We explore how these structures elucidate the molecular basis for drug actions, reveal novel pharmacological sites, and provide critical clues to future drug discovery.





Similar content being viewed by others
References
Galvani, L. De viribus electricitatis in motu musculari commentarius. Bon Sci. Art. Inst. Acad. Comm. 7, 363–418 (1791).
Cole, K. S. & Curtis, H. J. Electric impedance of the squid giant axon during activity. J. Gen. Physiol. 22, 649–670 (1939).
Hodgkin, A. L. & Huxley, A. F. Action potentials recorded from inside a nerve fibre. Nature 144, 710–711 (1939).
Hodgkin, A. L. & Huxley, A. F. Resting and action potentials in single nerve fibres. J. Physiol. 104, 176–195 (1945).
Hodgkin, A. L. & Huxley, A. F. Currents carried by sodium and potassium ions through the membrane of the giant axon of Loligo. J. Physiol. 116, 449–472 (1952).
Hodgkin, A. L. & Huxley, A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 117, 500–544 (1952).
Neher, E. & Sakmann, B. Single-channel currents recorded from membrane of denervated frog muscle fibres. Nature 260, 799–802 (1976).
Hamill, O. P., Marty, A., Neher, E., Sakmann, B. & Sigworth, F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflug. Arch. 391, 85–100 (1981).
Verkhratsky, A., Krishtal, O. A. & Petersen, O. H. From Galvani to patch clamp: the development of electrophysiology. Pflug. Arch. 453, 233–247 (2006). This review article summarizes the study history of biological electricity.
Strazzullo, P. & Leclercq, C. Sodium. Adv. Nutr. 5, 188–190 (2014).
Zacchia, M., Abategiovanni, M. L., Stratigis, S. & Capasso, G. Potassium: from physiology to clinical implications. Kidney Dis. 2, 72–79 (2016).
Ringer, S. A further contribution regarding the influence of the different constituents of the blood on the contraction of the heart. J. Physiol. 4, 29–42.23 (1883).
Reuter, H. The dependence of slow inward current in Purkinje fibres on the extracellular calcium-concentration. J. Physiol. 192, 479–492 (1967).
Bers, D. M. Cardiac excitation-contraction coupling. Nature 415, 198–205 (2002).
Clapham, D. E. Calcium signaling. Cell 131, 1047–1058 (2007).
Hursh, J. B. Conduction velocity and diameter of nerve fibers. Am. J. Physiol. 127, 131–139 (1939).
Waxman, S. G. Determinants of conduction velocity in myelinated nerve fibers. Muscle Nerve 3, 141–150 (1980).
Hille, B. Ion Channels of Excitable Membranes 3rd edn (Sinauer, 2001).
Bean, B. P. The action potential in mammalian central neurons. Nat. Rev. Neurosci. 8, 451–465 (2007).
Santos, R. et al. A comprehensive map of molecular drug targets. Nat. Rev. Drug Discov. 16, 19–34 (2017).
Bagal, S. K. et al. Ion channels as therapeutic targets: a drug discovery perspective. J. Med. Chem. 56, 593–624 (2013).
Frazao, B., Vasconcelos, V. & Antunes, A. Sea anemone (Cnidaria, Anthozoa, Actiniaria) toxins: an overview. Mar. Drugs 10, 1812–1851 (2012).
Llewellyn, L. E. Saxitoxin, a toxic marine natural product that targets a multitude of receptors. Nat. Prod. Rep. 23, 200–222 (2006).
Dolphin, A. C. A short history of voltage-gated calcium channels. Br. J. Pharmacol. 147, S56–S62 (2006). This brief review elegantly summarizes the research history and general knowledge of voltage-gated calcium channels.
Jan, L. Y. & Jan, Y. N. Voltage-gated potassium channels and the diversity of electrical signalling. J. Physiol. 590, 2591–2599 (2012).
Huang, X. & Jan, L. Y. Targeting potassium channels in cancer. J. Cell Biol. 206, 151–162 (2014).
Zamponi, G. W., Striessnig, J., Koschak, A. & Dolphin, A. C. The physiology, pathology, and pharmacology of voltage-gated calcium channels and their future therapeutic potential. Pharmacol. Rev. 67, 821–870 (2015). This is a comprehensive and updated review on voltage-gated calcium channels.
Ahern, C. A., Payandeh, J., Bosmans, F. & Chanda, B. The hitchhiker’s guide to the voltage-gated sodium channel galaxy. J. Gen. Physiol. 147, 1–24 (2016). This comprehensive review affords an updated overview of voltage-gated sodium channels.
Alsaloum, M., Higerd, G. P., Effraim, P. R. & Waxman, S. G. Status of peripheral sodium channel blockers for non-addictive pain treatment. Nat. Rev. Neurol. 16, 689–705 (2020). This is a timely review that summarizes efforts on drug discovery targeting peripheral voltage-gated sodium channels for pain relief.
Meisler, M. H., Hill, S. F. & Yu, W. Sodium channelopathies in neurodevelopmental disorders. Nat. Rev. Neurosci. 22, 152–166 (2021).
Brunklaus, A. et al. Gene variant effects across sodium channelopathies predict function and guide precision therapy. Brain 145, 4275–4286 (2022).
Kühlbrandt, W. The resolution revolution. Science 343, 1443–1444 (2014).
Payandeh, J., Scheuer, T., Zheng, N. & Catterall, W. A. The crystal structure of a voltage-gated sodium channel. Nature 475, 353–358 (2011).
Zhang, X. et al. Crystal structure of an orthologue of the NaChBac voltage-gated sodium channel. Nature 486, 130–134 (2012).
Gao, S. et al. Employing NaChBac for cryo-EM analysis of toxin action on voltage-gated Na+ channels in nanodisc. Proc. Natl Acad. Sci. USA 117, 14187–14193 (2020).
Lu, Y. et al. Structural basis for the activity regulation of a potassium channel AKT1 from Arabidopsis. Nat. Commun. 13, 5682 (2022).
Guo, J. et al. Structure of the voltage-gated two-pore channel TPC1 from Arabidopsis thaliana. Nature 531, 196–201 (2016).
Kintzer, A. F. & Stroud, R. M. Structure, inhibition and regulation of two-pore channel TPC1 from Arabidopsis thaliana. Nature 531, 258–262 (2016).
Hedrich, R. Ion channels in plants. Physiol. Rev. 92, 1777–1811 (2012).
Martinac, B., Saimi, Y. & Kung, C. Ion channels in microbes. Physiol. Rev. 88, 1449–1490 (2008).
de Lera Ruiz, M. & Kraus, R. L. Voltage-gated sodium channels: structure, function, pharmacology, and clinical indications. J. Med. Chem. 58, 7093–7118 (2015).
Long, S. B., Campbell, E. B. & MacKinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 309, 897–903 (2005). This research article reports the high-resolution crystal structure of a prototypical Kv channel.
Shen, H. et al. Structure of a eukaryotic voltage-gated sodium channel at near-atomic resolution. Science 355, eaal4326 (2017). This research article reports the first structure of a eukaryotic voltage-gated sodium channel.
Wu, J. P. et al. Structure of the voltage-gated calcium channel Cav1.1 complex. Science 350, eaad2395 (2015). This research article reports the first structure of a eukaryotic Cav channel complex.
Whicher, J. R. & MacKinnon, R. Structure of the voltage-gated K+ channel Eag1 reveals an alternative voltage sensing mechanism. Science 353, 664–669 (2016). This research article reports the first structure of a domain-non-swapped VGIC, expanding our understanding of the VGIC fold.
Lee, C. H. & MacKinnon, R. Structures of the human HCN1 hyperpolarization-activated channel. Cell 168, 111–120.e11 (2017).
Li, M. et al. Structure of a eukaryotic cyclic-nucleotide-gated channel. Nature 542, 60–65 (2017).
Tao, X., Hite, R. K. & MacKinnon, R. Cryo-EM structure of the open high-conductance Ca2+-activated K+ channel. Nature 541, 46–51 (2017).
Hite, R. K., Tao, X. & MacKinnon, R. Structural basis for gating the high-conductance Ca2+-activated K+ channel. Nature 541, 52–57 (2017).
Yellen, G. The moving parts of voltage-gated ion channels. Q. Rev. Biophys. 31, 239–295 (1998).
Noda, M. et al. Primary structure of Electrophorus electricus sodium channel deduced from cDNA sequence. Nature 312, 121–127 (1984).
Schoppa, N. E., McCormack, K., Tanouye, M. A. & Sigworth, F. J. The size of gating charge in wild-type and mutant Shaker potassium channels. Science 255, 1712–1715 (1992).
Yang, N. & Horn, R. Evidence for voltage-dependent S4 movement in sodium channels. Neuron 15, 213–218 (1995).
Aggarwal, S. K. & MacKinnon, R. Contribution of the S4 segment to gating charge in the Shaker K+ channel. Neuron 16, 1169–1177 (1996).
Tempel, B. L., Papazian, D. M., Schwarz, T. L., Jan, Y. N. & Jan, L. Y. Sequence of a probable potassium channel component encoded at Shaker locus of Drosophila. Science 237, 770–775 (1987).
Tao, X., Lee, A., Limapichat, W., Dougherty, D. A. & MacKinnon, R. A gating charge transfer center in voltage sensors. Science 328, 67–73 (2010).
Doyle, D. A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998). This landmark research article reports the Nobel-winning crystal structure o KcsA.
Aryal, P., Sansom, M. S. P. & Tucker, S. J. Hydrophobic gating in ion channels. J. Mol. Biol. 427, 121–130 (2015).
Liao, M., Cao, E., Julius, D. & Cheng, Y. Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature 504, 107–112 (2013). This reseach article exemplifes the resolution revolution of cryo-EM, which marks the beginning of a new era of structral biology.
Yan, Z. et al. Structure of the rabbit ryanodine receptor RyR1 at near-atomic resolution. Nature 517, 50–55 (2015).
Zalk, R. et al. Structure of a mammalian ryanodine receptor. Nature 517, 44–49 (2015).
Peng, W. et al. Structural basis for the gating mechanism of the type 2 ryanodine receptor RyR2. Science 354, aah5324 (2016).
Ren, D. et al. A sperm ion channel required for sperm motility and male fertility. Nature 413, 603–609 (2001).
Lin, S., Ke, M., Zhang, Y., Yan, Z. & Wu, J. Structure of a mammalian sperm cation channel complex. Nature 595, 746–750 (2021).
Julius, D. TRP channels and pain. Annu. Rev. Cell Dev. Biol. 29, 355–384 (2013).
Ramsey, I. S., Delling, M. & Clapham, D. E. An introduction to TRP channels. Annu. Rev. Physiol. 68, 619–647 (2006).
Woll, K. A. & Van Petegem, F. Calcium-release channels: structure and function of IP3 receptors and ryanodine receptors. Physiol. Rev. 102, 209–268 (2022).
Jentsch, T. J. & Pusch, M. CLC chloride channels and transporters: structure, function, physiology, and disease. Physiol. Rev. 98, 1493–1590 (2018).
DeCoursey, T. E. Voltage-gated proton channels: molecular biology, physiology, and pathophysiology of the H(V) family. Physiol. Rev. 93, 599–652 (2013).
Catterall, W. A., Goldin, A. L. & Waxman, S. G. International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels. Pharmacol. Rev. 57, 397–409 (2005).
Mosher, H. S., Fuhrman, F. A., Buchwald, H. D. & Fischer, H. G. Tarichatoxin-tetrodotoxin: a potent neurotoxin. Science 144, 1100–1110 (1964).
Reed, J. K. & Raftery, M. A. Properties of the tetrodotoxin binding component in plasma membranes isolated from Electrophorus electricus. Biochemistry 15, 944–953 (1976).
Satin, J. et al. A mutant of TTX-resistant cardiac sodium channels with TTX-sensitive properties. Science 256, 1202–1205 (1992).
Akopian, A. N., Sivilotti, L. & Wood, J. N. A tetrodotoxin-resistant voltage-gated sodium channel expressed by sensory neurons. Nature 379, 257–262 (1996).
Tate, S. et al. Two sodium channels contribute to the TTX-R sodium current in primary sensory neurons. Nat. Neurosci. 1, 653–655 (1998).
Goldin, A. L. Resurgence of sodium channel research. Annu. Rev. Physiol. 63, 871–894 (2001).
Watanabe, E. et al. Nav2/NaG channel is involved in control of salt-intake behavior in the CNS. J. Neurosci. 20, 7743–7751 (2000).
Noland, C. L. et al. Structure-guided unlocking of NaX reveals a non-selective tetrodotoxin-sensitive cation channel. Nat. Commun. 13, 1416 (2022).
Pan, X. et al. Structure of the human voltage-gated sodium channel Nav1.4 in complex with β1. Science 362, eaau2486 (2018). This research article reports the first structure of a human Nav channel, allowing precise mapping of many pathogenic mutations.
Yan, Z. et al. Structure of the Nav1.4-β1 complex from electric eel. Cell 170, 470–482.e11 (2017). This article reports the structure of a Nav channel isolated from electric eel, which exhibits a different conformation from that of NavPaS. An allosteric blocking mechanism was proposed to account for fast inactivation of Nav channels based on structural comparison of EeNav1.4 and NavPaS.
Pan, X. et al. Comparative structural analysis of human Nav1.1 and Nav1.5 reveals mutational hotspots for sodium channelopathies. Proc. Natl Acad. Sci. USA 118, e2100066118 (2021).
Shen, H., Liu, D., Wu, K., Lei, J. & Yan, N. Structures of human Nav1.7 channel in complex with auxiliary subunits and animal toxins. Science 363, 1303–1308 (2019).
Li, Z., Wu, Q. & Yan, N. A structural atlas of druggable sites on Nav channels. Channels 18, 2287832 (2024).
Pan, X. et al. Molecular basis for pore blockade of human Na+ channel Nav1.2 by the μ-conotoxin KIIIA. Science 363, 1309–1313 (2019).
O’Malley, H. A. & Isom, L. L. Sodium channel beta subunits: emerging targets in channelopathies. Annu. Rev. Physiol. 77, 481–504 (2015). This is a comprehensive review on the auxiliary subunits of Nav channels.
Catterall, W. A., Perez-Reyes, E., Snutch, T. P. & Striessnig, J. International Union of Pharmacology. XLVIII. Nomenclature and structure-function relationships of voltage-gated calcium channels. Pharmacol. Rev. 57, 411–425 (2005).
Nowycky, M. C., Fox, A. P. & Tsien, R. W. Three types of neuronal calcium channel with different calcium agonist sensitivity. Nature 316, 440–443 (1985).
Sather, W. A. et al. Distinctive biophysical and pharmacological properties of class A (BI) calcium channel alpha 1 subunits. Neuron 11, 291–303 (1993).
Randall, A. & Tsien, R. W. Pharmacological dissection of multiple types of Ca2+ channel currents in rat cerebellar granule neurons. J. Neurosci. 15, 2995–3012 (1995).
Williams, M. E. et al. Structure and functional expression of an omega-conotoxin-sensitive human N-type calcium channel. Science 257, 389–395 (1992).
Ellinor, P. T. et al. Functional expression of a rapidly inactivating neuronal calcium channel. Nature 363, 455–458 (1993).
Tsien, R. W., Lipscombe, D., Madison, D. V., Bley, K. R. & Fox, A. P. Multiple types of neuronal calcium channels and their selective modulation. Trends Neurosci. 11, 431–438 (1988).
Yao, X., Gao, S. & Yan, N. Structural biology of voltage-gated calcium channels. Channels 18, 2290807 (2024).
Wu, J. et al. Structure of the voltage-gated calcium channel Cav1.1 at 3.6 A resolution. Nature 537, 191–196 (2016).
Zhao, Y. et al. Cryo-EM structures of apo and antagonist-bound human Cav3.1. Nature 576, 492–497 (2019).
Gao, S., Yao, X. & Yan, N. Structure of human Cav2.2 channel blocked by the painkiller ziconotide. Nature 596, 143–147 (2021).
Yao, X. et al. Structures of the R-type human Cav2.3 channel reveal conformational crosstalk of the intracellular segments. Nat. Commun. 13, 7358 (2022).
Yao, X., Gao, S. & Yan, N. Structural basis for pore blockade of human voltage-gated calcium channel Cav1.3 by motion sickness drug cinnarizine. Cell Res. 32, 946–948 (2022).
He, L. et al. Structure, gating, and pharmacology of human CaV3.3 channel. Nat. Commun. 13, 2084 (2022).
Gao, S. et al. Structural basis for human Cav1.2 inhibition by multiple drugs and the neurotoxin calciseptine. Cell 186, 5363–5374.e16 (2023).
Chen, Z. et al. EMC chaperone-CaV structure reveals an ion channel assembly intermediate. Nature 619, 410–419 (2023).
Huang, J. et al. Structural basis for human Cav3.2 inhibition by selective antagonists. Cell Res. 34, 440–450 (2024).
Dolphin, A. C. Voltage-gated calcium channels and their auxiliary subunits: physiology and pathophysiology and pharmacology. J. Physiol. 594, 5369–5390 (2016). This is a comprehensive review on the auxiliary subunits of Cav channels.
Gutman, G. A. et al. International Union of Pharmacology. LIII. Nomenclature and molecular relationships of voltage-gated potassium channels. Pharmacol. Rev. 57, 473–508 (2005).
Brown, D. A. & Adams, P. R. Muscarinic suppression of a novel voltage-sensitive K+ current in a vertebrate neurone. Nature 283, 673–676 (1980).
Brown, B. S. & Yu, S. P. Modulation and genetic identification of the M channel. Prog. Biophys. Mol. Biol. 73, 135–166 (2000).
Bocksteins, E. & Snyders, D. J. Electrically silent Kv subunits: their molecular and functional characteristics. Physiology 27, 73–84 (2012).
Pongs, O. & Schwarz, J. R. Ancillary subunits associated with voltage-dependent K+ channels. Physiol. Rev. 90, 755–796 (2010).
Yamanouchi, D., Kasuya, G., Nakajo, K., Kise, Y. & Nureki, O. Dual allosteric modulation of voltage and calcium sensitivities of the Slo1-LRRC channel complex. Mol. Cell 83, 4555–4569.e4 (2023).
Isom, L. L., De Jongh, K. S. & Catterall, W. A. Auxiliary subunits of voltage-gated ion channels. Neuron 12, 1183–1194 (1994).
Bucher, D., Guidoni, L., Carloni, P. & Rothlisberger, U. Coordination numbers of K+ and Na+ ions inside the selectivity filter of the KcsA potassium channel: insights from first principles molecular dynamics. Biophys. J. 98, L47–L49 (2010).
Zhou, Y. F., Morais-Cabral, J. H., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K channel-Fab complex at 2.0 Å resolution. Nature 414, 43–48 (2001).
Zhou, M. & MacKinnon, R. A mutant KcsA K+ channel with altered conduction properties and selectivity filter ion distribution. J. Mol. Biol. 338, 839–846 (2004).
Cordero-Morales, J. F. et al. Molecular determinants of gating at the potassium-channel selectivity filter. Nat. Struct. Mol. Biol. 13, 311–318 (2006).
Flavell, R. R. & Muir, T. W. Expressed protein ligation (EPL) in the study of signal transduction, ion conduction, and chromatin biology. Acc. Chem. Res. 42, 107–116 (2009).
Kim, D. M. & Nimigean, C. M. Voltage-gated potassium channels: a structural examination of selectivity and gating. Cold Spring Harb. Perspect. Biol. 8, a029231 (2016).
Choe, S. Potassium channel structures. Nat. Rev. Neurosci. 3, 115–121 (2002).
MacKinnon, R. Potassium channels and the atomic basis of selective ion conduction (Nobel Lecture). Angew. Chem. Int. Ed. Engl. 43, 4265–4277 (2004).
Lyumkis, D. Challenges and opportunities in cryo-EM single-particle analysis. J. Biol. Chem. 294, 5181–5197 (2019).
Baker, L. A. & Rubinstein, J. L. Radiation damage in electron cryomicroscopy. Methods Enzymol. 481, 371–388 (2010).
Marques, M. A., Purdy, M. D. & Yeager, M. CryoEM maps are full of potential. Curr. Opin. Struct. Biol. 58, 214–223 (2019).
Yu, H. et al. Simulating monovalent and divalent ions in aqueous solution using a Drude polarizable force field. J. Chem. Theory Comput. 6, 774–786 (2010).
Gonzalez, C. et al. K+ channels: function-structural overview. Compr. Physiol. 2, 2087–2149 (2012).
Hille, B. & Schwarz, W. Potassium channels as multi-ion single-file pores. J. Gen. Physiol. 72, 409–442 (1978).
Kopfer, D. A. et al. Ion permeation in K+ channels occurs by direct Coulomb knock-on. Science 346, 352–355 (2014).
Noskov, S. Y., Berneche, S. & Roux, B. Control of ion selectivity in potassium channels by electrostatic and dynamic properties of carbonyl ligands. Nature 431, 830–834 (2004).
Berneche, S. & Roux, B. Energetics of ion conduction through the K+ channel. Nature 414, 73–77 (2001).
Morais-Cabral, J. H., Zhou, Y. & MacKinnon, R. Energetic optimization of ion conduction rate by the K+ selectivity filter. Nature 414, 37–42 (2001).
Hess, P., Lansman, J. B. & Tsien, R. W. Calcium channel selectivity for divalent and monovalent cations. Voltage and concentration dependence of single channel current in ventricular heart cells. J. Gen. Physiol. 88, 293–319 (1986).
Yang, J., Ellinor, P. T., Sather, W. A., Zhang, J. F. & Tsien, R. W. Molecular determinants of Ca2+ selectivity and ion permeation in L-type Ca2+ channels. Nature 366, 158–161 (1993).
Hess, P. & Tsien, R. W. Mechanism of ion permeation through calcium channels. Nature 309, 453–456 (1984).
Almers, W. & McCleskey, E. W. Non-selective conductance in calcium channels of frog muscle: calcium selectivity in a single-file pore. J. Physiol. 353, 585–608 (1984).
Tsien, R. W., Hess, P., McCleskey, E. W. & Rosenberg, R. L. Calcium channels: mechanisms of selectivity, permeation, and block. Annu. Rev. Biophys. Biophys. Chem. 16, 265–290 (1987).
Zhao, Y. Y. et al. Molecular basis for ligand modulation of a mammalian voltage-gated Ca2+ channel. Cell 177, 1495–1506.e12 (2019).
Chen, Z., Mondal, A. & Minor, D. L. Jr. Structural basis for CaVα2δ:gabapentin binding. Nat. Struct. Mol. Biol. 30, 735–739 (2023).
Li, Z. et al. Structural basis for different ω-agatoxin IVA sensitivities of the P-type and Q-type Cav2.1 channels. Cell Res. 34, 455–457 (2024).
Cibulsky, S. M. & Sather, W. A. The EEEE locus is the sole high-affinity Ca2+ binding structure in the pore of a voltage-gated Ca2+ channel: block by Ca2+ entering from the intracellular pore entrance. J. Gen. Physiol. 116, 349–362 (2000).
Talavera, K. et al. Aspartate residues of the Glu-Glu-Asp-Asp (EEDD) pore locus control selectivity and permeation of the T-type Ca2+ channel alpha(1G). J. Biol. Chem. 276, 45628–45635 (2001).
Favre, I., Moczydlowski, E. & Schild, L. On the structural basis for ionic selectivity among Na+, K+, and Ca2+ in the voltage-gated sodium channel. Biophys. J. 71, 3110–3125 (1996).
Zhang, J. R. et al. Simulating the ion permeation and ion selection for a eukaryotic voltage-gated sodium channel NavPaS. Protein Cell 9, 580–585 (2018).
Shen, H. et al. Structural basis for the modulation of voltage-gated sodium channels by animal toxins. Science 362, eaau2596 (2018).
Alberini, G. et al. Molecular dynamics simulations of ion permeation in human voltage-gated sodium channels. J. Chem. Theory Comput. 19, 2953–2972 (2023).
Zhorov, B. S. Possible mechanism of ion selectivity in eukaryotic voltage-gated sodium channels. J. Phys. Chem. B 125, 2074–2088 (2021).
Zhang, A., Yu, H., Liu, C. & Song, C. The Ca2+ permeation mechanism of the ryanodine receptor revealed by a multi-site ion model. Nat. Commun. 11, 922 (2020).
Liu, C., Zhang, A., Yan, N. & Song, C. Atomistic details of charge/space competition in the Ca2+ selectivity of ryanodine receptors. J. Phys. Chem. Lett. 12, 4286–4291 (2021).
Ives, C. M., Thomson, N. J. & Zachariae, U. A cooperative knock-on mechanism underpins Ca2+-selective cation permeation in TRPV channels. J. Gen. Physiol. 155, e202213226 (2023).
Vennekens, R. et al. Permeation and gating properties of the novel epithelial Ca2+ channel. J. Biol. Chem. 275, 3963–3969 (2000).
Long, S. B., Campbell, E. B. & MacKinnon, R. Voltage sensor of Kv1.2: structural basis of electromechanical coupling. Science 309, 903–908 (2005). This research article for the first time reports a classic structure of the voltage-sensing domain, setting the foundation for understanding electromechanical coupling mechanism in VGICs.
Benarroch, E. E. HCN channels: function and clinical implications. Neurology 80, 304–310 (2013).
Ye, W. et al. Activation and closed-state inactivation mechanisms of the human voltage-gated KV4 channel complexes. Mol. Cell 82, 2427–2442.e24 (2022).
Huang, G. et al. Unwinding and spiral sliding of S4 and domain rotation of VSD during the electromechanical coupling in Nav1.7. Proc. Natl Acad. Sci. USA 119, e2209164119 (2022).
Ye, F. et al. Voltage-gating and cytosolic Ca2+ activation mechanisms of Arabidopsis two-pore channel AtTPC1. Proc. Natl Acad. Sci. USA 118, e2113946118 (2021).
Lee, C. H. & MacKinnon, R. Voltage sensor movements during hyperpolarization in the HCN channel. Cell 179, 1582–1589.e7 (2019).
Mandala, V. S. & MacKinnon, R. The membrane electric field regulates the PIP2-binding site to gate the KCNQ1 channel. Proc. Natl Acad. Sci. USA 120, e2301985120 (2023).
Huang, G. et al. High-resolution structures of human Nav1.7 reveal gating modulation through α-π helical transition of S6IV. Cell Rep. 39, 110735 (2022).
Zubcevic, L. & Lee, S. Y. The role of π-helices in TRP channel gating. Curr. Opin. Struct. Biol. 58, 314–323 (2019).
Lee, S. Y., Banerjee, A. & MacKinnon, R. Two separate interfaces between the voltage sensor and pore are required for the function of voltage-dependent K channels. PLoS Biol. 7, 676–686 (2009).
Huang, W., Liu, M., Yan, S. F. & Yan, N. Structure-based assessment of disease-related mutations in human voltage-gated sodium channels. Protein Cell 8, 401–438 (2017).
Zheng, Y. et al. Structural insights into the lipid and ligand regulation of a human neuronal KCNQ channel. Neuron 110, 237–247.e4 (2022).
Ulbricht, W. Sodium channel inactivation: molecular determinants and modulation. Physiol. Rev. 85, 1271–1301 (2005).
Demo, S. D. & Yellen, G. The inactivation gate of the Shaker K+ channel behaves like an open-channel blocker. Neuron 7, 743–753 (1991).
Fan, C. et al. Ball-and-chain inactivation in a calcium-gated potassium channel. Nature 580, 288–293 (2020).
Zhang, J. et al. N-Type fast inactivation of a eukaryotic voltage-gated sodium channel. Nat. Commun. 13, 2713 (2022).
Hoshi, T., Zagotta, W. N. & Aldrich, R. W. Two types of inactivation in Shaker K+ channels: effects of alterations in the carboxy-terminal region. Neuron 7, 547–556 (1991).
Cuello, L. G., Jogini, V., Cortes, D. M. & Perozo, E. Structural mechanism of C-type inactivation in K+ channels. Nature 466, 203–208 (2010).
Reddi, R., Matulef, K., Riederer, E. A., Whorton, M. R. & Valiyaveetil, F. I. Structural basis for C-type inactivation in a Shaker family voltage-gated K+ channel. Sci. Adv. 8, eabm8804 (2022).
Armstrong, C. M. & Bezanilla, F. Inactivation of the sodium channel. II. Gating current experiments. J. Gen. Physiol. 70, 567–590 (1977).
Chen, L. Q., Santarelli, V., Horn, R. & Kallen, R. G. A unique role for the S4 segment of domain 4 in the inactivation of sodium channels. J. Gen. Physiol. 108, 549–556 (1996).
Vassilev, P. M., Scheuer, T. & Catterall, W. A. Identification of an intracellular peptide segment involved in sodium channel inactivation. Science 241, 1658–1661 (1988).
West, J. W. et al. A cluster of hydrophobic amino acid residues required for fast Na+-channel inactivation. Proc. Natl Acad. Sci. USA 89, 10910–10914 (1992).
Budde, T., Meuth, S. & Pape, H. C. Calcium-dependent inactivation of neuronal calcium channels. Nat. Rev. Neurosci. 3, 873–883 (2002).
Eckert, R. & Chad, J. E. Inactivation of Ca channels. Prog. Biophys. Mol. Biol. 44, 215–267 (1984).
Lee, K. S., Marban, E. & Tsien, R. W. Inactivation of calcium channels in mammalian heart cells: joint dependence on membrane potential and intracellular calcium. J. Physiol. 364, 395–411 (1985).
Peterson, B. Z., DeMaria, C. D., Adelman, J. P. & Yue, D. T. Calmodulin is the Ca2+ sensor for Ca2+-dependent inactivation of L-type calcium channels. Neuron 22, 549–558 (1999).
Zuhlke, R. D. & Reuter, H. Ca2+-sensitive inactivation of L-type Ca2+ channels depends on multiple cytoplasmic amino acid sequences of the α1C subunit. Proc. Natl Acad. Sci. USA 95, 3287–3294 (1998).
Chien, A. J., Carr, K. M., Shirokov, R. E., Rios, E. & Hosey, M. M. Identification of palmitoylation sites within the L-type calcium channel β2a subunit and effects on channel function. J. Biol. Chem. 271, 26465–26468 (1996).
Abderemane-Ali, F., Findeisen, F., Rossen, N. D. & Minor, D. L. Jr. A selectivity filter gate controls voltage-gated calcium channel calcium-dependent inactivation. Neuron 101, 1134–1149.e3 (2019).
Patil, P. G., Brody, D. L. & Yue, D. T. Preferential closed-state inactivation of neuronal calcium channels. Neuron 20, 1027–1038 (1998).
Dong, Y. et al. Closed-state inactivation and pore-blocker modulation mechanisms of human CaV2.2. Cell Rep. 37, 109931 (2021).
Hutchings, C. J., Colussi, P. & Clark, T. G. Ion channels as therapeutic antibody targets. MAbs 11, 265–296 (2019).
Banerjee, A., Lee, A., Campbell, E. & Mackinnon, R. Structure of a pore-blocking toxin in complex with a eukaryotic voltage-dependent K+ channel. eLife 2, e00594 (2013).
Selvakumar, P. et al. Structures of the T cell potassium channel Kv1.3 with immunoglobulin modulators. Nat. Commun. 13, 3854 (2022).
Tyagi, A. et al. Rearrangement of a unique Kv1.3 selectivity filter conformation upon binding of a drug. Proc. Natl Acad. Sci. USA 119, e2113536119 (2022).
Yao, X. et al. Structural basis for the severe adverse interaction of sofosbuvir and amiodarone on L-type Cav channels. Cell 185, 4801–4810.e13 (2022).
Asai, T. et al. Cryo-EM structure of K+-bound hERG channel complexed with the blocker astemizole. Structure 29, 203–212.e4 (2021).
Lenaeus, M. J., Burdette, D., Wagner, T., Focia, P. J. & Gross, A. Structures of KcsA in complex with symmetrical quaternary ammonium compounds reveal a hydrophobic binding site. Biochemistry 53, 5365–5373 (2014).
Wu, Q. et al. Structural mapping of Nav1.7 antagonists. Nat. Commun. 14, 3224 (2023).
Gao, S. & Yan, N. Structural basis of the modulation of the voltage-gated calcium ion channel Cav 1.1 by dihydropyridine compounds. Angew. Chem. Int. Ed. Engl. 60, 3131–3137 (2021).
Li, X. et al. Structural basis for modulation of human NaV1.3 by clinical drug and selective antagonist. Nat. Commun. 13, 1286 (2022).
Zhang, J. et al. Structural basis for NaV1.7 inhibition by pore blockers. Nat. Struct. Mol. Biol. 29, 1208–1216 (2022).
Li, T. et al. Structural basis for the modulation of human KCNQ4 by small-molecule drugs. Mol. Cell 81, 25–37.e4 (2021).
Li, X. et al. Molecular basis for ligand activation of the human KCNQ2 channel. Cell Res. 31, 52–61 (2021).
Huang, J., Fan, X., Jin, X., Teng, L. & Yan, N. Dual-pocket inhibition of Nav channels by the antiepileptic drug lamotrigine. Proc. Natl Acad. Sci. USA 120, e2309773120 (2023).
Cristino, L., Bisogno, T. & Di Marzo, V. Cannabinoids and the expanded endocannabinoid system in neurological disorders. Nat. Rev. Neurol. 16, 9–29 (2020).
Huang, J. et al. Cannabidiol inhibits Nav channels through two distinct binding sites. Nat. Commun. 14, 3613 (2023).
Catterall, W. A. et al. Voltage-gated ion channels and gating modifier toxins. Toxicon 49, 124–141 (2007).
Clairfeuille, T. et al. Structural basis of α-scorpion toxin action on Nav channels. Science 363, eaav8573 (2019).
Jiang, D. et al. Structural basis for voltage-sensor trapping of the cardiac sodium channel by a deathstalker scorpion toxin. Nat. Commun. 12, 128 (2021).
Ahuja, S. et al. Structural basis of Nav1.7 inhibition by an isoform-selective small-molecule antagonist. Science 350, aac5464 (2015).
Li, Y. et al. Structure of human NaV1.6 channel reveals Na+ selectivity and pore blockade by 4,9-anhydro-tetrodotoxin. Nat. Commun. 14, 1030 (2023).
Ma, D. et al. Ligand activation mechanisms of human KCNQ2 channel. Nat. Commun. 14, 6632 (2023).
Pan, Y. P. et al. Cortisone dissociates the family K channels from their β subunits. Nat. Chem. Biol. 4, 708–714 (2008).
Vega-Vela, N. E. et al. L-Type calcium channels modulation by estradiol. Mol. Neurobiol. 54, 4996–5007 (2017).
Lansky, S. et al. A pentameric TRPV3 channel with a dilated pore. Nature 621, 206–214 (2023).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Baek, M. et al. Accurate prediction of protein structures and interactions using a three-track neural network. Science 373, 871–876 (2021).
Krishna, R. et al. Generalized biomolecular modeling and design with RoseTTAFold All-Atom. Science 384, eadl2528 (2024).
Abramson, J. et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 630, 493–500 (2024).
Li, Z. Q. et al. Structure of human Nav1.5 reveals the fast inactivation-related segments as a mutational hotspot for the long QT syndrome. Proc. Natl Acad. Sci. USA 118, e2100069118 (2021).
Li, Z. Q. et al. Structural basis for pore blockade of the human cardiac sodium channel Nav1.5 by the antiarrhythmic drug quinidine. Angew. Chem. Int. Ed. Engl. 60, 11474–11480 (2021).
Jiang, D. et al. Open-state structure and pore gating mechanism of the cardiac sodium channel. Cell 184, 5151–5162.e11 (2021).
Jiang, D. et al. Structure of the cardiac sodium channel. Cell 180, 122–134.e10 (2020).
Lenaeus, M., Gamal El-Din, T. M., Tonggu, L., Zheng, N. & Catterall, W. A. Structural basis for inhibition of the cardiac sodium channel by the atypical antiarrhythmic drug ranolazine. Nat. Cardiovasc. Res. 2, 587–594 (2023).
Fan, X., Huang, J., Jin, X. & Yan, N. Cryo-EM structure of human voltage-gated sodium channel Nav1.6. Proc. Natl Acad. Sci. USA 120, e2220578120 (2023).
Kschonsak, M. et al. Cryo-EM reveals an unprecedented binding site for NaV1.7 inhibitors enabling rational design of potent hybrid inhibitors. eLife 12, e84151 (2023).
Xu, H. et al. Structural basis of Nav1.7 inhibition by a gating-modifier spider toxin. Cell 176, 702–715.e14 (2019).
Wisedchaisri, G. et al. Structural basis for high-affinity trapping of the NaV1.7 channel in its resting state by tarantula toxin. Mol. Cell 81, 38–48.e4 (2021).
Huang, X. et al. Structural basis for high-voltage activation and subtype-specific inhibition of human Nav1.8. Proc. Natl Acad. Sci. USA 119, e2208211119 (2022).
Wei, Y. et al. Structural bases of inhibitory mechanism of CaV1.2 channel inhibitors. Nat. Commun. 15, 2772 (2024).
Gao, Y. et al. Molecular insights into the gating mechanisms of voltage-gated calcium channel CaV2.3. Nat. Commun. 14, 516 (2023).
Botte, M. et al. Apo and ligand-bound high resolution Cryo-EM structures of the human Kv3.1 channel reveal a novel binding site for positive modulators. PNAS Nexus 1, pgac083 (2022).
Willegems, K. et al. Structural and electrophysiological basis for the modulation of KCNQ1 channel currents by ML277. Nat. Commun. 13, 3760 (2022).
Ma, D. et al. Structural mechanisms for the activation of human cardiac KCNQ1 channel by electro-mechanical coupling enhancers. Proc. Natl Acad. Sci. USA 119, e2207067119 (2022).
Zhang, S. Y. et al. A small-molecule activation mechanism that directly opens the KCNQ2 channel. Nat. Chem. Biol. https://doi.org/10.1038/s41589-023-01515-y (2024).
Smart, O. S., Neduvelil, J. G., Wang, X., Wallace, B. A. & Sansom, M. S. HOLE: a program for the analysis of the pore dimensions of ion channel structural models. J. Mol. Graph. 14, 354–360 (1996).
Thompson, J. D., Higgins, D. G. & Gibson, T. J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994).
Tan, X. F. et al. Structure of the Shaker Kv channel and mechanism of slow C-type inactivation. Sci. Adv. 8, eabm7814 (2022).
Mandala, V. S. & MacKinnon, R. Voltage-sensor movements in the Eag Kv channel under an applied electric field. Proc. Natl Acad. Sci. USA 119, e2214151119 (2022).
Li, Z. et al. Dissection of the structure-function relationship of Nav channels. Proc. Natl Acad. Sci. USA 121, e2322899121 (2024).
Wisedchaisri, G. et al. Resting-state structure and gating mechanism of a voltage-gated sodium channel. Cell 178, 993–1003.e12 (2019).
Wu, X., Howe, E. N. W. & Gale, P. A. Supramolecular transmembrane anion transport: new assays and insights. Acc. Chem. Res. 51, 1870–1879 (2018).
Wang, L. & Sigworth, F. J. Structure of the BK potassium channel in a lipid membrane from electron cryomicroscopy. Nature 461, 292–295 (2009).
Tonggu, L. & Wang, L. Structure of the human BK ion channel in lipid environment. Membranes 12, 758 (2022).
Yao, X., Fan, X. & Yan, N. Cryo-EM analysis of a membrane protein embedded in the liposome. Proc. Natl Acad. Sci. USA 117, 18497–18503 (2020).
Acknowledgements
The authors thank C. Song, Z. Li, G. Yang, S. Gao, X. Yao and H. Shen for their critical reading and input. We apologize to those whose remarkable contributions are not cited, constrained by page limitations. This work was funded by the National Natural Science Foundation of China (32330052, 32322039 and 32271252). N.Y. has been supported by the start-up funds of the University Professorship from Tsinghua University.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Molecular Cell Biology thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- π helix
-
A secondary protein structure, characterized by hydrogen bonding between the amide group of a residue with the carbonyl oxygen five residues earlier (i + 5 → i) in a helix, resulting in a bulge in a typical α helix.
- 310 helix
-
A secondary protein structure with three residues and ten backbone atoms in each helical turn, characterized by hydrogen bonding between residues i + 3 → i.
- Cation–π interaction
-
A type of noncovalent electrostatic interaction between a positive charge (cation) and the electron-rich aromatic π system.
- Chirality
-
The geometric property of a molecule or object that cannot be superposed onto its mirror image.
- Current–voltage (I–V) curve
-
A curve that depicts the relationship between the electric current, corresponding to ion flow, through the channels and the applied voltages.
- Dihydropyridine
-
(DHP). A class of chemical compounds that can act as chirality-specific antagonists or agonists on the L-type calcium channels. Many DHP compounds are in clinical use as calcium channel blockers.
- Hydration shell
-
A ‘hydration shell’ or ‘water shell’ refers to a continuous cluster of water molecules surrounding and, to a degree, attached to certain chemicals or biomolecules when they are dissolved in an aqueous solution.
- Membrane depolarization
-
Shift in membrane potential reducing negative intracellular charges, mainly caused by cation influx or anion efflux.
- Membrane potentials
-
The difference in electric charge, or voltage, between the interior and exterior of a cell or an organelle.
- Molecular dynamic simulation (MDS) analysis
-
A computer simulation method that predicts the movements of atoms over time based on physical models for interatomic interactions.
- Protein internal packing
-
The arrangement and interactions of amino acid residues within the three-dimensional structure of a protein, which are crucial for the stability, function and overall shape of the protein.
- Resting state
-
The relatively stable state with regard to the membrane potential of an excitable cell, such as a neuron or a muscle cell.
- Saxitoxin
-
(STX). The best-known paralytic shellfish toxin. It acts as a potent neurotoxin by blocking Nav channels.
- Tetrodotoxin
-
(TTX). A potent guanidinium neurotoxin found in some fish and other species, exemplified by pufferfish. It specifically blocks Nav channels with different potencies.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, J., Pan, X. & Yan, N. Structural biology and molecular pharmacology of voltage-gated ion channels. Nat Rev Mol Cell Biol (2024). https://doi.org/10.1038/s41580-024-00763-7
Accepted:
Published:
DOI: https://doi.org/10.1038/s41580-024-00763-7
- Springer Nature Limited