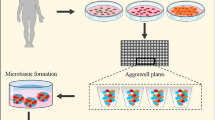
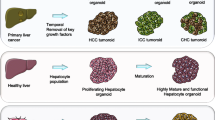
Abstract
Microphysiology systems (MPS), also called organs-on-chips and tissue chips, are miniaturized functional units of organs constructed with multiple cell types under a variety of physical and biochemical environmental cues that complement animal models as part of a new paradigm of drug discovery and development. Biomimetic human liver MPS have evolved from simpler 2D cell models, spheroids and organoids to address the increasing need to understand patient-specific mechanisms of complex and rare diseases, the response to therapeutic treatments, and the absorption, distribution, metabolism, excretion and toxicity of potential therapeutics. The parallel development and application of transdisciplinary technologies, including microfluidic devices, bioprinting, engineered matrix materials, defined physiological and pathophysiological media, patient-derived primary cells, and pluripotent stem cells as well as synthetic biology to engineer cell genes and functions, have created the potential to produce patient-specific, biomimetic MPS for detailed mechanistic studies. It is projected that success in the development and maturation of patient-derived MPS with known genotypes and fully matured adult phenotypes will lead to advanced applications in precision medicine. In this Review, we examine human biomimetic liver MPS that are designed to recapitulate the liver acinus structure and functions to enhance our knowledge of the mechanisms of disease progression and of the absorption, distribution, metabolism, excretion and toxicity of therapeutic candidates and drugs as well as to evaluate their mechanisms of action and their application in precision medicine and preclinical trials.
Key points
-
Liver in vitro experimental models have a long history involving the use of 2D and 3D models that continue to have valuable roles in our understanding of liver physiology and pathophysiology.
-
Human microphysiology systems (MPS) have evolved from simple cell-based experimental models and have the potential to meet the need for human experimental models for basic biomedical research and the development of therapeutics.
-
Human biomimetic liver MPS (HBL-MPS) aim to improve the efficiency of developing biomarkers, repurposed drugs and novel therapeutics by maximally recapitulating the structure and functions of the liver acinus.
-
HBL-MPS are evolving based either on liver organoids derived from patient cells that self-assemble and differentiate or on the directed assembly or bioprinting of matrix materials and cells into microfluidic devices.
-
Organoid-derived MPS and structured MPS are next-generation HBL-MPS that are projected to enable applications of precision medicine, including preclinical trials, either as stand-alone liver models or as coupled, multi-organ MPS.



Similar content being viewed by others
References
Alex, A., Harris, C. J., Keighley, W. W. & Smith, D. A. In Attrition in the Pharmaceutical Industry: Reasons, Implications, and Pathways Forward (eds Alex, A., Harris, C. J. & Smith, D. A.) 106–127 (Wiley, 2015).
Arrowsmith, J. & Miller, P. Trial watch: phase II and phase III attrition rates 2011-2012. Nat. Rev. Drug Discov. 12, 569 (2013).
Friedman, S. L., Neuschwander-Tetri, B. A., Rinella, M. & Sanyal, A. J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 24, 908–922 (2018).
Gribkoff, V. K. & Kaczmarek, L. K. The need for new approaches in CNS drug discovery: Why drugs have failed, and what can be done to improve outcomes. Neuropharmacology 120, 11–19 (2017).
Jardim, D. L., Groves, E. S., Breitfeld, P. P. & Kurzrock, R. Factors associated with failure of oncology drugs in late-stage clinical development: a systematic review. Cancer Treat. Rev. 52, 12–21 (2017).
Parasrampuria, D. A., Benet, L. Z. & Sharma, A. Why drugs fail in late stages of development: case study analyses from the last decade and recommendations. AAPS J. 20, 46 (2018).
Sanyal, A. J. Past, present and future perspectives in nonalcoholic fatty liver disease. Nat. Rev. Gastroenterol. Hepatol. 16, 377–386 (2019).
Taylor, D. L. et al. Harnessing human microphysiology systems as key experimental models for quantitative systems pharmacology. Handb. Exp. Pharmacol. 260, 327–367 (2019).
Eslam, M. & George, J. Genetic contributions to NAFLD: leveraging shared genetics to uncover systems biology. Nat. Rev. Gastroenterol. Hepatol. 17, 40–52 (2020).
Mullard, A. FDA rejects NASH drug. Nat. Rev. Drug Discov. 19, 501 (2020).
Roussel, R., Steg, P. G., Mohammedi, K., Marre, M. & Potier, L. Prevention of cardiovascular disease through reduction of glycaemic exposure in type 2 diabetes: a perspective on glucose-lowering interventions. Diabetes Obes. Metab. 20, 238–244 (2018).
Menon, V. et al. Fasiglifam-induced liver injury in patients with type 2 diabetes: results of a randomized controlled cardiovascular outcomes safety trial. Diabetes Care 41, 2603–2609 (2018).
Mosedale, M. & Watkins, P. B. Drug-induced liver injury: advances in mechanistic understanding that will inform risk management. Clin. Pharmacol. Ther. 101, 469–480 (2017).
Hartung, T. A toxicology for the 21st century - mapping the road ahead. Toxicol. Sci. 109, 18–23 (2009).
Sultana, J., Cutroneo, P. & Trifiro, G. Clinical and economic burden of adverse drug reactions. J. Pharmacol. Pharmacother. 4, S73–S77 (2013).
Bernal, W. Acute liver failure: review and update. Int. Anesthesiol. Clin. 55, 92–106 (2017).
Vernetti, L. et al. in Drug Efficacy, Safety, and Biologics Discovery: Emerging Technologies and Tools (eds Ekins, S. & Xu, J. J.) Ch. 3, 53–73 (Wiley & Sons, 2009).
Moore, T. J., Zhang, H., Anderson, G. & Alexander, G. C. Estimated costs of pivotal trials for novel therapeutic agents approved by the US Food and Drug Administration, 2015-2016. JAMA Intern. Med. 178, 1451–1457 (2018).
Wouters, O. J., McKee, M. & Luyten, J. Estimated research and development investment needed to bring a new medicine to market, 2009–2018. JAMA 323, 844–853 (2020).
Stern, A. M., Schurdak, M. E., Bahar, I., Berg, J. M. & Taylor, D. L. A perspective on implementing a quantitative systems pharmacology platform for drug discovery and the advancement of personalized medicine. J. Biomol. Screen. 21, 521–534 (2016).
Low, L. A. & Tagle, D. A. Tissue chips - innovative tools for drug development and disease modeling. Lab Chip 17, 3026–3036 (2017).
Watson, D. E., Hunziker, R. & Wikswo, J. P. Fitting tissue chips and microphysiological systems into the grand scheme of medicine, biology, pharmacology, and toxicology. Exp. Biol. Med. 242, 1559–1572 (2017).
Ewart, L. et al. Navigating tissue chips from development to dissemination: a pharmaceutical industry perspective. Exp. Biol. Med. 242, 1579–1585 (2017).
Bhatia, S. N. & Ingber, D. E. Microfluidic organs-on-chips. Nat. Biotechnol. 32, 760–772 (2014).
Ronaldson-Bouchard, K. & Vunjak-Novakovic, G. Organs-on-a-chip: a fast track for engineered human tissues in drug development. Cell Stem Cell 22, 310–324 (2018).
Low, L. A., Mummery, C., Berridge, B. R., Austin, C. P. & Tagle, D. A. Organs-on-chips: into the next decade. Nat. Rev. Drug Discov. https://doi.org/10.1038/s41573-020-0079-3 (2020).
Tagle, D. A. The NIH microphysiological systems program: developing in vitro tools for safety and efficacy in drug development. Curr. Opin. Pharmacol. 48, 146–154 (2019).
Sorger, P. K. et al. Quantitative and Systems Pharmacology in the Post-genomic Era: New Approaches to Discovering Drugs and Understanding Therapeutic Mechanisms (NIH, 2011).
Isoherranen, N., Madabushi, R. & Huang, S.-M. Emerging role of organ-on-a-chip technologies in quantitative clinical pharmacology evaluation. Clin. Transl. Sci. 12, 113–121 (2019).
European Medicines Agency. Meeting Report: First EMA workshop on non-animal approaches in support of medicinal product development – challenges and opportunities for use of micro-physiological systems (EMA/CHMP/SWP/250438/2018) (European Medicines Agency, 2018).
Fang, Y. & Eglen, R. M. Three-dimensional cell cultures in drug discovery and development. SLAS Discov. 22, 456–472 (2017).
Shamir, E. R. & Ewald, A. J. Three-dimensional organotypic culture: experimental models of mammalian biology and disease. Nat. Rev. Mol. Cell Biol. 15, 647–664 (2014).
Simian, M. & Bissell, M. J. Organoids: a historical perspective of thinking in three dimensions. J. Cell Biol. 216, 31–40 (2017).
Bhushan, A. et al. Towards a three-dimensional microfluidic liver platform for predicting drug efficacy and toxicity in humans. Stem Cell Res. Ther. 4 (Suppl. 1), S16 (2013).
Ma, L. D. et al. Design and fabrication of a liver-on-a-chip platform for convenient, highly efficient, and safe in situ perfusion culture of 3D hepatic spheroids. Lab Chip 18, 2547–2562 (2018).
Underhill, G. H. & Khetani, S. R. Advances in engineered human liver platforms for drug metabolism studies. Drug Metab. Dispos. 46, 1626–1637 (2018).
Foster, A. J. et al. Integrated in vitro models for hepatic safety and metabolism: evaluation of a human liver-chip and liver spheroid. Arch. Toxicol. https://doi.org/10.1007/s00204-019-02427-4 (2019).
Proctor, W. R. et al. Utility of spherical human liver microtissues for prediction of clinical drug-induced liver injury. Arch. Toxicol. 91, 2849–2863 (2017).
Wang, Y. et al. In situ differentiation and generation of functional liver organoids from human iPSCs in a 3D perfusable chip system. Lab Chip 18, 3606–3616 (2018).
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013).
Guye, P. et al. Genetically engineering self-organization of human pluripotent stem cells into a liver bud-like tissue using Gata6. Nat. Commun. 7, 10243 (2016).
Takebe, T., Zhang, B. & Radisic, M. Synergistic engineering: organoids meet organs-on-a-chip. Cell Stem Cell 21, 297–300 (2017).
Ho, B. X., Pek, N. M. Q. & Soh, B. S. Disease modeling using 3D organoids derived from human induced pluripotent stem cells. Int. J. Mol. Sci. 19 https://doi.org/10.3390/ijms19040936 (2018).
Fatehullah, A., Tan, S. H. & Barker, N. Organoids as an in vitro model of human development and disease. Nat. Cell Biol. 18, 246–254 (2016).
May, S., Evans, S. & Parry, L. Organoids, organs-on-chips and other systems, and microbiota. Emerg. Top. Life Sci. 1, 385–400 (2017).
Ouchi, R. et al. Modeling steatohepatitis in humans with pluripotent stem cell-derived organoids. Cell Metab. 30, 374–384.e6 (2019).
Lancaster, M. A. & Huch, M. Disease modelling in human organoids. Dis. Model. Mech. 12, dmm039347 (2019).
Prior, N., Inacio, P. & Huch, M. Liver organoids: from basic research to therapeutic applications. Gut 68, 2228–2237 (2019).
Prestigiacomo, V., Weston, A., Messner, S., Lampart, F. & Suter-Dick, L. Pro-fibrotic compounds induce stellate cell activation, ECM-remodelling and Nrf2 activation in a human 3D-multicellular model of liver fibrosis. PLoS ONE 12, e0179995 (2017).
Jang, M., Neuzil, P., Volk, T., Manz, A. & Kleber, A. On-chip three-dimensional cell culture in phaseguides improves hepatocyte functions in vitro. Biomicrofluidics 9, 034113 (2015).
Khetani, S. R. & Bhatia, S. N. Microscale culture of human liver cells for drug development. Nat. Biotechnol. 26, 120–126 (2008).
Davidson, M. D., Lehrer, M. & Khetani, S. R. Hormone and drug-mediated modulation of glucose metabolism in a microscale model of the human liver. Tissue Eng. Part C Methods 21, 716–725 (2015).
Davidson, M. D., Kukla, D. A. & Khetani, S. R. Microengineered cultures containing human hepatic stellate cells and hepatocytes for drug development. Integr. Biol. 9, 662–677 (2017).
Berger, D. R., Ware, B. R., Davidson, M. D., Allsup, S. R. & Khetani, S. R. Enhancing the functional maturity of induced pluripotent stem cell-derived human hepatocytes by controlled presentation of cell-cell interactions in vitro. Hepatology 61, 1370–1381 (2015).
Avila, A. M. et al. An FDA/CDER perspective on nonclinical testing strategies: classical toxicology approaches and new approach methodologies (NAMs). Regul. Toxicol. Pharmacol. 114, 104662 (2020).
Li, X., George, S. M., Vernetti, L., Gough, A. H. & Taylor, D. L. A glass-based, continuously zonated and vascularized human liver acinus microphysiological system (vLAMPS) designed for experimental modeling of diseases and ADME/TOX. Lab Chip 18, 2614–2631 (2018).
Ahadian, S. et al. Organ-on-a-chip platforms: a convergence of advanced materials, cells, and microscale technologies. Adv. Healthc. Mater. https://doi.org/10.1002/adhm.201700506 (2018).
Soto-Gutierrez, A., Gough, A., Vernetti, L. A., Taylor, D. L. & Monga, S. P. Pre-clinical and clinical investigations of metabolic zonation in liver diseases: the potential of microphysiology systems. Exp. Biol. Med. 242, 1605–1616 (2017).
Lee-Montiel, F. T. et al. Control of oxygen tension recapitulates zone-specific functions in human liver microphysiology systems. Exp. Biol. Med. 242, 1617–1632 (2017).
Bin Ramli, M. N. et al. Human pluripotent stem cell-derived organoids as models of liver disease. Gastroenterology 159, 1471–1486.e12 (2020).
Sharma, A., Sances, S., Workman, M. J. & Svendsen, C. N. Multi-lineage human iPSC-derived platforms for disease modeling and drug discovery. Cell Stem Cell 26, 309–329 (2020).
Vernetti, L. A. et al. A human liver microphysiology platform for investigating physiology, drug safety, and disease models. Exp. Biol. Med. 241, 101–114 (2016).
McAleer, C. W. et al. On the potential of in vitro organ-chip models to define temporal pharmacokinetic-pharmacodynamic relationships. Sci. Rep. 9, 9619 (2019).
Kizawa, H., Nagao, E., Shimamura, M., Zhang, G. & Torii, H. Scaffold-free 3D bio-printed human liver tissue stably maintains metabolic functions useful for drug discovery. Biochem. Biophys. Rep. 10, 186–191 (2017).
Feaver, R. E. et al. Development of an in vitro human liver system for interrogating nonalcoholic steatohepatitis. JCI Insight 1, e90954 (2016).
Kostrzewski, T. et al. A microphysiological system for studying nonalcoholic steatohepatitis. Hepatol. Commun. 4, 77–91 (2020).
Huch, M., Knoblich, J. A., Lutolf, M. P. & Martinez-Arias, A. The hope and the hype of organoid research. Development 144, 938–941 (2017).
Huch, M. et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160, 299–312 (2015).
Saito, Y. et al. Establishment of patient-derived organoids and drug screening for biliary tract carcinoma. Cell Rep. 27, 1265–1276.e1264 (2019).
Guan, Y. et al. Human hepatic organoids for the analysis of human genetic diseases. JCI Insight https://doi.org/10.1172/jci.insight.94954 (2017).
Broutier, L. et al. Human primary liver cancer-derived organoid cultures for disease modeling and drug screening. Nat. Med. 23, 1424–1435 (2017).
Velazquez, J. J. et al. Synthetic maturation of multilineage human liver organoids via genetically guided engineering. bioRxiv https://doi.org/10.1101/2020.05.10.087445 (2020).
Koike, H. et al. Modelling human hepato-biliary-pancreatic organogenesis from the foregut-midgut boundary. Nature 574, 112–116 (2019).
Kostrzewski, T. et al. Three-dimensional perfused human in vitro model of non-alcoholic fatty liver disease. World J. Gastroenterol. 23, 204–215 (2017).
Jang, K. J. et al. Reproducing human and cross-species drug toxicities using a Liver-Chip. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aax5516 (2019).
Beckwitt, C. H. et al. Liver ‘organ on a chip’. Exp. Cell. Res. 363, 15–25 (2018).
Senutovitch, N. et al. Fluorescent protein biosensors applied to microphysiological systems. Exp. Biol. Med. 240, 795–808 (2015).
Auner, A. W., Tasneem, K. M., Markov, D. A., McCawley, L. J. & Hutson, M. S. Chemical-PDMS binding kinetics and implications for bioavailability in microfluidic devices. Lab Chip 19, 864–874 (2019).
Tan, K. et al. A high-throughput microfluidic microphysiological system (PREDICT-96) to recapitulate hepatocyte function in dynamic, re-circulating flow conditions. Lab Chip 19, 1556–1566 (2019).
Wikswo, J. P. et al. Engineering challenges for instrumenting and controlling integrated organ-on-chip systems. IEEE Trans. Biomed. Eng. 60, 682–690 (2013).
Bavli, D. et al. Real-time monitoring of metabolic function in liver-on-chip microdevices tracks the dynamics of mitochondrial dysfunction. Proc. Natl Acad. Sci. USA 113, E2231–E2240 (2016).
Esch, M. B., Ueno, H., Applegate, D. R. & Shuler, M. L. Modular, pumpless body-on-a-chip platform for the co-culture of GI tract epithelium and 3D primary liver tissue. Lab Chip 16, 2719–2729 (2016).
Miller, P. G. & Shuler, M. L. Design and demonstration of a pumpless 14 compartment microphysiological system. Biotechnol. Bioeng. 113, 2213–2227 (2016).
Oleaga, C. et al. Multi-organ toxicity demonstration in a functional human in vitro system composed of four organs. Sci. Rep. 6, 20030 (2016).
Wang, Y. I., Carmona, C., Hickman, J. J. & Shuler, M. L. Multiorgan microphysiological systems for drug development: strategies, advances, and challenges. Adv. Healthc. Mater. 7, https://doi.org/10.1002/adhm.201701000 (2018).
Wang, Y., Wang, L., Guo, Y., Zhu, Y. & Qin, J. Engineering stem cell-derived 3D brain organoids in a perfusable organ-on-a-chip system. RSC Adv. 8, 1677–1685 (2018).
Park, S. E., Georgescu, A. & Huh, D. Organoids-on-a-chip. Science 364, 960–965 (2019).
Zhang, Y. S. et al. Multisensor-integrated organs-on-chips platform for automated and continual in situ monitoring of organoid behaviors. Proc. Natl Acad. Sci. USA 114, E2293–E2302 (2017).
Ehrlich, A., Duche, D., Ouedraogo, G. & Nahmias, Y. Challenges and opportunities in the design of liver-on-chip microdevices. Annu. Rev. Biomed. Eng. 21, 219–239 (2019).
Ehrlich, A. et al. Microphysiological flux balance platform unravels the dynamics of drug induced steatosis. Lab Chip 18, 2510–2522 (2018).
Kilic, T., Navaee, F., Stradolini, F., Renaud, P. & Carrara, S. Organs-on-chip monitoring: sensors and other strategies. Microphysiol. Syst. https://doi.org/10.21037/mps.2018.01.01 (2018).
Sung, J. H. Pharmacokinetic-based multi-organ chip for recapitulating organ interactions. Methods Cell. Biol. 146, 183–197 (2018).
Sung, J. H. et al. Using physiologically-based pharmacokinetic-guided “body-on-a-chip” systems to predict mammalian response to drug and chemical exposure. Exp. Biol. Med. 239, 1225–1239 (2014).
Vernetti, L. A., Vogt, A., Gough, A. & Taylor, D. L. Evolution of experimental models of the liver to predict human drug hepatotoxicity and efficacy. Clin. Liver Dis. 21, 197–214 (2017).
Ewart, L. et al. Application of microphysiological systems to enhance safety assessment in drug discovery. Annu. Rev. Pharmacol. Toxicol. 58, 65–82 (2018).
Wang, X., Cirit, M., Wishnok, J. S., Griffith, L. G. & Tannenbaum, S. R. Analysis of an integrated human multiorgan microphysiological system for combined tolcapone metabolism and brain metabolomics. Anal. Chem. 91, 8667–8675 (2019).
Herland, A. et al. Quantitative prediction of human pharmacokinetic responses to drugs via fluidically coupled vascularized organ chips. Nat. Biomed. Eng. https://doi.org/10.1038/s41551-019-0498-9 (2020).
Vernetti, L. et al. Functional coupling of human microphysiology systems: intestine, liver, kidney proximal tubule, blood-brain barrier and skeletal muscle. Sci. Rep. 7, 42296 (2017).
Wikswo, J. P. et al. Scaling and systems biology for integrating multiple organs-on-a-chip. Lab Chip 13, 3496–3511 (2013).
Koui, Y. et al. An in vitro human liver model by iPSC-derived parenchymal and non-parenchymal cells. Stem Cell Rep. 9, 490–498 (2017).
Prendergast, M. E. et al. Microphysiological systems: automated fabrication via extrusion bioprinting. Microphysiol. Syst. https://doi.org/10.21037/MPS.2018.03.01 (2018).
Velazquez, J. J., Su, E., Cahan, P. & Ebrahimkhani, M. R. Programming morphogenesis through systems and synthetic biology. Trends Biotechnol. 36, 415–429 (2018).
Ebrahimkhani, M. R. & Ebisuya, M. Synthetic developmental biology: build and control multicellular systems. Curr. Opin. Chem. Biol. 52, 9–15 (2019).
Johnson, M. B., March, A. R. & Morsut, L. Engineering multicellular systems: using synthetic biology to control tissue self-organization. Curr. Opin. Biomed. Eng. 4, 163–173 (2017).
Colnot, S. & Perret, C. in Molecular Pathology of Liver Diseases (ed. Satdarshan, S. & Monga, P.) 7–16 (Springer, 2011).
Marx, U. et al. Biology-inspired microphysiological system approaches to solve the prediction dilemma of substance testing. ALTEX 33, 272–321 (2016).
Baudy, A. R. et al. Liver microphysiological systems development guidelines for safety risk assessment in the pharmaceutical industry. Lab Chip 20, 215–225 (2020).
Hughes, D. J., Kostrzewski, T. & Sceats, E. L. Opportunities and challenges in the wider adoption of liver and interconnected microphysiological systems. Exp. Biol. Med. 242, 1593–1604 (2017).
Wheeler, S. E. et al. Spontaneous dormancy of metastatic breast cancer cells in an all human liver microphysiologic system. Br. J. Cancer 111, 2342–2350 (2014).
Sundaram, V. & Björnsson, E. S. Drug-induced cholestasis. Hepatol. Commun. 1, 726–735 (2017).
Sampaziotis, F. et al. Directed differentiation of human induced pluripotent stem cells into functional cholangiocyte-like cells. Nat. Protoc. 12, 814–827 (2017).
Sampaziotis, F. et al. Reconstruction of the mouse extrahepatic biliary tree using primary human extrahepatic cholangiocyte organoids. Nat. Med. 23, 954–963 (2017).
Sampaziotis, F. Building better bile ducts. Science 359, 1113 (2018).
Du, Y. et al. A bile duct-on-a-chip with organ-level functions. Hepatology 71, 1350–1363 (2020).
Leclerc, E. et al. Comparison of the transcriptomic profile of hepatic human induced pluripotent stem like cells cultured in plates and in a 3D microscale dynamic environment. Genomics 109, 16–26 (2017).
Grant, M. H. et al. Human adult hepatocytes in primary monolayer culture. Maintenance of mixed function oxidase and conjugation pathways of drug metabolism. Biochem. Pharmacol. 36, 2311–2316 (1987).
Guzelian, P. S., Bissell, D. M. & Meyer, U. A. Drug metabolism in adult rat hepatocytes in primary monolayer culture. Gastroenterology 72, 1232–1239 (1977).
Long, T. J. et al. Modeling therapeutic antibody-small molecule drug-drug interactions using a three-dimensional perfusable human liver coculture platform. Drug Metab. Dispos. 44, 1940–1948 (2016).
Tsamandouras, N. et al. Quantitative assessment of population variability in hepatic drug metabolism using a perfused three-dimensional human liver microphysiological system. J. Pharmacol. Exp. Ther. 360, 95–105 (2017).
Cirit, M. & Stokes, C. L. Maximizing the impact of microphysiological systems with in vitro-in vivo translation. Lab Chip 18, 1831–1837 (2018).
Shen, J. X., Youhanna, S., Shafagh, R. Z., Kele, J. & Lauschke, V. M. Organotypic and microphysiological models of liver, gut and kidney for studies of drug metabolism, pharmacokinetics and toxicity. Chem. Res. Toxicol. 33, 38–60 (2020).
Truskey, G. A. Human microphysiological systems and organoids as in vitro models for toxicological studies. Front. Public. Health 6, 185–185 (2018).
Zhou, Y., Shen, J. X. & Lauschke, V. M. Comprehensive evaluation of organotypic and microphysiological liver models for prediction of drug-induced liver injury. Front. Pharmacol. 10, 1093 (2019).
Terelius, Y. et al. Transcriptional profiling suggests that Nevirapine and Ritonavir cause drug induced liver injury through distinct mechanisms in primary human hepatocytes. Chem. Biol. Interact. 255, 31–44 (2016).
Schurdak, M. et al. Applications of the microphysiology systems database for experimental ADME-Tox and disease models. Lab Chip 20, 1472–1492 (2020).
Gough, A., Vernetti, L., Bergenthal, L., Shun, T. Y. & Taylor, D. L. The microphysiology systems database for analyzing and modeling compound interactions with human and animal organ models. Appl. In Vitro Toxicol. 2, 103–117 (2016).
Choudhury, Y. et al. Patient-specific hepatocyte-like cells derived from induced pluripotent stem cells model pazopanib-mediated hepatotoxicity. Sci. Rep. 7, 41238 (2017).
Koido, M. et al. Polygenic architecture informs potential vulnerability to drug-induced liver injury. Nat. Med. https://doi.org/10.1038/s41591-020-1023-0 (2020).
Adams, D. H., Ju, C., Ramaiah, S. K., Uetrecht, J. & Jaeschke, H. Mechanisms of immune-mediated liver injury. Toxicol. Sci. 115, 307–321 (2010).
Ahn, J. et al. Human three-dimensional in vitro model of hepatic zonation to predict zonal hepatotoxicity. J. Biol. Eng. 13, 22–22 (2019).
Tonon, F. et al. In vitro metabolic zonation through oxygen gradient on a chip. Sci. Rep. 9, 13557 (2019).
Halpern, K. B. et al. Single-cell spatial reconstruction reveals global division of labour in the mammalian liver. Nature 542, 352–356 (2017).
Prill, S. et al. Real-time monitoring of oxygen uptake in hepatic bioreactor shows CYP450-independent mitochondrial toxicity of acetaminophen and amiodarone. Arch. Toxicol. 90, 1181–1191 (2016).
Roden, M. & Shulman, G. I. The integrative biology of type 2 diabetes. Nature 576, 51–60 (2019).
Hardy, T., Oakley, F., Anstee, Q. M. & Day, C. P. Nonalcoholic fatty liver disease: pathogenesis and disease spectrum. Annu. Rev. Pathol. 11, 451–496 (2016).
Eslam, M., Sanyal, A.J., George, J. & International Consensus Panel. MAFLD: a consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology, 158, 1999–2014 (2020).
Pennisi, G. et al. Pharmacological therapy of non-alcoholic fatty liver disease: what drugs are available now and future perspectives. Int. J. Environ. Res. Public Health 16, https://doi.org/10.3390/ijerph16224334 (2019).
Danford, C. J., Yao, Z.-M. & Jiang, Z. G. Non-alcoholic fatty liver disease: a narrative review of genetics. J. Biomed. Res. 32, 389–400 (2018).
Sato, K. et al. Intercellular communication between hepatic cells in liver diseases. Int. J. Mol. Sci. 20 https://doi.org/10.3390/ijms20092180 (2019).
Boeckmans, J. et al. Human-based systems: mechanistic NASH modelling just around the corner? Pharmacol. Res. 134, 257–267 (2018).
Anstee, Q. M., Reeves, H. L., Kotsiliti, E., Govaere, O. & Heikenwalder, M. From NASH to HCC: current concepts and future challenges. Nat. Rev. Gastroenterol. Hepatol. 16, 411–428 (2019).
Mannaa, F. A. & Abdel-Wahhab, K. G. Physiological potential of cytokines and liver damages. Hepatoma Res. 2, 131–143 (2016).
Tacke, F. Targeting hepatic macrophages to treat liver diseases. J. Hepatol. 66, 1300–1312 (2017).
Musso, G., Cassader, M. & Gambino, R. Non-alcoholic steatohepatitis: emerging molecular targets and therapeutic strategies. Nat. Rev. Drug Discov. 15, 249–274 (2016).
Ezhilarasan, D., Sokal, E. & Najimi, M. Hepatic fibrosis: it is time to go with hepatic stellate cell-specific therapeutic targets. Hepatobiliary Pancreat. Dis. Int. 17, 192–197 (2018).
Higashi, T., Friedman, S. L. & Hoshida, Y. Hepatic stellate cells as key target in liver fibrosis. Adv. Drug Deliv. Rev. 121, 27–42 (2017).
Rao, S. S., Kondapaneni, R. V. & Narkhede, A. A. Bioengineered models to study tumor dormancy. J. Biol. Eng. 13, 3 (2019).
Lee, J. W. et al. Hepatocytes direct the formation of a pro-metastatic niche in the liver. Nature 567, 249–252 (2019).
Wells, A., Clark, A., Bradshaw, A., Ma, B. & Edington, H. The great escape: how metastases of melanoma, and other carcinomas, avoid elimination. Exp. Biol. Med. 243, 1245–1255 (2018).
Miedel, M. T. et al. Modeling the effect of the metastatic microenvironment on phenotypes conferred by estrogen receptor mutations using a human liver microphysiological system. Sci. Rep. 9, 8341 (2019).
Jia, S. et al. Clinically observed estrogen receptor alpha mutations within the ligand-binding domain confer distinguishable phenotypes. Oncology 94, 176–189 (2018).
Scherz-Shouval, R. et al. The reprogramming of tumor stroma by HSF1 is a potent enabler of malignancy. Cell 158, 564–578 (2014).
Clark, A. M. et al. A microphysiological system model of therapy for liver micrometastases. Exp. Biol. Med. 239, 1170–1179 (2014).
Clark, A. M. et al. A model of dormant-emergent metastatic breast cancer progression enabling exploration of biomarker signatures. Mol. Cell. Proteomics 17, 619–630 (2018).
Beckwitt, C. H. et al. Statins attenuate outgrowth of breast cancer metastases. Br. J. Cancer 119, 1094–1105 (2018).
Zhao, Y., Kankala, R. K., Wang, S. B. & Chen, A. Z. Multi-organs-on-chips: towards long-term biomedical investigations. Molecules https://doi.org/10.3390/molecules24040675 (2019).
Sung, J. H. et al. Recent advances in body-on-a-chip systems. Anal. Chem. 91, 330–351 (2019).
McAleer, C. W. et al. Multi-organ system for the evaluation of efficacy and off-target toxicity of anticancer therapeutics. Sci. Transl. Med. 11, eaav1386 (2019).
Vunjak-Novakovic, G., Bhatia, S., Chen, C. & Hirschi, K. HeLiVa platform: integrated heart-liver-vascular systems for drug testing in human health and disease. Stem Cell Res. Ther. 4 (Suppl. 1), S8 (2013).
Bricks, T. et al. Development of a new microfluidic platform integrating co-cultures of intestinal and liver cell lines. Toxicol. In Vitro 28, 885–895 (2014).
Bricks, T. et al. Investigation of omeprazole and phenacetin first-pass metabolism in humans using a microscale bioreactor and pharmacokinetic models. Biopharm. Drug Dispos. 36, 275–293 (2015).
Maschmeyer, I. et al. A four-organ-chip for interconnected long-term co-culture of human intestine, liver, skin and kidney equivalents. Lab Chip 15, 2688–2699 (2015).
Bauer, S. et al. Functional coupling of human pancreatic islets and liver spheroids on-a-chip: Towards a novel human ex vivo type 2 diabetes model. Sci. Rep. 7, 14620 (2017).
Tsamandouras, N. et al. Integrated gut and liver microphysiological systems for quantitative in vitro pharmacokinetic studies. AAPS J. https://doi.org/10.1208/s12248-017-0122-4 (2017).
Edington, C. D. et al. Interconnected microphysiological systems for quantitative biology and pharmacology studies. Sci. Rep. 8, 4530–4530 (2018).
Tilg, H., Moschen, A. R. & Roden, M. NAFLD and diabetes mellitus. Nat. Rev. Gastroenterol. Hepatol. 14, 32–42 (2017).
Ferreira, C. R., Cassiman, D. & Blau, N. Clinical and biochemical footprints of inherited metabolic diseases. II. Metabolic liver diseases. Mol. Genet. Metab. 127, 117–121 (2019).
Younossi, Z. M., Marchesini, G., Pinto-Cortez, H. & Petta, S. Epidemiology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: implications for liver transplantation. Transplantation 103, 22–27 (2019).
Younossi, Z. M., Henry, L., Bush, H. & Mishra, A. Clinical and economic burden of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Clin. Liver Dis. 22, 1–10 (2018).
Goldberg, D. et al. Changes in the prevalence of hepatitis C virus infection, nonalcoholic steatohepatitis, and alcoholic liver disease among patients with cirrhosis or liver failure on the waitlist for liver transplantation. Gastroenterology 152, 1090–1099.e1 (2017).
Mikolasevic, I. et al. Nonalcoholic fatty liver disease and liver transplantation - where do we stand? World J. Gastroenterol. 24, 1491–1506 (2018).
Mittal, S. et al. Hepatocellular carcinoma in the absence of cirrhosis in United States veterans is associated with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 14, 124–131.e1 (2016).
Singh, S. et al. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin. Gastroenterol. Hepatol. 13, 643–654.e1-9 (2015).
McPherson, S. et al. Evidence of NAFLD progression from steatosis to fibrosing-steatohepatitis using paired biopsies: implications for prognosis and clinical management. J. Hepatol. 62, 1148–1155 (2015).
Hoang, S. A. et al. Gene expression predicts histological severity and reveals distinct molecular profiles of nonalcoholic fatty liver disease. Sci. Rep. 9, 12541 (2019).
Younossi, Z. M. et al. Current and future therapeutic regimens for nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology 68, 361–371 (2018).
Sumida, Y. & Yoneda, M. Current and future pharmacological therapies for NAFLD/NASH. J. Gastroenterol. 53, 362–376 (2018).
Polyzos, S. A., Kountouras, J., Anastasiadis, S., Doulberis, M. & Katsinelos, P. Nonalcoholic fatty liver disease: Is it time for combination treatment and a diabetes-like approach? Hepatology 68, 389 (2018).
Dyson, J. et al. Hepatocellular cancer: the impact of obesity, type 2 diabetes and a multidisciplinary team. J. Hepatol. 60, 110–117 (2014).
Piscaglia, F. et al. Clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: a multicenter prospective study. Hepatology 63, 827–838 (2016).
Wong, V. W., Adams, L. A., de Lédinghen, V., Wong, G. L. & Sookoian, S. Noninvasive biomarkers in NAFLD and NASH - current progress and future promise. Nat. Rev. Gastroenterol. Hepatol. 15, 461–478 (2018).
Greene, C. M. et al. α1-antitrypsin deficiency. Nat. Rev. Dis. Primers 2, 16051 (2016).
Hazari, Y. M. et al. Alpha-1-antitrypsin deficiency: genetic variations, clinical manifestations and therapeutic interventions. Mutat. Res. 773, 14–25 (2017).
Moat, S. J. et al. Performance of laboratory tests used to measure blood phenylalanine for the monitoring of patients with phenylketonuria. J. Inherit. Metab. Dis. 43, 179–188 (2020).
Blau, N. Genetics of phenylketonuria: then and now. Hum. Mutat. 37, 508–515 (2016).
Bergenthal, L. M., Shun, T. Y., Vernetti, L., Taylor, D. L. & Gough, A. H. The Microphysiology Systems Database http://mps.csb.pitt.edu (2018).
Shi, Y., Inoue, H., Wu, J. C. & Yamanaka, S. Induced pluripotent stem cell technology: a decade of progress. Nat. Rev. Drug Discov. 16, 115–130 (2017).
Takahashi, K. & Yamanaka, S. A decade of transcription factor-mediated reprogramming to pluripotency. Nat. Rev. Mol. Cell Biol. 17, 183–193 (2016).
Pei, F. et al. Connecting neuronal cell protective pathways and drug combinations in a Huntington’s disease model through the application of quantitative systems pharmacology. Sci. Rep. 7, 17803 (2017).
Chow, S. C. & Chang, M. Adaptive design methods in clinical trials - a review. Orphanet J. Rare Dis. 3, 11 (2008).
Fagiuoli, S., Daina, E., D’Antiga, L., Colledan, M. & Remuzzi, G. Monogenic diseases that can be cured by liver transplantation. J. Hepatol. 59, 595–612 (2013).
Isabella, V. M. et al. Development of a synthetic live bacterial therapeutic for the human metabolic disease phenylketonuria. Nat. Biotechnol. 36, 857–864 (2018).
Ghouse, R., Chu, A., Wang, Y. & Perlmutter, D. H. Mysteries of alpha1-antitrypsin deficiency: emerging therapeutic strategies for a challenging disease. Dis. Model. Mech. 7, 411–419 (2014).
Turner, A. M. et al. Hepatic-targeted RNA interference provides robust and persistent knockdown of alpha-1 antitrypsin levels in ZZ patients. J. Hepatol. 69, 378–384 (2018).
Hidvegi, T. et al. An autophagy-enhancing drug promotes degradation of mutant alpha1-antitrypsin Z and reduces hepatic fibrosis. Science 329, 229–232 (2010).
Bouchecareilh, M., Conkright, J. J. & Balch, W. E. Proteostasis strategies for restoring alpha1-antitrypsin deficiency. Proc. Am. Thorac. Soc. 7, 415–422 (2010).
Tafaleng, E. N. et al. Induced pluripotent stem cells model personalized variations in liver disease resulting from α1-antitrypsin deficiency. Hepatology 62, 147–157 (2015).
Low, L. A. & Tagle, D. A. Microphysiological systems (tissue chips) and their utility for rare disease research. Adv. Exp. Med. Biol. 1031, 405–415 (2017).
Blumenrath, S. H., Lee, B. Y., Low, L., Prithviraj, R. & Tagle, D. Tackling rare diseases: clinical trials on chips. Exp. Biol. Med. 245, 1155–1162 (2020).
Pan, G. Roles of hepatic drug transporters in drug disposition and liver toxicity. Adv. Exp. Med. Biol. 1141, 293–340 (2019).
Nguyen, D. G. et al. Bioprinted 3D primary liver tissues allow assessment of organ-level response to clinical drug induced toxicity in vitro. PLoS ONE 11, e0158674 (2016).
Norona, L. M., Nguyen, D. G., Gerber, D. A., Presnell, S. C. & LeCluyse, E. L. Editor’s highlight: modeling compound-induced fibrogenesis in vitro using three-dimensional bioprinted human liver tissues. Toxicol. Sci. 154, 354–367 (2016).
Norona, L. M. et al. Bioprinted liver provides early insight into the role of Kupffer cells in TGF-β1 and methotrexate-induced fibrogenesis. PLoS ONE 14, e0208958 (2019).
Trietsch, S. J., Israels, G. D., Joore, J., Hankemeier, T. & Vulto, P. Microfluidic titer plate for stratified 3D cell culture. Lab Chip 13, 3548–3554 (2013).
Domansky, K. et al. Perfused multiwell plate for 3D liver tissue engineering. Lab Chip 10, 51–58 (2010).
Novik, E., Maguire, T. J., Chao, P., Cheng, K. C. & Yarmush, M. L. A microfluidic hepatic coculture platform for cell-based drug metabolism studies. Biochem. Pharmacol. 79, 1036–1044 (2010).
Chao, P., Maguire, T., Novik, E., Cheng, K. C. & Yarmush, M. L. Evaluation of a microfluidic based cell culture platform with primary human hepatocytes for the prediction of hepatic clearance in human. Biochem. Pharmacol. 78, 625–632 (2009).
Dash, A. et al. Hemodynamic flow improves rat hepatocyte morphology, function, and metabolic activity in vitro. Am. J. Physiol. Cell Physiol. 304, C1053–C1063 (2013).
Boeri, L. et al. Advanced organ-on-a-chip devices to investigate liver multi-organ communication: focus on gut, microbiota and brain. Bioengineering 6, 91 (2019).
Natarajan, V., Berglund, E. J., Chen, D. X. & Kidambi, S. Substrate stiffness regulates primary hepatocyte functions. RSC Adv. 5, 80956–80966 (2015).
Newman, R. H. & Zhang, J. The design and application of genetically encodable biosensors based on fluorescent proteins. Methods Mol. Biol. 1071, 1–16 (2014).
Collin de l’Hortet, A. et al. Generation of human fatty livers using custom-engineered induced pluripotent stem cells with modifiable SIRT1 metabolism. Cell Metab. 30, 385–401.e9 (2019).
Toepke, M. W. & Beebe, D. J. PDMS absorption of small molecules and consequences in microfluidic applications. Lab Chip 6, 1484–1486 (2006).
Regehr, K. J. et al. Biological implications of polydimethylsiloxane-based microfluidic cell culture. Lab Chip 9, 2132–2139 (2009).
Donato, M. T. & Tolosa, L. Stem-cell derived hepatocyte-like cells for the assessment of drug-induced liver injury. Differentiation 106, 15–22 (2019).
Rezvani, M., Grimm, A. A. & Willenbring, H. Assessing the therapeutic potential of lab-made hepatocytes. Hepatology 64, 287–294 (2016).
Tasnim, F. et al. Generation of mature kupffer cells from human induced pluripotent stem cells. Biomaterials 192, 377–391 (2019).
Coll, M. et al. Generation of hepatic stellate cells from human pluripotent stem cells enables in vitro modeling of liver fibrosis. Cell Stem Cell 23, 101–113.e7 (2018).
Parent, R. et al. An immortalized human liver endothelial sinusoidal cell line for the study of the pathobiology of the liver endothelium. Biochem. Biophys. Res. Commun. 450, 7–12 (2014).
Matsumura, T. et al. Establishment of an immortalized human-liver endothelial cell line with SV40T and hTERT. Transplantation 77, 1357–1365 (2004).
Maruyama, M. et al. Establishment of a highly differentiated immortalized human cholangiocyte cell line with SV40T and hTERT. Transplantation 77, 446–451 (2004).
Tabibian, J. H. et al. Characterization of cultured cholangiocytes isolated from livers of patients with primary sclerosing cholangitis. Lab. Invest. 94, 1126–1133 (2014).
Sampaziotis, F. et al. Cholangiocytes derived from human induced pluripotent stem cells for disease modeling and drug validation. Nat. Biotechnol. 33, 845–852 (2015).
Ghanekar, A. & Kamath, B. M. Cholangiocytes derived from induced pluripotent stem cells for disease modeling. Curr. Opin. Gastroenterol. 32, 210–215 (2016).
Poisson, J. et al. Liver sinusoidal endothelial cells: physiology and role in liver diseases. J. Hepatol. 66, 212–227 (2017).
Hammoutene, A. & Rautou, P. E. Role of liver sinusoidal endothelial cells in non-alcoholic fatty liver disease. J. Hepatol. 70, 1278–1291 (2019).
DeLeve, L. D. & Maretti-Mira, A. C. Liver sinusoidal endothelial cell: an update. Semin. Liver Dis. 37, 377–387 (2017).
Jang, S., Collin de l’Hortet, A. & Soto-Gutierrez, A. Induced pluripotent stem cell-derived endothelial cells: overview, current advances, applications, and future directions. Am. J. Pathol. 189, 502–512 (2019).
Li, J., Zhao, Y. R. & Tian, Z. Roles of hepatic stellate cells in acute liver failure: From the perspective of inflammation and fibrosis. World J. Hepatol. 11, 412–420 (2019).
Grunhut, J. et al. Macrophages in nonalcoholic steatohepatitis: friend or foe? Eur. Med. J. Hepatol. 6, 100–109 (2018).
Nishimura, T. & Nakauchi, H. Generation of antigen-specific T cells from human induced pluripotent stem cells. Methods Mol. Biol. 1899, 25–40 (2019).
Wang, G. et al. Modeling the mitochondrial cardiomyopathy of Barth syndrome with induced pluripotent stem cell and heart-on-chip technologies. Nat. Med. 20, 616–623 (2014).
Brown, J. A. et al. Metabolic consequences of inflammatory disruption of the blood-brain barrier in an organ-on-chip model of the human neurovascular unit. J. Neuroinflammation 13, 306 (2016).
Jenkins, R. W. et al. Ex vivo profiling of PD-1 blockade using organotypic tumor spheroids. Cancer Discov. 8, 196–215 (2018).
Peck, R. W., Hinojosa, C. D. & Hamilton, G. A. Organs-on-chips in clinical pharmacology: putting the patient into the center of treatment selection and drug development. Clin. Pharmacol. Ther. 107, 181–185 (2020).
Yi, H. G. et al. A bioprinted human-glioblastoma-on-a-chip for the identification of patient-specific responses to chemoradiotherapy. Nat. Biomed. Eng. 3, 509–519 (2019).
Aref, A. R. et al. 3D microfluidic ex vivo culture of organotypic tumor spheroids to model immune checkpoint blockade. Lab Chip 18, 3129–3143 (2018).
Shirure, V. S. et al. Tumor-on-a-chip platform to investigate progression and drug sensitivity in cell lines and patient-derived organoids. Lab Chip 18, 3687–3702 (2018).
Miller, C. P., Shin, W., Ahn, E. H., Kim, H. J. & Kim, D. H. Engineering microphysiological immune system responses on chips. Trends Biotechnol. 38, 857–872 (2020).
Chen, W. L. K. et al. Integrated gut/liver microphysiological systems elucidates inflammatory inter-tissue crosstalk. Biotechnol. Bioeng. 114, 2648–2659 (2017).
Li, G., Huang, K., Nikolic, D. & van Breemen, R. B. High-throughput cytochrome P450 cocktail inhibition assay for assessing drug-drug and drug-botanical interactions. Drug Metab. Dispos. 43, 1670–1678 (2015).
Siramshetty, V. B. et al. WITHDRAWN–a resource for withdrawn and discontinued drugs. Nucleic Acids Res. 44, D1080–D1086 (2016).
Wikswo, J. P. et al. Integrated human organ-on-chip microphysiolocal systems. US patent US 2015/0004077 A1 (2015).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
A.S.-G. is co-founder and D.L.T. is an adviser for Von Baer Wolff Inc., a company focused on biofabrication of autologous human hepatocytes using stem cell technology and genetic reprogramming to overcome liver failure. Their interests are managed by the Conflict of Interest Office at the University of Pittsburgh, USA, in accordance with their policies. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Gastroenterology & Hepatology thanks J. Hickman, Y. Zhang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
U.S. National Library of Medicine Clinical Trials.gov: https://clinicaltrials.gov/
Supplementary information
Glossary
- Absorption, distribution, metabolism, excretion and toxicity
-
(ADMET). Studies conducted during the drug discovery, lead optimization and preclinical development phases to provide information for characterization and ranking of compounds based on their properties and to predict their fate after administration into the human body.
- Micropatterned cell arrays
-
Methodologies, often based on nanofabrication, to fix one or more cell types on a substrate with precisely controlled spatial distributions.
- Spheroids
-
In vitro 3D spherical aggregates of cells of either a single cell type or a combination of cells generated by a variety of 3D culturing methods.
- Organoids
-
3D multicellular systems produced primarily from patient-specific stem cells and their progenies via in situ differentiation, cell sorting and self-organization processes.
- Plate-based platforms
-
Platforms designed around microplate standards from the Society of Biomolecular Sciences, available in 6–1,536-well formats.
- Fit-for-purpose
-
A drug development tool that has been accepted for use in a specific application based on thorough evaluation of the information provided.
- Synthetic biology
-
An interdisciplinary area of science focused on the (re)design and construction of biological systems in a bottom-up fashion, often through the engineering of well-characterized genetic components, modules and devices to attain new functions or to correct dysregulated ones.
- Secretome
-
A set of proteins expressed by cells (organs) and secreted into the extracellular space, including cytokines, growth factors, extracellular matrix proteins mediating autocrine, paracrine, endocrine (via circulation) and/or exocrine (via ducts) physiological regulation or pathophysiological dysregulation.
- Clearance
-
The collection of processes by which the body removes a drug, generally categorized as metabolism or elimination.
- Pharmacokinetic models
-
Quantitative models that predict how an organism influences the absorption, distribution, metabolism and excretion of a drug.
- Pharmacodynamic models
-
A quantitative integration of pharmacokinetics, pharmacological systems and (patho-) physiological processes to understand the intensity and time course of drug effects on the body.
Rights and permissions
About this article
Cite this article
Gough, A., Soto-Gutierrez, A., Vernetti, L. et al. Human biomimetic liver microphysiology systems in drug development and precision medicine. Nat Rev Gastroenterol Hepatol 18, 252–268 (2021). https://doi.org/10.1038/s41575-020-00386-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41575-020-00386-1
- Springer Nature Limited
This article is cited by
-
Microneedle array facilitates hepatic sinusoid construction in a large-scale liver-acinus-chip microsystem
Microsystems & Nanoengineering (2023)