Abstract
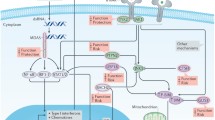
A perplexing feature of type 1 diabetes (T1D) is that the immune system destroys pancreatic β-cells but not neighbouring α-cells, even though both β-cells and α-cells are dysfunctional. Dysfunction, however, progresses to death only for β-cells. Recent findings indicate important differences between these two cell types. First, expression of BCL2L1, a key antiapoptotic gene, is higher in α-cells than in β-cells. Second, endoplasmic reticulum (ER) stress-related genes are differentially expressed, with higher expression levels of pro-apoptotic CHOP in β-cells than in α-cells and higher expression levels of HSPA5 (which encodes the protective chaperone BiP) in α-cells than in β-cells. Third, expression of viral recognition and innate immune response genes is higher in α-cells than in β-cells, contributing to the enhanced resistance of α-cells to coxsackievirus infection. Fourth, expression of the immune-inhibitory HLA-E molecule is higher in α-cells than in β-cells. Of note, α-cells are less immunogenic than β-cells, and the CD8+ T cells invading the islets in T1D are reactive to pre-proinsulin but not to glucagon. We suggest that this finding is a result of the enhanced capacity of the α-cell to endure viral infections and ER stress, which enables them to better survive early stressors that can cause cell death and consequently amplify antigen presentation to the immune system. Moreover, the processing of the pre-proglucagon precursor in enteroendocrine cells might favour immune tolerance towards this potential self-antigen compared to pre-proinsulin.
Key points
-
Pancreatic β-cells and α-cells are both dysfunctional in type 1 diabetes (T1D) but, while β-cells are killed, α-cells survive.
-
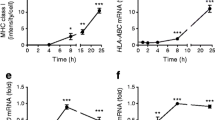
Exposure of islet cells to interferon-α (IFNα), a cytokine that is induced early in T1D pathogenesis, induces expression of both similar genes (such as HLA-related genes) and different genes (such as BCL2L1, endoplasmic reticulum (ER) stress-related genes, innate immune response genes and antiviral response genes) in β-cells and α-cells.
-
Expression of candidate genes for T1D shows major differences between β-cells and α-cells.
-
The antigen presentation capacity seems similar in β-cells and α-cells, but either α-cells are less antigenic than β-cells (perhaps owing to higher HLA-E expression) or their capacity to better endure viral infections and ER stress increases their survival when facing diabetogenic stressors and thus decreases antigen presentation.
-
Pre-proglucagon processing in enteroendocrine cells might favour immune tolerance towards glucagon and further limit α-cell immunogenicity.



Similar content being viewed by others
References
Carre, A. & Mallone, R. Making insulin and staying out of autoimmune trouble: the beta-cell conundrum. Front. Immunol. 12, 639682 (2021).
Anderson, A. M. et al. Human islet T cells are highly reactive to preproinsulin in type 1 diabetes. Proc. Natl Acad. Sci. USA 118, e2107208118 (2021).
Gonzalez-Duque, S. et al. Conventional and neo-antigenic peptides presented by β cells are targeted by circulating naive CD8+ T cells in type 1 diabetic and healthy donors. Cell Metab. 28, 946–960.e6 (2018).
Azoury, M. E. et al. Peptides derived from insulin granule proteins are targeted by CD8+ T cells across MHC class I restrictions in humans and NOD mice. Diabetes 69, 2678–2690 (2020).
Damond, N. et al. A map of human type 1 diabetes progression by imaging mass cytometry. Cell Metab. 29, 755–768.e5 (2019).
Coppieters, K. T. et al. Demonstration of islet-autoreactive CD8 T cells in insulitic lesions from recent onset and long-term type 1 diabetes patients. J. Exp. Med. 209, 51–60 (2012).
Campbell-Thompson, M. L. et al. The diagnosis of insulitis in human type 1 diabetes. Diabetologia 56, 2541–2543 (2013).
Carre, A., Richardson, S. J., Larger, E. & Mallone, R. Presumption of guilt for T cells in type 1 diabetes: lead culprits or partners in crime depending on age of onset? Diabetologia 64, 15–25 (2021).
Brissova, M. et al. α Cell function and gene expression are compromised in type 1 diabetes. Cell Rep. 22, 2667–2676 (2018).
Martinez, M. S. et al. The role of the α cell in the pathogenesis of diabetes: a world beyond the mirror. Int. J. Mol. Sci. 22, 9504 (2021).
Unger, R. H. & Orci, L. The role of glucagon in the endogenous hyperglycemia of diabetes mellitus. Annu. Rev. Med. 28, 119–130 (1977).
Raskin, P. & Unger, R. H. Hyperglucagonemia and its suppression. Importance in the metabolic control of diabetes. N. Engl. J. Med. 299, 433–436 (1978).
Müller, W. A., Faloona, G. R., Aguilar-Parada, E. & Unger, R. H. Abnormal alpha-cell function in diabetes. Response to carbohydrate and protein ingestion. N. Engl. J. Med. 283, 109–115 (1970).
Doliba, N. M. et al. α Cell dysfunction in islets from nondiabetic, glutamic acid decarboxylase autoantibody-positive individuals. J. Clin. Invest. 132, e156243 (2022).
Gromada, J., Chabosseau, P. & Rutter, G. A. The α-cell in diabetes mellitus. Nat. Rev. Endocrinol. 14, 694–704 (2018).
Camunas-Soler, J. et al. Patch-Seq links single-cell transcriptomes to human islet dysfunction in diabetes. Cell Metab. 31, 1017–1031.e14 (2020).
Bosco, D. et al. Unique arrangement of α- and β-cells in human islets of Langerhans. Diabetes 59, 1202–1210 (2010).
Campbell, J. E. & Newgard, C. B. Mechanisms controlling pancreatic islet cell function in insulin secretion. Nat. Rev. Mol. Cell Biol. 22, 142–158 (2021).
Bosi, E., Marchetti, P., Rutter, G. A. & Eizirik, D. L. Human alpha cell transcriptomic signatures of types 1 and 2 diabetes highlight disease-specific dysfunction pathways. iScience 25, 105056 (2022).
Eizirik, D. L., Pasquali, L. & Cnop, M. Pancreatic β-cells in type 1 and type 2 diabetes mellitus: different pathways to failure. Nat. Rev. Endocrinol. 16, 349–362 (2020).
Doyle, A. C. in The Complete Sherlock Holmes (Hamlyn, 1984).
Tesi, M. et al. Pro-inflammatory cytokines induce insulin and glucagon double positive human islet cells that are resistant to apoptosis. Biomolecules 11, 320 (2021).
Lahnemann, D. et al. Eleven grand challenges in single-cell data science. Genome Biol. 21, 31 (2020).
Benninger, R. K., Head, W. S., Zhang, M., Satin, L. S. & Piston, D. W. Gap junctions and other mechanisms of cell-cell communication regulate basal insulin secretion in the pancreatic islet. J. Physiol. 589, 5453–5466 (2011).
Moede, T., Leibiger, I. B. & Berggren, P. O. Alpha cell regulation of beta cell function. Diabetologia 63, 2064–2075 (2020).
Stuart, T. & Satija, R. Integrative single-cell analysis. Nat. Rev. Genet. 20, 257–272 (2019).
Cao, J. et al. The single-cell transcriptional landscape of mammalian organogenesis. Nature 566, 496–502 (2019).
Bergen, V., Lange, M., Peidli, S., Wolf, F. A. & Theis, F. J. Generalizing RNA velocity to transient cell states through dynamical modeling. Nat. Biotechnol. 38, 1408–1414 (2020).
Kaestner, K. H., Powers, A. C., Naji, A., Consortium, H. & Atkinson, M. A. NIH initiative to improve understanding of the pancreas, islet, and autoimmunity in type 1 diabetes: the Human Pancreas Analysis Program (HPAP). Diabetes 68, 1394–1402 (2019).
Fasolino, M. et al. Single-cell multi-omics analysis of human pancreatic islets reveals novel cellular states in type 1 diabetes. Nat. Metab. 4, 284–299 (2022).
Colli, M. L. et al. An integrated multi-omics approach identifies the landscape of interferon-α-mediated responses of human pancreatic beta cells. Nat. Commun. 11, 2584 (2020).
Ramos-Rodriguez, M. et al. The impact of proinflammatory cytokines on the β-cell regulatory landscape provides insights into the genetics of type 1 diabetes. Nat. Genet. 51, 1588–1595 (2019).
Szymczak, F. et al. Transcription and splicing regulation by NLRC5 shape the interferon response in human pancreatic β cells. Sci. Adv. 8, eabn5732 (2022).
Veres, A. et al. Charting cellular identity during human in vitro β-cell differentiation. Nature 569, 368–373 (2019).
Chandra, V. et al. The type 1 diabetes gene TYK2 regulates β-cell development and its responses to interferon-α. Nat. Commun. 13, 6363 (2022).
Leite, N. C., Pelayo, G. C. & Melton, D. A. Genetic manipulation of stress pathways can protect stem-cell-derived islets from apoptosis in vitro. Stem Cell Rep. 17, 766–774 (2022).
Sintov, E. et al. Whole-genome CRISPR screening identifies genetic manipulations to reduce immune rejection of stem cell-derived islets. Stem Cell Rep. 17, 1976–1990 (2022).
Demine, S. et al. Pro-inflammatory cytokines induce cell death, inflammatory responses, and endoplasmic reticulum stress in human iPSC-derived beta cells. Stem Cell Res. Ther. 11, 7 (2020).
Balboa, D. et al. Functional, metabolic and transcriptional maturation of human pancreatic islets derived from stem cells. Nat. Biotechnol. 40, 1042–1055 (2022).
Kallionpaa, H. et al. Standard of hygiene and immune adaptation in newborn infants. Clin. Immunol. 155, 136–147 (2014).
Oresic, M. et al. Cord serum lipidome in prediction of islet autoimmunity and type 1 diabetes. Diabetes 62, 3268–3274 (2013).
Heninger, A. K. et al. Activation of islet autoreactive naive T cells in infants is influenced by homeostatic mechanisms and antigen-presenting capacity. Diabetes 62, 2059–2066 (2013).
Tretina, K., Park, E. S., Maminska, A. & MacMicking, J. D. Interferon-induced guanylate-binding proteins: guardians of host defense in health and disease. J. Exp. Med. 216, 482–500 (2019).
Honkala, A. T., Tailor, D. & Malhotra, S. V. Guanylate-binding protein 1: an emerging target in inflammation and cancer. Front. Immunol. 10, 3139 (2019).
Pagani, I., Poli, G. & Vicenzi, E. TRIM22. A multitasking antiviral factor. Cells 10, 1864 (2021).
Jeong, S. I. et al. XAF1 forms a positive feedback loop with IRF-1 to drive apoptotic stress response and suppress tumorigenesis. Cell Death Dis. 9, 806 (2018).
Tessier, M. C. et al. Type 1 diabetes and the OAS gene cluster: association with splicing polymorphism or haplotype? J. Med. Genet. 43, 129–132 (2006).
Op de Beeck, A. & Eizirik, D. L. Viral infections in type 1 diabetes mellitus – why the β cells? Nat. Rev. Endocrinol. 12, 263–273 (2016).
Dunne, J. L. et al. Rationale for enteroviral vaccination and antiviral therapies in human type 1 diabetes. Diabetologia 62, 744–753 (2019).
Lloyd, R. E., Tamhankar, M. & Lernmark, A. Enteroviruses and type 1 diabetes: multiple mechanisms and factors? Annu. Rev. Med. 73, 483–499 (2022).
Nejentsev, S., Walker, N., Riches, D., Egholm, M. & Todd, J. A. Rare variants of IFIH1, a gene implicated in antiviral responses, protect against type 1 diabetes. Science 324, 387–389 (2009).
Winkler, C. et al. An interferon-induced helicase (IFIH1) gene polymorphism associates with different rates of progression from autoimmunity to type 1 diabetes. Diabetes 60, 685–690 (2011).
Marroqui, L. et al. Differential cell autonomous responses determine the outcome of coxsackievirus infections in murine pancreatic α and β cells. eLife 4, e06990 (2015).
Nigi, L. et al. Increased expression of viral sensor MDA5 in pancreatic islets and in hormone-negative endocrine cells in recent onset type 1 diabetic donors. Front. Immunol. 13, 833141 (2022).
Welsh, N. et al. Differences in the expression of heat-shock proteins and antioxidant enzymes between human and rodent pancreatic islets: implications for the pathogenesis of insulin-dependent diabetes mellitus. Mol. Med. 1, 806–820 (1995).
Lenzen, S. Chemistry and biology of reactive species with special reference to the antioxidative defence status in pancreatic β-cells. Biochim. Biophys. Acta Gen. Subj. 1861, 1929–1942 (2017).
Redondo, M. J. et al. Type 1 diabetes in diverse ancestries and the use of genetic risk scores. Lancet Diabetes Endocrinol. 10, 597–608 (2022).
Onengut-Gumuscu, S. et al. Fine mapping of type 1 diabetes susceptibility loci and evidence for colocalization of causal variants with lymphoid gene enhancers. Nat. Genet. 47, 381–386 (2015).
Szymczak, F., Colli, M. L., Mamula, M. J., Evans-Molina, C. & Eizirik, D. L. Gene expression signatures of target tissues in type 1 diabetes, lupus erythematosus, multiple sclerosis, and rheumatoid arthritis. Sci. Adv. 7, eabd7600 (2021).
Eizirik, D. L. et al. The human pancreatic islet transcriptome: expression of candidate genes for type 1 diabetes and the impact of pro-inflammatory cytokines. PLoS Genet. 8, e1002552 (2012).
Mawla, A. M. & Huising, M. O. Navigating the depths and avoiding the shallows of pancreatic islet cell transcriptomes. Diabetes 68, 1380–1393 (2019).
Marroqui, L. et al. BACH2, a candidate risk gene for type 1 diabetes, regulates apoptosis in pancreatic β-cells via JNK1 modulation and crosstalk with the candidate gene PTPN2. Diabetes 63, 2516–2527 (2014).
Rasschaert, J. et al. Toll-like receptor 3 and STAT-1 contribute to double-stranded RNA + interferon-γ-induced apoptosis in primary pancreatic β-cells. J. Biol. Chem. 280, 33984–33991 (2005).
Garcia, M. et al. Regulation and function of the cytosolic viral RNA sensor RIG-I in pancreatic beta cells. Biochim. Biophys. Acta 1793, 1768–1775 (2009).
Colli, M. L., Moore, F., Gurzov, E. N., Ortis, F. & Eizirik, D. L. MDA5 and PTPN2, two candidate genes for type 1 diabetes, modify pancreatic β-cell responses to the viral by-product double-stranded RNA. Hum. Mol. Genet. 19, 135–146 (2010).
Richardson, S. J., Morgan, N. G. & Foulis, A. K. Pancreatic pathology in type 1 diabetes mellitus. Endocr. Pathol. 25, 80–92 (2014).
Kallionpaa, H. et al. Innate immune activity is detected prior to seroconversion in children with HLA-conferred type 1 diabetes susceptibility. Diabetes 63, 2402–2414 (2014).
Ferreira, R. C. et al. A type I interferon transcriptional signature precedes autoimmunity in children genetically at risk for type 1 diabetes. Diabetes 63, 2538–2550 (2014).
Lundberg, M., Krogvold, L., Kuric, E., Dahl-Jorgensen, K. & Skog, O. Expression of interferon-stimulated genes in insulitic pancreatic islets of patients recently diagnosed with type 1 diabetes. Diabetes 65, 3104–3110 (2016).
Krogvold, L. et al. Detection of antiviral tissue responses and increased cell stress in the pancreatic islets of newly diagnosed type 1 diabetes patients: results from the DiViD study. Front. Endocrinol. 13, 881997 (2022).
Marroqui, L. et al. Interferon-α mediates human beta cell HLA class I overexpression, endoplasmic reticulum stress and apoptosis, three hallmarks of early human type 1 diabetes. Diabetologia 60, 656–667 (2017).
Cho, H. et al. Differential innate immune response programs in neuronal subtypes determine susceptibility to infection in the brain by positive-stranded RNA viruses. Nat. Med. 19, 458–464 (2013).
Lucas, T. M., Richner, J. M. & Diamond, M. S. The interferon-stimulated gene Ifi27l2a restricts West Nile virus infection and pathogenesis in a cell-type- and region-specific manner. J. Virol. 90, 2600–2615 (2015).
Croft, N. P. et al. Kinetics of antigen expression and epitope presentation during virus infection. PLoS Pathog. 9, e1003129 (2013).
Colli, M. L., Szymczak, F. & Eizirik, D. L. Molecular footprints of the immune assault on pancreatic beta cells in type 1 diabetes. Front. Endocrinol. 11, 568446 (2020).
Andre, P. et al. Anti-NKG2A mAb is a checkpoint inhibitor that promotes anti-tumor immunity by unleashing both T and NK cells. Cell 175, 1731–1743.e13 (2018).
Borst, L., van der Burg, S. H. & van Hall, T. The NKG2A-HLA-E axis as a novel checkpoint in the tumor microenvironment. Clin. Cancer Res. 26, 5549–5556 (2020).
Schuit, F. C., In’t Veld, P. A. & Pipeleers, D. G. Glucose stimulates proinsulin biosynthesis by a dose-dependent recruitment of pancreatic beta cells. Proc. Natl Acad. Sci. USA 85, 3865–3869 (1988).
Eizirik, D. L. & Cnop, M. ER stress in pancreatic β cells: the thin red line between adaptation and failure. Sci. Signal. 3, pe7 (2010).
Sahin, G. S., Lee, H. & Engin, F. An accomplice more than a mere victim: the impact of β-cell ER stress on type 1 diabetes pathogenesis. Mol. Metab. 54, 101365 (2021).
Yong, J., Johnson, J. D., Arvan, P., Han, J. & Kaufman, R. J. Therapeutic opportunities for pancreatic β-cell ER stress in diabetes mellitus. Nat. Rev. Endocrinol. 17, 455–467 (2021).
Marchetti, P. et al. The endoplasmic reticulum in pancreatic beta cells of type 2 diabetes patients. Diabetologia 50, 2486–2494 (2007).
Marhfour, I. et al. Expression of endoplasmic reticulum stress markers in the islets of patients with type 1 diabetes. Diabetologia 55, 2417–2420 (2012).
Engin, F. et al. Restoration of the unfolded protein response in pancreatic β cells protects mice against type 1 diabetes. Sci. Transl Med. 5, 211ra156 (2013).
Chen, C. W. et al. Adaptation to chronic ER stress enforces pancreatic β-cell plasticity. Nat. Commun. 13, 4621 (2022).
Marroqui, L. et al. Pancreatic α cells are resistant to metabolic stress-induced apoptosis in type 2 diabetes. EBioMedicine 2, 378–385 (2015).
James, E. A., Mallone, R., Kent, S. C. & DiLorenzo, T. P. T-cell epitopes and neo-epitopes in type 1 diabetes: a comprehensive update and reappraisal. Diabetes 69, 1311–1335 (2020).
Moriyama, H. et al. Evidence for a primary islet autoantigen (preproinsulin 1) for insulitis and diabetes in the nonobese diabetic mouse. Proc. Natl Acad. Sci. USA 100, 10376–10381 (2003).
Nakayama, M. et al. Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature 435, 220–223 (2005).
Krishnamurthy, B. et al. Responses against islet antigens in NOD mice are prevented by tolerance to proinsulin but not IGRP. J. Clin. Invest. 116, 3258–3265 (2006).
Kracht, M. J. et al. Autoimmunity against a defective ribosomal insulin gene product in type 1 diabetes. Nat. Med. 23, 501–507 (2017).
Delong, T. et al. Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science 351, 711–714 (2016).
Babon, J. A. et al. Analysis of self-antigen specificity of islet-infiltrating T cells from human donors with type 1 diabetes. Nat. Med. 22, 1482–1487 (2016).
Baker, R. L. et al. Hybrid insulin peptides are autoantigens in type 1 diabetes. Diabetes 68, 1830–1840 (2019).
Wiles, T. A. et al. Characterization of human CD4 T cells specific for a C-peptide/C-peptide hybrid insulin peptide. Front. Immunol. 12, 668680 (2021).
Skowera, A. et al. CTLs are targeted to kill beta cells in patients with type 1 diabetes through recognition of a glucose-regulated preproinsulin epitope. J. Clin. Invest. 118, 3390–3402 (2008).
Kronenberg-Versteeg, D. et al. Molecular pathways for immune recognition of preproinsulin signal peptide in type 1 diabetes. Diabetes 67, 687–696 (2018).
Wan, X. et al. Pancreatic islets communicate with lymphoid tissues via exocytosis of insulin peptides. Nature 560, 107–111 (2018).
Mallone, R. & Eizirik, D. L. Presumption of innocence for beta cells: why are they vulnerable autoimmune targets in type 1 diabetes? Diabetologia 63, 1999–2006 (2020).
Scotto, M. et al. Zinc transporter (ZnT)8186–194 is an immunodominant CD8+ T cell epitope in HLA-A2+ type 1 diabetic patients. Diabetologia 55, 2026–2031 (2012).
Culina, S. et al. Islet-reactive CD8+ T cell frequencies in the pancreas, but not in blood, distinguish type 1 diabetic patients from healthy donors. Sci. Immunol. 3, eaao4013 (2018).
Blancou, P. et al. Immunization of HLA class I transgenic mice identifies autoantigenic epitopes eliciting dominant responses in type 1 diabetes patients. J. Immunol. 178, 7458–7466 (2007).
Segerstolpe, A. et al. Single-cell transcriptome profiling of human pancreatic islets in health and type 2 diabetes. Cell Metab. 24, 593–607 (2016).
MacDonald, P. E. & Rorsman, P. Metabolic messengers: glucagon. Nat. Metab. 5, 186–192 (2023).
Foster, M. C., Leapman, R. D., Li, M. X. & Atwater, I. Elemental composition of secretory granules in pancreatic islets of Langerhans. Biophys. J. 64, 525–532 (1993).
Bach, J. F. The multi-faceted zinc dependency of the immune system. Immunol. Today 2, 225–227 (1981).
Prasad, A. S. Lessons learned from experimental human model of zinc deficiency. J. Immunol. Res. 2020, 9207279 (2020).
Haase, H. et al. Zinc signals are essential for lipopolysaccharide-induced signal transduction in monocytes. J. Immunol. 181, 6491–6502 (2008).
Yu, M. et al. Regulation of T cell receptor signaling by activation-induced zinc influx. J. Exp. Med. 208, 775–785 (2011).
Acevedo-Murillo, J. A. et al. Zinc supplementation promotes a Th1 response and improves clinical symptoms in fewer hours in children with pneumonia younger than 5 years old. A randomized controlled clinical trial. Front. Pediatr. 7, 431 (2019).
Beck, F. W., Prasad, A. S., Kaplan, J., Fitzgerald, J. T. & Brewer, G. J. Changes in cytokine production and T cell subpopulations in experimentally induced zinc-deficient humans. Am. J. Physiol. 272, E1002–E1007 (1997).
Hostens, K. et al. Exposure of human islets to cytokines can result in disproportionately elevated proinsulin release. J. Clin. Invest. 104, 67–72 (1999).
Rodriguez-Calvo, T. et al. Increase in pancreatic proinsulin and preservation of β-cell mass in autoantibody-positive donors prior to type 1 diabetes onset. Diabetes 66, 1334–1345 (2017).
Wasserfall, C. et al. Persistence of pancreatic insulin mRNA expression and proinsulin protein in type 1 diabetes pancreata. Cell Metab. 26, 568–575.e3 (2017).
Sims, E. K. et al. Elevations in the fasting serum proinsulin-to-C-peptide ratio precede the onset of type 1 diabetes. Diabetes Care 39, 1519–1526 (2016).
Sims, E. K., Evans-Molina, C., Tersey, S. A., Eizirik, D. L. & Mirmira, R. G. Biomarkers of islet beta cell stress and death in type 1 diabetes. Diabetologia 61, 2259–2265 (2018).
Coombes, J. L. et al. A functionally specialized population of mucosal CD103+ DCs induces Foxp3+ regulatory T cells via a TGF-β- and retinoic acid-dependent mechanism. J. Exp. Med. 204, 1757–1764 (2007).
Sun, C. M. et al. Small intestine lamina propria dendritic cells promote de novo generation of Foxp3 T reg cells via retinoic acid. J. Exp. Med. 204, 1775–1785 (2007).
Weiner, H. L., da Cunha, A. P., Quintana, F. & Wu, H. Oral tolerance. Immunol. Rev. 241, 241–259 (2011).
Hadjiyanni, I., Siminovitch, K. A., Danska, J. S. & Drucker, D. J. Glucagon-like peptide-1 receptor signalling selectively regulates murine lymphocyte proliferation and maintenance of peripheral regulatory T cells. Diabetologia 53, 730–740 (2010).
Thorel, F. et al. Normal glucagon signaling and β-cell function after near-total α-cell ablation in adult mice. Diabetes 60, 2872–2882 (2011).
Acknowledgements
D.L.E. acknowledges the support of grants from the Welbio-FNRS (Fonds National de la Recherche Scientifique) (WELBIO-CR-2019C-04), Belgium; the Dutch Diabetes Research Foundation (Innovate2CureType1), Netherlands; the JDRF (3-SRA-2022-1201-S-B); the National Institutes of Health Human Islet Research Network Consortium on Beta Cell Death & Survival from Pancreatic β-Cell Gene Networks to Therapy (HIRN-CBDS) (grant U01 DK127786). D.L.E. and R.M. acknowledge support from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreements 115797 (INNODIA) and 945268 (INNODIA HARVEST). These joint undertakings receive support from the European Union’s Horizon 2020 research and innovation programme and the European Federation of Pharmaceutical Industries and Associations (EFPIA), JDRF, and The Leona M. and Harry B. Helmsley Charitable Trust. F.S. is supported by a Research Fellow (Aspirant) fellowship from the Fonds National de la Recherche Scientifique (FNRS, Belgium). R.M. acknowledges the support of grants from Agence Nationale de la Recherche (ANR-19-CE15-0014-01), Fondation pour la Recherche Medicale (EQU20193007831), and from The Leona M. and Harry B. Helmsley Charitable Trust to INSERM.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, contributed substantially to discussion of the content, wrote sections of the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
D.L.E. received grant support from Eli Lilly, Indianapolis, IN, for research on new approaches to protect pancreatic β-cells in T1D (not directly related to the present study). The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Endocrinology thanks Maureen Gannon, Jason Collier and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Review criteria
Relevant publications were identified by searching the PubMed database (1 January 2005 to September 2022) using combinations of the following terms: ‘pancreatic beta cells’, ‘pancreatic alpha cells’, ‘pancreatic β-cells’, ‘pancreatic α-cells’, ‘pancreatic islets’, ‘insulin release’, ‘insulin secretion’, ‘glucagon release’, ‘glucagon secretion’, ‘diabetes’, ‘type 1 diabetes’, ‘type 2 diabetes’, ‘pathogenesis’, ‘histology’, ‘transcriptome’, ‘genetics’, ‘candidate genes’, ‘islet gene regulation’, ‘islet epigenomics’, ‘viral infection’, ‘endoplasmic reticulum stress’ and ‘apoptosis’. We preferentially selected publications from the past 5 years, plus earlier key publications for citation (of note, the literature on α-cells in T1D is rather limited). Some references cited in these papers or in relevant articles related to the fate of pancreatic β-cells and α-cells in diabetes were also searched manually. All selected papers were full-text articles in English. Review articles are often cited to provide the readers with additional references.
Glossary
- Antigens
-
Molecular structures (proteins, peptides, polysaccharides, lipids or nucleic acids) that can bind to an antigen receptor (for example, antibodies for B cells and T cell receptors for T cells) and trigger an immune response. Antigens can originate from within the body (self-antigens or autoantigens) or from the external environment (foreign antigens).
- Candidate genes
-
Genes related to particular traits that either increase or decrease the risk of disease, either as a result of their protein product or their position on a chromosome.
- Epitopes
-
The specific parts of the antigen (most commonly peptides) to which antigen receptors bind.
- Gene set enrichment analysis
-
Computational method to determine whether an a priori defined set of genes shows statistically significant differences between two biological states (for example, phenotypes).
- Immune tolerance
-
The state of unresponsiveness of the immune system to antigens that have the potential to induce an immune response. Immune tolerance to self-antigens is achieved through both central tolerance and peripheral tolerance mechanisms in the thymus and in the periphery, respectively.
- Insulitis
-
Inflammation of the islets of Langerhans, characterized by infiltration of immune cells within and at the periphery of islets.
- Leading edges
-
Subsets of genes in a gene set that contribute the most to the enrichment or depletion in a gene set enrichment analysis.
- Neoantigens
-
Peptide sequences not templated in the genome that can be preferentially recognized as non-self and trigger autoimmunity. Neoantigens can be generated by mis-initiated mRNA transcription, alternative mRNA splicing and post-translational modifications (that is, the addition of chemical groups to amino acid residues or the fusion of non-contiguous fragments from the same protein (cis-splicing) or of two fragments from different proteins (trans-splicing, generating so-called hybrid peptides)).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Eizirik, D.L., Szymczak, F. & Mallone, R. Why does the immune system destroy pancreatic β-cells but not α-cells in type 1 diabetes?. Nat Rev Endocrinol 19, 425–434 (2023). https://doi.org/10.1038/s41574-023-00826-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-023-00826-3
- Springer Nature Limited
This article is cited by
-
Identification of unique cell type responses in pancreatic islets to stress
Nature Communications (2024)
-
β-cells protected from T1DM by early senescence programme
Nature Reviews Endocrinology (2024)
-
The relationship between SARS-CoV-2 infection and type 1 diabetes mellitus
Nature Reviews Endocrinology (2024)
-
tRNA-derived fragments in T lymphocyte–beta cell crosstalk and in type 1 diabetes pathogenesis in NOD mice
Diabetologia (2024)