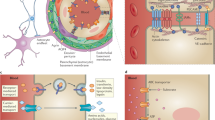
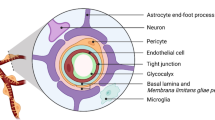
Abstract
For a blood-borne cancer therapeutic agent to be effective, it must cross the blood vessel wall to reach cancer cells in adequate quantities, and it must overcome the resistance conferred by the local microenvironment around cancer cells. The brain microenvironment can thwart the effectiveness of drugs against primary brain tumours as well as brain metastases. In this Review, we highlight the cellular and molecular components of the blood–brain barrier (BBB), a specialized neurovascular unit evolved to maintain brain homeostasis. Tumours are known to compromise the integrity of the BBB, resulting in a vasculature known as the blood–tumour barrier (BTB), which is highly heterogeneous and characterized by numerous distinct features, including non-uniform permeability and active efflux of molecules. We discuss the challenges posed by the BBB and BTB for drug delivery, how multiple cell types dictate BBB function and the role of the BTB in disease progression and treatment. Finally, we highlight emerging molecular, cellular and physical strategies to improve drug delivery across the BBB and BTB and discuss their impact on improving conventional as well as emerging treatments, such as immune checkpoint inhibitors and engineered T cells. A deeper understanding of the BBB and BTB through the application of single-cell sequencing and imaging techniques, and the development of biomarkers of BBB integrity along with systems biology approaches, should enable new personalized treatment strategies for primary brain malignancies and brain metastases.



Similar content being viewed by others
References
O’Brown, N. M., Pfau, S. J. & Gu, C. Bridging barriers: a comparative look at the blood–brain barrier across organisms. Genes Dev. 32, 466–478 (2018). This review describes the conservation and variation of BBB function across organisms.
Abbott, N. J. Blood–brain barrier structure and function and the challenges for CNS drug delivery. J. Inherit. Metab. Dis. 36, 437–449 (2013).
Hobbs, S. K. et al. Regulation of transport pathways in tumor vessels: role of tumor type and microenvironment. Proc. Natl Acad. Sci. USA 95, 4607–4612 (1998). This study presents the first systematic examination of the role of the brain versus the subcutaneous microenvironment on permeability of tumour vessels.
Monsky, W. L. et al. Role of host microenvironment in angiogenesis and microvascular functions in human breast cancer xenografts: mammary fat pad versus cranial tumors. Clin. Cancer Res. 8, 1008–1013 (2002). This study demonstrates the differential response of blood vessels in tumours growing in the brain versus in the subcutaneous microenvironment.
Pitz, M. W., Desai, A., Grossman, S. A. & Blakeley, J. O. Tissue concentration of systemically administered antineoplastic agents in human brain tumors. J. Neurooncol. 104, 629–638 (2011).
Sarkaria, J. N. et al. Is the blood–brain barrier really disrupted in all glioblastomas? A critical assessment of existing clinical data. Neuro. Oncol. 20, 184–191 (2018).
Saunders, N. R. et al. The rights and wrongs of blood–brain barrier permeability studies: a walk through 100 years of history. Front. Neurosci. 8, 404 (2014).
Daneman, R. & Prat, A. The blood–brain barrier. Cold Spring Harb. Perspect. Biol. 7, a020412 (2015).
Dombrowski, Y. et al. Regulatory T cells promote myelin regeneration in the central nervous system. Nat. Neurosci. 20, 674–680 (2017).
Prinz, M. & Priller, J. The role of peripheral immune cells in the CNS in steady state and disease. Nat. Neurosci. 20, 136–144 (2017).
Haddad-Tovolli, R., Dragano, N. R. V., Ramalho, A. F. S. & Velloso, L. A. Development and function of the blood–brain barrier in the context of metabolic control. Front. Neurosci 11, 224 (2017).
Sweeney, M. D., Zhao, Z., Montagne, A., Nelson, A. R. & Zlokovic, B. V. Blood–brain barrier: from physiology to disease and back. Physiol. Rev. 99, 21–78 (2018).
Zhao, Z., Nelson, A. R., Betsholtz, C. & Zlokovic, B. V. Establishment and dysfunction of the blood–brain barrier. Cell 163, 1064–1078 (2015).
Møllgård, K., Dziegielewska, K. M., Holst, C. B., Habgood, M. D. & Saunders, N. R. Brain barriers and functional interfaces with sequential appearance of ABC efflux transporters during human development. Sci. Rep. 7, 11603 (2017).
Phoenix, T. N. et al. Medulloblastoma genotype dictates blood brain barrier phenotype. Cancer Cell 29, 508–522 (2016). This paper demonstrates that the WNT subtype of medulloblastoma produces antagonists of WNT7A that cause fenestrations in the ECs of the BBB.
Triarico, S. et al. Improving the brain delivery of chemotherapeutic drugs in childhood brain tumors. Cancers 11, E824 (2019).
Griveau, A. et al. A glial signature and Wnt7 signaling regulate glioma–vascular interactions and tumor microenvironment. Cancer Cell 33, 874–889.e7 (2018). This study shows the molecular mechanisms of vessel co-option versus angiogenesis by different subtypes of brain tumours.
Kuhnert, F. et al. Essential regulation of CNS angiogenesis by the orphan G protein-coupled receptor GPR124. Science 330, 985–989 (2010).
Umans, R. A. et al. CNS angiogenesis and barriergenesis occur simultaneously. Dev. Biol. 425, 101–108 (2017).
Vanlandewijck, M. et al. A molecular atlas of cell types and zonation in the brain vasculature. Nature 554, 475–480 (2018). Using vascular single-cell transcriptomics, this paper provides molecular definitions for the principal types of blood vascular and vessel-associated cells in the adult mouse brain.
Saunders, A. et al. Molecular diversity and specializations among the cells of the adult mouse brain. Cell 174, 1015–1030.e16 (2018).
Sabbagh, M. F. et al. Transcriptional and epigenomic landscapes of CNS and non-CNS vascular endothelial cells. eLife 7, e36187 (2018).
Khan, S. et al. EndoDB: a database of endothelial cell transcriptomics data. Nucleic Acids Res. 47, D736–D744 (2019).
He, L. et al. Single-cell RNA sequencing of mouse brain and lung vascular and vessel-associated cell types. Sci. Data 5, 180160 (2018).
Kaisar, M. A. et al. New experimental models of the blood–brain barrier for CNS drug discovery. Expert Opin. Drug Discov. 12, 89–103 (2017).
Tietz, S. & Engelhardt, B. Brain barriers: crosstalk between complex tight junctions and adherens junctions. J. Cell Biol. 209, 493–506 (2015).
Stamatovic, S. M., Johnson, A. M., Keep, R. F. & Andjelkovic, A. V. Junctional proteins of the blood–brain barrier: new insights into function and dysfunction. Tissue Barriers 4, e1154641 (2016).
Thomsen, M. S., Routhe, L. J. & Moos, T. The vascular basement membrane in the healthy and pathological brain. J. Cereb. Blood Flow Metab. 37, 3300–3317 (2017).
Ayloo, S. & Gu, C. Transcytosis at the blood–brain barrier. Curr. Opin. Neurobiol. 57, 32–38 (2019).
Chow, B. W. & Gu, C. The molecular constituents of the blood–brain barrier. Trends Neurosci. 38, 598–608 (2015).
Robey, R. W. et al. Revisiting the role of ABC transporters in multidrug-resistant cancer. Nat. Rev. Cancer 18, 452 (2018).
Schinkel, A. H. et al. Disruption of the mouse mdr1a P-glycoprotein gene leads to a deficiency in the blood-brain barrier and to increased sensitivity to drugs. Cell 77, 491–502 (1994).
Oberoi, R. K. et al. Strategies to improve delivery of anticancer drugs across the blood–brain barrier to treat glioblastoma. Neuro Oncol. 18, 27–36 (2016).
Sanchez-Covarrubias, L., Slosky, L. M., Thompson, B. J., Davis, T. P. & Ronaldson, P. T. Transporters at CNS barrier sites: obstacles or opportunities for drug delivery? Curr. Pharm. Des. 20, 1422–1449 (2014).
Bell, R. D. et al. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 68, 409–427 (2010).
Armulik, A., Genové, G. & Betsholtz, C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev. Cell 21, 193–215 (2011).
Daneman, R., Zhou, L., Kebede, A. A. & Barres, B. A. Pericytes are required for blood–brain barrier integrity during embryogenesis. Nature 468, 562–566 (2010).
Armulik, A. et al. Pericytes regulate the blood–brain barrier. Nature 468, 557–561 (2010).
Ben-Zvi, A. et al. MSFD2A is critical for the formation and function of the blood–brain barrier. Nature 509, 507–511 (2014).
ElAli, A., Thériault, P. & Rivest, S. The role of pericytes in neurovascular unit remodeling in brain disorders. Int. J. Mol. Sci. 15, 6453–6474 (2014).
Giaume, C., Koulakoff, A., Roux, L., Holcman, D. & Rouach, N. Astroglial networks: a step further in neuroglial and gliovascular interactions. Nat. Rev. Neurosci. 11, 87–99 (2010).
Alvarez, J. I., Katayama, T. & Prat, A. Glial influence on the blood–brain barrier. Glia 61, 1939–1958 (2013).
Horng, S. et al. Astrocytic tight junctions control inflammatory CNS lesion pathogenesis. J. Clin. Invest. 127, 3136–3151 (2017).
Kubotera, H. et al. Astrocytic endfeet re-cover blood vessels after removal by laser ablation. Sci. Rep. 9, 1263 (2019).
Lannes, N., Eppler, E., Etemad, S., Yotovski, P. & Filgueira, L. Microglia at center stage: a comprehensive review about the versatile and unique residential macrophages of the central nervous system. Oncotarget 8, 114393–114413 (2017).
Shemer, A., Erny, D., Jung, S. & Prinz, M. Microglia plasticity during health and disease: an immunological perspective. Trends Immunol. 36, 614–624 (2015).
Sarelius, I. H. & Glading, A. J. Control of vascular permeability by adhesion molecules. Tissue Barriers 3, e985954 (2015).
Mizee, M. R. & de Vries, H. E. Blood–brain barrier regulation. Tissue Barriers 1, e26882 (2013).
Iadecola, C. The neurovascular unit coming of age: a journey through neurovascular coupling in health and disease. Neuron 96, 17–42 (2017).
Tominaga, N. et al. Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood–brain barrier. Nat. Commun. 6, 6716 (2015).
Seano, G. et al. Solid stress in brain tumours causes neuronal loss and neurological dysfunction and can be reversed by lithium. Nat. Biomed. Eng. 3, 230 (2019).
Quail, D. F. & Joyce, J. A. The microenvironmental landscape of brain tumors. Cancer Cell 31, 326–341 (2017).
Carmeliet, P. & Jain, R. K. Molecular mechanisms and clinical applications of angiogenesis. Nature 473, 298–307 (2011). This review discusses how tumours acquire blood vessels by six different mechanisms, including angiogenesis and vessel co-option.
Voutouri, C. et al. Experimental and computational analyses reveal dynamics of tumor vessel cooption and optimal treatment strategies. Proc. Natl Acad. Sci. USA 116, 2662–2671 (2019). This study presents a systematic analysis of vessel co-option by tumours and how to overcome this barrier to increase drug delivery.
Vakoc, B. J. et al. Three-dimensional microscopy of the tumor microenvironment in vivo using optical frequency domain imaging. Nat. Med. 15, 1219–1223 (2009).
Argaw, A. T. et al. IL-1β regulates blood–brain barrier permeability via reactivation of the hypoxia-angiogenesis program. J. Immunol. 177, 5574–5584 (2006).
Engelhardt, S., Patkar, S. & Ogunshola, O. O. Cell-specific blood–brain barrier regulation in health and disease: a focus on hypoxia. Br. J. Pharmacol. 171, 1210–1230 (2014).
Batchelor, T. T. et al. Improved tumor oxygenation and survival in glioblastoma patients who show increased blood perfusion after cediranib and chemoradiation. Proc. Natl Acad. Sci. USA 110, 19059–19064 (2013).
Emblem, K. E. et al. Vessel architectural imaging identifies cancer patient responders to anti-angiogenic therapy. Nat. Med. 19, 1178–1183 (2013).
Pàez-Ribes, M. et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15, 220–231 (2009).
Argaw, A. T., Gurfein, B. T., Zhang, Y., Zameer, A. & John, G. R. VEGF-mediated disruption of endothelial CLN-5 promotes blood–brain barrier breakdown. Proc. Natl Acad. Sci. USA 106, 1977–1982 (2009).
Argaw, A. T. et al. Astrocyte-derived VEGF-A drives blood–brain barrier disruption in CNS inflammatory disease. J. Clin. Invest. 122, 2454–2468 (2012).
van Tellingen, O. et al. Overcoming the blood–brain tumor barrier for effective glioblastoma treatment. Drug Resist. Updat. 19, 1–12 (2015).
Dubois, L. G. et al. Gliomas and the vascular fragility of the blood–brain barrier. Front. Cell. Neurosci. 8, 418 (2014).
Watkins, S. et al. Disruption of astrocyte–vascular coupling and the blood–brain barrier by invading glioma cells. Nat. Commun. 5, 4196 (2014). This study systematically investigates the interaction of invading glioma cells with host cerebral blood vessels during the early stages of disease.
Ratnam, N. M., Gilbert, M. R. & Giles, A. J. Immunotherapy in CNS cancers: the role of immune cell trafficking. Neuro Oncol. 21, 37–46 (2019).
Achrol, A. S. et al. Brain metastases. Nat. Rev. Dis. Primers 5, 5 (2019).
Yonemori, K. et al. Disruption of the blood–brain barrier by brain metastases of triple-negative and basal-type breast cancer but not HER2/neu-positive breast cancer. Cancer 116, 302–308 (2010).
Bassett, E. A. et al. Norrin/Frizzled4 signalling in the preneoplastic niche blocks medulloblastoma initiation. eLife 5, e16764 (2016).
Chang, J. et al. Gpr124 is essential for blood–brain barrier integrity in central nervous system disease. Nat. Med. 23, 450 (2017).
Lyle, L. T. et al. Alterations in pericyte subpopulations are associated with elevated blood–tumor barrier permeability in experimental brain metastasis of breast cancer. Clin. Cancer Res. 22, 5287–5299 (2016).
Lockman, P. R. et al. Heterogeneous blood–tumor barrier permeability determines drug efficacy in experimental brain metastases of breast cancer. Clin. Cancer Res. 16, 5664–5678 (2010).
Taskar, K. S. et al. Lapatinib distribution in HER2 overexpressing experimental brain metastases of breast cancer. Pharm. Res. 29, 770–781 (2012).
Tiwary, S. et al. Metastatic brain tumors disrupt the blood–brain barrier and alter lipid metabolism by inhibiting expression of the endothelial cell fatty acid transporter Mfsd2a. Sci. Rep. 8, 8267 (2018).
Gril, B. et al. Reactive astrocytic S1P3 signaling modulates the blood–tumor barrier in brain metastases. Nat. Commun. 9, 2705 (2018).
Yanagida, K. et al. Size-selective opening of the blood–brain barrier by targeting endothelial sphingosine 1-phosphate receptor 1. Proc. Natl Acad. Sci. USA 114, 4531–4536 (2017).
Cheng, L. et al. Glioblastoma stem cells generate vascular pericytes to support vessel function and tumor growth. Cell 153, 139–152 (2013).
Hambardzumyan, D. & Bergers, G. Glioblastoma: defining tumor niches. Trends Cancer 1, 252–265 (2015).
Boucher, Y., Salehi, H., Witwer, B., Harsh, G. R. & Jain, R. K. Interstitial fluid pressure in intracranial tumours in patients and in rodents. Br. J. Cancer 75, 829–836 (1997).
Zhou, W. et al. Targeting glioma stem cell-derived pericytes disrupts the blood–tumor barrier and improves chemotherapeutic efficacy. Cell Stem Cell 21, 591–603.e4 (2017).
Valiente, M. et al. The evolving landscape of brain metastasis. Trends Cancer 4, 176–196 (2018).
Kienast, Y. et al. Real-time imaging reveals the single steps of brain metastasis formation. Nat. Med. 16, 116–122 (2010).
Avraham, H. K. et al. Angiopoietin-2 mediates blood–brain barrier impairment and colonization of triple-negative breast cancer cells in brain. J. Pathol. 232, 369–381 (2014).
Bos, P. D. et al. Genes that mediate breast cancer metastasis to the brain. Nature 459, 1005–1009 (2009).
Sevenich, L. et al. Analysis of tumour- and stroma-supplied proteolytic networks reveals a brain-metastasis-promoting role for cathepsin S. Nat. Cell Biol. 16, 876–888 (2014).
Kodack, D. P., Askoxylakis, V., Ferraro, G. B., Fukumura, D. & Jain, R. K. Emerging strategies for treating brain metastases from breast cancer. Cancer Cell 27, 163–175 (2015). This review discusses recent advances in breast cancer brain metastasis therapy and potential approaches for successful treatment.
Kabraji, S. et al. Drug resistance in HER2-positive breast cancer brain metastases: blame the barrier or the brain? Clin Cancer Res 24, 1795–1804 (2018).
Ni, J. et al. Combination inhibition of PI3K and mTORC1 yields durable remissions in mice bearing orthotopic patient-derived xenografts of HER2-positive breast cancer brain metastases. Nat. Med. 22, 723–726 (2016).
Kodack, D. P. et al. The brain microenvironment mediates resistance in luminal breast cancer to PI3K inhibition through HER3 activation. Sci. Transl Med. 9, eaal4682 (2017). This paper systematically investigates how the brain microenvironment confers resistance to low and high molecular weight therapeutics — targeted against oncogenic pathways — even when these therapeutics accrue within brain metastases.
Lin, N. U. et al. Multicenter phase II study of lapatinib in patients with brain metastases from HER2-positive breast cancer. Clin. Cancer Res. 15, 1452–1459 (2009).
Banks, W. A. From blood–brain barrier to blood–brain interface: new opportunities for CNS drug delivery. Nat. Rev. Drug Discov. 15, 275–292 (2016).
Banks, W. A. Characteristics of compounds that cross the blood–brain barrier. BMC Neurol. 9, S3 (2009).
Nałęcz, K. A. Solute carriers in the blood–brain barrier: safety in abundance. Neurochem. Res. 42, 795–809 (2017).
Seelig, A. The role of size and charge for blood–brain barrier permeation of drugs and fatty acids. J. Mol. Neurosci. 33, 32–41 (2007).
Schinkel, A. H. P-Glycoprotein, a gatekeeper in the blood–brain barrier. Adv. Drug Delivery Rev. 36, 179–194 (1999).
Ni, Z., Bikadi, Z., Rosenberg, M. F. & Mao, Q. Structure and function of the human breast cancer resistance protein (BCRP/ABCG2). Curr. Drug Metab. 11, 603–617 (2010).
Smith, M. W. & Gumbleton, M. Endocytosis at the blood–brain barrier: from basic understanding to drug delivery strategies. J. Drug Target. 14, 191–214 (2006).
Deeken, J. F. & Löscher, W. The blood–brain barrier and cancer: transporters, treatment, and trojan horses. Clin. Cancer Res. 13, 1663–1674 (2007).
Lajoie, J. M. & Shusta, E. V. Targeting receptor-mediated transport for delivery of biologics across the blood–brain barrier. Annu. Rev. Pharmacol. Toxicol. 55, 613–631 (2015).
Pardridge, W. M. Drug transport across the blood–brain barrier. J. Cereb. Blood Flow Metab. 32, 1959–1972 (2012).
Drappatz, J. et al. Phase I study of GRN1005 in recurrent malignant glioma. Clin. Cancer Res. 19, 1567–1576 (2013).
Tang, S.-C. et al. ANG1005, a novel peptide-paclitaxel conjugate crosses the BBB and shows activity in patients with recurrent CNS metastasis from breast cancer, results from a phase II clinical study. Ann. Oncol. 27, 103–113 (2016).
Wang, J. et al. Glucose transporter GLUT1 expression and clinical outcome in solid tumors: a systematic review and meta-analysis. Oncotarget 8, 16875–16886 (2017).
Youland, R. S. et al. The role of LAT1 in 18F-DOPA uptake in malignant gliomas. J. Neurooncol. 111, 11–18 (2013).
de Gooijer, M. C. et al. Improved brain penetration and antitumor efficacy of temozolomide by inhibition of ABCB1 and ABCG2. Neoplasia 20, 710–720 (2018).
Lin, F. et al. ABCB1, ABCG2, and PTEN determine the response of glioblastoma to temozolomide and ABT-888 therapy. Clin. Cancer Res. 20, 2703–2713 (2014).
Wijaya, J., Fukuda, Y. & Schuetz, J. D. Obstacles to brain tumor therapy: key ABC transporters. Int. J. Mol. Sci. 18, 2544 (2017).
Dréan, A. et al. ATP binding cassette (ABC) transporters: expression and clinical value in glioblastoma. J. Neurooncol. 138, 479–486 (2018).
Durmus, S., Sparidans, R. W., Wagenaar, E., Beijnen, J. H. & Schinkel, A. H. Oral availability and brain penetration of the B-RAFV600E inhibitor vemurafenib can be enhanced by the P-GLYCOprotein (ABCB1) and breast cancer resistance protein (ABCG2) inhibitor elacridar. Mol. Pharm. 9, 3236–3245 (2012).
Kalvass, J. C. et al. Why clinical modulation of efflux transport at the human blood–brain barrier is unlikely: the ITC evidence-based position. Clin. Pharmacol. Ther. 94, 80–94 (2013).
Kim, M. et al. Barriers to effective drug treatment for brain metastases: a multifactorial problem in the delivery of precision medicine. Pharm. Res. 35, 177 (2018). This study provides direct evidence of heterogeneity in the BBB/BTB and its impact on the therapeutic efficacy of small-molecule inhibitors.
Osswald, M. et al. Impact of blood–brain barrier integrity on tumor growth and therapy response in brain metastases. Clin. Cancer Res. 22, 6078–6087 (2016).
Agarwal, S., Sane, R., Oberoi, R., Ohlfest, J. R. & Elmquist, W. Delivery of molecularly targeted therapy to malignant glioma, a disease of the whole brain. Expert Rev. Mol. Med. 13, e17 (2011).
Stuckey, D. W. & Shah, K. Stem cell-based therapies for cancer treatment: separating hope from hype. Nat. Rev. Cancer 14, 683–691 (2014).
Bovenberg, M. S. S., Degeling, M. H. & Tannous, B. A. Advances in stem cell therapy against gliomas. Trends Mol. Med. 19, 281–291 (2013).
Mooney, R., Hammad, M., Batalla-Covello, J., Majid, A. A. & Aboody, K. S. Concise review: neural stem cell-mediated targeted cancer therapies. Stem Cells Transl Med. 7, 740–747 (2018).
Liu, L. et al. From blood to the brain: can systemically transplanted mesenchymal stem cells cross the blood–brain barrier? Stem Cells Int. 2013, 435093 (2013).
Rüster, B. et al. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood 108, 3938–3944 (2006).
Aboody, K. S. et al. Neural stem cells display extensive tropism for pathology in adult brain: evidence from intracranial gliomas. Proc. Natl Acad. Sci. USA 97, 12846–12851 (2000). This paper is the first to demonstrate that NSCs can migrate into brain tumours.
Portnow, J. et al. Neural stem cell-based anticancer gene therapy: a first-in-human study in recurrent high-grade glioma patients. Clin. Cancer Res. 23, 2951–2960 (2017).
Sonabend, A. M. et al. Mesenchymal stem cells effectively deliver an oncolytic adenovirus to intracranial glioma. Stem Cells 26, 831–841 (2008).
Auffinger, B. et al. Drug-loaded nanoparticle systems and adult stem cells: a potential marriage for the treatment of malignant glioma? Oncotarget 4, 378–396 (2013).
Batrakova, E. V., Gendelman, H. E. & Kabanov, A. V. Cell-mediated drugs delivery. Expert Opin. Drug Deliv. 8, 415–433 (2011).
Lee, A. S., Tang, C., Rao, M. S., Weissman, I. L. & Wu, J. C. Tumorigenicity as a clinical hurdle for pluripotent stem cell therapies. Nat. Med. 19, 998–1004 (2013).
Aryal, M., Arvanitis, C. D., Alexander, P. M. & McDannold, N. Ultrasound-mediated blood–brain barrier disruption for targeted drug delivery in the central nervous system. Adv. Drug Deliv. Rev. 72, 94–109 (2014).
Hynynen, K., McDannold, N., Vykhodtseva, N. & Jolesz, F. A. Noninvasive MR imaging-guided focal opening of the blood–brain barrier in rabbits. Radiology 220, 640–646 (2001). This paper is the first to demonstrate that FUS with microbubbles can induce focal BBB disruption.
Meairs, S. Facilitation of drug transport across the blood–brain barrier with ultrasound and microbubbles. Pharmaceutics 7, 275–293 (2015).
Arvanitis, C. D. et al. Mechanisms of enhanced drug delivery in brain metastases with focused ultrasound-induced blood–tumor barrier disruption. Proc. Natl Acad. Sci. USA 115, E8717–E8726 (2018). This study demonstrates that BBB/BTB disruption mediated by FUS with microbubbles induces a shift from diffusive to convective transport in the tumour interstitial space.
Park, J., Aryal, M., Vykhodtseva, N., Zhang, Y.-Z. & McDannold, N. Evaluation of permeability, doxorubicin delivery, and drug retention in a rat brain tumor model after ultrasound-induced blood–tumor barrier disruption. J. Control. Release 250, 77–85 (2017).
Marty, B. et al. Dynamic study of blood–brain barrier closure after its disruption using ultrasound: a quantitative analysis. J. Cereb. Blood Flow Metab. 32, 1948–1958 (2012).
Choi, J. J., Wang, S., Tung, Y.-S., Morrison III, B. & Konofagou, E. E. Molecules of various pharmacologically-relevant sizes can cross the ultrasound-induced blood–brain barrier opening in vivo. Ultrasound Med. Biol. 36, 58–67 (2010).
McMahon, D. & Hynynen, K. Acute inflammatory response following increased blood–brain barrier permeability induced by focused ultrasound is dependent on microbubble dose. Theranostics 7, 3989–4000 (2017).
Kovacs, Z. I. et al. Disrupting the blood–brain barrier by focused ultrasound induces sterile inflammation. Proc. Natl Acad. Sci. USA 114, E75–E84 (2017).
Schmidt, M. M. & Wittrup, K. D. A modeling analysis of the effects of molecular size and binding affinity on tumor targeting. Mol. Cancer Ther. 8, 2861–2871 (2009).
McDannold, N., Arvanitis, C. D., Vykhodtseva, N. & Livingstone, M. S. Temporary disruption of the blood–brain barrier by use of ultrasound and microbubbles: safety and efficacy evaluation in rhesus macaques. Cancer Res. 72, 3652–3663 (2012).
Mainprize, T. et al. Blood–brain barrier opening in primary brain tumors with non-invasive MR-guided focused ultrasound: a clinical safety and feasibility study. Sci. Rep. 9, 321 (2019).
Carpentier, A. et al. Clinical trial of blood–brain barrier disruption by pulsed ultrasound. Sci. Transl Med. 8, 343re2 (2016).
Idbaih, A. et al. Safety and feasibility of repeated and transient blood–brain barrier disruption by pulsed ultrasound in patients with recurrent glioblastoma. Clin. Cancer Res. 25, 3793–3801 (2019).
Arvanitis, C. D., Crake, C., McDannold, N. & Clement, G. T. Passive acoustic mapping with the angular spectrum method. IEEE Trans. Med. Imaging 36, 983–993 (2017).
Jones, R. M. & Hynynen, K. Advances in acoustic monitoring and control of focused ultrasound-mediated increases in blood–brain barrier permeability. Br. J. Radiol. 92, 20180601 (2019).
Patel, A., Schoen, S. J. & Arvanitis, C. D. Closed loop spatial and temporal control of cavitation activity with passive acoustic mapping. IEEE Trans. Biomed. Eng. 66, 2022–2031 (2019).
Sun, T. et al. Closed-loop control of targeted ultrasound drug delivery across the blood–brain/tumor barriers in a rat glioma model. Proc. Natl Acad. Sci. USA 114, E10281–E10290 (2017).
Krug, S. M. et al. Angubindin-1, a novel paracellular absorption enhancer acting at the tricellular tight junction. J. Control. Release 260, 1–11 (2017).
Zeniya, S. et al. Angubindin-1 opens the blood–brain barrier in vivo for delivery of antisense oligonucleotide to the central nervous system. J. Control. Release 283, 126–134 (2018).
Greene, C. & Campbell, M. Tight junction modulation of the blood brain barrier: CNS delivery of small molecules. Tissue Barriers 4, e1138017 (2016).
Guerit, S. & Liebner, S. Blood–brain barrier breakdown determines differential therapeutic outcome in genetically diverse forms of medulloblastoma. Cancer Cell 29, 427–429 (2016).
Stapleton, S., Jaffray, D. & Milosevic, M. Radiation effects on the tumor microenvironment: implications for nanomedicine delivery. Adv. Drug Deliv. Rev. 109, 119–130 (2017).
Bouchet, A. et al. Permeability of brain tumor vessels induced by uniform or spatially microfractionated synchrotron radiation therapies. Int. J. Radiat. Oncol. Biol. Phys. 98, 1174–1182 (2017).
Teng, F., Tsien, C. I., Lawrence, T. S. & Cao, Y. Blood–tumor barrier opening changes in brain metastases from pre to one-month post radiation therapy. Radiother. Oncol. 125, 89–93 (2017).
Crowe, W. et al. MRI evaluation of the effects of whole brain radiotherapy on breast cancer brain metastasis. Int. J. Radiat. Biol. 95, 338–346 (2019).
Lemasson, B. et al. Monitoring blood–brain barrier status in a rat model of glioma receiving therapy: dual injection of low-molecular-weight and macromolecular MR contrast media. Radiology 257, 342–352 (2010).
Erel-Akbaba, G. et al. Radiation-induced targeted nanoparticle-based gene delivery for brain tumor therapy. ACS Nano 13, 4028–4040 (2019).
Mann, J., Ramakrishna, R., Magge, R. & Wernicke, A. G. Advances in radiotherapy for glioblastoma. Front. Neurol. 8, 748 (2018).
Zeng, Y. et al. Blood–brain barrier permeability of gefitinib in patients with brain metastases from non-small-cell lung cancer before and during whole brain radiation therapy. Oncotarget 6, 8366–8376 (2015).
Fang, L. et al. Whole-brain radiation fails to boost intracerebral gefitinib concentration in patients with brain metastatic non-small cell lung cancer: a self-controlled, pilot study. Cancer Chemother. Pharmacol. 76, 873–877 (2015).
Miller, M. A. et al. Radiation therapy primes tumors for nanotherapeutic delivery via macrophage-mediated vascular bursts. Sci. Transl Med. 9, eaal0225 (2017).
Kreuter, J. Drug delivery to the central nervous system by polymeric nanoparticles: what do we know? Adv. Drug Deliv. Rev. 71, 2–14 (2014).
Tapeinos, C., Battaglini, M. & Ciofani, G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J. Control. Release 264, 306–332 (2017).
Bertrand, N., Wu, J., Xu, X., Kamaly, N. & Farokhzad, O. C. Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv. Drug Deliv. Rev. 66, 2–25 (2014).
Wilhelm, S. et al. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 1, 16014 (2016).
Hau, P. et al. Pegylated liposomal doxorubicin-efficacy in patients with recurrent high-grade glioma. Cancer 100, 1199–1207 (2004).
Beier, C. P. et al. RNOP-09: pegylated liposomal doxorubicine and prolonged temozolomide in addition to radiotherapy in newly diagnosed glioblastoma — a phase II study. BMC Cancer 9, 308 (2009).
Ananda, S. et al. Phase 2 trial of temozolomide and pegylated liposomal doxorubicin in the treatment of patients with glioblastoma multiforme following concurrent radiotherapy and chemotherapy. J. Clin. Neurosci. 18, 1444–1448 (2011).
Johnsen, K. B. et al. Modulating the antibody density changes the uptake and transport at the blood–brain barrier of both transferrin receptor-targeted gold nanoparticles and liposomal cargo. J. Control. Release 295, 237–249 (2019).
Chauhan, V. P. & Jain, R. K. Strategies for advancing cancer nanomedicine. Nat. Mater. 12, 958–962 (2013).
Hare, J. I. et al. Challenges and strategies in anti-cancer nanomedicine development: an industry perspective. Adv. Drug Deliv. Rev. 108, 25–38 (2017).
Stylianopoulos, T. & Jain, R. K. Design considerations for nanotherapeutics in oncology. Nanomedicine 11, 1893–1907 (2015).
Thorne, R. G. & Nicholson, C. In vivo diffusion analysis with quantum dots and dextrans predicts the width of brain extracellular space. Proc. Natl Acad. Sci. USA 103, 5567–5572 (2006).
Stylianopoulos, T., Munn, L. L. & Jain, R. K. Reengineering the physical microenvironment of tumors to improve drug delivery and efficacy: from mathematical modeling to bench to bedside. Trends Cancer 4, 292–319 (2018).
Lyon, P. C. et al. Safety and feasibility of ultrasound-triggered targeted drug delivery of doxorubicin from thermosensitive liposomes in liver tumours (TARDOX): a single-centre, open-label, phase 1 trial. Lancet Oncol. 19, 1027–1039 (2018).
Lim, M., Xia, Y., Bettegowda, C. & Weller, M. Current state of immunotherapy for glioblastoma. Nat. Rev. Clin. Oncol. 15, 422–442 (2018).
Thorsson, V. et al. The immune landscape of cancer. Immunity 48, 812–830.e14 (2018).
Sampson, J. Brain immunology and immunotherapy in brain tumours. Nat. Rev. Cancer (in the press).
Cloughesy, T. F. et al. Neoadjuvant anti-PD-1 immunotherapy promotes a survival benefit with intratumoral and systemic immune responses in recurrent glioblastoma. Nat. Med. 25, 477 (2019).
Sevenich, L. Turning “cold” into “hot” tumors—opportunities and challenges for radio-immunotherapy against primary and metastatic brain cancers. Front. Oncol. 9, 163 (2019).
Curley, C. T., Sheybani, N. D., Bullock, T. N. & Price, R. J. Focused ultrasound immunotherapy for central nervous system pathologies: challenges and opportunities. Theranostics 7, 3608–3623 (2017).
Pham, C. D. et al. Differential immune microenvironments and response to immune checkpoint blockade among molecular subtypes of murine medulloblastoma. Clin. Cancer Res. 22, 582–595 (2016).
Okada, H. et al. Immunotherapy response assessment in neuro-oncology: a report of the RANO working group. Lancet Oncol. 16, e534–e542 (2015).
Kamoun, W. S. et al. Edema control by cediranib, a vascular endothelial growth factor receptor–targeted kinase inhibitor, prolongs survival despite persistent brain tumor growth in mice. J. Clin. Oncol. 27, 2542–2552 (2009).
Stegmayr, C. et al. Influence of blood–brain barrier permeability on O-(2-18F-fluoroethyl)-l-tyrosine uptake in rat gliomas. Eur. J. Nucl. Med. Mol. Imaging 44, 408–416 (2017).
Postow, M. A., Sidlow, R. & Hellmann, M. D. Immune-related adverse events associated with immune checkpoint blockade. N. Engl. J. Med. 378, 158–168 (2018).
Platten, M. & Reardon, D. A. Concepts for immunotherapies in gliomas. Semin. Neurol. 38, 62–72 (2018).
Brown, C. E. et al. Regression of glioblastoma after chimeric antigen receptor T-cell therapy. N. Engl. J. Med. 375, 2561–2569 (2016).
Brown, C. E. et al. Optimization of IL13Rα2-targeted chimeric antigen receptor T cells for improved anti-tumor efficacy against glioblastoma. Mol. Ther. 26, 31–44 (2018).
Kebir, H. et al. Human TH17 lymphocytes promote blood–brain barrier disruption and central nervous system inflammation. Nat. Med. 13, 1173 (2007).
Levin, V. A. et al. CNS anticancer drug discovery and development conference white paper. Neuro Oncol. 17, vi1–vi26 (2015).
Sorensen, A. G. et al. A “vascular normalization index” as potential mechanistic biomarker to predict survival after a single dose of cediranib in recurrent glioblastoma patients. Cancer Res. 69, 5296–5300 (2009).
Langen, K.-J., Galldiks, N., Hattingen, E. & Shah, N. J. Advances in neuro-oncology imaging. Nat. Rev. Neurol. 13, 279–289 (2017).
Li, J. et al. Quantitative and mechanistic understanding of AZD1775 penetration across human blood–brain barrier in glioblastoma patients using an IVIVE–PBPK modeling approach. Clin. Cancer Res. 23, 7454–7466 (2017).
Lee, E. Q. et al. Phase I and biomarker study of plerixafor and bevacizumab in recurrent high-grade glioma. Clin. Cancer Res. 24, 4643–4649 (2018).
Kennedy, R. T. Emerging trends in in vivo neurochemical monitoring by microdialysis. Curr. Opin. Chem. Biol. 17, 860–867 (2013).
Li, B. et al. Comprehensive analyses of tumor immunity: implications for cancer immunotherapy. Genome Biol. 17, 174 (2016).
Venkataramani, V. et al. Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature 573, 532–538 (2019).
Venkatesh, H. S. et al. Electrical and synaptic integration of glioma into neural circuits. Nature 573, 539–545 (2019).
Zeng, Q. et al. Synaptic proximity enables NMDAR signalling to promote brain metastasis. Nature 573, 526–531 (2019).
Morad, G. et al. Tumor-derived extracellular vesicles breach the intact blood–brain barrier via transcytosis. ACS Nano https://doi.org/10.1021/acsnano.9b04397 (2019).
Yang, F.-Y. et al. Focused ultrasound and interleukin-4 receptor-targeted liposomal doxorubicin for enhanced targeted drug delivery and antitumor effect in glioblastoma multiforme. J. Control. Release 160, 652–658 (2012).
Agarwal, S. et al. Active efflux of dasatinib from the brain limits efficacy against murine glioblastoma: broad implications for the clinical use of molecularly targeted agents. Mol. Cancer Ther. 11, 2183–2192 (2012).
Cha, S. et al. Dynamic, contrast-enhanced perfusion MRI in mouse gliomas: correlation with histopathology. Magn. Reson. Med. 49, 848–855 (2003).
Leten, C., Struys, T., Dresselaers, T. & Himmelreich, U. In vivo and ex vivo assessment of the blood–brain barrier integrity in different glioblastoma animal models. J. Neurooncol. 119, 297–306 (2014).
Jacobs, V. L., Valdes, P. A., Hickey, W. F. & De Leo, J. A. Current review of in vivo GBM rodent models: emphasis on the CNS-1 tumour model. ASN Neuro. 3, e00063 (2011).
Murrell, D. H. et al. Understanding heterogeneity and permeability of brain metastases in murine models of HER2-positive breast cancer through magnetic resonance imaging: implications for detection and therapy. Transl Oncol. 8, 176 (2015).
Kodack, D. P. et al. Combined targeting of HER2 and VEGFR2 for effective treatment of HER2-amplified breast cancer brain metastases. Proc. Natl Acad. Sci. USA 109, E3119–E3127 (2012).
Wyatt, E. A. & Davis, M. E. Method of establishing breast cancer brain metastases affects brain uptake and efficacy of targeted, therapeutic nanoparticles. Bioeng. Transl Med. 4, 30–37 (2019).
Henry, M. N., Chen, Y., McFadden, C. D., Simedrea, F. C. & Foster, P. J. In-vivo longitudinal MRI study: an assessment of melanoma brain metastases in a clinically relevant mouse model. Melanoma Res. 25, 127–137 (2015).
Zou, H. Y. et al. PF-06463922, an ALK/ROS1 inhibitor, overcomes resistance to first and second generation ALK inhibitors in preclinical models. Cancer Cell 28, 70–81 (2015).
Nduom, E. K., Yang, C., Merrill, M. J., Zhuang, Z. & Lonser, R. R. Characterization of the blood–brain barrier of metastatic and primary malignant neoplasms: laboratory investigation. J. Neurosurg. 119, 427 (2013).
Perreault, S. et al. MRI surrogates for molecular subgroups of medulloblastoma. Am. J. Neuroradiol. 35, 1263–1269 (2014).
Dangouloff-Ros, V. et al. Imaging features of medulloblastoma: conventional imaging, diffusion-weighted imaging, perfusion-weighted imaging, and spectroscopy: from general features to subtypes and characteristics. Neurochirurgie S0028-3770, 30178–30179 (2018).
Morikawa, A. et al. Capecitabine and lapatinib uptake in surgically resected brain metastases from metastatic breast cancer patients: a prospective study. Neuro Oncol. 17, 289–295 (2015).
Saleem, A. et al. Lapatinib access into normal brain and brain metastases in patients with Her-2 overexpressing breast cancer. EJNMMI Res. 5, 30 (2015).
Tamura, K. et al. 64Cu-DOTA-trastuzumab PET imaging in patients with HER2-positive breast cancer. J. Nucl. Med. 54, 1869–1875 (2013).
Lewis, G. P. et al. Trastuzumab uptake and its relation to efficacy in an animal model of HER2-positive breast cancer brain metastasis. Breast Cancer Res. Treat. 164, 581–591 (2017).
Askoxylakis, V. et al. Preclinical efficacy of ado-trastuzumab emtansine in the brain microenvironment. J. Natl Cancer Inst. 108, djv313 (2016).
Heye, A. K., Culling, R. D., Valdés, C. H. M., Thrippleton, M. J. & Wardlaw, J. M. Assessment of blood–brain barrier disruption using dynamic contrast-enhanced MRI. A systematic review. Neuroimage Clin. 6, 262–274 (2014).
Jahangiri, A. et al. Convection-enhanced delivery in glioblastoma: a review of preclinical and clinical studies. J. Neurosurg. 126, 191–200 (2017).
Lonser, R. R., Sarntinoranont, M., Morrison, P. F. & Oldfield, E. H. Convection-enhanced delivery to the central nervous system. J. Neurosurg. 122, 697–706 (2015).
Zhou, Z., Singh, R. & Souweidane, M. M. Convection-enhanced delivery for diffuse intrinsic pontine glioma treatment. Curr. Neuropharmacol. 15, 116–128 (2017).
Beauchesne, P. Intrathecal chemotherapy for treatment of leptomeningeal dissemination of metastatic tumours. Lancet Oncol. 11, 871–879 (2010).
Groothuis, D. R. The blood–brain and blood–tumor barriers: a review of strategies for increasing drug delivery. Neuro Oncol. 2, 45–49 (2000).
Chowdhary, S. A., Ryken, T. & Newton, H. B. Survival outcomes and safety of carmustine wafers in the treatment of high-grade gliomas: a meta-analysis. J. Neurooncol. 122, 367–382 (2015).
Bregy, A. et al. The role of Gliadel wafers in the treatment of high-grade gliomas. Expert Rev. Anticancer Ther. 13, 1453–1461 (2013).
Masi, B. C. et al. Intracranial MEMS based temozolomide delivery in a 9L rat gliosarcoma model. Biomaterials 33, 5768–5775 (2012).
Vellimana, A. K. et al. Combination of paclitaxel thermal gel depot with temozolomide and radiotherapy significantly prolongs survival in an experimental rodent glioma model. J. Neurooncol. 111, 229–236 (2013).
Bastiancich, C. et al. Lauroyl-gemcitabine-loaded lipid nanocapsule hydrogel for the treatment of glioblastoma. J. Control. Release 225, 283–293 (2016).
Wick, W. et al. Phase III study of enzastaurin compared with lomustine in the treatment of recurrent intracranial glioblastoma. J. Clin. Oncol. 28, 1168–1174 (2010).
Lau, D., Magill, S. T. & Aghi, M. K. Molecularly targeted therapies for recurrent glioblastoma: current and future targets. Neurosurg. Focus 37, E15 (2014).
Westphal, M. et al. Adenovirus-mediated gene therapy with sitimagene ceradenovec followed by intravenous ganciclovir for patients with operable high-grade glioma (ASPECT): a randomised, open-label, phase 3 trial. Lancet Oncol. 14, 823–833 (2013).
Jain, R. K., Munn, L. L. & Fukumura, D. Dissecting tumour pathophysiology using intravital microscopy. Nat. Rev. Cancer 2, 266–276 (2002).
Galldiks, N., Law, I., Pope, W. B., Arbizu, J. & Langen, K.-J. The use of amino acid PET and conventional MRI for monitoring of brain tumor therapy. Neuroimage Clin. 13, 386–394 (2017).
Dhermain, F. G., Hau, P., Lanfermann, H., Jacobs, A. H. & van den Bent, M. J. Advanced MRI and PET imaging for assessment of treatment response in patients with gliomas. Lancet Neurol. 9, 906–920 (2010).
Niemeijer, A. N. et al. Whole body PD-1 and PD-L1 positron emission tomography in patients with non-small-cell lung cancer. Nat. Commun. 9, 4664 (2018).
Keu, K. V. et al. Reporter gene imaging of targeted T cell immunotherapy in recurrent glioma. Sci. Transl Med. 9, eaag2196 (2017).
Acknowledgements
The authors would like to thank C. Betsholtz, S. Chatterjee, M. Datta, Z. Amoozgar and S. Krishnan for their helpful input. C.D.A.’s research in this area is supported by NIH (National Institutes of Health) Grant R00EB016971 (National Institute of Biomedical Imaging and Bioengineering), NSF (National Science Foundation) Grants 1933158 and 1830577 (Leading Engineering for America’s Prosperity, Health, and Infrastructure) and CURE Childhood Cancer. G.B.F. received a fellowship from Susan G. Komen for the Cure. R.K.J.’s research in this general area is supported by an Outstanding Investigator Award from the National Cancer Institute (R35CA197743) and grants from the National Foundation for Cancer Research, the Ludwig Center at Harvard, the Advanced Medical Research Foundation, the Ellison Foundation, the Jane’s Trust Foundation and the Koch Institute-Dana Farber/Harvard Cancer Center Bridge Project Award.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing Interests
R.K.J. received honorarium from Amgen; consultant fees from Chugai, Enlight, Ophthotech, SPARC and SynDevRx; owns equity in Enlight, Ophthotech and SynDevRx; and serves on the Boards of Trustees of Tekla Healthcare Investors, Tekla Life Sciences Investors, Tekla Healthcare Opportunities Fund and Tekla World Healthcare Fund. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Cancer thanks W. Elmquist, T. Phoenix and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Pericytes
-
Mural cells that regulate development, permeability and function of the microcirculation.
- Astrocytic endfeet
-
Extensions of astrocytes into the perivascular space, covering the majority of the central nervous system endothelium and regulating development and function of the microcirculation.
- Microglia
-
Resident myeloid cells in the central nervous system (CNS) that regulate CNS function and homeostasis.
- Astrocytes
-
Major glial cells in the central nervous system (CNS) that regulate CNS function and homeostasis.
- Mural cells
-
Vascular-associated cells that include pericytes and smooth muscle cells.
- Pinocytosis
-
Small particles or molecules suspended in extracellular fluid that are brought into the cell through cell membrane invagination.
- ATP-binding cassette transporters
-
(ABC transporters). Active transporters that use energy in the form of ATP to transport molecules across the cell membrane against their concentration gradient.
- Diffusive transport
-
The net movement of molecules from a region of higher concentration to a region of lower concentration.
- Convective transport
-
Mass transport mediated by bulk fluid flow that is driven by a pressure gradient.
- Fast electronic beam steering
-
Changing the direction of the ultrasound focus or pattern by changing the relative phases of the radiofrequency signals driving the transducer elements. Multiphase array transducers that are composed of hundreds of elements are able to perform electronic beam steering within a few microseconds.
Rights and permissions
About this article
Cite this article
Arvanitis, C.D., Ferraro, G.B. & Jain, R.K. The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nat Rev Cancer 20, 26–41 (2020). https://doi.org/10.1038/s41568-019-0205-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-019-0205-x
- Springer Nature Limited
This article is cited by
-
Involvement of the tumour necrosis factor receptor system in glioblastoma cell death induced by palbociclib-heptamethine cyanine dye conjugate
Cell Communication and Signaling (2024)
-
Personalized identification and characterization of genome-wide gene expression differences between patient-matched intracranial and extracranial melanoma metastasis pairs
Acta Neuropathologica Communications (2024)
-
Exosomes as drug delivery systems in glioma immunotherapy
Journal of Nanobiotechnology (2024)
-
Unravelling immune microenvironment features underlying tumor progression in the single-cell era
Cancer Cell International (2024)
-
IDH1 mutation is detectable in plasma cell-free DNA and is associated with survival outcome in glioma patients
BMC Cancer (2024)