Abstract
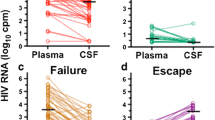
HIV-1 persists as a latent reservoir in people receiving suppressive antiretroviral therapy (ART). When ART is interrupted (treatment interruption/TI), rebound virus re-initiates systemic infection in the lymphoid system. During TI, HIV-1 is also detected in cerebrospinal fluid (CSF), although the source of this rebound virus is unknown. To investigate whether there is a distinct HIV-1 reservoir in the central nervous system (CNS), we compared rebound virus after TI in the blood and CSF of 11 participants. Peak rebound CSF viral loads vary and we show that high viral loads and the appearance of clonally amplified viral lineages in the CSF are correlated with the transient influx of white blood cells. We found no evidence of rebound macrophage-tropic virus in the CSF, even in one individual who had macrophage-tropic HIV-1 in the CSF pre-therapy. We propose a model in which R5 T cell-tropic virus is released from infected T cells that enter the CNS from the blood (or are resident in the CNS during therapy), with clonal amplification of infected T cells and virus replication occurring in the CNS during TI.




Similar content being viewed by others
Data availability
The sequences of the full-length env amplicons are available in GenBank (accession numbers ON599411–ON59572). The deep sequencing data are available in the Sequencing Read Archive (https://www.ncbi.nlm.nih.gov/sra/PRJNA880316). Source data are provided with this paper.
Code availability
Illumina MiSeq data were analysed using TCS pipeline version 2.5.1 (https://www.primer-id.org/?from_old).
References
Ping, L. H. et al. Comparison of viral Env proteins from acute and chronic infections with subtype C human immunodeficiency virus type 1 identifies differences in glycosylation and CCR5 utilization and suggests a new strategy for immunogen design. J. Virol. 87, 7218–7233 (2013).
Parrish, N. F. et al. Transmitted/founder and chronic subtype C HIV-1 use CD4 and CCR5 receptors with equal efficiency and are not inhibited by blocking the integrin α4β7. PLoS Pathog. 8, e1002686 (2012).
Joseph, S. B. & Swanstrom, R. The evolution of HIV-1 entry phenotypes as a guide to changing target cells. J. Leuk. Biol. 103, 421–431 (2018).
Colby, D. J. et al. Rapid HIV RNA rebound after antiretroviral treatment interruption in persons durably suppressed in Fiebig I acute HIV infection. Nat. Med. 24, 923–926 (2018).
Henrich, T. J. et al. HIV-1 persistence following extremely early initiation of antiretroviral therapy (ART) during acute HIV-1 infection: an observational study. PLoS Med. 14, e1002417 (2017).
Whitney, J. B. et al. Rapid seeding of the viral reservoir prior to SIV viraemia in rhesus monkeys. Nature 512, 74–77 (2014).
Treasure, G. C. et al. Relationship among viral load outcomes in HIV treatment interruption trials. J. Acquir. Immune Defic. Syndr. 72, 310–313 (2016).
Li, J. Z. et al. The size of the expressed HIV reservoir predicts timing of viral rebound after treatment interruption. AIDS 30, 343–353 (2016).
Bar, K. J. et al. Effect of HIV antibody VRC01 on viral rebound after treatment interruption. N. Engl. J. Med. 375, 2037–2050 (2016).
Bednar, M. M. et al. Diversity and tropism of HIV-1 rebound virus populations in plasma level after treatment discontinuation. J. Infect. Dis. 214, 403–407 (2016).
De Scheerder, M. A. et al. HIV rebound is predominantly fueled by genetically identical viral expansions from diverse reservoirs. Cell Host Microbe 26, 347–358 (2019).
Fisher, K. et al. Plasma-derived HIV-1 virions contain considerable levels of defective genomes. J. Virol. 96, e0201121 (2022).
Kearney, M. F. et al. Origin of rebound plasma HIV includes cells with identical proviruses that are transcriptionally active before stopping of antiretroviral therapy. J. Virol. 90, 1369–1376 (2016).
Rothenberger, M. K. et al. Large number of rebounding/founder HIV variants emerge from multifocal infection in lymphatic tissues after treatment interruption. Proc. Natl Acad. Sci. USA 112, E1126–E1134 (2015).
Andrade, V. M. et al. A minor population of macrophage-tropic HIV-1 variants is identified in recrudescing viremia following analytic treatment interruption. Proc. Natl Acad. Sci. USA 117, 9981–9990 (2020).
Chun, T. W. et al. In vivo fate of HIV-1-infected T cells: quantitative analysis of the transition to stable latency. Nat. Med. 1, 1284–1290 (1995).
Hiener, B. et al. Identification of genetically intact HIV-1 proviruses in specific CD4+ T cells from effectively treated participants. Cell Rep. 21, 813–822 (2017).
Chomont, N. et al. HIV reservoir size and persistence are driven by T cell survival and homeostatic proliferation. Nat. Med. 15, 893–900 (2009).
Shacklett, B. L., Ferre, A. L. & Kiniry, B. E. Defining T cell tissue residency in humans: implications for HIV pathogenesis and vaccine design. Curr. HIV/AIDS Rep. 17, 109–117 (2020).
Louveau, A., Harris, T. H. & Kipnis, J. Revisiting the mechanisms of CNS immune privilege. Trends Immunol. 36, 569–577 (2015).
Engelhardt, B., Vajkoczy, P. & Weller, R. O. The movers and shapers in immune privilege of the CNS. Nat. Immunol. 18, 123–131 (2017).
Iwasaki, A. Immune regulation of antibody access to neuronal tissues. Trends Mol. Med. 23, 227–245 (2017).
Burbelo, P. D. et al. Anti-human immunodeficiency virus antibodies in the cerebrospinal fluid: evidence of early treatment impact on central nervous system reservoir? J. Infect. Dis. 217, 1024–1032 (2018).
Sturdevant, C. B. et al. Compartmentalized replication of R5 T cell-tropic HIV-1 in the central nervous system early in the course of infection. PLoS Pathog. 11, e1004720 (2015).
Schnell, G. et al. HIV-1 replication in the central nervous system occurs in two distinct cell types. PLoS Pathog. 7, e1002286 (2011).
Joseph, S. B. et al. Quantification of entry phenotypes of macrophage-tropic HIV-1 across a wide range of CD4 densities. J. Virol. 88, 1858–1869 (2014).
Price, R. W. & Deeks, S. G. Antiretroviral drug treatment interruption in human immunodeficiency virus-infected adults: clinical and pathogenetic implications for the central nervous system. J. NeuroVirol. 10, 44–51 (2004).
Santangelo, P. J. et al. Whole-body immunoPET reveals active SIV dynamics in viremic and antiretroviral therapy-treated macaques. Nat. Methods 12, 427–432 (2015).
Honeycutt, J. B. et al. T cells establish and maintain CNS viral infection in HIV-infected humanized mice. J. Clin. Invest. 128, 2862–2876 (2018).
Honeycutt, J. B. et al. HIV persistence in tissue macrophages of humanized myeloid-only mice during antiretroviral therapy. Nat. Med. 23, 638–643 (2017).
Whitney, J. B. et al. Prevention of SIVmac251 reservoir seeding in rhesus monkeys by early antiretroviral therapy. Nat. Commun. 9, 5429 (2018).
Fennessey, C. M. et al. Genetically-barcoded SIV facilitates enumeration of rebound variants and estimation of reactivation rates in nonhuman primates following interruption of suppressive antiretroviral therapy. PLoS Pathog. 13, e1006359 (2017).
Obregon-Perko, V. et al. Dynamics and origin of rebound viremia in SHIV-infected infant macaques following interruption of long-term ART. JCI Insight 6, e152526 (2021).
Gama, L. et al. Reactivation of simian immunodeficiency virus reservoirs in the brain of virally suppressed macaques. AIDS 31, 5–14 (2017).
Avalos, C. R. et al. Brain macrophages in simian immunodeficiency virus-infected, antiretroviral-suppressed macaques: a functional latent reservoir. mBio 8, e01186-17 (2017).
Abreu, C. et al. Brain macrophages harbor latent, infectious simian immunodeficiency virus. AIDS 33, S181–S188 (2019).
Su, H. et al. Recovery of latent HIV-1 from brain tissue by adoptive cell transfer in virally suppressed humanized mice. J. Neuroimmune Pharmacol. 16, 796–805 (2021).
Dubé, K. et al. Ethical considerations for HIV cure-related research at the end of life. BMC Med. Ethics 19, 83 (2018).
de Almeida, S. N. et al. Dynamics of monocyte chemoattractant protein type one (MCP-1) and HIV viral load in human cerebrospinal fluid and plasma. J. Neuroimmunol. 169, 144–152 (2005).
Gianella, S. et al. Compartmentalized HIV rebound in the central nervous system after interruption of antiretroviral therapy. Virus Evol. 2, vew020 (2016).
Deeks, S. G. et al. Virologic and immunologic consequences of discontinuing combination antiretroviral-drug therapy in HIV-infected patients with detectable viremia. N. Engl. J. Med. 344, 472–480 (2001).
Price, R. W. et al. Cerebrospinal fluid response to structured treatment interruption after virological failure. AIDS 15, 1251–1259 (2001).
Gisslen, M. et al. Cerebrospinal fluid signs of neuronal damage after antiretroviral treatment interruption in HIV-1 infection. AIDS Res. Ther. 2, 6 (2005).
Zhou, S. et al. Deep sequencing of the HIV-1 env gene reveals discrete X4 lineages and linkage disequilibrium between X4 and R5 viruses in the V1/V2 and V3 variable regions. J. Virol. 90, 7142–7158 (2016).
Zhou, S. et al. Primer ID validates template sampling depth and greatly reduces the error rate of next-generation sequencing of HIV-1 genomic RNA populations. J. Virol. 89, 8540–8555 (2015).
Lustig, G. et al. T cell derived HIV-1 is present in the CSF in the face of suppressive antiretroviral therapy. PLoS Pathog. 17, e1009871 (2021).
Sharma, V. et al. Cerebrospinal fluid CD4+ T cell infection in humans and macaques during acute HIV-1. PLoS Pathog. 17, e1010105 (2021).
Slatkin, M. & Maddison, W. P. A cladistic measure of gene flow inferred from the phylogenies of alleles. Genetics 123, 603–613 (1989).
Adewumi, O. M. et al. HIV-1 central nervous system compartmentalization and cytokine interplay in non-subtype B HIV-1 infections in Nigeria and Malawi. AIDS Res. Hum. Retroviruses 36, 490–500 (2020).
Aamer, H. A. et al. Cells producing residual viremia during antiretroviral treatment appear to contribute to rebound viremia following interruption of treatment. PLoS Pathog. 16, e1008791 (2020).
Bailey, J. R. et al. Residual human immunodeficiency virus type 1 viremia in some patients on antiretroviral therapy is dominated by a small number of invariant clones rarely found in circulating CD4(+) T cells. J. Virol. 80, 6441–6457 (2006).
Cole, B. et al. In-depth single-cell analysis of translation-competent HIV-1 reservoirs identifies cellular sources of plasma viremia. Nat. Commun. 12, 3727 (2021).
Halvas, E. K. et al. HIV-1 viremia not suppressible by antiretroviral therapy can originate from large T cell clones producing infectious virus. J. Clin. Invest. 130, 5847–5857 (2020).
Rassler, S. et al. Prolonged persistence of a novel replication-defective HIV-1 variant in plasma of a patient on suppressive therapy. Virol. J. 13, 157 (2016).
Sahu, G. K., Sarria, J. C. & Cloyd, M. W. Recovery of replication-competent residual HIV-1 from plasma of a patient receiving prolonged, suppressive highly active antiretroviral therapy. J. Virol. 84, 8348–8352 (2010).
Simonetti, F. R. et al. Clonally expanded CD4+ T cells can produce infectious HIV-1 in vivo. Proc. Natl Acad. Sci. USA 113, 1883–1888 (2016).
Lu, C. L. et al. Relationship between intact HIV-1 proviruses in circulating CD4(+) T cells and rebound viruses emerging during treatment interruption. Proc. Natl Acad. Sci. USA 115, E11341–E11348 (2018).
Cohen, Y. Z. et al. Relationship between latent and rebound viruses in a clinical trial of anti-HIV-1 antibody 3BNC117. J. Exp. Med. 215, 2311–2324 (2018).
Liu, P. T. et al. Origin of rebound virus in chronically SIV-infected rhesus monkeys following treatment discontinuation. Nat. Commun. 11, 5412 (2020).
Johnston, S. H. et al. A quantitative affinity-profiling system that reveals distinct CD4/CCR5 usage patterns among human immunodeficiency virus type 1 and simian immunodeficiency virus strains. J. Virol. 83, 11016–11026 (2009).
Abrahams, M. R. et al. The replication-competent HIV-1 latent reservoir is primarily established near the time of therapy initiation. Sci. Transl. Med. 11, eaaw5589 (2019).
Imamichi, H. et al. Human immunodeficiency virus type 1 quasi species that rebound after discontinuation of highly active antiretroviral therapy are similar to the viral quasi species present before initiation of therapy. J. Infect. Dis. 183, 36–50 (2001).
Kearney, M. F. et al. Lack of detectable HIV-1 molecular evolution during suppressive antiretroviral therapy. PLoS Pathog. 10, e1004010 (2014).
Hagberg, L. et al. Cerebrospinal fluid neopterin: an informative biomarker of central nervous system immune activation in HIV-1 infection. AIDS Res. Ther. 7, 15 (2010).
Jessen Krut, J. et al. Biomarker evidence of axonal injury in neuroasymptomatic HIV-1 patients. PLoS ONE 9, e88591 (2014).
Norgren, N., Rosengren, L. & Stigbrand, T. Elevated neurofilament levels in neurological diseases. Brain Res. 987, 25–31 (2003).
Yilmaz, A. et al. Neurofilament light chain protein as a marker of neuronal injury: review of its use in HIV-1 infection and reference values for HIV-negative controls. Expert Rev. Mol. Diagn. 17, 761–770 (2017).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Joseph, S. B., Lee, B. & Swanstrom, R. Affinofile assay for identifying macrophage-tropic HIV-1. Bio Protoc. 4, e1184 (2014).
Acknowledgements
We thank the many participants who donated the specimens that were analysed in this study, and the UNC High Throughput Sequencing Facility for their assistance in generating the sequence data. This work was supported by NIH grant R01 NS094067 (R.W.P.), the UNC Center for AIDS Research (NIH award P30 AI050410 to R.S.), the UNC Lineberger Comprehensive Cancer Center (NIH award P30 CA16068 to R.S.) and by the Swedish state, under an agreement between the Swedish government and the county councils (ALF agreement ALFGBG-717531 to M.G.). H.Z. is a Wallenberg Scholar supported by grants from the Swedish Research Council (no. 2018-02532), the European Research Council (no. 681712) and the Swedish State Support for Clinical Research (no. ALFGBG-720931).
Author information
Authors and Affiliations
Contributions
R.S., R.W.P., M.G., H.Z. and S.G.D. conceived the study. S.Z. developed the methodology. L.P.K., M.M.G., B.M.H., S. Sizemore, C.D.G. and D.F. conducted the investigations. L.P.K., S.B.J., S.Spudich., M.G., R.W.P. and R.S. analysed the data. R.S. and S.B.J. wrote the manuscript with input from R.W.P., M.G., S. Spudich and L.P.K.
Corresponding author
Ethics declarations
Competing interests
UNC is pursuing IP protection for Primer ID and R.S. has received nominal royalties.
Peer review
Peer review information
Nature Microbiology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Statistical analysis of the link between CSF viral load and pleocytosis.
The ratio of CSF viral load to blood viral load was calculated for each time point shown in Fig. 1. This was done to normalize the CSF viral load as a fraction of the blood viral load thus reducing the variability in blood set point viral load; in this analysis the larger the value the closer the CSF viral load approached that of the blood. These values were then grouped as coming from time points where the white blood cell count was greater than 5/μl or less than or equal to 5/μl. The values of these two groups were compared using the two-sided Mann-Whitney test, with the p value included in the graph. In this analysis we did not correct for the fact that multiple values were collected for each participant.
Extended Data Fig. 2 Deep sequencing of HIV-1 populations from participant 51126.
Neighbor-joining phylogenetic tree containing a large sampling of template consensus sequences (TCS) from MiSeq/Primer ID sequencing from the pretherapy and TI timepoints. Sequences from the blood plasma pretherapy are shown in pink (1,734 TCS). Sequences from the CSF pretherapy are shown in light blue (1,734 TCS). Sequences from the blood plasma post TI are shown in red (2,233 TCS). Sequences from the CSF post TI are shown in dark blue (2,233 TCS). Also included in gray are 50 sequences from the blood and CSF from each of the two intermediate decay timepoints. On the right the tree is expanded to show the portion where the macrophage-tropic virus lineage was found in the CSF pretherapy.
Extended Data Fig. 3 Marker analysis during TI.
Further analysis of markers was done for the five participants shown in Fig. 4. Two additional graphs are presented in vertical columns for each of the participants. In the top graph is shown the level of NfL as a function of time post TI/enrollment (purple circles). Also included is the QNPZ4 score test of neurocognition (tan diamonds). The vertical dashed line is drawn at the time point of the initial peak CSF viral load for each participant (Fig. 1). In the lower graph of the pair, the values of sCD163 are shown (light blue circles), and sCD14 (orange squares).
Extended Data Fig. 4 Statistical analysis of links between biomarkers and pleocytosis during TI.
Selected biomarker data presented in Fig. 4 and Extended Data Fig. 3 were pooled based on the presence or absence of pleocytosis (WBC count greater than or less than 5/μl, respectively) in the CSF. These two groups were compared for IP-10 (a), MMP9 (b), neopterin (c), NfL (d), or the neurocognitive score QNPZ4 (e). The groups were compared using the two-sided Mann-Whitney test, with the p values indicated on each graph. A Bonferroni correction for multiple comparisons indicates a significant p value cutoff of 0.01. No corrections were made for the fact that multiple values were included for each participant.
Supplementary information
Source data
Source Data Fig. 2
Infectivity data including biological replicates.
Source Data Fig. 3
Infectivity data including biological replicates.
Source Data Extended Data Fig. 1
Statistical source data for VL ratio comparison.
Source Data Extended Data Fig. 4
Statistical source data for biomarker comparisons.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kincer, L.P., Joseph, S.B., Gilleece, M.M. et al. Rebound HIV-1 in cerebrospinal fluid after antiviral therapy interruption is mainly clonally amplified R5 T cell-tropic virus. Nat Microbiol 8, 260–271 (2023). https://doi.org/10.1038/s41564-022-01306-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-022-01306-6
- Springer Nature Limited
This article is cited by
-
More than the Infinite Monkey Theorem: NHP Models in the Development of a Pediatric HIV Cure
Current HIV/AIDS Reports (2024)
-
Fentanyl dysregulates neuroinflammation and disrupts blood-brain barrier integrity in HIV-1 Tat transgenic mice
Journal of NeuroVirology (2024)
-
Modeling Antiretrovial Treatment to Mitigate HIV in the Brain: Impact of the Blood-Brain Barrier
Bulletin of Mathematical Biology (2023)