Abstract
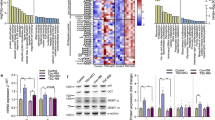
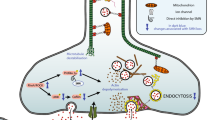
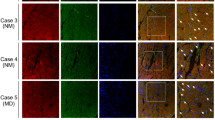
The ubiquitin proteasome system and autophagy are major protein turnover mechanisms in muscle cells, which ensure stemness and muscle fibre maintenance. Muscle cells contain a high proportion of cytoskeletal proteins, which are prone to misfolding and aggregation; pathological processes that are observed in several neuromuscular diseases called proteinopathies. Despite advances in deciphering the mechanisms underlying misfolding and aggregation, little is known about how muscle cells manage cytoskeletal degradation. Here, we describe a process by which muscle cells degrade the misfolded intermediate filament proteins desmin and vimentin by the proteasome. This relies on the MTM1–UBQLN2 complex to recognize and guide these misfolded proteins to the proteasome and occurs prior to aggregate formation. Thus, our data highlight a safeguarding function of the MTM1–UBQLN2 complex that ensures cytoskeletal integrity to avoid proteotoxic aggregate formation.








Similar content being viewed by others
References
Lu, K., Psakhye, I. & Jentsch, S. Autophagic clearance of polyQ proteins mediated by ubiquitin–Atg8 adaptors of the conserved CUET protein family. Cell 158, 549–563 (2014).
Hjerpe, R. et al. UBQLN2 mediates autophagy-independent protein aggregate clearance by the proteasome. Cell 166, 935–949 (2016).
Menzies, F. M., Moreau, K. & Rubinsztein, D. C. Protein misfolding disorders and macroautophagy. Curr. Opin. Cell. Biol. 23, 190–197 (2011).
Feng, Y., He, D., Yao, Z. & Klionsky, D. J. The machinery of macroautophagy. Cell. Res. 24, 24–41 (2014).
Smith, M. H., Ploegh, H. L. & Weissman, J. S. Road to ruin: targeting proteins for degradation in the endoplasmic reticulum. Science 334, 1086–1090 (2011).
Araki, K. & Nagata, K. Protein folding and quality control in the ER. Cold Spring Harb. Perspect. Biol. 4, a015438 (2012).
Wang, M. & Kaufman, R. J. Protein misfolding in the endoplasmic reticulum as a conduit to human disease. Nature 529, 326–335 (2016).
Hetz, C. The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat. Rev. Mol. Cell. Biol. 13, 89–102 (2012).
Gardner, B. M., Pincus, D., Gotthardt, K., Gallagher, C. M. & Walter, P. Endoplasmic reticulum stress sensing in the unfolded protein response. Cold Spring Harb. Perspect. Biol. 5, a013169 (2013).
Balchin, D., Hayer-Hartl, M. & Hartl, F. U. In vivo aspects of protein folding and quality control. Science 353, aac4354 (2016).
Wang, Z. V. & Hill, J. A. Protein quality control and metabolism: bidirectional control in the heart. Cell. Metab. 21, 215–226 (2015).
Hnia, K., Ramspacher, C., Vermot, J. & Laporte, J. Desmin in muscle and associated diseases: beyond the structural function. Cell. Tissue Res. 360, 591–608 (2015).
Sandri, M., Coletto, L., Grumati, P. & Bonaldo, P. Misregulation of autophagy and protein degradation systems in myopathies and muscular dystrophies. J. Cell. Sci. 126, 5325–5333 (2013).
Nowak, K. J., Ravenscroft, G. & Laing, N. G. Skeletal muscle α-actin diseases (actinopathies): pathology and mechanisms. Acta Neuropathol. 125, 19–32 (2013).
Kley, R. A., Olive, M. & Schröder, R. New aspects of myofibrillar myopathies. Curr. Opin. Neurol. 29, 628–634 (2016).
Capetanaki, Y., Papathanasiou, S., Diokmetzidou, A., Vatsellas, G. & Tsikitis, M. Desmin related disease: a matter of cell survival failure. Curr. Opin. Cell. Biol. 32, 113–120 (2015).
Goldfarb, L. G. & Dalakas, M. C. Tragedy in a heartbeat: malfunctioning desmin causes skeletal and cardiac muscle disease. J. Clin. Invest. 119, 1806–1813 (2009).
Herrmann, H. & Aebi, U. Intermediate filaments: structure and assembly. Cold Spring Harb. Perspect. Biol. 8, a018242 (2016).
Laporte, J. et al. A gene mutated in X-linked myotubular myopathy defines a new putative tyrosine phosphatase family conserved in yeast. Nat. Genet. 13, 175–182 (1996).
Goebel, H. H. & Blaschek, A. Protein aggregation in congenital myopathies. Semin. Pediatr. Neurol. 18, 272–276 (2011).
Wang, X. et al. Expression of R120G–αB-crystallin causes aberrant desmin and αB-crystallin aggregation and cardiomyopathy in mice. Circ. Res. 89, 84–91 (2001).
Johnson, A. E., Shu, H., Hauswirth, A. G., Tong, A. & Davis, G. W. VCP-dependent muscle degeneration is linked to defects in a dynamic tubular lysosomal network in vivo. eLife 4, e07366 (2015).
Hnia, K., Vaccari, I., Bolino, A. & Laporte, J. Myotubularin phosphoinositide phosphatases: cellular functions and disease pathophysiology. Trends Mol. Med. 18, 317–327 (2012).
Lim, P. J. et al. Ubiquilin and p97/VCP bind erasin, forming a complex involved in ERAD. J. Cell. Biol. 187, 201–217 (2009).
Deng, H. X. et al. Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature 477, 211–215 (2011).
Bargagna-Mohan, P. et al. The tumor inhibitor and antiangiogenic agent withaferin A targets the intermediate filament protein vimentin. Chem. Biol. 14, 623–634 (2007).
Blokhuis, A. M. et al. Comparative interactomics analysis of different ALS-associated proteins identifies converging molecular pathways. Acta Neuropathol. 132, 175–196 (2016).
Bar, H., Strelkov, S. V., Sjoberg, G., Aebi, U. & Herrmann, H. The biology of desmin filaments: how do mutations affect their structure, assembly, and organisation? J. Struct. Biol. 148, 137–152 (2004).
Liu, J., Tang, M., Mestril, R. & Wang, X. Aberrant protein aggregation is essential for a mutant desmin to impair the proteolytic function of the ubiquitin–proteasome system in cardiomyocytes. J. Mol. Cell. Cardiol. 40, 451–454 (2006).
Bornheim, R. et al. A dominant vimentin mutant upregulates Hsp70 and the activity of the ubiquitin–proteasome system, and causes posterior cataracts in transgenic mice. J. Cell. Sci. 121, 3737–3746 (2008).
Christianson, J. C. et al. Defining human ERAD networks through an integrative mapping strategy. Nat. Cell. Biol. 14, 93–105 (2011).
Eggers, D. K., Welch, W. J. & Hansen, W. J. Complexes between nascent polypeptides and their molecular chaperones in the cytosol of mammalian cells. Mol. Biol. Cell. 8, 1559–1573 (1997).
Sandri, M. & Robbins, J. Proteotoxicity: an underappreciated pathology in cardiac disease. J. Mol. Cell. Cardiol. 71, 3–10 (2014).
Mahammad, S. et al. Giant axonal neuropathy-associated gigaxonin mutations impair intermediate filament protein degradation. J. Clin. Invest. 123, 1964–1975 (2013).
Lowery, J. et al. Abnormal intermediate filament organization alters mitochondrial motility in giant axonal neuropathy fibroblasts. Mol. Biol. Cell. 27, 608–616 (2016).
Furukawa, M., He, Y. J., Borchers, C. & Xiong, Y. Targeting of protein ubiquitination by BTB–cullin 3–Roc1 ubiquitin ligases. Nat. Cell. Biol. 5, 1001–1007 (2003).
Pintard, L., Willems, A. & Peter, M. Cullin-based ubiquitin ligases: Cul3–BTB complexes join the family. Embo J. 23, 1681–1687 (2004).
Cohen, S., Zhai, B., Gygi, S. P. & Goldberg, A. L. Ubiquitylation by Trim32 causes coupled loss of desmin, Z-bands, and thin filaments in muscle atrophy. J. Cell. Biol. 198, 575–589 (2012).
Wong, E. S. et al. Autophagy-mediated clearance of aggresomes is not a universal phenomenon. Hum. Mol. Genet. 17, 2570–2582 (2008).
Bhuiyan, M. S. et al. Enhanced autophagy ameliorates cardiac proteinopathy. J. Clin. Invest. 123, 5284–5297 (2013).
Winter, L. et al. Chemical chaperone ameliorates pathological protein aggregation in plectin-deficient muscle. J. Clin. Invest. 124, 1144–1157 (2014).
Wang, X. et al. αB-crystallin modulates protein aggregation of abnormal desmin. Circ. Res. 93, 998–1005 (2003).
Selcen, D. et al. Mutation in BAG3 causes severe dominant childhood muscular dystrophy. Ann. Neurol. 65, 83–89 (2009).
Ganassi, M. et al. A surveillance function of the HSPB8–BAG3–HSP70 chaperone complex ensures stress granule integrity and dynamism. Mol. Cell. 63, 796–810 (2016).
Arndt, V. et al. Chaperone-assisted selective autophagy is essential for muscle maintenance. Curr. Biol. 20, 143–148 (2010).
Chen, Y. et al. Bcl2-associated athanogene 3 interactome analysis reveals a new role in modulating proteasome activity. Mol. Cell. Proteom. 12, 2804–2819 (2013).
Falcone, S. et al. N-WASP is required for amphiphysin-2/BIN1-dependent nuclear positioning and triad organization in skeletal muscle and is involved in the pathophysiology of centronuclear myopathy. EMBO Mol. Med. 6, 1455–1475 (2014).
Hnia, K. et al. Myotubularin controls desmin intermediate filament architecture and mitochondrial dynamics in human and mouse skeletal muscle. J. Clin. Invest. 121, 70–85 (2011).
Barry, S. C. et al. Lentivirus vectors encoding both central polypurine tract and posttranscriptional regulatory element provide enhanced transduction and transgene expression. Hum. Gene Ther. 12, 1103–1108 (2001).
Liu, H., Sadygov, R. G. & Yates, J. R. III A model for random sampling and estimation of relative protein abundance in shotgun proteomics. Anal. Chem. 76, 4193–4201 (2004).
Colinge, J., Chiappe, D., Lagache, S., Moniatte, M. & Bougueleret, L. Differential proteomics via probabilistic peptide identification scores. Anal. Chem. 77, 596–606 (2005).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Herrmann, H. & Aebi, U. Structure, assembly, and dynamics of intermediate filaments. Subcell. Biochem. 31, 319–362 (1998).
Hofmann, I., Herrmann, H. & Franke, W. W. Assembly and structure of calcium-induced thick vimentin filaments. Eur. J. Cell. Biol. 56, 328–341 (1991).
Wanker, E. E. et al. Membrane filter assay for detection of amyloid-like polyglutamine-containing protein aggregates. Methods Enzymol. 309, 375–386 (1999).
Kisselev, A. F. & Goldberg, A. L. Monitoring activity and inhibition of 26S proteasomes with fluorogenic peptide substrates. Methods Enzymol. 398, 364–378 (2005).
Buj-Bello, A. et al. The lipid phosphatase myotubularin is essential for skeletal muscle maintenance but not for myogenesis in mice. Proc. Natl Acad. Sci. USA 99, 15060–15065 (2002).
Acknowledgements
We thank the IGBMC technical platforms (animal facility, electron and confocal microscopy) and C. Kretz for their technical assistance, C. Sellier, N. Dali-Youssef, F. Alpy and H. E. Magliarelli for sharing antibodies and other materials, L. Beilscmidt for technical assistance, M. Mendoza Parra and F. Colin for their help with modelling, F. Gaits-Iacovoni, A. Lewis for manuscript editing and B. Payrastre for his support. We acknowledge funding from INSERM (K.H.), the Scientific Council of the University of Strasbourg (K.H.), the AFM (Association Française contre la Myopathy) (AFM-20879 to K.H., AFM-15352 to J.L.) and grant ANR-10-LABX-0030-INRT, awarded as part of the Investissements d’Avenir ANR-10-IDEX-0002-02 framework.
Author information
Authors and Affiliations
Contributions
C.G. and L.L. performed experiments, analysed data, interpreted results and edited the manuscript. N.M. performed TEM experiments and interpreted the results. J.L. conceived the Y2H experiment, provided material, discussed the data and edited the manuscript. J.V., R.S. and I.S. provided materials, discussed the data and edited the manuscript. K.H. conceived the project, designed and performed experiments, interpreted results, wrote and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–9 and Supplementary Table legends
Supplementary Table 1
MTM1 interactors in skeletal muscle.
Supplementary Table 2
List of antibodies used in this study.
Supplementary Table 3
List of primers.
Rights and permissions
About this article
Cite this article
Gavriilidis, C., Laredj, L., Solinhac, R. et al. The MTM1–UBQLN2–HSP complex mediates degradation of misfolded intermediate filaments in skeletal muscle. Nat Cell Biol 20, 198–210 (2018). https://doi.org/10.1038/s41556-017-0024-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-017-0024-9
- Springer Nature Limited
This article is cited by
-
A tumor suppressor protein encoded by circKEAP1 inhibits osteosarcoma cell stemness and metastasis by promoting vimentin proteasome degradation and activating anti-tumor immunity
Journal of Experimental & Clinical Cancer Research (2024)
-
FRMD3 inhibits the growth and metastasis of breast cancer through the ubiquitination-mediated degradation of vimentin and subsequent impairment of focal adhesion
Cell Death & Disease (2023)
-
X-linked myotubular myopathy is associated with epigenetic alterations and is ameliorated by HDAC inhibition
Acta Neuropathologica (2022)
-
Desmin aggrephagy in rat and human ischemic heart failure through PKCζ and GSK3β as upstream signaling pathways
Cell Death Discovery (2021)
-
Molecular and cellular basis of genetically inherited skeletal muscle disorders
Nature Reviews Molecular Cell Biology (2021)