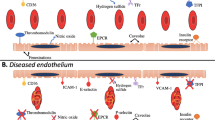
Abstract
Bone-marrow endothelial cells in the haematopoietic stem-cell niche form a network of blood vessels that regulates blood-cell traffic as well as the maintenance and function of haematopoietic stem and progenitor cells. Here, we report the design and in vivo performance of systemically injected lipid–polymer nanoparticles encapsulating small interfering RNA (siRNA), for the silencing of genes in bone-marrow endothelial cells. In mice, nanoparticles encapsulating siRNA sequences targeting the proteins stromal-derived factor 1 (Sdf1) or monocyte chemotactic protein 1 (Mcp1) enhanced (when silencing Sdf1) or inhibited (when silencing Mcp1) the release of stem and progenitor cells and of leukocytes from the bone marrow. In a mouse model of myocardial infarction, nanoparticle-mediated inhibition of cell release from the haematopoietic niche via Mcp1 silencing reduced leukocytes in the diseased heart, improved healing after infarction and attenuated heart failure. Nanoparticle-mediated RNA interference in the haematopoietic niche could be used to investigate haematopoietic processes for therapeutic applications in cancer, infection and cardiovascular disease.








Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within this paper and its Supplementary Information. The raw and analysed datasets generated during the study are too large to be publicly shared, but they are available for research purposes from the corresponding authors on reasonable request.
References
Morrison, S. J. & Scadden, D. T. The bone marrow niche for haematopoietic stem cells. Nature 505, 327–334 (2014).
Mendelson, A. & Frenette, P. S. Hematopoietic stem cell niche maintenance during homeostasis and regeneration. Nat. Med. 20, 833–846 (2014).
Crane, G. M., Jeffery, E. & Morrison, S. J. Adult haematopoietic stem cell niches. Nat. Rev. Immunol. 17, 573–590 (2017).
Kaplan, R. N., Psaila, B. & Lyden, D. Niche-to-niche migration of bone-marrow-derived cells. Trends Mol. Med. 13, 72–81 (2007).
Sipkins, D. A. et al. In vivo imaging of specialized bone marrow endothelial microdomains for tumour engraftment. Nature 435, 969–973 (2005).
Butler, J. M. et al. Endothelial cells are essential for the self-renewal and repopulation of Notch-dependent hematopoietic stem cells. Cell Stem Cell 6, 251–264 (2010).
Katayama, Y. et al. Signals from the sympathetic nervous system regulate hematopoietic stem. Cell 124, 407–421 (2006).
Gratwohl, A. et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 303, 1617–1624 (2010).
Giralt, S. et al. Optimizing autologous stem cell mobilization strategies to improve patient outcomes: consensus guidelines and recommendations. Biol. Blood Marrow Transpl. 20, 295–308 (2014).
Swirski, F. K. & Nahrendorf, M. Leukocyte behavior in atherosclerosis, myocardial infarction and heart failure. Science 339, 161–166 (2013).
Calado, R. T. & Young, N. S. Telomere maintenance and human bone marrow failure. Blood 111, 4446–4455 (2008).
Seo, A. et al. Bone marrow failure unresponsive to bone marrow transplant is caused by mutations in THPO. Blood 130, 875–880 (2017).
Whitehead, K. A., Langer, R. & Anderson, D. G. Knocking down barriers: advances in siRNA delivery. Nat. Rev. Drug Discov. 8, 129–138 (2009).
Titze-de-Almeida, R., David, C. & Titze-de-Almeida, S. S. The race of 10 synthetic RNAi-based drugs to the pharmaceutical market. Pharm. Res. 34, 1339–1363 (2017).
Anselmo, A. C. & Mitragotri, S. Nanoparticles in the clinic. Bioeng. Transl. Med. 1, 10–29 (2016).
Yin, H. et al. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 15, 541–555 (2014).
Kanasty, R., Dorkin, J. R., Vegas, A. & Anderson, D. Delivery materials for siRNA therapeutics. Nat. Mater. 12, 967–977 (2013).
Leuschner, F. et al. Therapeutic siRNA silencing in inflammatory monocytes in mice. Nat. Biotechnol. 29, 1005–1010 (2011).
Dahlman, J. E. et al. In vivo endothelial siRNA delivery using polymeric nanoparticles with low molecular weight. Nat. Nanotechnol. 9, 648–655 (2014).
Dong, Y. et al. Lipopeptide nanoparticles for potent and selective siRNA delivery in rodents and nonhuman primates. Proc. Natl Acad. Sci. USA 111, 3955–3960 (2014).
Love, K. T. et al. Lipid-like materials for low-dose, in vivo gene silencing. Proc. Natl Acad. Sci. USA 107, 1864–1869 (2010).
Khan, O. F. et al. Ionizable amphiphilic dendrimer-based nanomaterials with alkyl-chain-substituted amines for tunable siRNA delivery to the liver endothelium in vivo. Angew. Chem. Int. Ed. 53, 14397–14401 (2014).
Khan, O. F. et al. Dendrimer-inspired nanomaterials for the in vivo delivery of siRNA to lung vasculature. Nano Lett. 15, 3008–3016 (2015).
Whitehead, K. A. et al. Degradable lipid nanoparticles with predictable in vivo siRNA delivery activity. Nat. Commun. 5, 4277 (2014).
Khan, O. F. et al. Endothelial siRNA delivery in nonhuman primates using ionizable low-molecular weight polymeric nanoparticles. Sci. Adv. 4, eaar8409 (2018).
Chen, D. et al. Rapid discovery of potent siRNA-containing lipid nanoparticles enabled by controlled microfluidic formulation. J. Am. Chem. Soc. 134, 6948–6951 (2012).
Gref, R. et al. Stealth corona-core nanoparticles surface modified by polyethylene glycol (PEG): influences of the corona (PEG chain length and surface density) and of the core composition on phagocytic uptake and plasma protein adsorption. Colloids Surf. B Biointerfaces 18, 301–313 (2000).
Owens, D. E. III & Peppas, N. A. Opsonization, biodistribution and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
Heidt, T. et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 20, 754–758 (2014).
Xu, C. et al. Stem cell factor is selectively secreted by arterial endothelial cells in bone marrow. Nat. Commun. 9, 2449 (2018).
Cheng, M. & Qin, G. Progenitor cell mobilization and recruitment: SDF-1, CXCR4, α4-integrin and c-kit. Prog. Mol. Biol. Transl. Sci. 111, 243–264 (2012).
Furze, R. C. & Rankin, S. M. The role of the bone marrow in neutrophil clearance under homeostatic conditions in the mouse. FASEB J. 22, 3111–3119 (2008).
Whitehead, K. A., Dahlman, J. E., Langer, R. S. & Anderson, D. G. Silencing or stimulation? siRNA delivery and the immune system. Annu. Rev. Chem. Biomol. Eng. 2, 77–96 (2011).
Serbina, N. V. & Pamer, E. G. Monocyte emigration from bone marrow during bacterial infection requires signals mediated by chemokine receptor CCR2. Nat. Immunol. 7, 311–317 (2006).
Nahrendorf, M., Pittet, M. J. & Swirski, F. K. Monocytes: protagonists of infarct inflammation and repair after myocardial infarction. Circulation 121, 2437–2445 (2010).
Swirski, F. K. et al. Ly-6Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J. Clin. Invest. 117, 195–205 (2007).
Tacke, F. et al. Monocyte subsets differentially employ CCR2, CCR5 and CX3CR1 to accumulate within atherosclerotic plaques. J. Clin. Invest. 117, 185–194 (2007).
Yvan-Charvet, L. et al. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 328, 1689–1693 (2010).
Nahrendorf, M. et al. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J. Exp. Med. 204, 3037–3047 (2007).
Panizzi, P. et al. Impaired infarct healing in atherosclerotic mice with Ly-6Chi monocytosis. J. Am. Coll. Cardiol. 55, 1629–1638 (2010).
Itkin, T. et al. Distinct bone marrow blood vessels differentially regulate haematopoiesis. Nature 532, 323–328 (2016).
Broxmeyer, H. E. et al. Rapid mobilization of murine and human hematopoietic stem and progenitor cells with AMD3100, a CXCR4 antagonist. J. Exp. Med. 201, 1307–1318 (2005).
Ferraro, F. et al. Diabetes impairs hematopoietic stem cell mobilization by altering niche function. Sci. Transl. Med. 3, 104ra101 (2011).
Langen, U. H. et al. Cell-matrix signals specify bone endothelial cells during developmental osteogenesis. Nat. Cell Biol. 19, 189–201 (2017).
Peranteau, W. H., Hayashi, S., Hsieh, M., Shaaban, A. F. & Flake, A. W. High-level allogeneic chimerism achieved by prenatal tolerance induction and postnatal nonmyeloablative bone marrow transplantation. Blood 100, 2225–2234 (2002).
Kwarteng, E. O. & Heinonen, K. M. Competitive transplants to evaluate hematopoietic stem cell fitness. J. Vis. Exp. 114, e54345 (2016).
Maryanovich, M. et al. The ATM–BID pathway regulates quiescence and survival of haematopoietic stem cells. Nat. Cell Biol. 14, 535–541 (2012).
Acknowledgements
This work was funded in part by the National Institutes of Health (NIH) (nos. HL125428, HL131495 and T32HL076136), the European Union’s Horizon 2020 research and innovation programme (grant no. 667837) and the MGH Research Scholar Program. M.K.-G., M.J.S., D.R., S.C. and F.F.H. were supported by the Deutsche Forschungsgemeinschaft (nos. KR4613/1-1, SCHL 2221/1-1, RO5071/1-1, CR603/11 and HO5279/1-2). M.J.M. is supported by a Burroughs Wellcome Fund Career Award at the Scientific Interface, a Ruth L. Kirschstein National Research Service Award (no. F32CA200351) from the NIH, a fellowship from the Max Planck Society and a grant from the Burroughs Wellcome Fund (no. 1015145). P.P.G.G. is supported by a CNPq postdoctoral fellowship (no. 202856/2015-1) and a Fundação Estudar fellowship. This work is further supported in part by NIH grant no. R37-EB000244-30 (to R.L.), NIH contract no. HHSN268201000045C (to R.L.), a Koch–Prostate Cancer Foundation Award in Nanotherapeutics (to R.L.), the Koch Institute Marble Center for Cancer Nanomedicine and a Cancer Center Support (core) grant P30-CA14051 from the National Cancer Institute. We acknowledge the use of resources at the Koch Institute Swanson Biotechnology Center (and technical support), as well as the W.M. Keck Biological Imaging Facility (Whitehead Institute) and thank S. Mordecai at the flow, image and mass cytometry core of the MGH Department of Pathology. We thank K. Joyes for editing the manuscript.
Author information
Authors and Affiliations
Contributions
M.K.-G., M.J.M., M.J.S., D.G.A. and M.N. designed the experiments. M.J.S., O.F.K., G.C., P.P.G.G., S.C., Y.S., M.T., J.W., K.W., D.R., P.S.K., R.N., V.F., M.H., A.C., F.F.H., Y.I., S.P.S. and G.R.W. performed experiments and collected data. M.K.-G., M.J.M., F.K.S., R.L., D.G.A. and M.N. discussed the results and strategy. M.K.-G., M.J.M., D.G.A. and M.N. wrote the manuscript, which was edited by all co-authors. D.G.A. and M.N. supervised, directed and managed the study.
Corresponding authors
Ethics declarations
Competing interests
R.L. has served as an advisor to Alnylam Pharmaceuticals. For a list of entities with which R.L. is involved, compensated or uncompensated, see www.dropbox.com/s/yc3xqb5s8s94v7x/Rev%20Langer%20COI.pdf?dl=0. M.N. has received consulting fees unrelated to this work from Verseau, Gimv and IMF Therapeutics. The remaining authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures.
Rights and permissions
About this article
Cite this article
Krohn-Grimberghe, M., Mitchell, M.J., Schloss, M.J. et al. Nanoparticle-encapsulated siRNAs for gene silencing in the haematopoietic stem-cell niche. Nat Biomed Eng 4, 1076–1089 (2020). https://doi.org/10.1038/s41551-020-00623-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-00623-7
- Springer Nature Limited
This article is cited by
-
Nanoparticle-based DNA vaccine protects against SARS-CoV-2 variants in female preclinical models
Nature Communications (2024)
-
mRNA nanodelivery systems: targeting strategies and administration routes
Biomaterials Research (2023)
-
Engineering biomaterials to tailor the microenvironment for macrophage–endothelium interactions
Nature Reviews Materials (2023)
-
Passive, active and endogenous organ-targeted lipid and polymer nanoparticles for delivery of genetic drugs
Nature Reviews Materials (2023)
-
MicroRNA-208a silencing against myocardial ischemia/reperfusion injury mediated by reversibly camouflaged biomimetic nanocomplexes
Nano Research (2023)