Abstract
Herein, we disclose a highly efficient cobalt-catalyzed cross-electrophile alkynylation of a broad range of unactivated chlorosilanes with alkynyl sulfides as a stable and practical alkynyl electrophiles. Strategically, employing easily synthesized alkynyl sulfides as alkynyl precursors allows access to various alkynylsilanes in good to excellent yields. Notably, this method avoids the utilization of strong bases, noble metal catalysts, high temperature and forcing reaction conditions, thus presenting apparent advantages, such as broad substrate scope (72 examples, up to 97% yield), high Csp-S chemo-selectivity and excellent functional group compatibility (Ar-X, X = Cl, Br, I, OTf, OTs). Moreover, the utilities of this method are also illustrated by downstream transformations and late-stage modification of structurally complex natural products and pharmaceuticals. Mechanistic studies elucidated that the cobalt catalyst initially reacted with alkynyl sulfides, and the activation of chlorosilanes occurred via an SN2 process instead of a radical pathway.
Similar content being viewed by others
Introduction
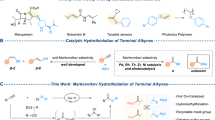
Transition-metal catalyzed cross-electrophile coupling (XEC) has become a prominent method for synthesizing molecules, attracting significant attention in the field1,2,3,4,5,6. These reactions offer several advantages, including the avoidance of pre-generation and handling of sensitive organometallic reagents, high efficiency in terms of steps, and excellent compatibility with various functional groups compared to traditional methods. The groundbreaking studies by Weix and Gosmini using nickel and cobalt catalysis paved the way for advancements in C-C bond formation7,8,9,10,11,12,13,14,15,16. Despite the significant progress in C-C bond formation through XEC, there remains a significant gap in the development of XEC reactions between carbon electrophiles and readily available chlorosilanes for producing organosilicon compounds, highlighting a pressing need for further research in this area17,18,19.
Organosilicon compounds have unique properties that are broadly applicable to synthetic chemistry, medicinal chemistry, materials science and other fields20,21,22,23. Very recently, the Shu, Oestreich and other group respectively reported several elegant Ni-catalyzed cross-electrophile protocols to form C-Si bond, in which a variety of aryl/vinyl/alkyl electrophiles underwent a highly efficient coupling with activated chlorosilanes (R = vinyl, H, Fig. 1a)24,25,26,27,28,29. Subsequently, Fe- and Cr-catalyzed XEC of activated chlorosilanes had also been developed using different catalytic systems30,31. Despite formidable advances, no examples of XEC of alkynyl electrophiles with silyl electrophiles by transition-metal catalysis to form Csp-Si bond, have been reported until now.
We speculated that there were mainly two reasons. Firstly, alkynyl electrophiles, such as bromoalkynes, were too active to undergo homocoupling to form 1,3-diynes instead of the cross-coupling products32,33,34. Meanwhile, the low reactivity and the high bond dissociation energy (BDE) of Si-Cl bond35 (113 kcal/mol) compared to the C-Cl bond36 (84 kcal/mol) were adverse to its XEC with reactive alkynyl electrophiles. Secondly, the low nucleophilicity of Csp-M intermediate compared with aryl- or alkyl-M species exhibited low reactivity towards chlorosilanes. To verify this hypothesis, bromoalkyne 1-a and TMSCl were utilized as the starting materials to conduct preliminary experimental exploration (Fig. 1a)24,25,26,28. Mainly the dimer product 4-a was obtained due to the mismatched reaction rates. Thus, utilizing an appropriate alkynyl electrophile and metal catalyst was the key to achieve the XEC between alkynyl electrophiles and chlorosilanes.
Easily synthesized and air-stable alkynyl sulfides37,38 had sporadically been utilized as alkynyl electrophiles for metal catalyzed cross-coupling39,40, while was unknown for XEC. In this report, we disclose a highly efficient cobalt-catalyzed cross-electrophile alkynylation of a broad range of unactivated chlorosilanes with alkynyl sulfides as a stable and practical alkynyl electrophiles (Fig.1b). This method generates various alkynylsilanes (72 examples, up to 97% yield) under mild condition, which avoids the use of strong bases, noble metal catalysts and high temperature, providing an alternative and complementary approach41,42,43.
Results
Investigation of reaction conditions
We started with our investigations by using alkynyl sulfide alkynyl sulfide 1a and commercially available TMSCl as the model substrates. Extensive examination of the reaction parameters revealed that the combination of 10 mol% of CoI2 as the catalyst and 15 mol% of L8 (tpy) as ligand with zinc as the reductant in DMF at room temperature was the optimal conditions to deliver the desired product 3a in 85% isolated yield (Table 1, entry 1, see Suppelementary Tables for details). Different Co(II) precursors were tested, CoCl2, CoBr2 and Co(acac)2 gave the product 3a in high yields but with less efficiency (entries 2-3). Meanwhile, the cross-selectivity decreased dramatically and a certain amount of 1,3-diyne was obtained when NiI2 or CrCl2 was utilized instead of CoI2 as the catalyst (entry 4)24,28,31. The alkynyl sulfide 1a was fully recovered in the presence of iron salt precatalyst (entry 5)30. Solvent screening demonstrated that acetonitrile (MeCN) or tetrahydrofuran (THF) shut down the reaction completely (entry 6). The replacement of Zn to Mn resulted no conversion of this reaction (entry 7). When 1 equiv of ZnI2 was added together with Mn, the reaction generated the product 3a in 67% yield (entry 8). This indicated that Zn not only acted as reductant, but also the in-situ formed zinc salt as a thiophilic reagent to decrease the inhibition of the leaving sulfide part to catalyst44. Control experiments showed that the stoichiometric reductant zinc, the cobalt catalyst and ligand were all essential for this transformation (entry 9). The effect of ligand indicated that bidentate ligands such as bipyridine and phenanthroline derivatives, though were not as good as terpyridine L8, still gave the desired product in reduced yields. Conversely, the sterically hindered ligand L7 failed to give the desired product 3a.
Substrate scope
With the optimized reaction conditions in hand, we explored the scope of alkynyl sulfides for this cobalt-catalyzed XEC with TMSCl or PhMe2SiCl, and the results were summarized in Fig. 2. Alkynyl sulfides with different electronic properties or substitution patterns (3a-t) were tolerated. Firstly, alkynyl sulfides with diverse electron-donating groups at the para-position of the aryl moiety, including ether (3a-c), -OAc (3 f), -OCF3 (3 g), -OTIPS (3 h) and -NHAc(3j), were converted into the corresponding products in good yields (41–93%). The method also demonstrated high Csp-S chemo-selectivity as reactions with an alkynyl sulfide containing an aryl sulfide moiety to give the product 3i in 91% yield45,46,47,48. Additionally, the electron-withdrawing groups, such as -F (3k-l), -CN (3 m), -CO2Me (3n), -CONMe2 (3o), and -CO2H (3p) were all compatible with the method and delivered the desired products in moderate to excellent yields (55-93%). Although the presence of a terminal alkene or alkyne was challenging to the silylation procedure49,50, the corresponding products 3q and 3r were successfully obtained in 75% and 88% yield, respectively. Moreover, a free alcohol (3 s) or amine (3t), which could react directly with chlorosilanes, was well tolerated under the standard conditions. When alkyl or alkenyl substituted alkynyl sulfides were selected as substrates, the alkynylsilylation reactions occurred as expected to afford the corresponding products in excellent yields (3u-y). The reactions of alkynyl sulfides containing polyaromatic or heteroaromatic ring proceeded smoothly to produce alkynylsilanes (3z-aj) in moderate to good yields, including fluorene (3z), naphthalene (3aa) thiophene (3ab, 3ac), pyridine (3ad), quinoline (3ae), benzofuran (3af), indole (3ag), benzothiophene (3ah), carbazole (3ai) and dibenzofuran (3aj). Given the broad substrate scope and good functionality tolerance of our strategy, we further explored this transformation to for late-stage functionalization with more complex molecules. Alkynyl sulfides derived from Donepezil (3ak), Desloratadine (3al), Citronellol (3am), DL-Menthol (3an), Gemfibrozil (3ao), and Cholesterol (3ap), were all suitable substrates, providing the corresponding products in 41-87% yields.
Next, we explored the reactivity of chlorosilanes by varying the substituents on silicon for these transformations (Fig. 3). A series of chlorodimethyl(alkyl)silanes with varied chain lengths and steric properties reacted with alkynyl sulfide 1a, giving the corresponding alkynylsilylation products in moderate to high yields. For instance, chlorodimethyl(ethyl) silane (4a), chlorodimethyl(butyl) silane (4b), chlorodimethyl(3-phenylpropyl) silane (4c) and chlorodimethyl-(butanenitrile) silane (4d) furnished the desired products in 83% to 96% yields. Chlorosilanes bearing steric substituents, such as diaryl (4 f), isopropyl (4 g), cyclohexanyl (4 h) and TMS (4i), all worked well for this cobalt-catalyzed system, affording the corresponding products with good efficiency. The presence of allylic and vinyl functionalities on the chlorosilanes was well-tolerated, offering opportunities for further diversification of the resulting products (4j-l). It is worth noting that several chlorotrialkylsilanes with larger steric hindrance were also tested, yielding the corresponding products in the range of 69% to 71% (4m-n).
To further exhibit the excellent chemo-selectivity of alkynyl sulfides as alkynyl electrophiles for these reactions, we conducted the XEC with alkynyl sulfides containing various electrophilic and nucleophilic reaction sites and the results were summarized in Fig. 4. These studies demonstrated that this Co-catalyzed silylation of alkynyl sulfides offered orthogonal selectivity towards other well-established cross-coupling methods. Aryl electrophiles, including chloride (3aq), bromides (3ar–as), iodide (3at), tosylate (3au), and triflates (3av, 3aw), were all tolerated under the standard conditions. In addition, alkynyl sulfides containing additional electrophilic functional groups were converted to alkynylsilanes in good yields (3ax-bc), which provided the chance to further modify these molecules. An alkynyl sulfide containing an alkyl chloride moiety was selectively silylated at the Csp-S site (3bd). Alkynylsilane containing a boronic ester group (3be) was constructed in 85% yield for this Co-catalyzed XEC.
Alkynylsilanes are synthetically useful intermediates, which have been widely used in synthetic chemistry51,52,53,54,55,56,57,58,59. To show the synthetic utility of this protocol, we conducted a series of further transformation of the desired alkynylsilane. First, a gram-scale reaction of alkynyl sulfide 1ay with a reduced catalyst loading (5 mol%) to generate alkynylsilane 3ay in 71% yield. Tetrabutylammonium fluoride catalyzed the addition of 3ay to trifluoromethyl ketone, producing the CF3-substituted tertiary propargylic alcohol 5a in moderate yield (Fig. 5a-a). A Cu-catalyzed three-component coupling reaction of 3ay, o-hydroxybenzaldehyde and amine, formed the corresponding benzofuran 5b in 76% yield, involving an intramolecular 5-exo-dig cyclization (Fig. 5a, b). A copper-catalyzed three-component coupling reaction of alkynylsilane 3ay, aldehyde and amine generated the propargylic amine 5c in 84% yield (Fig. 5a–c). A ZnCl2-catalyzed Diels-Alder/retro-Diels-Alder reaction between electron-deficient 2-pyrone and alkynylsilane 3ay enabled the synthesis of arylsilane 5d in 73% yield (Fig. 5a–d). The gold-catalyzed reaction of o-alkynylbenzaldehydes with alkynylsilane 3ay gave naphthylsilane 5e in 49% yield (Fig. 5a–e). And the Cobalt(I)–diphosphine catalyzed dehydrogenative annulation between alkynylsilane 3ay and salicylaldehyde afforded the corresponding 2-aryl-3-silylchromone 5 f in 69% yield (Fig. 5a–f). Moreover, a Co-catalyzed regioselective [3 + 2] annulation of ortho-functionalized arylboronic acid with 3ay gave the corresponding cyclized products 5 g and 5 h in 81% and 65% yield respectively Fig. 5ag, h). To further demonstrate the synthetic utility of this developed methodology, the efficient synthesis of 6-fluoroflavone, an anti-rhinovirus agent, was carried out using current method as the key step (Fig. 5b). Finally, we extended our method to several examples of the synthesis of alkynylgermanes, all of which had good yields (Fig. 5c).
a Gram-scale reaction and transformations of alkynylsilane; b Synthesis of 6-fluoroflavone; c Extended to synthesis of alkynylgermanes. See Supplementary Information for detailed experimental conditions.
To elucidate the underlying mechanism of these reactions, a set of experiments was conducted as shown in Fig. 6. Firstly, to reveal whether a radical process was involved in the activation of R3Si-Cl, several radical acceptors (3.0 equiv) were added to the standard reaction conditions and 4e was obtained in 69-82% yields with no sign of the expected side product 7 from radical trapping (Fig. 6a). A radical clock experiment of chlorosilane 2o was performed, and the directly silylated product 4o was obtained in 87% yield with no cyclized derivative detected (Fig. 6b). These results suggest that Si-Cl is not activated through a radical process. Subsequently, alkynyl sulfide 1a was transformed to the homodimer (diyne) product 1a-1 in 89% yield in the absence of chlorosilane, which indicated that oxidative addition of alkynyl sulfide with low valent Co to form Csp-Co species was possible. Moreover, in the absence of alkynyl sulfide, chlorosilane 2 l failed to dimerize. In addition, adding 3 equivalents of water to the reaction system under standard conditions, the corresponding terminal alkyne 1a-2 was generated in 72% yield. These results further indicated that the low valant cobalt catalyst might activate alkynyl sulfide firstly and generate the Csp-Co species (Fig. 6c). Considering whether our reaction involves alkynylzinc species, we monitored the reaction progress and found that the conversion rate of 1a was basically consistent with the yield of 3a, and no corresponding iodide product 1a-3 formation after I2 quenched the aliquots at different time periods (Fig. 6d). This suggests that the alkynylzinc intermediate should not be involved in our reaction. To understand which oxidation state of cobalt reacts with 1a to initiate the reaction, we performed cyclic voltammetry experiments of CoII(tpy) and in-situ generated low valant Co in DMF. The cyclic voltammogram of B revealed a 2-electron reduction wave at E = −1.55 V (Fig. 6e), which corresponds to the CoI /Co-I couple. And that was consistent with Gosmini’s observation16. Finally, we investigated the reactivity of the in-situ-formed CoI(tpy) towards alkynyl sulfide 1a. Both the stoichiometric reaction of CoI(tpy) with 1a and using CoI(tpy) to catalyze the reaction had been carried out (Fig. 6f), and the experimental results indicated that: 1) alkynyl sulfide 1a can be consumed by equivalent CoI(tpy); 2) the in-situ-formed CoI(tpy) is the active intermediate of the reaction.
a Radical trapping experiments. b Radical clock experiment. c Control experiment. d Alkynylzinc species exploration experiment. e Cyclic voltammograms of CoII(tpy) and in-situ generated CoI(tpy) in DMF. f The reactivity of in-situ-formed Co(I) towards alkynyl sulfide 1a. See Supplementary Information for detailed experimental conditions.
On the basis of these experimental results and previous reports24,25,27, a catalytic cycle was proposed in Fig. 7. A low-valent CoI species was generated in-situ via reduction of CoII precursor with zinc60,61. Then, the alkynyl sulfide 1 reacted with CoI via oxidative addition to give Csp-CoIII Int. A, which was subsequently reduced by Zn to provide a more nucleophilic alkynyl CoI Int. B62,63. The SN2 oxidative addition of chlorosilanes to Int. B64,65, possibly through a five-coordinate cobalt intermediate C25,66,67, to deliver alkynyl CoIII D. Reductive elimination of complex D would afford the desired product 3 with generation of CoI to entered next catalytic cycle.
Discussion
In conclusion, we reported the cobalt-catalyzed cross-electrophile coupling between alkynyl electrophiles with chlorosilanes to construct the C(sp)-Si bonds in the presence of commercially available cobalt catalyst with zinc as the reductant. The utilization of easily prepared alkynyl sulfides by our previous reported work as the alkynyl electrophile was key to achieving cross selectivity with various chlorosilanes. These cobalt-catalyzed XEC between alkynyl sulfides with chlorosilanes exhibited excellent chemoselectivity towards various electrophiles such as, aryl chloride, bromide, iodide, triflate, tosylate and alkyl chloride. Moreover, the efficacy of this approach is further demonstrated through late-stage modification of structurally intricate natural products and drugs. Mechanistic studies revealed that the cobalt catalyst reacts with alkynyl sulfides first and it is likely the chlorosilanes react through a SN2 process via a non-radical pathway. Developing other cross-electrophile coupling of alkynyl sulfides as a stable and practical alkynyl electrophiles are ongoing in our laboratory.
Methods
General procedure for cobalt-catalyzed cross-electrophile coupling of alkynyl sulfides with unactivated chlorosilanes
The procedure was conducted in a nitrogen-filled glove box. To a reaction vial equipped with a magnetic stir bar was added CoI2 (6.3 mg, 0.02 mmol), L8 (7.0 mg, 0.03 mmol), Zn (26.2 mg, 2 equiv). A solution of 1 (0.2 mmol) and chlorosilane 2 (0.4 mmol) in DMF (2.0 mL) was added. The reaction vial was sealed and removed from the glove box. The mixture was stirred at room temperature for 4 h, subsequently quenched with water (10.0 mL) and extracted with ethyl acetate (3 × 15.0 mL). The combined organic layers were washed with water, brine, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel to afford the desired product.
General procedure for cobalt-catalyzed cross-electrophile coupling of alkynyl sulfides with Me3GeCl
The procedure was conducted in a nitrogen-filled glove box. To a reaction vial equipped with a magnetic stir bar was added CoI2 (6.3 mg, 0.02 mmol), L8 (7.0 mg, 0.03 mmol), Zn (26.2 mg, 2 equiv). A solution of 1 (0.2 mmol) and Me3GeCl (0.4 mmol) in DMF (2.0 mL) was added. The reaction vial was sealed and removed from the glove box. The mixture was stirred at room temperature for 4 h, subsequently quenched with water (10.0 mL) and extracted with ethyl acetate (3 × 15.0 mL). The combined organic layers were washed with water, brine, dried over anhydrous Na2SO4, and concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel to afford the desired product.
Data availability
Data related to materials and methods, optimization of conditions, experimental procedures, mechanistic experiments, and spectra are provided in the Supplementary information. All data are available from the corresponding authors upon request.
References
Knappke, C. E. I. et al. Reductive cross-coupling reactions between two electrophiles. Chem. Eur. J. 20, 6828–6842 (2014).
Everson, D. A. & Weix, D. J. Cross-electrophile coupling: principles of reactivity and selectivity. J. Org. Chem. 79, 4793–4798 (2014).
Wang, X., Dai, Y. & Gong, H. Nickel-catalyzed reductive couplings. Top. Curr. Chem. 374, 43 (2016).
Poremba, K. E., Dibrell, S. E. & Reisman, S. E. Nickel-catalyzed enantioselective reductive cross-coupling reactions. ACS Catal 10, 8237–8246 (2020).
Liu, J., Ye, Y., Sessler, J. L. & Gong, H. Cross-electrophile couplings of activated and sterically hindered halides and alcohol derivatives. Acc. Chem. Res. 53, 1833–1845 (2020).
Yi, L., Ji, T., Chen, K., Chen, X. & Rueping, M. Nickel-catalyzed reductive cross-couplings: new opportunities for carbon-carbon bond formations through photochemistry and electrochemistry. CCS Chem 4, 9–30 (2022).
Everson, D. A., Jones, B. A. & Weix, D. J. Replacing conventional carbon nucleophiles with electrophiles: nickel-catalyzed reductive alkylation of aryl bromides and Chlorides. J. Am. Chem. Soc. 134, 6146–6159 (2012).
Zhang, P., Le, C. C. & MacMillan, D. W. C. Silyl radical activation of alkyl halides in metallaphotoredox catalysis: a unique pathway for cross-electrophile coupling. J. Am. Chem. Soc. 138, 8084–8087 (2016).
Salgueiro, D. C., Chi, B. K., Guzei, I. A., Garcia-Reynaga, P. & Weix, D. J. Control of redox-active ester reactivity enables a general cross-electrophile approach to access arylated strained rings. Angew. Chem., Int. Ed. 61, e202205673 (2022).
Kang, K., Huang, L. & Weix, D. J. Sulfonate versus sulfonate: nickel and palladium multimetallic cross-electrophile coupling of aryl triflates with aryl tosylates. J. Am. Chem. Soc. 142, 10634–10640 (2020).
Huang, L., Ackerman, L. K. G., Kang, K., Parsons, A. M. & Weix, D. J. LiCl-accelerated multimetallic cross-coupling of aryl chlorides with aryl triflates. J. Am. Chem. Soc. 141, 10978–10983 (2019).
He, R.-D. et al. Reductive alkylation of alkenyl acetates with alkyl bromides by nickel catalysis. Angew. Chem., Int. Ed. 61, e202114556 (2022).
Gomes, P., Gosmini, C. & Périchon, J. New chemical cross-coupling between aryl halides and allylic acetates using a cobalt catalyst. Org. Lett. 5, 1043–1045 (2003).
Amatore, M. & Gosmini, C. Efficient cobalt-catalyzed formation of unsymmetrical biaryl compounds and its application in the synthesis of a sartan intermediate. Angew. Chem., Int. Ed. 47, 2089–2092 (2008).
Qian, X., Auffrant, A., Felouat, A. & Gosmini, C. Cobalt-catalyzed reductive allylation of alkyl halides with allylic acetates or carbonates. Angew. Chem., Int. Ed. 50, 10402–10405 (2011).
Dorval, C., Tricoire, M., Begouin, J.-M., Gandon, V. & Gosmini, C. Cobalt-catalyzed C(sp2)-CN bond activation: cross-electrophile coupling for biaryl formation and mechanistic insight. ACS Catal 10, 12819–12827 (2020).
Pang, X., Su, P.-F. & Shu, X.-Z. Reductive cross-coupling of unreactive electrophiles. Acc. Chem. Res. 55, 2491–2509 (2022).
Pang, X. & Shu, X.-Z. Nickel-catalyzed reductive coupling of chlorosilanes. Chem. Eur. J. 29, e202203362 (2023).
Yang, Y.-H., Pang, X. & Shu, X.-Z. Transition-metal-catalyzed cross-coupling of chlorosilanes. Synthesis 55, 868–876 (2023).
Ito, Y. et al. Crystalline ultrasmooth self-assembled monolayers of alkylsilanes for organic field-effect transistors. J. Am. Chem. Soc. 131, 9396–9404 (2009).
Franz, A. K. & Wilson, S. O. Organosilicon molecules with medicinal applications. J. Med. Chem. 56, 388–405 (2013).
Ramesh, R. & Reddy, D. S. Quest for novel chemical entities through incorporation of silicon in drug scaffolds. J. Med. Chem. 61, 3779–3798 (2018).
Zuo, Y., Gou, Z., Quan, W. & Lin, W. Silicon-assisted unconventional fluorescence from organosilicon materials. Coord. Chem. Rev. 438, 213887 (2021).
Duan, J. et al. Cross-electrophile C(sp2)−Si coupling of vinyl chlorosilanes. Angew. Chem., Int. Ed. 59, 23083–23088 (2020).
Zhao, Z.-Z., Pang, X., Wei, X.-X., Liu, X.-Y. & Shu, X.-Z. Nickel-catalyzed reductive C(sp2)−Si coupling of chlorohydrosilanes via Si−Cl cleavage. Angew. Chem., Int. Ed. 61, e202200215 (2022).
Duan, J. et al. Nickel-catalyzed cross-electrophile C(sp3)−Si coupling of unactivated alkyl bromides with vinyl chlorosilanes. Org. Lett. 23, 7855–7859 (2021).
Su, P.-F. et al. Nickel-catalyzed reductive C-Ge coupling of aryl/alkenyl electrophiles with chlorogermanes. Angew. Chem., Int. Ed. 60, 26571–26576 (2021).
Zhang, L. & Oestreich, M. Nickel-catalyzed, reductive C(sp3)-Si cross-coupling of α-Cyano alkyl electrophiles and chlorosilanes. Angew. Chem., Int. Ed. 60, 18587–18590 (2021).
Sun, J. et al. Regioselective Ni-catalyzed reductive alkylsilylation of acrylonitrile with unactivated alkyl bromides and chlorosilanes. Nat. Commun. 13, 7093–7102 (2022).
Lin, Y. et al. Iron-catalyzed cross-electrophile coupling of bromostyrenes and chlorosilanes. Org. Chem. Front. 10, 3052–3060 (2023).
Li, C., Yang, S. & Zeng, X. Cross-electrophile silylation of aryl carboxylic esters with hydrochlorosilanes by SiH-directed and Cr-catalyzed couplings. ACS Catal 13, 12062–12073 (2023).
Huang, L., Olivares, A. M. & Weix, D. J. Reductive decarboxylative alkynylation of N-hydroxyphthalimide esters with bromoalkynes. Angew. Chem., Int. Ed. 56, 11901–11905 (2017).
Jiang, Y., Pan, J., Yang, T., Zhao, Y. & Koh, M. J. Nickel-catalyzed site- and stereoselective reductive alkylalkynylation of alkynes. Chem 7, 993–1005 (2021).
Pan, R. et al. Nickel-catalyzed reductive 1,2-Dialkynylation of alkenes bearing an 8-aminoquinoline directing group. Org. Lett. 21, 8915–8920 (2019).
Walsh, R. Bond dissociation energy values in silicon-containing compounds and some of their implications. Acc. Chem. Res. 14, 246–252 (1981).
Ji, C.-L. et al. Photoinduced activation of alkyl chlorides. Chem. Soc. Rev. 52, 6120–6138 (2023).
Xing, D. et al. Alkyne/thio umpolung tactic replacement: synthesis of alkynyl sulfides via capturing the in situ formed alkynylthiolate anion. Org. Chem. Front. 10, 3603–3611 (2023).
Cao, Y., Huang, Y. & Blakemore, P. R. Synthesis of Thioalkynes by Desilylative Sonogashira cross-coupling of Aryl Iodides and 1-Methylthio-2-(trimethylsilyl)ethyne. Eur. J. Org. Chem. 2022, e202200498 (2022).
Savarin, C., Srogl, J. & Liebeskind, L. S. Substituted alkyne synthesis under nonbasic conditions: copper carboxylate-mediated, palladium-catalyzed thioalkyne-boronic acid cross-coupling. Org. Lett. 3, 91–93 (2001).
Melzig, L., Metzger, A. & Knochel, P. Pd- and Ni-catalyzed cross-coupling reactions of functionalized organozinc reagents with unsaturated Thioethers. Chem. Eur. J. 17, 2948–2956 (2011).
Toutov, A. A. et al. Alkali metal-hydroxide-catalyzed C(sp)-H bond silylation. J. Am. Chem. Soc. 139, 1668–1674 (2017).
Ma, Y. et al. B(C6F5)3/Amine-catalyzed C(sp)-H silylation of terminal alkynes with hydrosilanes: experimental and theoretical studies. Angew. Chem., Int. Ed. 57, 15222–15226 (2018).
Helbert, H., Visser, P., Hermens, J. G. H., Buter, J. & Feringa, B. L. Palladium-catalysed cross-coupling of lithium acetylides. Nat. Catal. 3, 664–671 (2020).
Liebeskind, L. S., Srogl, J., Savarin, C. & Polanco, C. Bioinspired organometallic chemistry. Pure Appl. Chem. 74, 115–122 (2002).
Ma, Y., Cammarata, J. & Cornella, J. Ni-catalyzed reductive Liebeskind-Srogl alkylation of heterocycles. J. Am. Chem. Soc. 141, 1918–1922 (2019).
Isshiki, R., Kurosawa, M. B., Muto, K. & Yamaguchi, J. Ni-catalyzed aryl sulfide synthesis through an aryl exchange reaction. J. Am. Chem. Soc. 143, 10333–10340 (2021).
Ma, N.-N. et al. Nickel-catalyzed direct cross-coupling of aryl thioether with aryl bromide. Org. Lett. 25, 1771–1775 (2023).
Begouin, J.-M., Rivard, M. & Gosmini, C. Cobalt-catalyzed C-SMe bond activation of heteroaromatic thioethers. Chem. Commun. 46, 5972–5974 (2010).
McAtee, J. R. et al. The first example of nickel-catalyzed silyl-Heck reactions: direct activation of silyl triflates without iodide additives. Tetrahedron 70, 4250–4256 (2014).
Matsumoto, K. et al. Direct silyl-heck reaction of chlorosilanes. Org. Lett. 20, 2481–2484 (2018).
Larson, G. L. Some aspects of the chemistry of alkynylsilanes. Synthesis 50, 2433–2462 (2018).
Asao, N., Takahashi, K., Lee, S., Kasahara, T. & Yamamoto, Y. AuCl3-catalyzed benzannulation: synthesis of naphthyl ketone derivatives from o-alkynylbenzaldehydes with alkynes. J. Am. Chem. Soc. 124, 12650–12651 (2002).
Sakai, N., Uchida, N. & Konakahara, T. Facile and selective synthesis of propargylic amines and 1,6-diynes: one-pot three-component coupling reactions of alkynylsilanes, aldehydes and Amines by a cooperative catalytic system comprised of CuCl and Cu(OTf)2. Synlett, 1515–1519 https://doi.org/10.1055/s-2008-1077790 (2008).
Sakai, N., Uchida, N. & Konakahara, T. Facile and efficient synthesis of polyfunctionalized benzofurans:: three-component coupling reactions from an alkynylsilane, an o-hydroxybenzaldehyde derivative, and a secondary amine by a Cu(I)-Cu(II) cooperative catalytic system. Tetrahedron Lett 49, 3437–3440 (2008).
Chintareddy, V. R., Wadhwa, K. & Verkade, J. G. Tetrabutylammonium fluoride (TBAF)-catalyzed addition of substituted trialkylsilylalkynes to aldehydes, ketones, and trifluoromethyl ketones. J. Org. Chem. 76, 4482–4488 (2011).
Ueda, M., Ueno, T., Suyama, Y. & Ryu, I. Synthesis of 2,3-disubstituted indenones by cobalt-catalyzed 3+2 annulation of o-methoxycarbonylphenylboronic acid with alkynes. Chem. Commun. 52, 13237–13240 (2016).
Yang, J. & Yoshikai, N. Cobalt-catalyzed annulation of Salicylaldehydes and alkynes to form chromones and 4-chromanones. Angew. Chem., Int. Ed. 55, 2870–2874 (2016).
Ueda, M., Ueno, T., Suyama, Y. & Ryu, I. Cobalt-catalyzed regioselective 3+2 annulation of ortho-formyl and acetyl substituted phenylboronic acids with alkynes. Tetrahedron Lett 58, 2972–2974 (2017).
Song, L. et al. Ru-catalyzed migratory geminal semihydrogenation of internal alkynes to terminal Olefinsl. J. Am. Chem. Soc. 141, 17441–17451 (2019).
Gandeepan, P. & Cheng, C.-H. Cobalt catalysis involving π components in organic synthesis. Acc. Chem. Res. 48, 1194–1206 (2015).
Aresta, M., Rossi, M. & Sacco, A. Tetrahedral complexes of cobalt(I). Inorg. Chim. Acta 3, 227–231 (1969).
Cahiez, G. & Moyeux, A. Cobalt-catalyzed cross-coupling reactions. Chem. Rev. 110, 1435–1462 (2010).
Ueda, Y., Tsurugi, H. & Mashima, K. Cobalt-catalyzed alkynylation of organic compounds: hydroalkynylation, dehydrogenative alkynylation, and reductive alkynylation. Synlett 34, 990–1000 (2023).
Levy, C. J. & Puddephatt, R. J. Rapid reversible oxidative addition of group 14 halide bonds to platinum(II): Rates, equilibria, and bond energies. J. Am. Chem. Soc. 119, 10127–10136 (1997).
Kameo, H. et al. Experimental and theoretical studies of Si-Cl and Ge-Cl σ-bond activation reactions by iridium-hydride. Dalton Trans 45, 7570–7580 (2016).
Morita, E. et al. Copper-catalyzed arylation of chlorosilanes with grignard reagents. Bull. Chem. Soc. Jpn. 82, 1012–1014 (2009).
Murakami, K., Hirano, K., Yorimitsu, H. & Oshima, K. Silver-catalyzed transmetalation between chlorosilanes and aryl and alkenyl Grignard reagents for the synthesis of tetraorganosilanes. Angew. Chem., Int. Ed. 47, 5833–5835 (2008).
Acknowledgements
The authors thank National Natural Science Foundation of China (No. 21971074, L.H.; No. 22001076, L.H.), Natural Science Foundation of Guangdong Province (No. 2019A1515010006, L.H.; No. 2021A1515010159, L.H.) for the financial support. We thank Dr. Matthew J. Goldfogel (Bristol Myers Squibb) for a helpful discussion.
Author information
Authors and Affiliations
Contributions
D.X. and L.H. designed the experiments. D.X. performed experiments. J.L., D.C. and B.H. assisted with melting points determination, some substrates synthesis and validation. D.X. wrote the paper and L.H. revised reviewed & edited the paper. H.J. contributed to discussions. All authors discussed the results and commented on the manuscript. L.H. directed the whole project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xing, D., Liu, J., Cai, D. et al. Cobalt-catalyzed cross-electrophile coupling of alkynyl sulfides with unactivated chlorosilanes. Nat Commun 15, 4502 (2024). https://doi.org/10.1038/s41467-024-48873-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-48873-2
- Springer Nature Limited