Abstract
We investigated whether the association between a history of hypertensive disorders of pregnancy (HDP) and hypertension in later life varies by age group and the effect of obesity on the association between a history of HDP and hypertension in later life. This cross-sectional population-based study was conducted at the Tohoku Medical Megabank Project in Miyagi and Iwate, Japan. The study subjects were 33,412 parous women of 20 years of age and older. We used multivariate logistic regression analysis to assess the association between a history of HDP and hypertension. We constructed a composite variable that combined a history of HDP (±) and overweight/obesity (BMI ≥ 25 kg/m2) (±), resulting in four categories, and analyzed the risks of each category by multivariate logistic regression analysis. In total, 1585 (4.7%) women had a history of HDP. The prevalence of hypertension was higher in women with HDP (51.4%) than in those without HDP (36.8%; p < 0.01). The adjusted odds ratios (ORs) for hypertension in women with HDP in their 30s, 40s, 50s, 60s, and 70s or older were 3.63, 1.84, 2.15, 1.48, and 1.86, respectively. In the interaction analysis, the association between a history of HDP and hypertension was stronger in women in their 30s–50s than in women who were 60 or older (p = 0.057). The adjusted ORs for hypertension were higher in overweight/obese women with HDP than in their nonoverweight/obese counterparts in all age groups (30s: 27.17 vs. 2.22; 70s: 4.75 vs. 1.90). In conclusion, the association between HDP and later hypertension was stronger in younger women and in obese women in the 30–70 age group.
Similar content being viewed by others
Introduction
Hypertension is a common disease, affecting ~31% of the adult population worldwide [1]. Hypertension is the leading contributor to the global disease burden [2], particularly the burdens associated with cardiovascular disease [3] and chronic kidney disease; [4] hypertension is also associated with dementia [5] and with age-related declines in activities of daily living [6].
Hypertension is a multifactorial disease: lifestyle factors such as obesity and excess intake of sodium and alcohol, underlying conditions such as diabetes and dyslipidemia, and genetic factors all play important roles [7]. The prevalence of obesity is increasing worldwide, and as a result, the impact of obesity on hypertension has increased [8, 9].
Interestingly, the association between obesity and hypertension is stronger in women than in men [10, 11]. The difference in body fat distribution is presumed to be associated with this observed sex difference [11].
Women have other specific risk factors for hypertension, such as hypertensive disorders of pregnancy (HDP) [12, 13] and menopause [7, 14]. Accumulating evidence suggests that a history of HDP is a risk factor for hypertension, metabolic syndrome, and cardiovascular disease later in life [15, 16]. Reportedly, mortality from cardiovascular disease is higher in women with a history of early onset preeclampsia than in women with late-onset preeclampsia [17]. Although HDP is a good predictor of future hypertension, there are few guidelines regarding reducing the risk for future hypertension in women with a history of HDP. It is unclear whether the association between a history of HDP and hypertension in later life varies by age group. If the effect of HDP on hypertension is stronger in younger women, we should recommend that women with a history of HDP undergo regular checkups from a young age.
It also remains unclear which is more strongly correlated with hypertension in women: a history of HDP or obesity. Likewise, it is unknown whether the risk for hypertension is equivalent in obese and nonobese women of any age group who have a history of HDP. If the risk for hypertension after a pregnancy complicated by HDP is higher in obese women than in nonobese women, we might be able to use this information to motivate these women to maintain an appropriate body weight.
The aim of this study is to clarify whether the association between a history of HDP and hypertension in later life varies by age. We also aimed to determine whether the association with hypertension is equivalent in obese and nonobese women with a history of HDP by conducting an age-group analysis.
Materials and methods
Study design and data source
The Tohoku Medical Megabank Project (TMM) [18], conducted at Tohoku University; TMM Organization; and Iwate Medical University, Iwate TMM Organization in northern Japan, was launched to bring about creative reconstruction and to solve medical problems in the aftermath of the Great East Japan Earthquake of 2011. A population-based cohort study, the TMM CommCohort Study, recruited over 80,000 participants of 20 years of age and older in Miyagi and Iwate Prefectures between May 2013 and March 2016. Participants underwent a health examination and completed a self-administered questionnaire. The health examination included measurements of height, weight, and blood pressure as well as laboratory blood analyses. Information on each individual’s medical history, family history, pregnancy history (e.g., age at first and last delivery, number of children), pregnancy complications such as HDP and gestational diabetes mellitus, and lifestyle factors (e.g., smoking history, alcohol consumption, physical activity) was collected using a self-administered questionnaire.
Study population
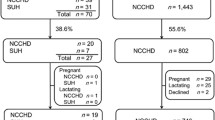
A cross-sectional population-based study was conducted as part of the TMM CommCohort Study. A total of 41,331 women participated in this study. Figure 1 shows the flow chart illustrating participant selection. Men (n = 25,612), individuals with no data on age at delivery (n = 7900), those with an age at delivery ≤9 years old (n = 2), and those with no data on blood pressure (n = 17) were excluded; finally, 33,412 women were included in this analysis.
Measurements
Women who provided an age at first delivery on the questionnaire were regarded as parous, and women who did not answer the question were considered nulliparous. The history of HDP was assessed by questionnaire using the question, “Do you have a history of hypertensive disorders of pregnancy or toxemia?” Height and weight were measured at the time of the health examination, and body mass index (BMI) was calculated as the weight divided by the height squared (kg/m2). Overweight/obesity was defined as a BMI ≥ 25 kg/m2, according to the cutoff point recommended by the World Health Organization [19]. Blood pressure was measured by nurses while the participants were seated in health check-up settings. The values were recorded twice (if required) by an automated blood pressure measuring device. Hypertension was defined as either a systolic blood pressure ≥ 140 mm Hg or a diastolic blood pressure ≥ 90 mm Hg [20] at the time of health examination or current use of medication for hypertension reported on the questionnaire. The questionnaire also asked about a history of hypertension in the parents of the participants. Alcohol consumption status was determined using the questionnaire and was divided into four categories: never, past, current, and unknown use.
Statistical analysis
Study population characteristics were described using the mean and standard deviation for continuous variables and number and frequency for categorical variables. The prevalence of hypertension in women with and without a history of HDP was compared using the chi-square test or Fisher’s exact test in each 10-year age range group (20–29, 30–39, 40–49, 50–59, 60–69, and 70 years of age and older). Multivariate logistic regression analysis was performed, adjusting for age, BMI, family history of hypertension, and alcohol consumption status in each age group, to calculate the adjusted odds ratios (ORs) and 95% confidence intervals of a history of HDP for hypertension in later life. We also examined the interaction between a history of HDP and two age categories (30s–50s and 60s or older) by multivariate logistic regression analysis, adjusting for age, BMI, family history of hypertension, and alcohol intake, to confirm age-related changes in the association between a history of HDP and hypertension. These two age categories were chosen to create a premenopausal and a postmenopausal cohort.
Finally, we constructed a composite variable that combined a history of HDP (±) and overweight–obesity (±), resulting in four categories. The category of nonobese women without a history of HDP was used as a reference, and we calculated the adjusted ORs for hypertension in the other three categories using multivariate logistic regression analysis, adjusting for age, family history of hypertension, and alcohol consumption status, to compare the ORs for hypertension between women with a history of HDP, overweight–obese women, and overweight–obese women with a history of HDP.
All statistical analyses were carried out using SAS, version 9.4 (SAS Inc, Cary, NC). A p value of <0.05 was considered statistically significant.
Results
The baseline patient characteristics are shown in Table 1. The mean age of the study population was 60.3 years (±10.6 years standard deviation). A total of 1585 women (4.7%) had a history of HDP, and 12,524 women (37.5%) were affected by hypertension.
The prevalence and adjusted ORs for hypertension in each age group according to a history of HDP are shown in Table 2. The prevalence of hypertension was significantly higher in women with a history of HDP (814/1585, 51.4%) than in those without (11,710/31,827, 36.8%; p < 0.01). There were no hypertensive women in their 20s. Women in all other age groups (30s–70s) had a significantly higher prevalence of hypertension if a history of HDP was reported (all p < 0.01). The adjusted ORs for hypertension in women with a history of HDP were significantly higher in women in all age groups except for women in their 20s. The adjusted ORs for hypertension in women with a history of HDP tended to be higher in younger age groups; the adjusted OR in women in their 30s–50s was 1.88 (1.57–2.26) and that in women older than 60 was 1.51 (1.32–1.74) (p for interaction = 0.057).
We calculated the adjusted ORs for hypertension in the categories of the combination of HDP history and obesity in all age groups except for the 20s group because there were no hypertensive women in the 20s group, and the results are shown in Table 3. Overweight/obese women with a history of HDP had the highest OR for hypertension in all age groups. Overweight/obese women without a history of HDP had a higher OR for hypertension than nonoverweight/obese women with a history of HDP in all age groups. Among women with a history of HDP, overweight/obese women had a higher OR for hypertension than nonoverweight/obese women in all age groups.
Discussion
This cross-sectional analysis revealed a significant association between a history of HDP and hypertension in later life in all age groups; although the difference was not statistically significant, the adjusted ORs of a history of HDP for hypertension were higher in the 30s–50s group than in the 60s and older group. When we considered overweight/obesity status, participants with normal body weight had a lower risk for hypertension than overweight/obese women, even if they had a history of HDP.
To the best of our knowledge, this is the first study to address age-related changes in the association between HDP history and hypertension. Although the difference was not statistically significant, our subgroup analysis showed that the adjusted ORs of a history of HDP for hypertension were higher in the 30s–50s group than in the 60s and older group. This may be because aging, menopause, and declining estrogen levels play an important role in endothelial dysfunction and oxidative stress, resulting in hypertension in older women [14, 21]. The reported average age of hypertension onset is 7.7 years earlier after a pregnancy complicated by HDP than in a woman without this history [22]. In our study, the prevalence of hypertension in women with a history of HDP was almost 16%, even for women in their 30s. Therefore, continuous follow-up immediately after the postpartum period is important for women with a history of HDP to enable early detection of hypertension. This follow-up could take the form of aperiodic health examination and home blood pressure measurement.
Of course, regular measurement of blood pressure is recommended for all adults aged 20 years or over by the US preventive service task force [23]. We considered that a more intensive recommendation should be required for women with a history of HDP. Heidema et al. compared women with and without a history of HDP in three BMI classes (BMI 19.5–24.9, 25.0–29.9, 30.0 kg/m2) 12 months after delivery and concluded that HDP is a more important risk factor for hypertension and cardiac syndrome than obesity [24]. However, their study was limited by the fact that they evaluated women immediately after delivery, and pregnancy-related metabolic abnormalities may not yet have resolved. Our study included women in a wide range of age groups, and we found that obesity has a stronger association with hypertension than a history of HDP in all age groups. A previous literature review suggests that lifestyle interventions after a pregnancy complicated by HDP are effective at decreasing cardiovascular risk [25]. Therefore, even after a pregnancy complicated by HDP, weight control might be an effective method of reducing the risk for hypertension. In our study, the prevalence of hypertension in participants with a history of HDP without obesity was lower than that in those with obesity who did not have a history of HDP.
The underlying mechanisms of HDP are reduced placental perfusion secondary to abnormal implantation and subsequent reduced placental vascularization. The exacerbation of hypoxia–ischemia may induce the overproduction of antiangiogenic factors and result in vascular endothelial dysfunction [26]. Several possibilities have been proposed for the mechanisms underlying the association between HDP and future hypertension [27, 28]. One theory is the “failed stress test.” Hypertension and HDP share several common risk factors, such as obesity and diabetes mellitus [29], which may lead to both HDP and hypertension in later life. Another explanation is that HDP itself may induce irreversible metabolic and vascular abnormalities that contribute to the risk of developing hypertension and cardiovascular disease later in life [30]. Because of the overlapping and complex pathways, the precise causal relationship between HDP and hypertension in later life remains incompletely understood, and it is unclear which is the greater risk factor for future hypertension: HDP itself or the underlying metabolic conditions. Romundstad et al. reported that the positive association between preeclampsia and post pregnancy cardiovascular risk factors is attenuated after adjustment for prepregnancy BMI and high-density cholesterol and triglyceride levels, and they suggest that post pregnancy cardiovascular risk factors are mainly shared prepregnancy risk factors rather than the HDP state itself [31]. In our study, obesity had a stronger association with hypertension than a history of HDP; this finding may support the hypothesis that common risk factors for HDP and hypertension have a greater effect on hypertension in later life than HDP itself.
Our study had several strengths. Since a wide range of age groups is represented in our study, we were able to conduct age-group analysis and assess the change in the association of HDP, obesity, and hypertension with age. As this population-based study had a large sample size, we could assess prevalence and associations in the general study population.
This study has several limitations. First, the history of HDP was obtained using a questionnaire. Agreement between a history of HDP by self-reporting vs. medical records has been assessed in several studies, and the sensitivity and specificity are 60–70% and >90%, respectively [32,33,34,35]. The sensitivity of recall is relatively low, and the effect of our method of gathering information could lead to an underestimation of HDP. The false negative rate was reported to be higher in older age groups [35], and this might result in an underestimation of the effect of HDP, especially in older age groups. Furthermore, the diagnostic criteria of HDP changed over time, and it was also possible that the diagnosis was not adequately communicated to the women by the health care personnel in the past, which might affect the recall rate of HDP in each age group. Second, we had no information about the severity or onset of HDP. Early onset HDP is often considered to be a disease of placental dysfunction, whereas late-onset HDP is proposed to be caused primarily by maternal factors [36]. The influences of early and late-onset HDP on hypertension in later life may therefore be caused by different mechanisms. Further study is needed to clarify the influence of early and late-onset HDP on hypertension. Finally, we did not mention any other chronic conditions. Although we focused on the relationship between a history of HDP and hypertension later in life, HDP will contribute to other conditions related to hypertension and obesity later in life. Further study is required to clarify the relationship between HDP and these conditions, such as diabetes, chronic kidney disease, and metabolic syndrome.
In conclusion, our findings suggest that there was a significant association between a history of HDP and hypertension in women in the 30s–70s age group. Therefore, physicians and other health care providers should recommend regular health examinations and home blood pressure monitoring after the postpartum period. Our study also demonstrated that women with normal body weight had a lower risk for hypertension than overweight–obese women, even if they had a history of HDP. Therefore, even after a pregnancy complicated by HDP, weight control may reduce the risk for future hypertension. Although pregnancy represents a chance to predict future cardiovascular disease risk, the best method of reducing the risk for hypertension in women after a pregnancy complicated by HDP has not been well discussed. Further study is needed to evaluate the preventive effect of lifestyle interventions, such as exercise and dietary habits, on hypertension in women with a history of HDP.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Mills KT, Bundy JD, Kelly TN, Reed JE, Kearney PM, Reynolds K, et al. Global disparities of hypertension prevalence and control: a systematic analysis of population-based studies from 90 countries. Circulation. 2016;134:441–50.
GBD 2015 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388:1659–724.
Rapsomaniki E, Timmis A, George J, Pujades-Rodriguez M, Shah AD, Denaxas S, et al. Blood pressure and incidence of twelve cardiovascular diseases: lifetime risks, healthy life-years lost, and age-specific associations in 1.25 million people. Lancet. 2014;383:1899–911.
Tozawa M, Iseki K, Iseki C, Kinjo K, Ikemiya Y, Takishita S. Blood pressure predicts risk of developing end-stage renal disease in men and women. Hypertension. 2003;41:1341–5.
Gorelick PB, Scuteri A, Black SE, Decarli C, Greenberg SM, Iadecola C, et al. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the american heart association/american stroke association. Stroke. 2011;42:2672–713.
Hozawa A, Okamura T, Murakami Y, Kadowaki T, Okuda N, Takashima N, et al. High blood pressure in middle age is associated with a future decline in activities of daily living. NIPPON DATA80. J Hum Hypertens. 2009;23:546–52.
Benjamin EJ, Blaha MJ, Chiuve SE, Cushman M, Das SR, Deo R, et al. Heart disease and stroke statistics-2017 update: a report from the American Heart Association. Circulation. 2017;135:e146–e603.
NCD-RisC. Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet. 2016;387:1377–96.
Yatsuya H, Li Y, Hilawe EH, Ota A, Wang C, Chiang C, et al. Global trend in overweight and obesity and its association with cardiovascular disease incidence. Circ J. 2014;78:2807–18.
Wakabayashi I. Stronger associations of obesity with prehypertension and hypertension in young women than in young men. J Hypertens. 2012;30:1423–9.
Fujita M, Hata A. Sex and age differences in the effect of obesity on incidence of hypertension in the Japanese population: a large historical cohort study. J Am Soc Hypertens. 2014;8:64–70.
Drost JT, Maas AH, van Eyck J, van der Schouw YT. Preeclampsia as a female-specific risk factor for chronic hypertension. Maturitas. 2010;67:321–6.
Mito A, Arata N, Qiu D, Sakamoto N, Murashima A, Ichihara A, et al. Hypertensive disorders of pregnancy: a strong risk factor for subsequent hypertension 5 years after delivery. Hypertens Res. 2018;41:141–6.
Coylewright M, Reckelhoff JF, Ouyang P. Menopause and hypertension: an age-old debate. Hypertension. 2008;51:952–9.
Brown MC, Best KE, Pearce MS, Waugh J, Robson SC, Bell R. Cardiovascular disease risk in women with pre-eclampsia: systematic review and meta-analysis. Eur J Epidemiol. 2013;28:1–19.
Bellamy L, Casas JP, Hingorani AD, Williams DJ. Pre-eclampsia and risk of cardiovascular disease and cancer in later life: systematic review and meta-analysis. BMJ. 2007;335:974.
Cirillo PM, Cohn BA. Pregnancy complications and cardiovascular disease death: 50-year follow-up of the child health and development studies pregnancy cohort. Circulation. 2015;132:1234–42.
Kuriyama S, Yaegashi N, Nagami F, Arai T, Kawaguchi Y, Osumi N, et al. The tohoku medical megabank project: design and mission. J Epidemiol. 2016;26:493–511.
WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–63.
Whitworth JA, World Health Organization, ISoHWG. 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J Hypertens. 2003;21:1983–92.
Zhou Y, Zhou X, Guo X, Sun G, Li Z, Zheng L, et al. Prevalence and risk factors of hypertension among pre- and post-menopausal women: a cross-sectional study in a rural area of northeast China. Maturitas. 2015;80:282–7.
Heida KY, Franx A, van Rijn BB, Eijkemans MJ, Boer JM, Verschuren MW, et al. Earlier age of onset of chronic hypertension and type 2 diabetes mellitus after a hypertensive disorder of pregnancy or gestational diabetes mellitus. Hypertension. 2015;66:1116–22.
Siu AL, US Preventive Services Task Force. Screening for high blood pressure in adults: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015;163:778–86.
Heidema WM, Scholten RR, Lotgering FK, Spaanderman ME. History of preeclampsia is more predictive of cardiometabolic and cardiovascular risk factors than obesity. Eur J Obstet Gynecol Reprod Biol. 2015;194:189–93.
Berks D, Hoedjes M, Raat H, Duvekot JJ, Steegers EA, Habbema JD. Risk of cardiovascular disease after pre-eclampsia and the effect of lifestyle interventions: a literature-based study. BJOG. 2013;120:924–31.
Seki H. Balance of antiangiogenic and angiogenic factors in the context of the etiology of preeclampsia. Acta Obstet Gynecol Scand. 2014;93:959–64.
Garovic VD, August P. Preeclampsia and the future risk of hypertension: the pregnant evidence. Curr Hypertens Rep. 2013;15:114–21.
Scantlebury DC, Hayes SN. How does preeclampsia predispose to future cardiovascular disease? Curr Hypertens Rep. 2014;16:472.
Egeland GM, Klungsoyr K, Oyen N, Tell GS, Naess O, Skjaerven R. Preconception cardiovascular risk factor differences between gestational hypertension and preeclampsia: cohort norway study. Hypertension. 2016;67:1173–80.
Melchiorre K, Sutherland GR, Liberati M, Thilaganathan B. Preeclampsia is associated with persistent postpartum cardiovascular impairment. Hypertension. 2011;58:709–15.
Romundstad PR, Magnussen EB, Smith GD, Vatten LJ. Hypertension in pregnancy and later cardiovascular risk: common antecedents? Circulation. 2010;122:579–84.
Coolman M, de Groot CJ, Jaddoe VW, Hofman A, Raat H, Steegers EA. Medical record validation of maternally reported history of preeclampsia. J Clin Epidemiol. 2010;63:932–7.
Klemmensen AK, Olsen SF, Osterdal ML, Tabor A. Validity of preeclampsia-related diagnoses recorded in a national hospital registry and in a postpartum interview of the women. Am J Epidemiol. 2007;166:117–24.
Gresham E, Forder P, Chojenta CL, Byles JE, Loxton DJ, Hure AJ. Agreement between self-reported perinatal outcomes and administrative data in New South Wales, Australia. BMC Pregnancy Childbirth. 2015;15:161.
Falkegard M, Schirmer H, Lochen ML, Oian P, Acharya G. The validity of self-reported information about hypertensive disorders of pregnancy in a population-based survey: the Tromso Study. Acta Obstet Gynecol Scand. 2015;94:28–34.
Craici IM, Wagner SJ, Weissgerber TL, Grande JP, Garovic VD. Advances in the pathophysiology of pre-eclampsia and related podocyte injury. Kidney Int. 2014;86:275–85.
Acknowledgements
The authors would like to thank all participants and all the staff in Tohoku Medical Megabank Organization, Tohoku University (a full list of members is available at http://www.megabank.tohoku.ac.jp/english/a151201/) and Iwate Tohoku Medical Megabank Organization.
Funding
This work was supported in part by the Tohoku Medical Megabank Project from the Japan Agency for Medical Research and Development (AMED) and the Ministry of Education, Culture, Sports, Science and Technology (MEXT).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was conducted in accordance with the Declaration of Helsinki. Approval was obtained from the TMM Organization Institutional Review Board (2016-4-054) and the Iwate TMM Organization Institutional Review Board (HG H25-2).
Informed consent
All participants gave written informed consent for study inclusion.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wagata, M., Kogure, M., Nakaya, N. et al. Hypertensive disorders of pregnancy, obesity, and hypertension in later life by age group: a cross-sectional analysis. Hypertens Res 43, 1277–1283 (2020). https://doi.org/10.1038/s41440-020-0463-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-020-0463-8
- Springer Nature Singapore Pte Ltd.
Key words
This article is cited by
-
Association of physiological factors with grip and leg extension strength: tohoku medical megabank community-based cohort study
BMC Public Health (2024)
-
Relationship between parity and the prevalence of chronic kidney disease in Japan considering hypertensive disorders of pregnancy and body mass index
BMC Nephrology (2024)
-
The association of breast feeding for at least six months with hemodynamic and metabolic health of women and their children aged three years: an observational cohort study
International Breastfeeding Journal (2023)
-
Preeclampsia up to date—What’s going on?
Hypertension Research (2023)
-
Annual reports on hypertension research 2020
Hypertension Research (2022)