Abstract
Oxidative stress is implicated in hypertension, carotid wall thickening, and renal dysfunction. Oxidative stress is linked to cardiovascular pathology in the black South African individuals who have a high prevalence of hypertension and early vascular aging. However, there are limited data relating changes in oxidative stress with vascular and renal deterioration over time. We aimed to investigate whether changes in oxidative stress over 3 years are associated with target organ damage in black (N = 89) and white (N = 91) men. Carotid intima-media thickness was measured using the SonoSite Micromaxx ultrasound system, and cross-sectional wall area (CSWA) was calculated. The estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease formula. The percentage change (%∆) in oxidative stress markers was calculated and included reactive oxygen species (ROS), superoxide dismutase (SOD), glutathione peroxidase (GPx), and glutathione reductase (GR). Over 3 years, black men exhibited decreased ROS, SOD, and GR, while white men revealed decreased SOD and GPx. Black men displayed positive associations of CSWA with %∆ ROS (β = 0.28; p = 0.017) and %∆ SOD (β = 0.24; p = 0.047). White men displayed a negative association of CSWA with %∆ SOD (β = −0.22; p = 0.042) and positive associations of eGFR with %∆ GPx (β = 0.33; p = 0.001) and %∆ GR (β = 0.39; p < 0.001). In white men, the association of CSWA with decreased SOD activity suggests oxidative-stress-related carotid remodeling, while associations of eGFR with the glutathione system suggests a postponement of microvascular deterioration. In black men, associations of oxidative stress markers with CSWA suggest that a sufficiently functioning antioxidant system may delay target organ damage.
Similar content being viewed by others
Introduction
Cardiovascular disease is one of the leading risk factors for mortality both globally and in developing countries [1,2,3,4]. In South Africa [5, 6], especially among the urban black population, the prevalence of hypertension is a mounting concern [7,8,9]. Oxidative stress is implicated in the development of hypertension [10, 11], and associations of oxidative stress and blood pressure have already been confirmed in black South Africans [12, 13]. Under normal physiological conditions, reactive oxygen species (ROS) act as signaling molecules that maintain endothelial function and vascular tone [14, 15]. However, excessive ROS production or ineffective functioning of the antioxidant system enhances inflammation, vascular remodeling, and endothelial dysfunction, all of which may lead to cardiovascular disease and related comorbidities, such as renal disease, especially with advanced age [14, 16,17,18]. Previous results within the South African population have linked oxidative stress with pulse pressure (PP) (as an indicator of early vascular changes) [19], arterial stiffness [12, 20], and carotid wall thickening [13, 21]. However, it is not clear whether changes in oxidative stress over time are associated with target organ damage, such as the deterioration of vascular structure and renal function. We aimed to investigate the relationship between changes in markers of oxidative stress over 3 years with measures of target organ damage (glomerular filtration rate and carotid wall thickening) in a black and white South African cohort.
Methods
Study population
This study was embedded in the Sympathetic activity and Ambulatory Blood Pressure in Africans (SABPA) study. This was a prospective cohort study of black and white teachers with similar socioeconomic statuses from the Dr Kenneth Kuanda Education District in the North-West Province of South Africa. Details on the study population and protocol have been reported elsewhere [22]. In summary, participants were between the ages of 20 and 65 years at baseline, and participants who were pregnant or lactating, exhibited elevated ear temperature (>37 °C), used alpha or beta blockers, had confirmed psychotropic substance abuse or had a history of blood donation or vaccination in the 3 months prior to the commencement of the study were excluded. Phase I (baseline) of the SABPA study was conducted between 2008 and 2009, while phase II (follow-up) was conducted between 2011 and 2012. The protocols of Phase I and Phase II were designed to be similar. A 3-year successful follow-up rate of 87.8% provided a sample of 359 participants in the follow-up phase that were subsequently divided into black men (N = 89), white men (N = 91), black women (N = 84), and white women (N = 95).
The SABPA study adhered to all the requirements of the Declaration of Helsinki regarding investigations of human participants and was further approved by the Health Research Ethics Committee of North-West University (NWU-00036-07-S6). Participants were informed of the study in their native language both verbally and in a written manner. Written informed consent was acquired before the commencement of the study.
Questionnaires
A general health questionnaire was completed by each participant with the help of a researcher to obtain demographic data and information on behavioral risk factors, such as alcohol use.
Anthropometric and physical activity measurements
All anthropometric measurements were performed using standardized methods after calibration [23], and these measurements included body height (Invicta Stadiometer, IP 1465, Invicta, London, UK), body weight (Precision Health Scale, A&D Company, Tokyo, Japan), and waist circumference (Holtain unstretchable flexible 7-mm-wide metal tape, Crosswell, Wales). Body mass index (BMI) was calculated as kg/m2. Each participant was equipped with an Actical® omnidirectional accelerometer (Actical® activity monitor, Mini Mitter Co., Inc., Bend, OR; Montreal, Quebec, Canada) for 24 h during the baseline phase and an ActiHeart physical activity monitor (CamNtech Ltd, England, UK) for 7 days during the follow-up phase to monitor total energy expenditure (TEE).
Cardiovascular measurements
Ambulatory blood pressure measurements were taken for 24 h at 30-min intervals during the day (07:00–22:00) and at 1-h intervals during the night (22:00–06:00) using the Cardiotens device (CE0120, Meditech, Budapest, Hungary). Ambulatory measurements included systolic blood pressure (24-h SBP), diastolic blood pressure (24-h DBP), pulse pressure (24-h PP), and mean arterial pressure (24-h MAP). The device was fitted with a correctly sized cuff on the nondominant arm, and participants continued with their normal daily activities while reporting any abnormalities (such as headaches, nausea, or stress) on their ambulatory diary cards.
The carotid intima-media thickness (cIMT) of the left common carotid artery was measured using the B-mode ultrasonography with the high resolution SonoSite Micromaxx ultrasound system (SonoSite Inc., Bothell, WA, USA) and a 6–13 MHz linear array transducer according to the Mannheim Consensus [24]. Still-frame images from at least two optimal angles with the clearest viewing of the left and right common carotid arteries were obtained at maximal dilation (as determined by a three-lead ECG recording). The results were interpreted with a semiautomated program, namely, the Artery Measurement Systems (AMS) II v1.139 (Gothenburg, Sweden), to obtain a maximal 10 mm segment with good image quality proximal to the carotid bifurcation for use in offline analyses. The program automatically identifies the borders from the trailing edge of the media-adventitia at the proximal wall to the leading edge of the distal wall media-adventitia and calculates the cIMT and diameter using ~100 measurements within the chosen segment. The same optimal angles were used in both the baseline and follow-up phases, and in this study, the distal wall measurements of cIMT were used. The analysis of cIMT was performed by a single observer, and the intraobserver variability was 0.04 mm between the two measurements made 4 weeks apart (N = 10). Carotid cross-sectional wall area (CSWA) was calculated to confirm structural changes in luminal diameter using the formula CSWA = π(d/2 + cIMT)2 – π(d/2)2, where d denotes luminal diameter [25, 26].
Biochemical measurements
A fasting blood sample was obtained from each participant by a registered nurse from the antebrachial vein branches using a winged infusion set and syringes.
Serum and plasma were prepared using the standardized procedures, after which they were immediately aliquoted into cryovials in the on-site laboratory and stored in biofreezers at −80 °C until analysis to maintain efficient long-term stability of antioxidant enzymes [27]. In both phases of the SABPA study, antioxidant enzyme activities, including glutathione peroxidase (GPx), glutathione reductase (GR), and superoxide dismutase (SOD), were measured using assay kits (Cayman Chemical Company, Ann Arbor, MI, USA) and a Synergy H4 hybrid microplate reader (BioTek, Winooski, VT, USA). We measured γ-glutamyl transferase (γ-GT) with the Unicel DXC 800 (Beckman and Coulter, Germany) and a Konelab™ 20I Sequential Multiple Analyzer Computer (Thermo Scientific, Vantaa, Finland) during baseline and the Cobas Integra 400 plus (Roche, Basel, Switzerland) during follow-up. Additional oxidative stress markers measured during both phases of the study include total glutathione (tGSH) (Kit: BIOXYTECH GSH/GSSG-412, Oxis International Inc, CA, USA) and ROS (Synergy HT microplate reader, BioTek), which are measured as serum peroxides and reported in units, where 1 mg H2O2/L is equivalent to 1 unit [28].
Serum total cholesterol and triglycerides were measured with the Unicel DXC 800 (Beckman and Coulter) and a Konelab™ 20I Sequential Multiple Analyzer Computer (Thermo Scientific) in the baseline phase and the Cobas Integra 400 plus (Roche) in the follow-up phase. Serum high-density lipoprotein cholesterol (HDL) and fluoride plasma glucose levels were measured using the Unicel DXC 800 (Beckman and Coulter) in the baseline phase and the Cobas Integra 400 plus (Roche) in the follow-up phase. Interleukin-6 (IL-6) was measured using a high-sensitivity Quantikine ELISA kit (R&D systems, Minneapolis, MN, USA) analyzed on a Synergy H4 hybrid microplate reader (BioTek). An immunoassay (Automated Modular, Roche) was used to determine serum cotinine levels. Serum creatinine was measured with the Unicel DXC 800 (Beckman and Coulter) and a Konelab™ 20I Sequential Multiple Analyzer Computer (Thermo Scientific) in the baseline phase and with an enzymatic colorimetric test on the Cobas Integra 400 plus (Roche) in the follow-up phase. Serum creatinine was used for the calculation of the estimated glomerular filtration rate (eGFR) according to the Modification of Diet in Renal Disease formula [29].
Statistical analyses
Statistica version 13.2 (Dell, TX, USA) was used to perform the statistical analyses in this study. The normal distribution of data was analyzed based on visual inspection of the symmetry of the bell-shaped histogram curves. The central tendency and spread of normally distributed variables are expressed as arithmetic mean and standard deviation, while skewed variables were logarithmically transformed (glucose, triglycerides, IL-6, GR, SOD, and γ-GT). The central tendency and spread of logarithmically transformed variables are expressed as the median and the interquartile range. Changes in oxidative stress markers were calculated as the percentage change (% ∆) from baseline to follow-up phase. Interactions of race and sex were tested in the relationships between the main dependent variables (blood pressure, macrovascular, and microvascular measurements) and oxidative stress markers using multiple regression analyses. Continuous variables were compared using T-tests of dependent samples (repeated measures T-tests). Single regression analyses were performed to evaluate unadjusted associations between follow-up cardiovascular variables and % ∆ in ROS, tGSH, GPx, GR, SOD, and γ-GT from baseline to follow-up. Partial correlations were assessed to evaluate the associations between variables while adjusting for % ∆ in BMI. Associations of cIMT and CSWA were additionally adjusted for % ∆ in 24-h SBP. Multiple regression analyses were performed to evaluate the independent associations between follow-up cardiovascular variables and 3-year changes in oxidative stress markers in black and white men and women. Covariates entered into each model included % ∆ in the oxidative stress marker, the follow-up cardiovascular variable, % ∆ in age, % ∆ in BMI, % ∆ in TEE, % ∆ in cotinine, % ∆ in glucose, % ∆ in triglycerides, % ∆ in IL-6, and change in self-reported alcohol use. Changes in alcohol use were coded no change in behavior, started using alcohol or stopped using alcohol between baseline and the follow-up phase. Measurements of cIMT and CSWA were additionally adjusted for % ∆ in 24-h SBP. In post hoc analyses, the achieved power (1−β) was determined in a cohort with a sample size of N = 89 for black men and N = 91 for white men using multiple regression models with either 9 (both 1− β = 0.98) or 10 (both 1− β = 0.98) covariates (as in the case of CSWA) (G*power v3.1.9.2) [30].
Results
Characteristics of the study population
Interactions of race were found on the association of CSWA with SOD (β = −0.884; p < 0.001), DBP with GR (β = 0.587; p = 0.030), and MAP with GR (β = 0.613; p = 0.023). In accordance with the interactions found, as well as the aims of this study and previous results within the South African population, this study was stratified by both race and sex [12, 13]. Analyses among women revealed similar characteristics as those observed in male cohorts; however, no significant associations between follow-up cardiovascular variables and 3-year changes in oxidative stress markers were found. (Supplementary Tables 1 and 2)
Changes in characteristics of the black and white men from baseline to follow-up are summarized in Table 1. Both black and white men experienced significant increases in BMI (p < 0.001), while black men displayed increases in IL-6 (p < 0.001), and TEE (p < 0.001). White men displayed a decline in lipid markers from baseline to follow-up, including HDL cholesterol (p < 0.001), total cholesterol (p < 0.001), and triglycerides (p = 0.018), while black men also showed a decrease in HDL cholesterol over 3 years (p < 0.001).
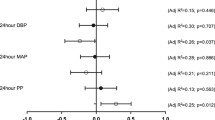
Changes in vascular structure and renal function markers over the 3-year study period in men are illustrated in Fig. 1. Increases in cIMT and CSWA (all p < 0.001) were noted in white men, while eGFR increased in black men (p < 0.001).
Black men revealed decreases in ROS (p < 0.001), tGSH (p < 0.001), GR activity (p < 0.001), SOD activity (p = 0.018), and γ-GT activity (p = 0.003) over 3 years. In white men, tGSH (p < 0.001) increased, while GPx activity and SOD activity (both p < 0.001) decreased from baseline to follow-up (Fig. 2).
Regression analyses
Single regression analyses (Fig. 3) revealed positive associations of cIMT and CSWA with changes in ROS (CSWA: r = 0.24, p = 0.028) and SOD (borderline) (cIMT: r = 0.20, p = 0.076; CSWA: r = 0.20, p = 0.068) in black men. On the other hand, white men displayed negative associations of cIMT and CSWA with changes in ROS (cIMT: r = −0.21, p = 0.048; CSWA (borderline): r = −0.19, p = 0.077). In white men, these associations were not significant (cIMT: r = −0.12, p = 0.299; CSWA: r = −0.13, p = 0.228). However, upon full adjustments in multiple regression analyses, the association of CSWA and change in SOD reached significance (R2 = 0.22; β = −0.22; p = 0.042).
Single (Supplementary Table 3), partial (Table 2), and multiple regression analyses (Table 3) revealed positive associations of CSWA with changes in ROS (R2 = 0.04; β = 0.28; p = 0.017) and SOD activity (R2 = 0.01; β = 0.24; p = 0.047) in black men. The negative association of CSWA with a change in SOD activity (R2 = 0.22; β = −0.22; p = 0.042) became significant in white men after full adjustments were made.
In white men, positive associations of eGFR were found in single, partial and multiple regression analyses with changes in GPx activity (R2 = 0.21; β = 0.33; p = 0.001) and changes in GR activity (R2 = 0.24; β = 0.39; p < 0.001) (Table 4).
Positive associations of 24-h DBP (R2 = 0.07; β = 0.32; p = 0.006) and 24-h MAP (R2 = 0.06; β = 0.31; p = 0.007) with increased GR activity in white men were confirmed to be independent of various confounders (results not shown). All other associations indicated in single and partial regression analyses were no longer significant once full adjustments were made.
Sensitivity analyses
Multiple regression analyses of cIMT and CSWA were repeated after adding % ∆ HDL cholesterol and total cholesterol separately as additional covariates into the models. Similar results were obtained in sensitivity analyses, and the additional lipid biomarkers did not significantly contribute to these models.
Discussion
We aimed to investigate whether changes in markers of oxidative stress over 3 years are associated with follow-up measures of target organ damage in black and white South African participants.
In both black and white men, SOD activity (along with ROS in black men) decreased from baseline to follow-up, while CSWA remained similar in black men but increased in white men over time. Despite these similarities in oxidative stress profiles, CSWA was positively related to changes in ROS and SOD in black men, whereas an inverse association of CSWA with changes in SOD was evident in white men. The opposing results of CSWA with decreased SOD activity in the black and white groups of this study are surprising. Superoxide plays an important role in maintaining optimal nitric oxide availability, and in turn, nitric oxide plays an important role within the vascular system to maintain vascular tone and to prevent the development of atherosclerosis [10, 31, 32]. Although there was a decrease in SOD in both groups, the decrease in the black group may not be enough to affect CSWA. However, in the white group, SOD activity may have decreased to such an extent that it resulted in insufficient inactivation of superoxide and lower NO availability and hence may, in part, explain the association with vascular deterioration (increased CSWA over time) [33, 34]. While diminished SOD activity may play a role in carotid wall thickening, previous results in men have found that serum SOD activity increases in direct relation to the pathological progression of plaque formation as a marker of heightened oxidative stress in the carotid artery [35]. This suggests a need for SOD activity to be highly adaptable to superoxide levels to prevent vascular remodeling.
Despite no change in eGFR, a measure of renal microvascular health, positive associations of eGFR with decreased GPx and GR activity were found in white men only. Studies have confirmed an inverse relationship between oxidative stress and eGFR, as shown by increased oxidative stress markers and decreased antioxidant enzyme activity with advancing stages of chronic kidney disease [18, 36,37,38,39]. However, eGFR was maintained in white men over time despite diminished GPx activity. This may suggest that this group may currently be in a stable physiological state, but positive associations of markers of the glutathione system with eGFR in white men may suggest that further fluctuations in the glutathione system may negatively impact renal function in this group. Interestingly, the black men displayed a mean eGFR above 150 ml/min/1.73 m2 in the follow-up phase [36, 40, 41], which is a well-documented predictor of glomerular hypertension and albuminuria and has also been shown to occur during states of oxidative stress [40, 42, 43]. However, the lack of associations between renal function and oxidative stress in the black men in our study uncovered the possible role of alternative confounding factors, such as early vascular aging, in the deterioration of renal microvascular function in this group [44].
This study must be interpreted within the context of its strengths and limitations. Our study population was based on participants from the Potchefstroom area in the North-West Province of South Africa, and it may not be representative of the population as a whole. Despite the relatively small sample size, the achieved power was 98%, which was sufficient to test these hypotheses. However, the link between changes in oxidative stress and cardiovascular variables needs to be investigated in larger cohorts. While this was a prospective study, our results are based on associations, and causality cannot be inferred. Although our results were consistent after multiple adjustments, we cannot exclude any unknown interactions that may play a role in the development of deteriorated cardiovascular function and structure.
However, to the best of our knowledge, this is the first study investigating the associations of changes in oxidative stress with cardiovascular variables in black and white cohorts over a 3-year period. Our study was well planned and executed under strict conditions in a fully equipped research facility, allowing us to explore in depth the possible mechanisms involving oxidative stress in the promotion of target organ damage over time.
In conclusion, the inverse association of CSWA with SOD activity in white men highlights the atherogenic effects of oxidative stress in the macrovasculature. Associations of CSWA with ROS and SOD activity in black men and associations of eGFR with the glutathione system in white men may suggest a role of adequate antioxidant enzyme activity in delaying target organ damage in these groups.
References
Lim SS, Vos T, Flaxman AD, Danaei G, Shibuya K, Adair-Rohani H, et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2224–60.
Campbell NR, Bovet P, Schutte AE, Lemogoum D, Nkwescheu AS. High blood pressure in sub‐Saharan Africa: why prevention, detection, and control are urgent and important. J Clin Hypertens. 2015;17:663–7.
Roth GA, Johnson C, Abajobir A, Abd-Allah F, Abera SF, Abyu G, et al. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J Am Coll Cardiol. 2017;70:1–25.
Frieden TR, Jaffe MG. Saving 100 million lives by improving global treatment of hypertension and reducing cardiovascular disease risk factors. J Clin Hyperten. 2018;20:208–11.
Keates AK, Mocumbi AO, Ntsekhe M, Sliwa K, Stewart S. Cardiovascular disease in Africa: epidemiological profile and challenges. Nat Rev Cardiol. 2017;14:273.
Ogah OS, Rayner BL. Recent advances in hypertension in sub-Saharan Africa. Heart. 2013;99:1390–7.
Addo J, Smeeth L, Leon DA. Hypertension in sub-Saharan Africa: a systematic review. Hypertension. 2007;50:1012–8.
Kandala N, Tigbe W, Manda SO, Stranges S. Geographic variation of hypertension in sub-Saharan Africa: a case study of South Africa. Am J Hypertens. 2013;26:382–91.
Schutte AE, Schutte R, Huisman HW, van Rooyen JM, Fourie CM, Malan NT, et al. Are behavioural risk factors to be blamed for the conversion from optimal blood pressure to hypertensive status in Black South Africans? A 5-year prospective study. Int J Epidemiol. 2012;41:1114–23.
Montezano AC, Dulak-Lis M, Tsiropoulou S, Harvey A, Briones AM, Touyz RM. Oxidative stress and human hypertension: vascular mechanisms, biomarkers, and novel therapies. Can J Cardiol. 2015;31:631–41.
Briones AM, Touyz RM. Oxidative stress and hypertension: current concepts. Curr Hypertens Rep. 2010;12:135–42.
Kruger R, Schutte R, Huisman HW, Van Rooyen JM, Malan NT, Fourie CMT, et al. Associations between reactive oxygen species, blood pressure and arterial stiffness in black South Africans: the SABPA study. J Hum Hypertens. 2012;26:91.
van Zyl C, Huisman HW, Mels CM. Antioxidant enzyme activity is associated with blood pressure and carotid intima media thickness in black men and women: the SABPA study. Atherosclerosis. 2016;248:91–6.
Touyz RM, Briones AM. Reactive oxygen species and vascular biology: implications in human hypertension. Hypertens Res. 2011;34:5.
Brown DI, Griendling KK. Regulation of signal transduction by reactive oxygen species in the cardiovascular system. Circ Res. 2015;116:531–49.
Nakano Y, Nakatani Y, Takami M, Taniyama Y, Arima S. Diverse associations between oxidative stress and thromboxane A 2 in hypertensive glomerular injury. Hypertens Res. 2019;42:450.
Higashi Y, Kihara Y, Noma K. Endothelial dysfunction and hypertension in aging. Hypertens Res. 2012;35:1039.
Cachofeiro V, Goicochea M, De Vinuesa SG, Oubiña P, Lahera V, Luño J. Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease: New strategies to prevent cardiovascular risk in chronic kidney disease. Kidney Int. 2008;74:S4–S9.
Myburgh C, Huisman HW, Mels CM. The relation of blood pressure and carotid intima-media thickness with the glutathione cycle in a young bi-ethnic population: the African-PREDICT study. J Hum Hypertens. 2018;32:268.
Mokhaneli MC, Fourie CMT, Botha S, Mels CMC. The association of oxidative stress with arterial compliance and vascular resistance in a bi-ethnic population: the SABPA study. Free Radic Res. 2016;50:920–8.
Schutte R, Schutte AE, Huisman HW, Van Rooyen JM, Malan NT, Péter S, et al. Blood glutathione and subclinical atherosclerosis in African men: the SABPA Study. Am J Hypertens. 2009;22:1154–9.
Malan L, Hamer M, Frasure-Smith N, Steyn HS, Malan NT. Cohort profile: sympathetic activity and ambulatory blood pressure in Africans (SABPA) prospective cohort study. Int J Epidemiol. 2014;44:1814–22.
Marfell-Jones MJ, Stewart A, de Ridder J. International standards for anthropometric assessment. Wellington, New Zealand: International Society for the Advancement of Kinanthropometry; 2012.
Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N, et al. Mannheim carotid intima-media thickness and plaque consensus (2004–2006–2011). An update on behalf of the advisory board of the 3rd, 4th and 5th watching the risk symposia, at the 13th, 15th and 20th European Stroke Conferences, Mannheim, Germany, 2004, Brussels, Belgium, 2006, and Hamburg, Germany, 2011. Cerebrovasc Dis. 2012;34:290–6.
Roman MJ, Pickering TG, Schwartz JE, Pini R, Devereux RB. Relation of arterial structure and function to left ventricular geometric patterns in hypertensive adults. J Am Coll Cardiol. 1996;28:751–6.
Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D, et al. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 2006;27:2588–605.
Abiaka C, Al-Awadi F, Olusi S. Effect of prolonged storage on the activities of superoxide dismutase, glutathione reductase, and glutathione peroxidase. Clin Chem. 2000;46:560–76.
Hayashi I, Morishita Y, Imai K, Nakamura M, Nakachi K, Hayashi T. High-throughput spectrophotometric assay of reactive oxygen species in serum. Mutat Res Genet Toxicol Environ Mutagen. 2007;631:55–61.
Levey AS, Coresh J, Greene T, Stevens LA, Zhang YL, Hendriksen S, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247–54.
Faul F, Erdfelder E, Lang A, Buchner A. G* Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods. 2007;39:175–91.
Li H, Horke S, Förstermann U. Vascular oxidative stress, nitric oxide and atherosclerosis. Atherosclerosis. 2014;237:208–19.
Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O. Oxidative stress and antioxidant defense. World Allergy Organ J. 2012;5:9.
Taniyama Y, Griendling KK. Reactive oxygen species in the vasculature: molecular and cellular mechanisms. Hypertension. 2003;42:1075–81.
Goyal T, Mitra S, Khaidakov M, Wang X, Singla S, Ding Z, et al. Current concepts of the role of oxidized LDL receptors in atherosclerosis. Curr Atheroscler Rep. 2012;14:150–9.
Isogawa A, Yamakado M, Yano M, Shiba T. Serum superoxide dismutase activity correlates with the components of metabolic syndrome or carotid artery intima-media thickness. Diabetes Res Clin Pract. 2009;86:213–8.
Dounousi E, Papavasiliou E, Makedou A, Ioannou K, Katopodis KP, Tselepis A, et al. Oxidative stress is progressively enhanced with advancing stages of CKD. Am J Kidney Dis. 2006;48:752–60.
Karamouzis I, Sarafidis PA, Karamouzis M, Iliadis S, Haidich AB, Sioulis A, et al. Increase in oxidative stress but not in antioxidant capacity with advancing stages of chronic kidney disease. Am J Nephrol. 2008;28:397–404.
Terawaki H, Yoshimura K, Hasegawa T, Matsuyama Y, Negawa T, Yamada K, et al. Oxidative stress is enhanced in correlation with renal dysfunction: examination with the redox state of albumin. Kidney Int. 2004;66:1988–93.
Johnson-Davis KL, Fernelius C, Eliason NB, Wilson A, Beddhu S, Roberts WL. Blood enzymes and oxidative stress in chronic kidney disease: a cross sectional study. Ann Clin Lab Sci. 2011;41:331–9.
Modlinger PS, Wilcox CS, Aslam S. Nitric oxide, oxidative stress, and progression of chronic renal failure. Semin Nephrol. 2004;24:354–65.
Cottone S, Lorito MC, Riccobene R, Nardi E, Mule G, Buscemi S, et al. Oxidative stress, inflammation and cardiovascular disease in chronic renal failure. J Nephrol. 2008;21:175–9.
Palatini P. Glomerular hyperfiltration: a marker of early renal damage in pre-diabetes and pre-hypertension glomerular hyperfiltration: a marker of early renal damage in pre-diabetes and pre-hypertension. Nephrol Dial Transpl. 2012;27:1708–14.
Annuk M, Zilmer M, Lind L, Linde T, Fellstrom B. Oxidative stress and endothelial function in chronic renal failure. J Am Soc Nephrol. 2001;12:2747–52.
Schutte R, Schutte AE, Huisman HW, Glyn MC, Van Rooyen JM, Malan NT, et al. Arterial stiffness, ambulatory blood pressure and low-grade albuminuria in non-diabetic African and Caucasian men: the SABPA study. Hypertens Res. 2011;34:862.
Acknowledgements
The SABPA study would not have been possible without the volunteers who took part in this study. The authors would like to thank all the researchers, support staff and students of the Hypertension Research and Training Clinic at North-West University for their dedication and support. We would also like to acknowledge Mrs Tina Scholtz, Dr Szabolcs Péter and Sr. Chrissie Lessing for their technical assistance throughout the SABPA study. The SABPA study was supported by the North-West University, Potchefstroom; the South African Medical Research Council (SAMRC), the National Research Foundation (NRF); the NRF Thuthuka (80643); ROCHE Diagnostics, South Africa; the Department of Education, North-West Province; and the Metabolic Syndrome Institute, France. Any opinion, findings, and conclusions or recommendations expressed in this material are those of the authors, and therefore, the NRF do not accept any liability in this regard.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Any opinions expressed and conclusions stated are those of the authors alone. The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Myburgh, C., Huisman, H.W. & M.C. Mels, C. Three-year change in oxidative stress markers is linked to target organ damage in black and white men: the SABPA study. Hypertens Res 42, 1961–1970 (2019). https://doi.org/10.1038/s41440-019-0325-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-019-0325-4
- Springer Nature Singapore Pte Ltd.