Abstract
Following the formulation of operational criteria for the diagnosis of psychosis in Parkinson’s disease, a neurodegenerative disorder, the past decade has seen increasing interest in such nonmotor psychopathology that appears to be independent of dopaminergic therapy. Similarly, there has been a resurgence of interest in motor aspects of the neurodevelopmental disorder of schizophrenia, including spontaneous parkinsonism that appears to be independent of antipsychotic treatment. This review first addresses the clinical and nosological challenges of these superficially paradoxical insights and then considers pathobiological challenges. It proposes that diverse modes of disturbance to one or more element(s) in a cortical-striatal-thalamocortical neuronal network, whether neurodegenerative or neurodevelopmental, can result in movement disorder, psychosis or both. It then proposes that time- and site-dependent dysfunction in such a neuronal network may be a generic substrate for the emergence of psychosis not only in Parkinson’s disease and schizophrenia-spectrum disorders but also in other neuropsychiatric disorders in which psychosis, and sometimes movement disorders, can be encountered; these include substance abuse, cerebrovascular disease, cerebral trauma, cerebral neoplasia, epilepsy, Huntington’s disease, frontotemporal dementia, Alzheimer’s disease and multiple sclerosis.
Similar content being viewed by others
Introduction
Psychosis intrinsic to Parkinson’s disease
Over the past decade, following the formulation of operational criteria for the diagnosis of psychosis in Parkinson’s disease (PD) [1], there has been a resurgence of interest in this ‘nonmotor’ aspect of the disorder [2]. With the historical literature on PD failing to clearly identify a typical psychotic syndrome in the pre-levodopa era in the absence of other known precipitating factors [3, 4], the onset of psychosis in PD was initially related to treatment with levodopa and, subsequently, with dopamine receptor agonists [5].
However, definitively disentangling the effects of medication and disease requires contemporary studies of patients with untreated disease over the longitudinal course [6], which presents both ethical and practical challenges. In relation to PD, these challenges can be overcome through the assessment of drug-naive patients at their first presentation and, in developing countries where health service provision is as yet insufficient, over the continuing course of untreated illness. Critically, findings in these drug-naive states indicate that psychosis can be detected in PD at first presentation [7] and for several years thereafter [8]. Thus, echoing earlier theorizing [9], contemporary theory posits that levodopa/dopamine (DA) D2 receptor agonist treatment interacts with the disease process in the manner of a ‘modifier’ that ‘unmasks’ psychosis intrinsic to PD [2]. Importantly, this same review [2] recognizes the unmet need for an analysis of psychotic psychopathology in PD and its putative pathobiology vis-à-vis psychosis across other conditions. The present article presents such an analysis in juxtaposition with schizophrenia spectrum and related psychotic disorders, the essential elements of which are summarized in Table 1.
Parkinsonism intrinsic to schizophrenia spectrum psychotic disorders
In recent decades, there has been a resurgence of interest in the ‘motor’ aspects of schizophrenia and related psychotic disorders, culminating in a special issue of the journal Schizophrenia Research devoted to this topic [10]. Although the historical literature on schizophrenia (now schizophrenia-spectrum psychosis, for which schizophrenia is used here as shorthand) in the preneuroleptic era clearly identified movement disorders as an intrinsic component of the disease process [11,12,13], the introduction of neuroleptic (DA D2 receptor antagonist) drugs [now first-generation antipsychotics (FGAs)] in the 1950s resulted in this literature being usurped by the presumption that movement disorders in schizophrenia, including parkinsonism, constitute an anti-DAergic side effect of such medication [14,15,16]; furthermore, this proposition has endured into the era of second-generation antipsychotics (SGAs) [17, 18].
However, as for psychosis in PD, definitively disentangling the roles of disease and medication requires contemporary studies of untreated schizophrenia over its longitudinal course, which presents comparable ethical and practical challenges. These challenges can be overcome through the assessment of drug-naive schizophrenia patients at their first presentation and, in developing countries where health service provision is as yet insufficient, over the continuing course of untreated illness. Critically, findings in these drug-naive states indicate that movement disorders, both parkinsonism and hyperkinesia, can be detected in schizophrenia at the first presentation and for several years thereafter [19,20,21,22,23,24]. Thus, we have posited that DA D2 receptor antagonist antipsychotics, both FGAs and, although perhaps less so, SGAs, interact with the disease process in the manner of a modifier that ‘precipitates’ and ‘accentuates’ movement disorders intrinsic to schizophrenia [20, 25].
Nosological challenges
That both psychosis in the movement disorder PD and parkinsonism in the psychotic disorder schizophrenia were initially related to their respective medications but are now recognized to be intrinsic to each disorder clearly indicates overlapping phenomenology. There might appear to be limited scope for diagnostic uncertainty in distinguishing clinically between these two disorders, with the neurodegenerative disorder PD emerging primarily in old age [26] and the neurodevelopmental disorder schizophrenia emerging primarily in young adulthood [27, 28]. However, in reality, these present challenges to operational diagnostic algorithms that are further accentuated by the increasing recognition of early-onset (both juvenile and young onset) PD [29, 30] and both late- and very-late-onset schizophrenia [31, 32].
The Movement Disorder Society Clinical Diagnostic Criteria for Parkinson’s Disease [33] proceed in two stages: (i) a clinical, phenomenological diagnosis of parkinsonism and (ii) the absence of nine exclusion criteria. These exclusion criteria include the following: “5. Treatment with a dopamine receptor blocker or a dopamine-depleting agent in a dose and time-course consistent with drug-induced parkinsonism”, which would exclude cases of schizophrenia with parkinsonism in the course of treatment with antipsychotics, and “9. Documentation of an alternative condition known to produce parkinsonism and plausibly connected to the patient’s symptoms, or, the expert evaluating physician, based on the full diagnostic assessment feels that an alternative syndrome is more likely than PD”, which could, but would not necessarily, exclude cases of schizophrenia with parkinsonism that are not receiving treatment with antipsychotics. The presence of psychosis in PD requires the exclusion of other neuropsychiatric conditions in which psychosis is encountered, including schizophrenia-spectrum disorders and [nonmedical] substance-induced psychosis, with the presence or absence of medical treatment with levodopa and/or dopamine receptor agonists specified as an associated feature of psychosis in PD [1].
Both DSM-IV and DSM-5 recognize the difficulty in distinguishing iatrogenic movement disorders in schizophrenia, such as antipsychotic drug-induced parkinsonism, from the development of idiopathic movement disorders, such as PD. This is exemplified by the long-standing conundrum of whether the emergence of parkinsonism in older patients with schizophrenia who are receiving treatment with antipsychotics reflects antipsychotic drug-induced parkinsonism or the coemergence of PD [34].
Furthermore, it is well recognized (DSM-IV/5) that psychosis similar to that observed in schizophrenia spectrum psychotic disorders can be encountered in a variety of other medical circumstances [subject to the exclusion of substance-induced psychotic disorder] under the diagnostic criteria for psychotic disorder due to a general medical condition; specific examples given include cerebral neoplasia, cerebral trauma, cerebrovascular disease, epilepsy, Huntington’s disease, multiple sclerosis, and central nervous system infections. In our own study of 432 subjects experiencing their first psychotic episode, we identified 13 patients with a DSM-IV diagnosis of psychotic disorder due to a general medical condition; these patients had cerebral neoplasia, cerebral trauma, cerebrovascular disease, epilepsy and multiple sclerosis [35, 36]. As an example, psychosis following a stroke is well recognized as falling within the category of psychotic disorder due to a general medical condition [37], which prompts the following question: why is psychosis in PD not included among the range of conditions exemplifying this category or encountered in our own real-world clinical study? This may reflect, at least in part, the fact that psychosis in PD arises and is most commonly managed within neurology and geriatric services [38]; in contrast, child and adolescent, adult, old-age or liaison psychiatric services commonly provide care for schizophrenia and psychosis in patients with general medical conditions other than PD. Nevertheless, nosological challenges remain.
Pathobiological challenges
Presumption of heterogeneity
At the level of phenomenology, both psychosis in the movement disorder PD and parkinsonism in the psychotic disorder schizophrenia are now recognized to be intrinsic to each disorder in a manner that can be unrelated to adverse effects of medication. The nosological challenges that this presents are predicated on a presumption that diagnostic criteria seek to distinguish between two independent disorders having distinct pathobiologies and treatments.
Psychosis
In schizophrenia, the cardinal diagnostic symptoms of psychosis (DSM-IV/5) involve hallucinations, commonly auditory but extending to multimodal experiences, and delusions [39,40,41]. In PD, visual hallucinations (VH) have been reported to occur more commonly than auditory hallucinations (AH), with the most recent meta-analysis of data from 1970 to 2017, using diverse criteria, indicating that VH occur in 28% and AH occur in 9% of PD patients; for comparison, this meta-analysis also indicates that VH occur in 62% and AH occur in 31% of patients with Lewy body dementia [42]. However, all but two of the studies reviewed did not involve the NINDS-NIMH diagnostic criteria for psychosis in PD [1]. The two studies applying these criteria reported (i) one or more hallucinations (in any modality) and/or delusions in 43% of cases, with AH in 18%, VH in 16%, tactile in 12%, olfactory in 11%, and delusions in 4% [43], and (ii) one or more hallucinations (in any modality) and/or delusions in 14%, with VH in 7%, AH in 4%, olfactory in 2%, tactile in 1%, and delusions in 3% [44].
The most recent systematic reviews on the spectrum of psychosis in PD further indicate that the prior accentuation of VH may have been overstated, necessitating a broader, more multimodal concept of hallucinations and delusions [45, 46]. It should be emphasized that any differences in hallucinations and/or delusions between PD and schizophrenia are quantitative variations rather than qualitative distinctions, such that essentially all forms and permutations of such symptoms can be evident in both diagnoses with no points of rarity between them [41].
Movement disorder
In PD, the cardinal diagnostic symptoms of this movement disorder (Movement Disorder Society Clinical Diagnostic Criteria for Parkinson’s Disease) involve bradykinesia/akinesia, rigidity, resting tremor and abnormalities of posture and gait [26, 33]. In drug-naive schizophrenia, parkinsonism is characterized by these same features [19,20,21,22,23,24]. However, movement disorders in drug-naive schizophrenia can also include abnormal involuntary movements (AIM) that overlap phenomenologically with what would be described as tardive dyskinesia in schizophrenia patients who have received long-term treatment with antipsychotics [12, 19,20,21,22,23,24] and dyskinesia in PD patients who have received long-term treatment with L-dopa [26]. Furthermore, parkinsonism and AIM can occasionally cooccur in patients with drug-naive schizophrenia; in such instances, it appears that while parkinsonism is evident primarily in peripheral regions, AIM have a primarily orofacial topography [19,20,21,22,23,24].
Broadening the relationships
There are additional examples of relationships between PD and schizophrenia. At the level of phenomenology, it appears that hallucinations in PD and parkinsonism in drug-naive schizophrenia may each be associated with cognitive deterioration [47, 48]. At the level of genetics, while a recent meta-analysis of genome-wide association studies (GWASs) across 25 brain disorders failed to indicate shared genetic risk between PD and schizophrenia [49], microdeletions at 22q11.2 are associated with an increased risk for both PD-like movement disorder [50, 51] and schizophrenia [52].
These relationships between PD and schizophrenia-spectrum psychosis present fundamental pathobiological challenges: in PD, each postmortem neurochemical measure of DA and in vivo neuroimaging of fluorodopa and the DA transporter indicate degeneration of nigrostriatal DAergic neurons in the substantia nigra pars compacta with loss of DA from the caudate and putamen [26, 53], while in schizophrenia, the same measures indicate that nigrostriatal DAergic neurons are intact and hyperactive [54, 55]. However, an alternative perspective embraces these phenomenological commonalities and interrelationships and invokes a broader concept of systems/network-based pathobiology that seeks integration within an overarching schema.
Overlapping network dysfunction
While psychosis involves domains of psychopathology that may appear to differ across various circumstances [e.g., VH are more prominent in PD, while AH and delusions are more prominent in schizophrenia; complex, multimodal hallucinations and delusions occur in epilepsy, and delusions follow stroke; VH can occur consequent to lysergic acid diethylamide treatment, and schizophrenia-like hallucinations and delusions can occur consequent to treatment with phencyclidine], the evidence [2, 37, 39, 56,57,58,59,60] shows that these variations are quantitative rather than qualitative, with essentially all domains of psychosis being encountered to some extent in each condition [41]. This implies some commonality in the neuronal dysfunction that underlies psychosis across such a breadth of circumstances. Furthermore, in each condition, no neuronal pathway functions in isolation; rather, each pathway functions within one or more neuronal networks of interacting regions, the overall functional integrity of which can be disrupted via disturbances in any one or more of the multiple regions involved in that network.
Using schizophrenia as an example, the actions of the DA-releasing drug amphetamine to induce schizophrenia-like psychosis and DA receptor antagonists to exert antipsychotic activity initially led to the proposal that the pathophysiology of psychosis involved DAergic hyperactivity; furthermore, given the classic presumption of a primary role for the nigrostriatal DAergic pathway and the striatum (caudate, putamen, and globus pallidus) in motor function, such hyperactivity in psychosis was related to mesolimbic/mesocortical DAergic pathways [61]. However, regardless of how persuasive the indirect evidence appeared, these issues remained conjectural in the absence of direct measurements of DAergic function in living patients.
When such measurements became available through positron emission tomography (PET)- and single photon emission computed tomography (SPECT)-based techniques, the results were unexpected: while DAergic hyperfunction in schizophrenia was confirmed, primarily through the D2 receptor, this was most evident in the caudate and putamen, with the greater resolution of the most recent studies indicating a large increase in the dorsal (associative and sensorimotor) regions and less so in the ventral (limbic) subdivision of the striatum [62]. In parallel, the normal functional anatomy of the striatum was recognized to involve (a) midbrain DAergic projections from the ventral tegmental area and medial substantia nigra to the limbic striatum and from the central/ventrolateral substantia nigra to the associative and sensorimotor striatum and (b) three corticostriatal loops that can act independently, interact with each other, and participate in cortico-striato-thalamocortical loops: premotor/motor cortex projections to the caudal putamen; dorsolateral prefrontal cortex (PFC) projections to the associative and sensorimotor striatum; and limbic area (amygdala, hippocampus, ventromedial PFC, and orbitomedial PFC) projections to the limbic striatum [55, 63]. These findings complement and elaborate extensive evidence from structural and functional magnetic resonance imaging (MRI) studies, including diffusion-tensor imaging tractography, and a broader range of PET, SPECT and neuropsychological investigations; integration of these findings now implicates dysfunction in a prefrontal/anterior cingulate cortical-striatal-hippocampal neuronal network in the pathobiology of schizophrenia and antipsychotic drug action [55, 64,65,66,67].
Against this background, it is notable that not only has the DAergic pathobiology of PD become better understood but it is also increasingly recognized to extend beyond DAergic processes. The greatest loss of DAergic neurons is from the ventrolateral substantia nigra with the attendant loss of projections to the dorsal putamen, resulting in the primary movement disorder in PD and relative preservation of DAergic terminals in the caudate [26, 30]. However, the loss of DAergic neurons can extend to the dorsal substantia nigra and ventral tegmental area with a loss of DA beyond the striatum to include the limbic and cortical regions; these abnormalities can be accompanied by the striatal and extrastriatal loss of serotonergic, noradrenergic and cholinergic neurons [68,69,70,71]. Importantly, extranigral and extrastriatal dysfunction in PD is further indicated by the presence of Lewy body pathology in the amygdala, hippocampus and frontal, temporal, parietal, cingulate and occipital cortices and pathways [26]. While it has been suggested that VH in PD may involve Lewy body deposition in the occipital cortex and associated visual pathways [2, 72], it is evident that Lewy bodies are also deposited in several components of the cortical-striatal-thalamocortical network discussed here, and there is evidence that hallucinations in PD are associated with striatal DAergic dysfunction [73].
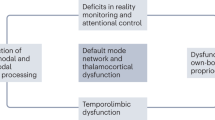
Given the long-standing recognition of cortical-striatal-thalamocortical loops [63], a recent study has integrated and interpreted these features in the pathobiology of PD [74, 75] in the following terms: (a) a dysfunctional cortical-subcortical neuronal network that includes the putamen and mediates predominantly motor dysfunction and (b) a dysfunctional cortico-striato-thalamocortical neuronal network that includes the caudate, frontal association cortex, limbic system and other neocortical areas, and is involved in mediating both motor and nonmotor features intrinsic to PD, such as psychosis [2, 30]. This model overlaps with dysfunction in the prefrontal/anterior cingulate cortical-striatal-hippocampal neuronal network posited in the pathobiology of psychosis and antipsychotic drug action in schizophrenia [55, 64,65,66,67]. In a complementary manner, it has been proposed that hypokinetic movement disorders intrinsic to schizophrenia, such as parkinsonism, involve dysfunction in the cingulate motor cortex/supplementary motor area/motor cortical-striatal-thalamocortical neuronal network [76].
Critically, there is a precedent for both DAergic hypofunction, a characteristic of PD, and DAergic hyperfunction, a characteristic of schizophrenia, having a common functional effect on an element of the cortico-striato-thalamocortical neuronal network: working memory and executive function mediated via the PFC is critically dependent on the level of prefrontal DAergic activity in the manner of an inverted U-shaped relationship, such that either DAergic hypofunction or DAergic hyperfunction leads to PFC dysfunction and impairment in these domains of cognition [77]. Similar relationships may be relevant for the present issues of movement disorders and psychosis in PD and schizophrenia. A recent meta-analysis of the neuroimaging literature further indicates that hallucinations in neurodegenerative disorders (including PD) and psychiatric disorders (including schizophrenia) appear to involve structural networks that each include elements of the cortico-striato-thalamocortical network at issue here [78].
Synthesis
As overall functional integrity in any cortico-striatal-thalamocortical network can be disrupted via disturbances in any one or more of the regions participating in that network, it follows that both parkinsonian features and psychosis in both PD and schizophrenia could arise through disturbances in different elements of such network circuitry. Following the conceptual and initial systematic outline of cortical-striatal-thalamocortical networks [63], subsequent detailed elaborations are available descriptively and diagrammatically in the contexts of both PD [74, 75] and schizophrenia [55]. Furthermore, variations in the relative preponderance of individual parkinsonian features and modality of hallucinations and nature of delusions might be expected due to variations in the location and severity of those disturbances. Notably, cortical-striatal-thalamocortical neuronal network dysfunction has been implicated not only in PD and schizophrenia but also in substance-induced psychosis [79], psychosis subsequent to stroke [37] and psychosis in epilepsy [80]. In addition, it has been proposed for psychotic illness on the basis of the juxtaposition of clinical findings with preclinical and computational neuroscience considerations [81].
It appears that the following varying modes of disturbance to one or more element(s) in the cortical-striatal-thalamocortical neuronal network can result in movement disorders, psychosis or both: neurodegenerative, neurodevelopmental, substance abuse, cerebrovascular disease, or seizure activity. This would also appear to be relevant to other forms of cerebral abnormality: trauma, where we have reported the most common finding with schizophrenia-like psychosis to be damage to the temporal cortex [82]; multiple sclerosis, where the most common finding with psychosis is white matter lesions in fronto-temporal regions [83]; and Alzheimer’s disease, where the most common finding with psychosis is abnormalities in dorsolateral PFC, with some reports also implicating anterior cingulate cortex and ventral striatal regions [84, 85]. In other disorders in which disruption to elements in a cortical-striatal-thalamocortical neuronal network is pathognomonic and psychosis can be encountered, such as frontotemporal dementia [86, 87] and Huntington’s disease [88], it will be a task for future studies to further clarify the relationship between psychosis and such network disruption. This challenge also applies to psychosis that is consequent to cerebral neoplasia [89] and the extent to which such psychosis might be associated with movement disorders.
While these systems-based considerations relate to overt neurological and psychiatric disorders in which movement disorders and psychosis can be encountered, they are also of interest in the context of other overlapping clinical features of PD and schizophrenia. In PD, it is well known that nonmotor features extend beyond psychosis to include depression [72], while in schizophrenia, nonpsychotic features are well known to extend beyond movement disorders to include depression [90]. Notably, recent evidence indicates psychosis and depression involve shared network dysfunction that overlaps with the cortical-striatal-thalamocortical network posited above [64, 67, 91, 92].
Subclinical features of disorders might facilitate early detection and hence early intervention to improve long-term outcomes. Thus, it is striking that (a) ‘minor’ (subclinical) psychotic experiences/ideation and depression may be early nonmotor symptoms in younger persons that can predate the onset of overt PD [7, 93], and (b) (subclinical) psychotic experiences/ideation and depression can be present in younger persons in the general population and may be prodromal indicators of the risk for overt schizophrenia [90, 94, 95]. Such findings further elaborate the relationship between PD and schizophrenia, summarized in Table 1, that might be best accommodated within a model of time- and site-dependent variations in the severity of dysfunction within a common cortical-striatal-thalamocortical neuronal network.
References
Ravina B, Marder K, Fernandez HH, Friedman JH, McDonald W, Murphy D, et al. Diagnostic criteria for psychosis in Parkinson’s disease: report of an NINDS, NIMH work group. Mov Disord. 2015;30:1061–8.
ffytche DH, Creese B, Politis M, Chaudhuri KR, Weintraub D, Ballard C, et al. The psychosis spectrum in Parkinson disease. Nat Rev Neurol. 2017;13:81–95.
Parkinson J. An essay on the shaking palsy. London: Sherwood, Neely, and Jones; 1817.
Fenelon G, Goetz CG, Karenberg A. Hallucinations in Parkinson disease in the prelevodopa era. Neurology. 2006;66:930–98.
Factor SA, Molho ES, Podskalny GD, Brown D. Parkinson’s disease: drug-induced psychiatric states. Adv Neurol. 1995;65:115–38.
Waddington JL, Lane A, Larkin C, O’Callaghan E. The neurodevelopmental basis of schizophrenia: clinical clues from cerebro-craniofacial dysmorphogenesis, and the roots of a lifetime trajectory of disease. Biol Psychiatry. 1999;46:31–9.
Pagonabarraga J, Martinez-Horta S, Fernandez de Bobadilla R, Perez J, Ribosa-Nogue R, Marin J, et al. Minor hallucinations occur in drug-naïve Parkinson’s disease patients, even from the premotor phase. Mov Disord. 2016;31:45–52.
Dotchin CL, Jusabani A, Walker RW. Non-motor symptoms in a prevalent population with Parkinson’s disease in Tanzania. Parkinsonism Relat Disord. 2009;15:457–60.
Moskovitz C, Moses H, Klawans HL. Levodopa-induced psychosis: a kindling phenomenon. Am J Psychiatry. 1978;135:669–75.
Cuesta MJ, Peralta V. Modeling neuromotor pathology in schizophrenia: a window to brain mechanisms and clinical insight into the syndrome. Schizophr Res. 2018;200:1–4.
Kraepelin E. Dementia praecox and paraphrenia, translated by Barclay RM. Edinburgh: Livingstone; 1919.
Waddington JL, Crow TJ. Abnormal involuntary movements and psychosis in the preneuroleptic era and in unmedicated patients: implications for the concept of tardive dyskinesia. In: Wolf M, Mosnaim AD, editors. Tardive dyskinesia: biological mechanisms and clinical aspects. Washington, DC: American Psychiatric Press; 1988. p. 51–66.
Kendler KS. Phenomenology of schizophrenia and the representativeness of modern diagnostic criteria. JAMA Psychiatry. 2016;73:1082–92.
Lader MH. Drug-induced extrapyramidal syndromes. J R Coll Phys Lond. 1970;5:87–98.
Marsden CD, Jenner P. The pathophysiology of extrapyramidal side-effects of neuroleptic drugs. Psychol Med. 1980;10:55–72.
Casey DE. Neuroleptic drug-induced extrapyramidal syndromes and tardive dyskinesia. Schizophr Res. 1991;4:109–20.
Leucht S, Corves C, Arbter D, Engel RR, Li C, Davis JM. Second-generation versus first generation antipsychotic drugs for schizophrenia: a meta-analysis. Lancet. 2009;373:31–41.
Leucht S, Cipriani A, Spineli L, Mavridis D, Orey D, Richter F, et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: a multiple-treatments meta-analysis. Lancet. 2013;382:951–62.
Pappa S, Dazzan P. Spontaneous movement disorders in antipsychotic-naive patients with first-episode psychoses: a systematic review. Psychol Med. 2009;39:1065–76.
Whitty PF, Owoeye O, Waddington JL. Neurological signs and involuntary movements in schizophrenia: intrinsic to and informative on systems pathobiology. Schizophr Bull. 2009;35:415–24.
Koning JPF, Tenback DE, van Os J, Aleman A, Kahn RS, van Harten PN. Dyskinesia and parkinsonism in antipsychotic-naïve patients with schizophrenia, first-degree relatives and healthy controls: a meta-analysis. Schizophr Bull. 2010;36:723–31.
Peralta V, Campos MS, De Jalon EG, Cuesta MJ. Motor behaviour abnormalities in drug-naïve patients with schizophrenia spectrum disorders. Mov Disord. 2010;25:1068–76.
Ayehu M, Shibre T, Milkias B, Fekadu A. Movement disorders in neuroleptic-naïve patients with schizophrenia spectrum disorders. BMC Psychiatry. 2014;14:280.
Ojagbemi A, Chiliza B, Bello T, Asmal L, Esan O, Emsley R, et al. Neurological soft signs, spontaneous and treatment emergent extrapyramidal syndromes in black Africans with first episode schizophrenia. Front Psychiatry. 2018;9:172.
Waddington JL, O’Tuathaigh CM. Modelling the neuromotor abnormalities of psychotic illness: putative mechanisms and systems dysfunction. Schizophr Res. 2018;200:12–9.
Kalia LV, Lang AE. Parkinson’s disease. Lancet. 2015;386:896–912.
Waddington JL, Hennessy RJ, O'Tuathaigh CMP, Owoeye O, Russell V. Schizophrenia and the lifetime trajectory of psychotic illness:developmental neuroscience and pathobiology, redux. In: Brown AS, Patterson PH, editors. The origins of schizophrenia. New York: Columbia University Press; 2012. p. 3–21.
Weinberger DR. Future of days past: neurodevelopment and schizophrenia. Schizophr Bull. 2017;43:1164–8.
Schrag A, Schott JM. Epidemiological, clinical, and genetic characteristics of early-onset parkinsonism. Lancet Neurol. 2006;5:355–63.
Liu SY, Wu JJ, Zhao J, Huang SF, Wang YX, Ge JJ, et al. Onset-related subtypes of Parkinson’s disease differ in the patterns of striatal dopaminergic dysfunction: a positron emission tomography study. Parkinsonism Relat Disord. 2015;21:1448–53.
Van der Werf M, Hanssen M, Kohler S, Verkaaik M, Verhey FR, RISE Investigators, et al. Systematic review and collaborative recalculation of 133693 incident cases of schizophrenia. Psychol Med. 2014;44:9–16.
Van Assche LV, Morrens M, Luyten P, Van de Ven L, Vandenbulcke M. The neuropsychology and neurobiology of late-onset schizophrenia and very-late-onset schizophrenia-like psychosis: a critical review. Neurosci Biobehav Rev. 2017;83:604–21.
Postuma RB, Berg D, Stern M, Poewe W, Olanow CW, Oertel W, et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord. 2015;30:1591–601.
Crow TJ, Johnstone EC, McClelland HA. The coincidence of schizophrenia and Parkinsonism: some neurochemical implications. Psychol Med. 1976;6:227–33.
Baldwin P, Browne D, Scully PJ, Quinn JF, Morgan MJ, Kinsella A, et al. Epidemiology of first-episode psychosis: illustrating the challenges across diagnostic boundaries through the Cavan/Monaghan study at 8 years. Schizophr Bull. 2005;31:624–38.
Nkire N, Scully PJ, Browne DJ, Baldwin PA, Kingston T, Owoeye O, et al. Systematic epidemiological and clinical comparisons across all 12 DSM-IV psychotic diagnoses in the Cavan-Monaghan First Episode Psychosis Study (CAMFEPS). Psychol Med. 2019. https://doi.org/10.1017/S0033291719003520.
Joyce EM. Organic psychosis: the pathobiology and treatment of delusions. CNS Neurosci Ther. 2018;24:598–603.
Lizarraga KJ, Fox SH, Strafella AP, Lang AE. Hallucinations, delusions and impulse control disorders in Parkinson disease. Clin Geriatr Med. 2020;36:105–18.
Arango C, Carpenter WT. The schizophrenia construct: symptomatic presentation. In: Weinberger DR, Harrison PJ, editors. Schizophrenia. Oxford: Wiley-Blackwell; 2011. p. 9–23.
McCarthy-Jones S, Smailes D, Corvin A, Gill M, Morris DW, Dinan TG, et al. Occurrence and co-occurrence of hallucinations by modality in schizophrenia-spectrum disorders. Psychiat Res. 2017;252:154–60.
Waters F, Fernyhough C. Hallucinations: a systematic review of points of similarity and difference across diagnostic classes. Schizophr Bull. 2017;43:32–43.
Eversfield CL, Orton LD. Auditory and visual hallucination prevalence in Parkinson’s disease and dementia with Lewy bodies: a systematic review and meta-analysis. Psychol Med. 2018;49:2342–53.
Fenelon G, Soulas T, Zenasni F, de Langavant LC. The changing face of Parkinson’s disease-associated psychosis: a cross-sectional study based on the new NINDS-NIMH criteria. Mov Disord. 2010;25:763–6.
Mack J, Rabins P, Anderson K, Goldstein S, Grill S, Hirsch ES, et al. Prevalence of psychotic symptoms in a community-based Parkinson disease sample. Am J Geriatr Psychiatry. 2012;20:123–32.
Frei K, Truong DD. Hallucinations and the spectrum of psychosis in Parkinson’s disease. J Neurol Sci. 2017;374:56–62.
Warren N, O’Gorman C, Hume Z, Kisely S, Siskind D. Delusions in Parkinson’s disease: a systematic review of published cases. Neuropsychol Rev. 2018;28:310–6.
Guo Y, Xu W, Liu FT, Li JQ, Cao XP, Tan L, et al. Modifiable risk factors for cognitive impairment in Parkinson’s disease: a systematic review and meta-analysis of prospective cohort studies. Mov Disord. 2019;34:876–83.
Cuesta MJ, Sanchez-Torres AM, de Jalon EG, Campos MS, Ibanez B, Moreno-Izco L, et al. Spontaneous parkinsonism is associated with cognitive impairment in antipsychotic-naïve patients with first-episode psychosis: a 6-month follow-up study. Schizophr Bull. 2014;40:1164–73.
Brainstorm Consortium. Analysis of shared heritability in common disorders of the brain. Science. 2018;360:eaap8757. https://doi.org/10.1126/science.aap8757.
Mok KY, Sheerin U, Simon-Sanchez J, Salaka A, Chester L, Escott-Price V, et al. Deletions at 22q11.2 in idiopathic Parkinson’s disease: a combined analysis of genome-wide association data. Lancet Neurol. 2016;15:585–96.
Boot E, Butcher NJ, Udow S, Marras C, Mok KY, Kaneko S, et al. Typical features of Parkinson disease and diagnostic challenges with microdeletion 22q11.2. Neurology. 2018;90:e2059–67.
Schneider M, Debbane M, Bassett AS, Chow EW, Fung WL, van den Bree M, et al. Psychiatric disorders from childhood to adulthood in 22q11.2 deletion syndrome: results from the International Consortium on Brain and Behavior in 22q11.2 Deletion Syndrome. Am J Psychiatry. 2014;171:627–39.
Weingarten CP, Sundman MH, Hickey P, Chen NK. Neuroimaging of Parkinson’s disease: expanding views. Neurosci Biobehav Rev. 2015;59:16–52.
Fusar-Poli P, Meyer-Lindenberg A. Striatal presynaptic dopamine in schizophrenia, Part 1: meta-analysis of dopamine active transporter (DAT) density. Schizophr Bull. 2013;39:22–32.
McCutcheon RA, Abi-Dargham A, Howes OD. Schizophrenia, dopamine and the striatum: from biology to symptoms. Trends Neurosci. 2019;42:205–20.
Elliott B, Joyce E, Shorvon S. Delusions, illusions and hallucinations in epilepsy: 1. Elementary phenomena. Epilepsy Res. 2009;85:162–71.
Elliott B, Joyce E, Shorvon S. Delusions, illusions and hallucinations in epilepsy: 2. Complex phenomena and psychosis. Epilepsy Res. 2009;85:172–86.
Javitt DC, Zukin SR. Recent advances in the phencyclidine model of schizophrenia. Am J Psychiatry. 1991;148:1301–8.
Rosse RB, Collins JP, Fay-McCarthy M, Alim TN, Wyatt RJ, Deutsch SI. Phenomenological comparison of the idiopathic psychosis of schizophrenia and drug-induced cocaine and phencyclidine psychoses: a retrospective study. Clin Neuropharmacol. 1994;17:359–69.
Murray RM, Paparelli A, Morrison PD, Marconi A, Di Forti M. What can we learn about schizophrenia from studying the human model, drug-induced psychosis? Am J Med Genet Neuropsychiatr Genet. 2013;162B:661–70.
Meltzer HY, Stahl SM. The dopamine hypothesis of schizophrenia: a review. Schizophr Bull. 1976;2:19–76.
McCutcheon RA, Beck K, Jauhar S, Howes OD. Defining the locus of dopaminergic dysfunction in schizophrenia: a meta-analysis and test of the mesolimbic hypothesis. Schizophr Bull. 2018;44:1301–11.
Alexander GE, DeLong MR, Strick PL. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci. 1986;9:357–81.
Goodkind M, Eickhoff SB, Oathes DJ, Jiang Y, Chang A, Jones-Hagata LB, et al. Identification of a common neurobiological substrate for mental illness. JAMA Psychiatry. 2015;72:305–15.
Sarpal DK, Robinson DG, Lencz T, Argyelan M, Ikuta T, Karlsgodt K, et al. Antipsychotic treatment and functional connectivity of the striatum in first-episode schizophrenia. JAMA Psychiatry. 2015;72:5–13.
Kraguljac NV, White DM, Hadley N, Hadley JA, Ver Hoef L, Davis E, et al. Abberant hippocampal connectivity in unmedicated patients with schizophrenia and effects of antipsychotic medication: a longitudinal resting state functional MRI study. Schizophr Bull. 2016;42:1046–55.
Sheffield JM, Kandala S, Tamminga CA, Pearlson GD, Keshavan MS, Sweeney JA, et al. Transdiagnostic associations between functional brain network integrity and cognition. JAMA Psychiatry. 2017;74:605–13.
Moore RY, Whone AL, Brooks DJ. Extrastriatal monoamine neuron function in Parkinson’s disease: an 18F-dopa PET study. Neurobiol Dis. 2008;29:381–90.
Bohnen NI, Kane P, Muller MLMT. Molecular imaging of the cholinergic system in Parkinson’s disease. Int Rev Neurobiol. 2018;141:211–50.
Morris R, Martini DN, Madhyastha T, Kelly VE, Grabowski TJ, Nutt J, et al. Overview of the cholinergic contribution to gait, balance and falls in Parkinson’s disease. Parkinsonism Relat Disord. 2019;63:20–30.
Pilotto A, di Cola FS, Premi E, Grasso R, Turrone R, Gipponi S, et al. Extrastriatal dopaminergic and serotonergic pathways in Parkinson’s disease and in dementia with Lewy bodies: a 123I-FP-CIT SPECT study. Eur J Nuc Med Mol Imaging. 2019;46:1642–51.
Schapira AHV, Chaudhuri KR, Jenner P. Non-motor features of Parkinson disease. Nat Rev Neurosci. 2017;18:435–50.
Jaakkola E, Jousta J, Makinen E, Johansson J, Kaasinen V. Ventral striatal dopaminergic defect is associated with hallucinations in Parkinson’s disease. Eur J Neurol. 2017;24:1341–7.
Obeso JA, Rodriguez-Oroz MC, Benitez-Temino B, Blesa FJ, Guridi J, Marin C, et al. Functional organization of the basal ganglia: therapeutic implications for Parkinson’s disease. Mov Disord. 2008;23(Suppl 3):S548–59.
Poston KL, Eidelberg D. Functional brain networks and abnormal connectivity in the movement disorders. Neuroimage. 2012;62:2261–70.
Walther S. Psychomotor symptoms of schizophrenia map on the cerebral motor circuit. Psychiat Res. 2015;233:293–8.
Seamans JK, Yang CR. The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol. 2004;74:1–58.
Rollins CPE, Garrison JR, Simons JS, Rowe JB, O’Callaghan C, Murray GK, et al. Meta-analytic evidence for the plurality of mechanisms in transdiagnostic structural MRI studies of hallucination status. EClinical Med. 2019;8:57–71.
Khokhar JY, Dwiel LL, Henricks AM, Doucette WT, Green AI. The link between schizophrenia and substance use disorder: a unifying hypothesis. Schizophr Res. 2018;194:78–85.
Allebone J, Kanaan R, Wilson SJ. Systematic review of structural and functional brain alterations in psychosis of epilepsy. J Neurol Neurosurg Psychiatry. 2018;89:611–7.
Heinz A, Murray GK, Schlagenhauf F, Sterzer P, Grace AA, Waltz JA. Towards a unifying cognitive, neurophysiological and computational neuroscience account of schizophrenia. Schizophr Bull. 2019;45:1092–100.
Buckley P, Stack JP, Madigan C, O’Callaghan E, Larkin C, Redmond O, et al. Magnetic resonance imaging of schizophrenia-like psychoses associated with cerebral trauma: clinicopathological correlates. Am J Psychiatry. 1993;150:146–8.
Camara-Lemarroy CR, Ibarra-Yruegas BE, Rodriguez-Gutierrez R, Berrrios-Morales I, Ionete C, Riskind P. The varieties of psychosis in multiple sclerosis: a systematic review of cases. Mult Scler Relat Disord. 2017;12:9–14.
Mega MS, Lee L, Dinov ID, Mishkin F, Toga AW, Cummings JL. Cerebral correlates of psychotic symptoms in Alzheimer’s disease. J Neurol Neurosurg Psychiatry. 2000;69:167–71.
Murray PS, Kumar S, Demichele-Sweet MA, Sweet RA. Psychosis in Alzheimer’s disease. Biol Psychiatry. 2014;75:542–52.
Waddington JL, Youssef HA, Farrell MA, Toland J. Initial ‘schizophrenia-like’ psychosis in Pick’s disease: case study with neuroimaging and neuropathology, and implications for frontotemporal dysfunction in schizophrenia. Schizophr Res. 1995;18:79–82.
Galimberti D, Dell’Osso B, Altamura AC, Scarpini E. Psychiatric symptoms in frontotemporal dementia: epidemiology, phenotypes, and differential diagnosis. Biol Psychiatry. 2015;78:684–92.
Rocha NP, Mwangi B, Gutierrez-Candano CA, Sampaio C, Furr Stimming E, Teixeira AL. The clinical picture of psychosis in manifest Huntington’s disease: A comprehensive analysis of the Enroll-HD database. Front Neurol. 2018;9:930.
Madhusoodanan S, Sting MB, Farah T, Ugur U. Psychiatric aspects of brain tumors: a review. World J Psychiatry. 2015;5:273–85.
Upthegrove R, Marwaha S, Birchwood M. Depression and schizophrenia: cause, consequence or trans-diagnostic issue? Schizophr Bull. 2017;34:240–4.
Tu PC, Bai YM, Li CT, Chen MH, Lin WC, Chang WC, et al. Identification of common thalamocortical dysconnectivity in four major psychiatric disorders. Schizophr Bull. 2019;45:1143–51.
Ma Q, Tang Y, Wang F, Liao X, Jiang X, Wei S, et al. Transdiagnostic dysfunctions in brain modules across patients with schizophrenia, bipolar disorder, and major depressive disorder: a connectome-based study. Schizophr Bull 2019. https://doi.org/10.1093/schbul/sbz111.
Wang S, Mao S, Xiang D, Fang C. Association between depression and the subsequent risk of Parkinson’s disease: a meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry. 2018;86:186–92.
Fusar-Poli P, Borgwardt S, Bechdolf A, Addington J, Riecher-Rossler A, Schultze-Lutter F, et al. The psychosis high-risk state: a comprehensive state-of-the-art review. JAMA Psychiatry. 2013;70:107–20.
van Os J, Reininghaus U. Psychosis as a transdiagnostic and extended phenotype in the general population. World Psychiatry. 2016;15:118–24.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Rights and permissions
About this article
Cite this article
Waddington, J.L. Psychosis in Parkinson’s disease and parkinsonism in antipsychotic-naive schizophrenia spectrum psychosis: clinical, nosological and pathobiological challenges. Acta Pharmacol Sin 41, 464–470 (2020). https://doi.org/10.1038/s41401-020-0373-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41401-020-0373-y
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
Drug contraindications in comorbid diseases: a protein interactome perspective
Network Modeling Analysis in Health Informatics and Bioinformatics (2024)
-
Facial and upper-limb movement abnormalities in individuals with psychotic-like experiences: a motion analysis study
European Archives of Psychiatry and Clinical Neuroscience (2023)
-
Extrapyramidal symptoms predict cognitive performance after first-episode psychosis
Schizophrenia (2022)
-
Characterizing the sensorimotor domain in schizophrenia spectrum disorders
European Archives of Psychiatry and Clinical Neuroscience (2022)
-
RNA-seq analysis of gene expression profiles in posttraumatic stress disorder, Parkinson’s disease and schizophrenia identifies roles for common and distinct biological pathways
Discover Mental Health (2022)