Abstract
Background
Large-for-gestational age (LGA), a marker of fetal overgrowth, has been linked to obesity in adulthood. Little is known about how infancy growth trajectories affect adiposity in early childhood in LGA.
Methods
In the Shanghai Birth Cohort, we followed up 259 LGA (birth weight >90th percentile) and 1673 appropriate-for-gestational age (AGA, 10th–90th percentiles) children on body composition (by InBody 770) at age 4 years. Adiposity outcomes include body fat mass (BFM), percent body fat (PBF), body mass index (BMI), overweight/obesity, and high adiposity (PBF >85th percentile).
Results
Three weight growth trajectories (low, mid, and high) during infancy (0–2 years) were identified in AGA and LGA subjects separately. BFM, PBF and BMI were progressively higher from low- to mid-to high-growth trajectories in both AGA and LGA children. Compared to the mid-growth trajectory, the high-growth trajectory was associated with greater increases in BFM and the odds of overweight/obesity or high adiposity in LGA than in AGA children (tests for interactions, all P < 0.05).
Conclusions
Weight trajectories during infancy affect adiposity in early childhood regardless of LGA or not. The study is the first to demonstrate that high-growth weight trajectory during infancy has a greater impact on adiposity in early childhood in LGA than in AGA subjects.
Impact
-
Large-for-gestational age (LGA), a marker of fetal overgrowth, has been linked to obesity in adulthood, but little is known about how weight trajectories during infancy affect adiposity during early childhood in LGA subjects.
-
The study is the first to demonstrate a greater impact of high-growth weight trajectory during infancy (0–2 years) on adiposity in early childhood (at age 4 years) in subjects with fetal overgrowth (LGA) than in those with normal birth size (appropriate-for-gestational age).
-
Weight trajectory monitoring may be a valuable tool in identifying high-risk LGA children for close follow-ups and interventions to decrease the risk of obesity.
Similar content being viewed by others
Introduction
Childhood obesity rates have been on the rise over the last five decades globally.1 Children in East and South Asia—comprised mostly of middle- and low-income countries—have experienced a faster increase in body mass index than in other regions. From 1975 to 2016, the global age-standardized obesity prevalence increased from 0.7% to 5.6% for girls and from 0.9% to 7.8% for boys.2 Obesity in childhood increases the risks of high blood pressure and high cholesterol—the major risk factors of coronary heart disease and type 2 diabetes.3,4
Large-for-gestational age (LGA), an indicator of excessive fetal growth, is commonly defined as birth weight that exceeds the 90th percentile of the reference values for infants of the same sex and gestational age.5 The rapid economic development in China over recent decades is accompanied by rapid increases in maternal obesity and LGA births according to the WHO birth weight standards.6 LGA is associated with elevated risks of obesity and related metabolic dysfunctional disorders in childhood and later life.7,8,9,10
Growth pattern in early postnatal life may be an important determinant of adiposity development. Rapid weight gain during infancy (the first 2 years of age) has been associated with an increased risk of overweight or obesity in childhood, adolescence, and adulthood.11,12,13,14,15 Rapid and stable-to-high growth patterns during infancy have been associated with an elevated risk of overweight or obesity in childhood or adolescence.16,17,18 Assessing the effects of growth trajectory pattern as a whole could be informative for identifying infants at high risk for childhood obesity.16,19 There is a lack of knowledge on growth trajectories during infancy in relation to adiposity in early childhood in LGA children. In the present study, we sought to assess the impacts of infancy growth trajectories on adiposity in early childhood (age 4 years) in LGA and birth weight appropriate for gestational age (AGA, control) children.
Methods
Study design and population
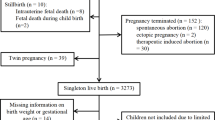
This was a follow-up cohort study in the Shanghai Birth Cohort (SBC).20 The SBC is a prospective birth cohort including 4127 pregnant women in Shanghai, 2013–2016. The study was approved by the research ethics committees of Xinhua Hospital (the coordination center, ref no. M2013-010) and all participating hospitals in Shanghai. Written informed consent was obtained from all study participants.
There were a total of 2207 singleton children with follow-up data at age 4 years in the SBC. We excluded children with missing data on sex, birth weight or gestational age (n = 138), as well as SGA infants (n = 104), leaving 263 LGA and 1702 AGA children for the present study. We further excluded children with birth defects (n = 17) or deliveries at gestational age <34 weeks (n = 16; very preterm infants may have differential growth patterns). The final study sample included 259 LGA and 1673 AGA children. A flowchart illustrating the selection of study subjects is presented in Fig. S1.
Growth measurements and definitions
LGA was defined as birth weight >90th percentile, and appropriate-for-gestational age (AGA) as birth weight between 10th and 90th percentiles, according to the 2015 Chinese sex- and gestational age-specific birth weight standards.21 Gestational age was ascertained according to the date of last menstruation period and confirmed by first-trimester ultrasound dating. If the difference between the two estimates was more than 2 weeks, the ultrasound dating-based estimate was used.
Length (age 0–2 years, by seca 416 infantometer, Seca Netherlands, Hamburg) or height (age 4 years, by a wall-mounted standiometer) and weight (by an electronic weighing device at birth, by seca 376 at age 6 weeks to 2 years, or seca 799 weighing scale at age 4 years) were measured in infants/children during physical examinations following standardized operation protocols at birth and follow-ups at ages 6 weeks, 6 months, 1, 2 and 4 years. Length or height was measured to the nearest millimeter. Weight was measured to the nearest gram at birth, and the nearest 100 g from age 6 weeks to 4 years. Body mass index (BMI) was calculated as weight in kg/(height in meter)2.2
Body composition in children at age 4 years was measured by bioelectrical impedance analysis (BIA) using the InBody720 (InBody Ltd., Seoul, KOREA). The measurements include body fat mass (BFM), fat free mass (FFM), and percent body fat (PBF).
Overweight/obesity in early childhood was defined as BMI>85th percentile at age 4 years according to sex-specific cutoffs for Chinese children.22 High adiposity in early childhood was defined as PBF>85th percentile, according to the sex-specific cut-off values among term AGA children at age 4 years (n = 1606) in the study cohort.
Outcomes
The primary outcomes were BFM, PBF at age 4 years. Secondary outcomes included BMI, FFM, high adiposity (PBF>85th percentile), and overweight/obesity at age 4 years.
Statistical analysis
All analyses were performed using SAS (Statistical Analysis System) version 9.4 (SAS Institute Inc., Cary, NC). Mean ± standard deviation (SD) and frequency (%) were presented for continuous and categorical study variables, respectively. The SAS macro package of the World Health Organization (WHO) was used in calculating sex-specific weight-for-age z (WAZ) scores and BMI-for-age z scores. The SAS PROC TRAJ23,24 was used to model the weight growth trajectories during infancy (birth to age 2 years) in LGA and AGA subjects separately. The group-based trajectory modeling assumes there are discrete underlying groups (trajectories) in the study population. The optimal number of trajectories and shape (constant, linear, quadratic, cubic, or quadratic terms) were selected by the Bayesian Information Criterion (BIC). A lower Bayesian information criterion value indicates a more parsimonious model. The suitability of the model was confirmed by evaluating the average posterior probability of trajectory assignments (>0.70) and the odds of correct classification (>5.0).
In addition to the primary analyses using the growth trajectory analysis approach, we also analyzed the effects of infancy growth patterns using the conventional approach comparing growth deceleration or acceleration vs. normal growth during early infancy (birth to 6 months) and late infancy (6 months to 2 years), based on the changes in WAZ scores: deceleration (ΔWAZ < −0.67), normal (ΔWAZ −0.67 to 0.67), acceleration (ΔWAZ > 0.67).15
There were some missing data for maternal pre-pregnancy BMI (12.7%) and child’s weights between birth and age 4 years (9.8–14.8%). Multiple imputations with the fully conditional specification (FCS) method25 were conducted to handle missing data. The pooled statistical parameter estimates from 25 imputed datasets were presented. Generalized linear models were used to assess the differences in continuous adiposity outcomes (BMI, PBF, FFM, and BFM) at age 4 years comparing infancy growth trajectories or patterns in LGA and AGA infants separately. Logistic regression models were fitted to estimate the odds ratios (ORs) of overweight/obesity and high adiposity at age 4 years. The adjusted effect estimates were controlled for maternal and infant characteristics that might confound the relationships. The adjusted effect estimates were controlled for pre-specified known risk factors of child adiposity/obesity, including maternal BMI, gestational diabetes, c-section delivery, infant sex, and breastfeeding. Other factors would be included in the adjusted analyses if they affected any adiposity outcome (BMI, PBF) in early childhood (age 4 years) at P < 0.2. Interaction effects between growth trajectory or pattern and LGA status were checked in regression models. P values <0.05 were considered statistically significant.
Results
Maternal and infant/child characteristics
Table 1 presents the descriptive statistics on maternal and infant/child characteristics. Maternal age, ethnicity, education, parity, pre-pregnancy BMI, and smoking were similar in LGA and AGA groups. As expected, weight and length or height were significantly higher in LGA vs. AGA infants/children from birth to age 4 years.
Adiposity and body composition at early childhood
At age 4 years, BMI (crude value or z score), PBF, FFM, and BFM were higher, while overweight/obesity was much more frequent (29.7% vs 17.0%) comparing LGA vs. AGA groups (Table 2).
Infancy growth trajectories
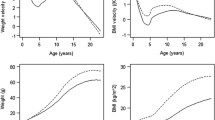
The weight growth trajectories during infancy (the first 2 years of life) in LGA and AGA infants are presented in Fig. 1. In both LGA and AGA infants, three growth trajectories were identified: low-, mid-, and high-growth trajectories. In AGA infants, the high-growth trajectory showed a rise in weight z scores from birth to age 12 months, reaching high weight z scores (mean >2) at age 12 months, and later a mild fall between age 1 and 2 years, while the mid- and low-growth trajectories were relatively steady in weight z scores from birth to age 2 years. In LGA infants, the high trajectory was characterized by persistently high weight z scores from birth to age 2 years, while the low- and mid-growth trajectories showed a fall in weight z scores from birth to age 6 months, and thereafter relatively steady to age 2 years.
Infancy growth trajectory in relation to adiposity at early childhood
Average BMI (crude value or z score), PBF, BFMS, FFM, as well as the rates of overweight/obesity and high adiposity at age 4 years were progressively higher from low- to mid- to high-growth trajectories during infancy in both AGA and LGA infants (all P < 0.01, Table 3 and Fig. 2). In LGA children, the high-growth trajectory during infancy was associated with greater increases in PBF (4.58%, 95% CI: 0.25–8.91%) at age 4 years relative to the mid-growth trajectory than in AGA children (3.29%, 95% CI: 1.51–5.07%) (P for interaction = 0.08). Similarly, the high-growth trajectory during infancy was associated with greater increases in BFM at age 4 years in LGA children (BFM: 1.31 kg, 95% CI: 0.19–2.43 kg) than in AGA children (BFM: 1.02 kg, 95% CI: 0.58–1.45 kg) (P for interaction <0.001). In AGA children, the low-growth trajectory was associated with lower BMI, and the high-growth trajectory with higher BMI relative to the mid-growth trajectory at age 4 years, while in LGA children, the associations were in the same direction but did not reach statistical significance for the low-growth trajectory.
The high-growth trajectory during infancy was associated with increased risks of overweight/obesity and high adiposity at age 4 years relative to the mid-growth trajectory in both LGA and AGA children (Table 4). The risk increases were greater in LGA children than in AGA children for either overweight (AGA: OR = 2.67; LGA: OR = 4.30) or high adiposity (AGA: OR = 2.18; LGA: OR = 2.57) (P for interaction <0.001 for both).
The effects of growth acceleration or deceleration during early and late infancy
The growth acceleration during late infancy (6-24 months) was associated with greater increase in BFM, PBF, BMI (crude value or z score) at age 4 years in LGA children than in AGA children (tests for interaction, all P < 0.001), while there were no differential associations for growth acceleration or deceleration during early infancy (tests for interactions, all P > 0.05) (Appendix Table S1). The estimated adjusted ORs suggested greater impacts of growth acceleration during early or later infancy on the risks of overweight/obesity and high adiposity at age 4 year in LGA children than in AGA children, but most effect estimates did not reach statistical significance with wide confidence intervals in the LGA group (Appendix Table S2). The previous growth trajectory analysis approach demonstrated a clearer picture of subjects at risk of obesity/high adiposity in early childhood than the conventional growth acceleration/deceleration analysis approach.
Discussion
Main findings
This is the first study on the impact of infancy (age 0–2 years) growth trajectory on PBF and BFM in early childhood in LGA subjects. Infancy growth trajectories were strongly predictive of PBF, BFM and BMI at age 4 years, and the prevalence rates of early childhood obesity or high adiposity rose progressively from low to mid to high-growth trajectories regardless of LGA or AGA. However, the high-growth trajectory was associated with greater increases in adiposity and the risk of overweight/obesity or high adiposity in early childhood in LGA than AGA subjects. The growth trajectory analysis approach appears to better capture LGA infants at risk of obesity/ high adiposity in early childhood than the conventional deceleration or acceleration vs. normal growth analysis approach.
Data interpretation
At age 4 years, LGA children were much more likely to be obese than AGA children, and had higher PBF, BFM, FFM, and BMI than AGA children. This is in line with previous studies demonstrating a connection between birth weight and BMI, body fat percentage, and fat mass in childhood, adolescence, and adults,26,27,28 and between LGA and obesity in adulthood.29
Only a few studies have examined how infancy growth trajectories affect adiposity in early childhood. In a population-based birth cohort, “persistent weight gain” trajectory (persistently higher weight than average) was associated with higher BMI at early childhood (age 4 years), and higher BMI and body fat (by dual‐energy X‐ray absorptiometry (DXA)) at late childhood (age 7 years).30 In LGA children, those with persistently high weight levels without catch-down growth during the first 2–3 years of life had an increased risk of obesity in early childhood (age 4 years)31 or late childhood (age 7 years).19 Our study is the first to assess infancy growth trajectory in relation to PBF and BFM in early childhood (age 4 years) in LGA subjects. PBF and BFM are more objective measurements of adiposity than BMI. Notably, the high-growth trajectory was found to be associated with higher PBF and BFM in both AGA and LGA children at age 4 years, but the increases in PBF and BFM were to a much greater extent in LGA children than in AGA children. Consistent with our finding in LGA children, small-for-gestational age (SGA) children who had a high-growth trajectory during infancy (the first 2 years of life) tend to have a higher BMI and an increased risk of overweight/obesity in early childhood (at age 5 years).16
Accelerated growth in infancy (0–2 years) has been associated with an increased risk of overweight/obesity from childhood to adulthood (pooled OR = 3.66) in a meta-analysis.13 Consistent with our data, accelerated growth during infancy (0–24 months) has been associated with higher BMI and increased likelihood of overweight (OR: 3.1) in early childhood (at age 4) even in children with normal birth weight (AGA).31 Moreover, we observed a greater impact of accelerated growth during infancy on adiposity in early childhood (at age 4) in LGA than AGA children (Table S1), especially for accelerated growth during late infancy (6–24 months). Accelerated growth during early infancy (0 to 6 or 12 months) has been linked to greater BMI, fat mass, and percent body fat in adolescents and young adults.32,33 The impact on adiposity (BMI, subcutaneous fat) in late childhood (age 6 years) has been reported to be greater for weight z score gains in late infancy (1–2 years) than in early infancy (0–6 months).15 Similarly, the impact on adiposity (% fat mass) in adolescence (at age 12 years) has been reported to be greater for accelerated growth during late infancy (6–18 months, +2.6%) than during early infancy (0–6 months, +1.4%).34
Our study is the first to report a greater impact of accelerated growth during late infancy on adiposity (PBF) in early childhood in LGA than AGA children. An individual-level data meta-analysis of 10 cohorts reported similarly elevated risks of childhood obesity (OR close to 2) for accelerated growth (one weight z score increment) during infancy (0–1 year) regardless of birth weight (low, normal or high).35 We observed in general comparable effect point estimates for accelerated growth (0.67 weight z score increment) during early infancy (0–6 months) and late infancy (6–24 months) in AGA children (OR for overweight/obesity: close to 1.9), but larger point effect estimates in LGA children (OR for overweight/obesity: close to 4), suggesting a greater impact in LGA subjects, but caution is warranted in data interpretation due to the wide confidence intervals.
Limitations
The study is observational in nature, and reverse causality could not be ruled out. The study endpoints were adiposity indicators in early childhood at age 4 years. Further studies are warranted to understand whether the observed effects of infancy growth trajectories may persist into late childhood and beyond. All study subjects were Chinese of Han ethnicity. This would enhance the power to detect an association with infancy growth trajectory or pattern due to a homogenous study population, but more studies in other ethnic populations are warranted to understand the generalizability of the study findings.
Conclusions
Infancy growth trajectories affect adiposity in early childhood regardless of LGA or AGA. Weight high-growth trajectory during infancy may have a greater impact on adiposity in early childhood in LGA than AGA children. Weight growth trajectory monitoring in infants is convenient in clinical settings, and may be a valuable tool in identifying high-risk children for close follow-ups and interventions to reduce the risk of obesity.
Data availability
As access to the deidentified participant research data must be approved by the research ethics board on a case-by-case basis, please contact the corresponding authors (zc.luo@utoronto.ca; feili@shsmu.edu.cn; srachel@126.com) for assistance in data access request.
References
Ng, M. et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384, 766–781 (2014).
Abarca-Gómez, L. et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet 390, 2627–2642 (2017).
Falkner, B. & Gidding, S. Life-course implications of pediatric risk factors for cardiovascular disease. Can. J. Cardiol. 37, 766–775 (2021).
Marcus, C., Danielsson, P. & Hagman, E. Pediatric obesity-long-term consequences and effect of weight loss. J. Intern. Med. 292, 870–891 (2022).
Bocca-Tjeertes, I. F. et al. Growth patterns of large for gestational age children up to age 4 years. Pediatrics 133, e643–e649 (2014).
Harvey, L., van Elburg, R. & van der Beek, E. M. Macrosomia and large for gestational age in Asia: one size does not fit all. J. Obstet. Gynaecol. Res. 47, 1929–1945 (2021).
Palatianou, M. E., Simos, Y. V., Andronikou, S. K. & Kiortsis, D. N. Long-term metabolic effects of high birth weight: a critical review of the literature. Horm. Metab. Res. 46, 911–920 (2014).
Yu, Z. B. et al. Birth weight and subsequent risk of obesity: a systematic review and meta-analysis. Obes. Rev. 12, 525–542 (2011).
Johannsson, E., Arngrimsson, S. A., Thorsdottir, I. & Sveinsson, T. Tracking of overweight from early childhood to adolescence in cohorts born 1988 and 1994: overweight in a high birth weight population. Int. J. Obes. 30, 1265–1271 (2006).
Gu, S. et al. Risk factors and long-term health consequences of macrosomia: a prospective study in Jiangsu Province, China. J. Biomed. Res. 26, 235–240 (2012).
Woo, J. G. Infant growth and long-term cardiometabolic health: a review of recent findings. Curr. Nutr. Rep. 8, 29–41 (2019).
Corvalan, C., Kain, J., Weisstaub, G. & Uauy, R. Impact of growth patterns and early diet on obesity and cardiovascular risk factors in young children from developing countries. Proc. Nutr. Soc. 68, 327–337 (2009).
Zheng, M. et al. Rapid weight gain during infancy and subsequent adiposity: a systematic review and meta-analysis of evidence. Obes. Rev. 19, 321–332 (2018).
Gatjens, I. et al. Relationship between birth weight, early growth rate, and body composition in 5- to 7-year-old children. Obes. Facts 15, 519–527 (2022).
Gishti, O. et al. Fetal and infant growth patterns associated with total and abdominal fat distribution in school-age children. J. Clin. Endocrinol. Metab. 99, 2557–2566 (2014).
Shi, H. et al. Insights into infancy weight gain patterns for term small-for-gestational-age babies. Nutr. J. 17, 1–9 (2018).
Norris, T. et al. Early childhood weight gain: latent patterns and body composition outcomes. Paediatr. Perinat. Epidemiol. 35, 557–568 (2021).
Matsumoto, N. et al. Trajectory of body mass index and height changes from childhood to adolescence: a nationwide birth cohort in Japan. Sci. Rep. 11, 1–10 (2021).
Lei, X. et al. Childhood health outcomes in term, large-for-gestational-age babies with different postnatal growth patterns. Am. J. Epidemiol. 187, 507–514 (2018).
Zhang, J. et al. Cohort profile: the Shanghai Birth Cohort. Int. J. Epidemiol. 48, 1–8 (2019).
Zhu, L. et al. [Chinese neonatal birth weight curve for different gestational age]. Chin. J. Pediatr. 53, 97–103 (2015).
Li, H., Ji, C., Zong, X. & Zhang, Y. [Body mass index growth curves for Chinese children and adolescents aged 0 to 18 years]. Chin. J. Pediatr. 47, 493–498 (2009).
Tasdelen, B., Ozge, A., Kaleagasi, H., Erdogan, S. & Mengi, T. Determining of migraine prognosis using latent growth mixture models. Chin. Med. J. 124, 1044–1049 (2011).
Arrandale, V., Koehoorn, M., MacNab, Y. & Kennedy, S. M. How to use SAS® Proc Traj and SAS® Proc Glimmix in respiratory epidemiology. https://open.library.ubc.ca/media/stream/pdf/52383/1.0048205/1 (2006).
van Buuren, S. Multiple imputation of discrete and continuous data by fully conditional specification. Stat. Methods Med. Res. 16, 219–242 (2007).
Hill, D. J. et al. Relationship between birth weight and metabolic status in obese adolescents. ISRN Obes. 2013, 1–8 (2013).
Olander, R. F. W., Sundholm, J. K. M., Suonsyrja, S. & Sarkola, T. Arterial health during early childhood following abnormal fetal growth. BMC Pediatr. 22, 1–12 (2022).
Honda, M. et al. Birth weight was associated positively with gluteofemoral fat mass and inversely with 2-h postglucose insulin concentrations, a marker of insulin resistance, in young normal-weight Japanese women. Diabetol. Int. 13, 375–380 (2022).
Derraik, J. G. B. et al. Large-for-gestational-age phenotypes and obesity risk in adulthood: a study of 195,936 women. Sci. Rep. 10, 1–9 (2020).
Santos-Silva, R. et al. Persistent weight gain between 0 and 4 years of age is associated with higher dehydroepiandrosterone sulphate levels at 7 years old: data from the Generation XXI birth cohort. Clin. Endocrinol. 97, 588–595 (2022).
Taal, H. R., Vd Heijden, A. J., Steegers, E. A., Hofman, A. & Jaddoe, V. W. Small and large size for gestational age at birth, infant growth, and childhood overweight. Obesity 21, 1261–1268 (2013).
Salgin, B. et al. Even transient rapid infancy weight gain is associated with higher BMI in young adults and earlier menarche. Int. J. Obes. 39, 939–944 (2015).
Ekelund, U. et al. Upward weight percentile crossing in infancy and early childhood independently predicts fat mass in young adults: the Stockholm Weight Development Study (SWEDES). Am. J. Clin. Nutr. 83, 324–330 (2006).
Samaranayake, D. et al. Association between early weight gain and later adiposity in Sri Lankan adolescents. J. Dev. Orig. Health Dis. 12, 250–259 (2021).
Druet, C. et al. Prediction of childhood obesity by infancy weight gain: an individual-level meta-analysis. Paediatr. Perinat. Epidemiol. 26, 19–26 (2012).
Acknowledgements
We gratefully acknowledged all research staff who had contributed to patient recruitment and data collection in the Shanghai Birth Cohort.
Funding
This study was supported by grants from the Ministry of Science and Technology of China (2019YFA0802501), National Natural Science Foundation of China (81930095, 82125032, 82204064, 81903323, 81803244, 81761128035), the Shanghai Science and Technology Commission (19410713500 and 2018SHZDZX01), the Shanghai Municipal Health Commission (GWV-10.1-XK07, 2020CXJQ01, 2018YJRC03), the Shanghai Clinical Key Subject Construction Project (shslczdzk02902), the Shanghai Pujiang Program (22PJD045), and the Canadian Institutes of Health Research (158616). The funders have no role in all aspects of the study, including study design, data collection and analysis, the preparation of the manuscript and the decision for publication.
Author information
Authors and Affiliations
Consortia
Contributions
Z.-C.L., F.L., X.-H.S., J.Z., and F.O. conceived the study. M.-Y.T., X.L., Z.-L.C., M.-N.Y., Y.-J.X., H.H., F.F., Q.C., X.-X.M., J.Z., F.O., X.-H.S., F.L., and Z.-C.L. contributed to the acquisition of research data. M.-Y.T. and X.L. conducted the literature review, data analysis, and drafted the manuscript. All authors contributed in revising the article critically for important intellectual content, and approved the final version for publication. Z.-C.L. is the guarantor who takes full responsibility for the work as a whole.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
The study was approved by the research ethics committees of Xinhua Hospital (the coordination center, reference number M2013-010) and all participating hospitals. Written informed consent was obtained from all study participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tao, MY., Liu, X., Chen, ZL. et al. Fetal overgrowth and weight trajectories during infancy and adiposity in early childhood. Pediatr Res 95, 1372–1378 (2024). https://doi.org/10.1038/s41390-023-02991-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-023-02991-7
- Springer Nature America, Inc.