Abstract
Objectives
To evaluate punctate white matter lesion (PWML) influence in preterm infants on the long-term neurodevelopmental outcome (NDO).
Methods
PubMed and EMBASE were searched from January 1, 2000, to May 31, 2021. Studies were included in which PWML in preterm infants on MRI around term-equivalent age (TEA) and NDO at ≥12 months were reported. Study and patient characteristics and NDO on motor, cognitive, and behavioral domains were extracted. The quality of studies was assessed using the Cochrane-approved Quality in Prognosis Studies tool.
Results
This analysis included nine studies with a total of 1655 patients. Mean incidence of isolated PWML was 22.1%. All studies showed a relationship between PWML and motor delay. Two studies found a significant correlation between cognitive and behavioral outcomes and PWML. Number and PWML location are related to severity and impairment types.
Limitations
PWML were not always separately described from generalized WMI, only studies with imaging around TEA were included, and studies were heterogenic in design and quality.
Conclusions
PWML is common in preterm infants and predictive of adverse NDO, in particular on motor outcomes and less on cognitive and behavioral outcomes. The type and severity of impairments are related to the number and location of PMWL.
Impact
-
PWML is common in preterm infants and seems predictive of adverse NDO.
-
DWI and SWI MRI sequences are informative because the different patterns suggest a difference in the underlying pathology.
-
The type and severity of impairments are related to the number and location of PMWL.
-
Our review can inform clinicians and parents about the NDO of preterm infants with a diagnosis of PWML.
-
Prospective neuroimaging case–control cohort studies are recommended.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Approximately one in ten babies worldwide is born prematurely (<37 weeks of gestation). This translates to an estimated 15 million preterm births every year.1,2 Despite improved survival rates due to major advances in fetal and neonatal care, preterm infants still face a high risk of neurodevelopmental impairments.3,4,5,6 In long-term follow-up, 1–7%, 5–10%, and 15–50% of the preterm infants develop cerebral palsy (CP), exhibit minor motor deficiencies, and have cognitive, behavioral, and social difficulties, respectively.2,6,7,8 There is an inverse relationship between gestational age and risk for neurodevelopmental impairment.6,8,9,10,11 However, the neuropathology underlying these reported disabilities remains largely unclear. Thus, accurately predicting neurodevelopmental outcomes (NDOs) based on brain injury patterns diagnosed with neuroimaging is often difficult for clinicians.8
Two types of overt brain injury in infants born preterm are (cystic) periventricular leukomalacia (c-PVL) and severe intraventricular hemorrhage (IVH). Both are associated with major adverse motor and cognitive outcomes. However, they do not account for all of the reported adverse NDO following preterm birth because the incidence of these severe complications of preterm birth is relatively low.8,12,13 More subtle white matter (WM) injury, such as punctate white matter lesions (PWML), has a far higher incidence than c-PVL and severe IVH, especially in late preterm infants, and also seems to be associated with long-term cognitive and behavioral problems.14,15
Cranial ultrasound (cUS) is the most readily available technique for routine (serial) neonatal brain imaging. It reliably detects major lesions, including c-PVL and IVH, but is less accurate in diagnosing diffuse and more subtle WM lesions (e.g., PWML). Several studies showed that magnetic resonance imaging (MRI) of the brain around term-equivalent age (TEA) provides complementary information to serial cUS and clinical examination and can improve the prediction of long-term NDO.14,15,16,17,18,19 Initially, conventional image sequences (e.g., T1- and T2-weighted sequences), and, recently, more advanced sequences, such as diffusion tensor imaging (DTI) and susceptibility-weighted imaging (SWI), have provided new insights into brain development and pathology in infants born preterm.15,20 With these more advanced MRI sequences, the focus on brain imaging research has shifted to the detection of more subtle abnormalities such as focal PWML.16,21,22
PWML, first described by Cornette et al.,23 are small, focal lesions of the cerebral WM typically seen on MRI as hyperintense and hypointense on T1- and T2-weighted imaging, respectively (Fig. 1). Different PWML forms have been described, including variations in pattern (isolated, linear, or in clusters20) location (along the corona radiata, in the centrum semiovale, in the posterior periventricular WM, or along the optic radiation), DWI or SWI visibility, and number or size.7,8 The reported PWML incidence ranges from 20 to >50% in premature infants.15,24,25,26 The exact PWML pathophysiology remains unresolved, although they might represent milder PVL forms.15 Histological studies of focal WM injuries show microhemorrhage changes, forming microcalcifications and necrotic areas as well as cellular reactions of glial cells, which are related to PVL.21,27,28,29 Wagenaar et al.25 investigated the PWML risk factors to understand the pathophysiology better. They identified several risk factors (e.g., greater birth weight, IVH grades 2–3, and center of birth). Moreover, Parodi et al. and Malova et al.30,31 showed a variation in risk factors depending on the visibility of the lesions on the SWI sequence.
PWML are now more commonly recognized and the prognosis of these typical lesions is determined by early management, particularly in the first year.32 After a diagnosis of brain injury appropriate referrals can be made (e.g., physiotherapist) that could allow the application of targeted interventions. Several studies have reported the PWML outcome, and therefore this study aims to perform a systematic review of the existing literature to evaluate the relationship between PWML in preterm infants and the NDO in infants at a minimum age of 12 months, specifically focusing on the motor, behavioral, and cognitive outcomes.
Methods
The study complied with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.33
Eligibility criteria
Articles fulfilled inclusion criteria when infants born preterm (i.e., born <37 weeks of gestation) were diagnosed with PWML on MRI scans made around the term date and when the NDO (minimum age of 12 months) with a standardized and validated scale was reported. PWML were defined as isolated areas of high or low signal on T1- and T2- weighted imaging, respectively. Only articles with WM abnormalities reported separately from other overt brain injuries were included. Overt brain injuries were defined as IVH ≥grade 3 (according to Papile et al.34), (posthemorrhagic) ventricular dilatation, c-PVL, and cerebellar hemorrhages. Case reports and articles written in languages other than English were excluded. Despite most studies of NDO use 24 months as follow-up age, we chose 12 months so potentially valuable articles were not excluded on the basis of a follow-up period that is too short.
Search
PubMed (MEDLINE) and EMBASE were searched in April 2019 for relevant papers in English starting from January 2000 with similar search strategies. An updated search in June 2021 did not provide any new articles. For PubMed, the search was (((((((((birth, preterm[MeSH Terms] OR infant, premature[MeSH Terms] OR intensive care units, neonatal[MeSH Terms] OR prematur*[Title/Abstract] OR preterm*[Title/Abstract]))))) AND ((((pwml[Title/Abstract]) OR (((punctat*[Title/Abstract]) AND white matter[Title/Abstract]) AND lesion*[Title/Abstract])) OR ((white matter[Title/Abstract]) AND ((lesion*[Title/Abstract]) OR injury*[Title/Abstract]))))))) AND ((((“Neurodevelopmental Disorders”[Mesh] OR neurodevelopment*[Title/Abstract] OR cognit*[Title/Abstract] OR motor*[Title/Abstract] OR behavior*[Title/Abstract] OR behavior*[Title/Abstract]))))). Reference lists of selected articles were screened for additional studies.
Study selection
After duplicate removal, two authors (C.A.M.d.B. and S.D.M.) independently screened the titles and abstracts to evaluate potential eligibility in the study using Rayyan QCRI (2016, Qatar).35 The title and abstract screening was followed by analyses of full texts while assessing the inclusion and exclusion criteria.
Data collection process and data items
Study design, cohort year, inclusion and exclusion criteria, number of patients with PWML, PWML incidence, number of selected infants, neuroimaging, definitions used for WMI, NDO scale and definitions for adverse outcomes, patient characteristics (gestational age at delivery, birth weight, patient’s age at MRI, and children’s age at the time of neurodevelopmental evaluation), and study results (association between PWML and NDO and adjusted confounders) were the study characteristics extracted using a data extraction form.
Only PWML diagnoses using TEA MRI were used if studies reported different PWML incidences due to different timing of MRI. The incidence of isolated PWML was calculated as the number of patients with isolated WM lesions on MRI divided by the total number of patients for each study and all studies combined. Only the patients with PWML and follow-up data were described under the number of selected infants. Patients with other types of brain injury, except IVH grades 1–2, were not included. Not all studies specifically reported the absence of other brain lesions besides PWML. Patients were excluded if different forms of WMI were combined in a scoring system, and the number of patients with PWML could not be traced. No authors were contacted to obtain missing data.
Risk of bias in individual and across studies
The quality of the studies was assessed with the Cochrane-approved Quality in Prognosis Studies tool.36 All domains were scored as low, moderate, or high risk. Each domain consisted of multiple items. A domain was considered at low risk of bias if most items were properly covered. To synthesize the quality of the studies and to be able to conclude the relationship between PWML and NDO, a best-evidence synthesis was applied consisting of the following three levels: (1) strong evidence—consistent findings in multiple (≥2) high-quality studies; (2) moderate evidence—consistent findings in one high-quality and at least one moderate- or low-quality study, or consistent findings in multiple moderate- or low-quality studies; and (3) insufficient evidence—only one study available or inconsistent findings in multiple (≥2) studies.37
Summary measures
The principal summary measure was the NDO at a minimum age of 12 months, specifically focusing on the motor, cognitive, and behavioral impairments. Data were often presented as the number of patients with an adverse outcome and a mean score of the total scale. In addition, different outcome measures were used. This made it difficult to summarize the outcome for each domain. A meta-analysis could not be performed because of the heterogeneity in the included studies. Therefore, the results are written in a narrative.
Results
Study selection
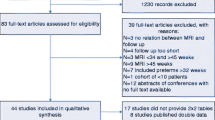
The original PubMed and EMBASE search obtained 2993 results. After removing duplicates (n = 501), 2492 articles were screened for title and abstract. Of these, 80 articles potentially met the inclusion criteria. After analyzing the full text, nine articles were included in the analysis. Figure 2 shows the reasons for exclusion.
Study characteristics
Table 1 summarizes the characteristics of the nine included studies. From a total cohort of 1655 patients, 365 patients had PWMLs, leading to an incidence of 22.1% (range in individual studies, 7.1–59%). The study sample size ranged from 9 to 120 infants, with a follow-up period ranging from 18 to 40 months.
Different classification systems for WMI were used in the studies. The most common definition for PWML was “spots of hyperintensity on T1-weighted images and hypointensity on T2-weighted images.” However, the cutoff value to evaluate PWML severity differed between studies. Chau et al.38, Guo et al.,22 and Miller et al.12 had a cutoff number (i.e., 3) and size (i.e., 2 mm) of the lesions and percent hemispheric involvement. Other studies only took the number (Bruïne et al.,39 Martinez et al.,40 and Tusor et al.41) or PWML size (Arberet et al.,42 Jeon et al.,7 and Kidokoro et al.8) into consideration. Furthermore, different MRI scanners (e.g., field strengths of 1.5 and 3T) and different MRI sequence parameters (e.g., slice thickness ranging from 0.8 to 5 mm) were used.
Also, multiple NDO scales were used. The Gross Motor Function Classification System43 was most commonly used to describe CP presence. Moreover, the Bayley Scale of Infant and Toddler Development44 was used most often (both the second and third editions). In addition, a general neurological examination was commonly described, and two studies (Martinez et al.40 and Arberet et al.42) reported the age at walking. Bruïne et al.39 used the Child Behavior Checklist45 to describe the behavioral outcome, while Arberet et al.42 used a nonstandardized interview. Arberet et al. described school type, numbers, and levels of repeated classes, paramedical or psychological support requirement, and need for medical equipment and uses of the Health Status Classification System Preschool46 to describe the overall quality of life. The severity of the impairments was also differently described. Five studies7,12,38,39,40 described a score of 1 standard deviation (SD) below the mean on the Bayley Scales of Infant Development as mild impairment and a score more than 2 SD below the mean as a severe impairment. The other four studies only made a distinction between the presence and absence of impairments.8,21,41,42
Risk of bias in individual studies and across studies
Figures 3 and 4 show the risk of bias of individuals and across studies, respectively. Five studies were of low bias and high quality, and four studies were of moderate bias and moderate quality. No studies were excluded because of the risk of bias analysis.
Results of individual studies
Table 2 shows the summary of the findings. The motor delay was the most frequently described impairment in PWML. Arberet et al.42 showed a significantly higher age at walking, and Martinez et al.40 had similar results when at least six lesions were present. In addition, Arberet et al.42 also described a significant mobility and dexterity impairment, dyspraxia, special care needs, and a higher chance to repeat a school year. Martinez et al.40 reported a significantly higher CP incidence in the presence of any lesions. Bruïne et al.39 described significantly higher motor, cognitive, and behavior delays when more than six PWML were diagnosed. Chau et al.38 reported remarkable motor and cognitive delays only when more than 5% of a hemisphere was involved. Guo et al.22 delineated a significant motor delay and cognitive impairment especially when PWML were located in the frontal lobe, highlighting the importance of lesion location as a key to predicting adverse cognitive and motor outcomes. Jeon et al.7 reported a higher, but not significant, severe motor and cognitive delay incidence as a PWML result and a significantly higher CP incidence. Kidokoro et al.8 showed significant motor and cognitive delays and a higher CP incidence when PWML is bilateral in the cortical spinal tract (CST) or when there are more than three lesions per hemisphere. Miller et al.12 mentioned that PWML are significantly related to adverse NDO, but the domains were not divided. Finally, Tusor et al.41 demonstrated a higher incidence of motor delay and CP when PWML were present, which increases if more than 20 PWML were documented, leading to a clear significant correlation. Tusor et al.41 showed that infants with PWML have reasonable results in terms of motor outcomes but did not report adjustment for potential confounders.
In summary, all studies showed a relationship between PWML and motor delay at >12 months. Six studies showed a significantly higher CP incidence in preterm infants with PWML.7,12,39,40,41,42 Cognitive delay was reported in seven out of nine studies, but only significant in two.21,39 Behavior impairment was only reported in two studies39,42 which both showed a significant delay. Arberet et al.42 reported more need for special care in the group with PWML, despite no difference in frequency in behavioral disorders. Bruïne et al.39 reported more total and externalizing behavior problems in the group with PWML. Furthermore, there was a directly proportional relationship between the number of lesions and the severity of impairments.8,21,38,39,40,41 Lesions in the CST and frontal lobes more often showed motor and cognitive impairments, respectively.22
Based on the findings of five12,22,38,39,41 high-quality and four7,8,40,42 moderate-quality studies, strong evidence for a relationship between the PWML presence on TEA brain MRI in preterm infants and long-term motor impairment seem to exist. Based on the findings of two high-quality studies, strong evidence for a relationship between PWML and long-term cognitive impairment seem to exist.22,39 Based on the findings of one high-quality39 and one moderate-quality study,42 insufficient evidence for a relationship between PWML and behavioral impairment was noted.
Discussion
Summary of evidence
To the best of our knowledge, this is the first systematic literature review to investigate the relationship between isolated PWML (without other overt brain injuries) and NDOs in preterm infants. In total, nine papers met the inclusion criteria of the current systematic review. The mean incidence of isolated PWML in the included studies was 22.1%. All nine studies showed a significantly higher incidence of motor impairment in preterm infants diagnosed with PWML on TEA brain MRI. Seven studies reported on cognitive outcomes in preterm infants with PWML, two of which found a significant correlation between PWML and adverse cognitive outcomes. Two studies evaluated behavioral problems in preterm infants diagnosed with PWML, and both found significant associations.39,42 A considerable heterogeneity was noted between the studies in terms of the interpretation of WMI and NDO. All measures used were different, including different NDO scales, PWML definitions and classifications, the cutoff value of lesion severity and impairment, and MRI setups (e.g., MRI scanner type, MRI field strength, slice thickness).
The incidence of motor delay varied between 11 and 35.7% and the incidence of CP varied between 3.4 and 25%. There are a number of possible explanations for this wide range worth mentioning. For example, a higher PWML load predicted adverse outcomes in several papers.40,41,47,48,49 Moreover, Guo et al.,22 Pavaine et al.,21 Tusor et al.,41 and Kersbergen et al.20 all reported the influence of PWML location on NDO. Guo et al.22 reported that PWML associated with adverse outcomes were more anteriorly distributed, while the PWML related to a more favorable outcome were more posteriorly distributed. Tusor et al.41 and Martinez et al.40 reported a significant correlation between PWML disrupting the corticospinal tract and motor deficits. Pavaine et al.21 reported a favorable outcome when the subplate zone and the thalamocortical fibers are spared. In addition, the distribution of PWML appears to be of importance. PWML can appear either in clusters, defined as more solitary lesions, with a rounded shape and somewhat larger, often located deeper in the WM, or linear, defined as multiple PWML close to another, often adjacent to the ventricles. Kersbergen et al.20 reported that all infants who develop CP had cluster PWML appearance and none of the infants with a linear pattern developed CP, suggesting that PWML appearance may influence the outcome as well.
The PWML patterns may reflect differences in the underlying pathophysiology.31 PWML with a cluster appearance had normal signal intensity (SI) on SWI, suggesting a non-hemorrhagic origin. DWI findings often showed high and low SI on the ADC map, suggesting an underlying restricted diffusion with a possible hypoxic-ischemic origin. PWML with a cluster appearance were most pronounced on T1-weighted imaging and generally more florid than PWML with a linear appearance, possibly due to early gliosis. In contrast, lesions with a linear appearance were often found to have low SI on SWI, suggestive of a hemorrhagic origin. Some studies also encountered a difference between the lesion load and appearance on early MRI and TEA scans.12,15,20,50,51 This evolution may be due to the tissue reaction or partial resolution of the PWML.20 Between an early and TEA scan, PWML were often less clear or decreased in number, with a milder pattern at TEA than the first MRI. However, PWML first seen on the TEA scan were described too. Also, the appearance could change, and cluster and mixed PWML would especially change appearance.52
In this study, PWML presence in preterm infants was more often associated with motor than cognitive impairment. This may be related to the PWML location, which is more commonly found in the perirolandic region.22,40,41 Frontal lesions were more often associated with cognitive impairment.22,40 There were several other factors than WMI that influence cognitive outcome. Chau et al.38 suggested that diffuse microstructural and metabolic abnormalities were more associated with early cognitive outcomes than the PWML visible on MRI. Furthermore, neurodevelopmental assessments performed in early childhood may not be a good predictor of cognitive function at school age and after that. This is either because of the limited reliability of assessment tools or because the cognitive function may change under the influence of both intrinsic and extrinsic factors such as brain plasticity, environment, and education.11,21,42,53
This study had some limitations. Firstly, the focus was on NDO in preterm infants with isolated PWML presence on TEA MRI brain scans. However, PWML is often part of a spectrum of brain injury, and PWML is rarely reported as a solitary finding. Most of the studies relating to neonatal brain MRI findings with the NDO were based on different scoring systems consisting of several items. For instance, Woodward et al. provided an overall score, which makes it difficult to assess the relative contribution of isolated PWML on NDO.54 Therefore, articles with potentially very relevant information had been excluded from this review.21,54,55,56,57 This led to a smaller study population and probable underreporting of PWML incidence and importance. Secondly, the current study only focused on MRI performed around TEA, and several studies described the difference in lesion load and appearance between 30 and 40 weeks.12,15,20,50 Therefore, some PWML cases apparent only on an early MRI scan may have been missed. In addition, some of the studies used thicker MRI slices, and therefore also some PWML may have been missed. Besides this, some PWML may be preceded by periventricular cysts, which are no longer seen at TEA.51,58 Since c-PVL is more likely to result in CP and adverse outcomes, this could lead toward a more negative outcome. Furthermore, only Arberet et al. specified the number, type, and location of the lesions. The other studies mentioned this in their discussion, but did not give specific numbers. As described earlier, this can be of great importance for the outcome.
Thirdly, most of the follow-up studies focused on the risks of severe impairment, excluding mild and moderate impairment, while the latter seemed to have a strong link with PWML.56 Only Arberet et al.42 reported milder impairments. Besides, assessments of outcomes for isolated PWML were mostly restricted to early childhood. That is why we have chosen a follow-up period from 12 months onwards. NDO data of infants born preterm with isolated PMWL in school-age children are currently lacking. Several studies suggested that PWML in preterm infants may be related to milder forms of long-term cognitive and behavioral problems, becoming more apparent in school-age children.20,46 For example, Pavaine et al.21 reported a cognitive impairment at 4 years old, which was not evident at 2 years old. Arberet et al.42 was the only study in this analysis with a longer follow-up period (age, 9–14 years old). Moreover, cognitive delay was notoriously difficult to test at this young age and was subjected to the influence of several factors.7,21 Subtle influences on NDO could therefore not be excluded. Future studies should investigate longer follow-up terms to answer this matter.
Finally, most of the studies in the current review included very preterm infants (<32 GA) instead of moderate to late preterm infants (32–37 GA), while PWML often occurs in the latter group. Martinez et al.40 was the only study that also included moderate to late preterm infants.
Conclusion
Isolated PWML are very common on TEA MRI brain scans of infants born preterm, with an incidence of 22%. All studies in the current review show a relationship between PWML and long-term NDO impairments, with motor delay being the most common finding. Cognitive delay and behavioral problems seem to be less commonly studied and may therefore be under-reported. The identification of brain injury and dysmaturation is important for clinicians and families. Early identification of neurodevelopmental risks can lead to neurorehabilitative interventions in a critical stage of brain plasticity.32 MRI and cUS can both diagnose overt brain injuries. However, only MRI can show less overt injuries to the brain (including altered brain maturation). Performing both early (e.g., 30 weeks of gestation) and TEA MRI brain scans in infants at risk for PWML may improve prognosis prediction (i.e., better informed about the full extent of the lesion load). The combination of both conventional MRI sequences and advanced techniques could be most informative because the different patterns suggest a difference in the underlying pathology and severity of impairments. Studies have shown the complementary value of cUS, MRI, and clinical examination to predict NDO.19 Current guidelines on routine neuroimaging differ both nationally and internationally, future research is needed to support an optimal imaging protocol. A large prospective cohort study with a clear definition of PWML, the absence of other types of brain injury, and standardized NDO testing in different domains using case controls seems needed.
Data availability
All data generated or analyzed during this review study are included in this published article.
References
Fynch-Edmondson, M., Morgan, C., Hunt, R. W. & Novak, I. Emergent prophylactic, reparative and restorative brain interventions for infants born preterm with cerebral palsy. Front. Physiol. 10, 15 (2019).
Liu, Y. et al. White matter abnormalities are related to microstructural changes in preterm neonates at term-equivalent age: a diffusion tensor imaging and probabilistic tractography study. Am. J. Neuroradiol. 33, 839–845 (2012).
Pavlova, M. A. & Krageloh-Mann, I. Limitations on the developing preterm brain: impact of periventricular white matter lesions on brain connectivity and cognition. Brain 136, 998–1011 (2013).
El Ters, N. M., Vesoulis, Z. A., Liao, S. M., Smyser, C. D. & Mathur, A. M. Term-equivalent functional brain maturational measures predict neurodevelopmental outcomes in premature infants. Early Hum. Dev. 119, 68–72 (2018).
Fawke, J. Neurological outcomes following preterm birth. Semin. Fetal Neonatal Med. 12, 374–382 (2007).
Pierrat, V. et al. Neurodevelopmental outcome at 2 years for preterm children born at 22 to 34 weeks’ gestation in France in 2011: EPIPAGE-2 cohort study. BMJ 358, j3448 (2017).
Jeon, T. Y. et al. Neurodevelopmental outcomes in preterm infants: comparison of infants with and without diffuse excessive high signal intensity on MR images at near-term-equivalent age. Radiology 263, 518–526 (2012).
Kidokoro, H. et al. Brain injury and altered brain growth in preterm infants: predictors and prognosis. Pediatrics 134, e444–e453 (2014).
Hintz, S. R. et al. Preterm neuroimaging and school-age cognitive outcomes. Pediatrics 142, e20174058 (2018).
Estep, M. E. et al. Diffusion tractography and neuromotor outcome in very preterm children with white matter abnormalities. Pediatr. Res. 76, 86–92 (2014).
Iwata, S. et al. Qualitative brain MRI at term and cognitive outcomes at 9 years after very preterm birth. Pediatrics 129, e1138–e1147 (2012).
Miller, S. P. et al. Early brain injury in premature newborns detected with magnetic resonance imaging is associated with adverse early neurodevelopmental outcome. J. Pediatr. 147, 609–616 (2015).
Back, S. A. & Miller, S. P. Brain injury in premature neonates: a primary cerebral dysmaturation disorder? Ann. Neurol. 75, 469–486 (2014).
De Vries, L. S., Benders, M. J. & Groenendaal, F. Progress in neonatal neurology with a focus on neuroimaging in the preterm infant. Neuropediatrics 46, 234–241 (2015).
Rutherford, M. A. et al. Magnetic resonance imaging of white matter diseases of prematurity. Neuroradiology 52, 505–521 (2010).
Leijser, L. M. et al. Brain imaging findings in very preterm infants throughout the neonatal period: part I. Incidences and evolution of lesions, comparison between ultrasound and MRI. Early Hum. Dev. 85, 101–119 (2009).
Leijser, L. M. et al. Comparing brain white matter on sequential cranial ultrasound and MRI in very preterm infants. Neuroradiology 50, 799–811 (2008).
Hart, A. R., Whitby, E. W., Griffiths, P. D. & Michael, F. S. Magnetic resonance imaging and developmental outcome following preterm birth: review of current evidence. Dev. Med. Child Neurol. 50, 655–663 (2008).
Inder, T. E. et al. Neuroimaging of the preterm brain: review and recommendations. J. Peadiatr. 237, 276–287 (2021).
Kersbergen, K. J. et al. Different patterns of punctate white matter lesions in serially scanned preterm infants. PLoS One 9, e108904 (2014).
Pavaine, J. et al. Diffusion tensor imaging-based assessment of white matter tracts and visual-motor outcomes in very preterm neonates. Neuroradiology 58, 301–310 (2016).
Guo, T. et al. Quantitative assessment of white matter injury in preterm neonates. Neurology 88, 614–622 (2017).
Cornette, L. G. et al. Magnetic resonance imaging of the infant brain: anatomical characteristics and clinical significance of punctate lesions. Arch. Dis. Child. Fetal Neonatal Ed. 86, 171–177 (2002).
Benders, M. J., Kersbergen, K. J. & De Vries, L. S. Neuroimaging of white matter injury, intraventricular and cerebellar hemorrhage. Clin. Perinatol. 41, 69–82 (2014).
Wagenaar, N. et al. Clinical risk factors for punctate white matter lesions on early magnetic resonance imaging in preterm newborns. J. Pediatr. 182, 34–40 (2017).
Ramenghi, L. A. et al. Magnetic resonance imaging assessment of brain maturation in preterm neonates with punctate white matter lesions. Neuroradiology 49, 161–167 (2007).
Nanba, Y. et al. Magnetic resonance imaging regional T1 abnormalities at term accurately predict motor outcome in preterm infants. Pediatrics 120, e10–e19 (2007).
Van Wezel-Meijler, G. et al. Predictive value of neonatal MRI as compared to ultrasound in premature infants with mild periventricular white matter changes. Neuropediatrics 30, 231–238 (1999).
Volpe, J. J. Brain injury in premature infants: a complex amalgam of destructive and developmental disturbances. Lancet Neurol. 8, 110–124 (2009).
Parodi, A. et al. Punctate white matter lesions of preterm infants: risk factor analysis. Eur. J. Paediatr. Neurol. 23, 733–739 (2019).
Malova, M. et al. Nosological differences in the nature of punctate white matter lesions in preterm infants. Front. Neurol. 12, 657461 (2021).
Hughes, A. J., Redsell, S. A. & Glazebrook, C. Motor development interventions for preterm infants: a systematic review and meta-analysis. Pediatrics 138, e20160147 (2016).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: the PRISMA statement. Int. J. Surg. 118, 536–548 (2006).
Papile, L. A., Burstein, J., Burstein, R. & Koffler, H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weight less than 1,500 gm. J. Pediatr. 92, 529–534 (1978).
Ouzzani, M., Hammady, H., Fedorowicz, Z. & Elmagarmid, A. Rayyan—a web and mobile app for systematic reviews. Syst. Rev. 5, 210 (2016).
Higgins, J. P. T., Churchill, R., Chandler, J. & Cumpston, M. S. Assessing risk of bias in included studies. In Cochrane Handbook for Systematic Reviews of Interventions: Version 5.2.0 (eds Higgins, J. P. T. & Green, S.) (Cochrane, 2017).
Hayden, J. A., Côté, P. & Bombardier, C. Evaluation of the quality of prognosis studies in systematic reviews. Ann. Intern. Med. 144, 427–437 (2006).
Chau, V. et al. Abnormal brain maturation in preterm neonates associated with adverse developmental outcomes. Neurology 81, 2082–2089 (2013).
De Bruine, F. T. et al. Clinical implications of MR imaging findings in the white matter in very preterm infants: a 2-year follow-up study. Radiology 261, 899–906 (2011).
Martinez-Biarge, M. et al. Neurodevelopmental outcomes in preterm infants with white matter injury using a new MRI classification. Neonatology 116, 227–235 (2019).
Tusor, N. et al. Punctate white matter lesions associated with altered brain development and adverse motor outcome in preterm infants. Sci. Rep. 7, 13250 (2017).
Arberet, C. et al. Isolated neonatal MRI punctate white matter lesions in very preterm neonates and quality of life at school age. J. Neonatal Perinat. Med. 10, 257–266 (2017).
Palisano, R. J. et al. Validation of a model of gross motor function for children with cerebral palsy. Phys. Ther. 80, 974–985 (2000).
Bayley, N. Bayley Scales of Infant and Toddler Development – Third Edition. J. Psychoeduc. Assess. 25, 180–198 (2007).
Achenbach, T. M. & Ruffle, T. M. The Child Behavior Checklist and related forms for assessing behavioral/emotional problems and competencies. Pediatr. Rev. 21, 265–271 (2000).
Saigal, S. et al. Development, reliability and validity of a new measure of overall health for pre-school children. Qual. Life Res. 14, 243–257 (2005).
Young, J. M. et al. Associations of perinatal clinical and magnetic resonance imaging measures with developmental outcomes in children born very preterm. J. Pediatr. 170, 90–96 (2016).
Sie, L. T. et al. Predictive value of neonatal MRI with respect to late MRI findings and clinical outcome. A study in infants with periventricular densities on neonatal ultrasound. Neuropediatrics 36, 78–89 (2005).
De Bruïne, F. T. et al. Prognostic value of gradient echo T2* sequences for brain MR imaging in preterm infants. Pediatr. Radiol. 44, 305–312 (2014).
Dyet, L. E. et al. Natural history of brain lesions in extremely preterm infants studied with serial magnetic resonance imaging from birth and neurodevelopmental assessment. Pediatrics 118, 536–548 (2006).
Martinez-Biarge, M. et al. MRI based preterm white matter injury classification: the importance of sequential imaging in determining severity of injury. PLoS One 11, e0156245 (2016).
Kersbergen, K. J. et al. Microstructural brain development between 30 and 40 weeks corrected age in a longitudinal cohort of extremely preterm infants. Neuroimage 103, 214–224 (2014).
Hack, M. et al. Poor predictive validity of the Bayley scales of infant development for cognitive function of extremely low birth weight children at school age. Pediatrics 116, 333–341 (2005).
Woodward, L. J., Anderson, P. J., Austin, N. C., Howard, K. & Inder, T. E. Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. N. Engl. J. Med. 355, 685–694 (2006).
Vesoulis, Z. A., El Ters, N. M., Herco, M., Whitehead, H. V. & Mathur, A. M. A web-based calculator for the prediction of severe neurodevelopmental impairment in preterm infants using clinical and imaging characteristics. Children (Basel) 5, 151 (2018).
Woodward, L. J., Clark, C. A. C., Bora, S. & Inder, T. E. Neonatal white matter abnormalities an important predictor of neurocognitive outcome for very preterm children. PLoS One 7, e51879 (2012).
Spittle, A. J. et al. Neonatal white matter abnormality predicts childhood motor impairment in very preterm children. Dev. Med. Child Neurol. 53, 1000–1006 (2011).
Niwa, T. et al. Punctate white matter lesions in infants: new insight using susceptibility-weighted imaging. Neuroradiology 53, 669–679 (2011).
Acknowledgements
The authors would like to thank “Eu-Brain” non-profit association for the technical support in the editing of the article.
Author information
Authors and Affiliations
Contributions
C.A.M.d.B. collected data, carried out the initial analyses, and took the lead in writing the manuscript with support from other authors. S.D.M. collected data, wrote the manuscript with support from other authors, and critically reviewed and revised the manuscript. M.L.T. conceptualized the study, and critically reviewed and revised the manuscript. L.A.R., A.R., M.M., and M.B. critically reviewed and revised the manuscript. A.v.d.H. conceptualized and designed the study, coordinated and supervised data collection, and critically reviewed and revised the manuscript. J.D. conceptualized and designed the study, coordinated and supervised data collection, and critically reviewed the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Bruijn, C.A.M., Di Michele, S., Tataranno, M.L. et al. Neurodevelopmental consequences of preterm punctate white matter lesions: a systematic review. Pediatr Res 93, 1480–1490 (2023). https://doi.org/10.1038/s41390-022-02232-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-022-02232-3
- Springer Nature America, Inc.