Abstract
Abnormalities within frontal lobe gray and white matter of bipolar disorder (BD) patients have been consistently reported in adult and pediatric studies, yet little is known about the neurochemistry of the anterior white matter (AWM) in pediatric BD and how medication status may affect it. The present cross-sectional 3T 1H MRS study is the first to use a multivoxel approach to study the AWM of BD youth. Absolute metabolite levels from four bilateral AWM voxels were collected from 49 subjects between the ages of 8 and 18 (25 healthy controls (HC); 24 BD) and quantified. Our study found BD subjects to have lower levels of N-acetylaspartate (NAA) and glycerophosphocholine plus phosphocholine (GPC + PC), metabolites that are markers of neuronal viability and phospholipid metabolism and have also been implicated in adult BD. Further analysis indicated that the observed patterns were mostly driven by BD subjects who were medicated at the time of scanning and had an ADHD diagnosis. Although limited by possible confounding effects of mood state, medication, and other mood comorbidities, these findings serve as evidence of altered neurochemistry in BD youth that is sensitive to medication status and ADHD comorbidity.
Similar content being viewed by others
Introduction
Bipolar disorder (BD) is a debilitating psychiatric illness that affects 1–2% of youth and continues into adulthood [1]. Along with other mood disorders, BD is regarded as a neurodevelopmental illness but remains, however, not fully understood [2]. Structural neuroimaging studies have sought to define the neuroanatomical profile of the disorder. Magnetic resonance imaging (MRI) studies have consistently reported smaller prefrontal cortex volumes in BD children and adults compared to healthy controls (HCs) [3,4,5]. The anterior white matter (AWM) has also been implicated by diffusion tensor imaging studies showing lower fractional anisotropy values, which reflects reduced anisotropic behavior of water diffusion in the WM microstructure [6, 7]. This, in turn, suggests that the signal conductance of neural information of these WM tracts to the frontal cortex, which is necessary for processes such as cognitive control and emotional regulation [8, 9], may be compromised. While these neuroimaging studies help to better understand the extent of structural alterations, little is known as to whether the neurochemistry in the AWM of BD patients is also impacted. This information may serve to further characterize neuropathology, predict illness severity, and guide treatment.
Proton magnetic resonance spectroscopy (1H MRS) is a well-established technique that can access the neurochemistry in vivo by quantifying levels of key metabolites [10, 11]. These include: N-acetylaspartate (NAA), a marker of functioning neuroaxonal tissue that includes aspects of the formation and/or maintenance of myelin [12, 13]; glutamate (Glu), the main excitatory neurotransmitter [14]; myo-inositol (MI), a metabolite that is exclusively localized in astrocytes and therefore, a marker of gliosis [15, 16]; phosphocreatine plus creatine (PCr + Cr), are high-energy phosphate metabolites that reflect total energy reserves [10, 17]; and glycerophosphocholine plus phosphocholine (GPC + PC), metabolites that are part of the catabolic and anabolic pathways of membrane phospholipids, respectively [18].
Compared to HCs, BD adults have been reported to have different metabolite levels in the frontal lobe, with some studies noting lower NAA and higher GPC + PC levels in BD [19,20,21,22]. Although 1H MRS studies have reported similar variations in metabolite levels in pediatric BD patients [23,24,25], the literature is relatively scant with no studies examining the AWM. There are, however, two 1H MRS studies assessing AWM differences in adult BDs, with one reporting decreases only in the NAA/PCr + Cr ratio and the other reporting decreases in NAA, GPC + PC, MI, and PCr + Cr in AWM voxels of older adults with BD along with increases in Glu plus glutamine and decreases in NAA levels in younger BD adults [26, 27]. Therefore, it remains unclear as to whether AWM areas in pediatric BD patients also exhibit differences in neurochemistry, to what extent, and how these putative changes relate to disorder severity and symptomatology. Furthermore, BD youth are often prescribed psychoactive medications [28], emphasizing the importance of also assessing effects related to medication status in which the reporting is mixed [29,30,31,32]. In addition to the complexities raised by medication status, pediatric BD patients have high rates of mood disorder comorbidities [33], thus the accounting of the effects of comorbidity is integral for a better understanding of the neurochemistry of BD.
In the present multivoxel 1H MRS study, the aims were to: (1) investigate differences in the neurochemistry of AWM areas of BD youth and HCs, (2) examine the relationship between neurochemistry and clinical severity scores, and (3) investigate possible effects of medication and comorbidity. Guided by the limited number of 1H MRS studies of the frontal cortex in pediatric BD and of the AWM in BD adults, we hypothesized BD youth would have lower NAA levels than HCs. We predicted lower levels of NAA to correspond with increased BD severity measures and medicated youth with BDs to have higher NAA levels than unmedicated youth with BD.
Materials and methods
Subjects
Our cross-sectional study included 49 subjects between the ages of 8 and 18 (25 HC, 24 BD). Subjects were recruited using flyers, radio, and newspaper advertisements from the local community and psychiatric clinics. BD diagnoses and clinical characteristics were assessed with the K-SADS-PL [34] administered by fully trained research assistants or postdoctoral fellows who were supervised by an experienced research psychiatrist. HCs had no history of Axis-I disorders, no first-degree relatives with an Axis-I disorder, and had not used any psychoactive medication within 2 weeks of the study. Exclusion criteria for all subjects included past head trauma that resulted in 15+ min of unconsciousness and/or a prior diagnosis of a severe traumatic brain injury, neurological disorders, and/or any major medical conditions. Subjects were evaluated through a socio-demographic history form for age, gender, and years of education. The Children’s Depression Rating Scale (CDRS) [35] and the Young Mania Rating Scale (YMRS) [36] were also used to assess symptom severity. All subjects and their parent or legal guardian gave written informed consent. This study was approved by the University of North Carolina at Chapel Hill IRB.
Imaging
A multivoxel approach was used to acquire the ¹H MRS data on a 3T Siemens system and postprocessing followed to extract and quantify the ¹H MRS spectra from four bilateral voxels in the AWM (Fig. 1). Regarding the protocol, scout MRI images were first acquired followed by a set of T1-weighted MRI images using the magnetization prepared rapid acquisition gradient-echo sequence (TR = 1750 ms, TE = 4.38 ms, flip angle = 8°, field of view (FOV) = 256 × 256 mm2, number of axial slices = 160, slice thickness = 1 mm, number of excitations = 1, matrix size = 256 × 208). The 1H MRS measurement was acquired using a three-dimensional (3D) multivoxel acquisition scheme, which combined the point resolved spectroscopy (PRESS) sequence with chemical shift imaging (CSI). The PRESS volume of interest (VOI) was located in the frontal lobe (70 × 50 × 40 mm3), which included the four voxels of interest in the AWM. The acquisition parameters included the following: TR = 1410 ms, TE = 80 ms [37], FOV = 160 × 160 × 80 mm3, acquired CSI matrix = 14 × 14 × 8 and zero-filled to 16 × 16 × 8, nominal pixel dimension = 10 × 10 × 10 mm3, complex spectral data points = 2048, spectral bandwidth = 2.0 kHz, water suppression using CHESS pulses [38], and number of averages = 1. Additionally, an unsuppressed water measurements for absolute quantification was acquired using the identical parameters except the acquired CSI matrix was 8 × 8 × 8 and zero-filled to 16 × 16 × 8.
Postprocessing included extracting and quantifying the 1H MRS signal from four different left and right AWM voxel locations and estimating the tissue fraction within those extracted 1H MRS voxels [i.e., the percent gray and white matter and cerebrospinal fluid (CSF)]. The procedure was completely automated and independent of operator inputs, using Linux shell scripts, and FreeSurfer and FSL tools (e.g., FLIRT, NU_CORRECT, BET, and FAST) [39, 40]. The four AWM voxels of interest were predefined anatomically on an MNI template brain image, and the voxels were mapped to the subject space using the co-registration transformation between the T1-weighted images and the template image. The coordinates of the voxel location in subject space were then used to shift the 3D multivoxel grid to ensure a voxel was centered at that location and then the 1H MRS signal of that voxel was extracted for quantification. This innovative procedure consistently and systematically placed the voxels in the specified anatomical locations between subjects, and has been demonstrated to be accurate and reliable [41]. These voxel locations were also mapped on the tissue-segmented images, in which the tissue fraction within each voxel was estimated.
GPC + PC, NAA, PCr + Cr, MI, and Glu levels were quantified using the Linear Combination Model software with a simulated basis set for the a priori knowledge [42]. The following inclusion criteria were applied to the fitted spectrum of each voxel: the signal-to-noise ratio equal to or greater than 5, CRLB values equal to of <20%, absolute data shift ⩽0.1 p.p.m., and absolute first-order phase correction equal to or less than 30°/p.p.m. The unsuppressed water signal along with the appropriate correction factors related to the proportions of gray matter, white matter and CSF within the voxel were applied to obtain absolute quantification values with institutional units (IU) [43].
Statistics
The General Estimated Equations (GEE) approach with repeated measures was used to investigate group effects. Specifically, GEEs with repeated measurements were chosen due to their suitability for analyzing data with multiple measurements per subject of which measurements may be correlated and without punity on missing data points. All statistical analyses were performed using RStudio Version 1.2.1335 based on R version 3.6.0.
GEEs were generated using the geeglm function in the geepack library. For each metabolite, a GEE model was created to test the main overall effect of subject group independent of voxel locations while controlling for age, gender, and hemisphere. To determine whether certain voxels differ differently between groups, a second GEE was conducted that accounted for group X voxel location interactions while also controlling for age, gender, and hemisphere. FDR corrections were used to adjust for the testing of multiple metabolites.
In order to test the effect of medication at time of scanning, we compared metabolite levels of unmedicated BDs, medicated BDs, and HCs. For each metabolite, we created two GEE models that covaried for age, gender, and hemisphere: one model contained the grouping variable while the other was a “null” model with no grouping data. Then, an ANOVA tested whether the models were significantly different from one another, thus indicating whether there was a significant effect of medication group. FDR corrections were used to adjust for multiple comparisons. We also conducted GEE analyses that tested for medication status X voxel location interactions. Chi-square tests were used to evaluate whether the medicated and unmedicated groups had differing comorbidities.
For all significant metabolites, the average metabolite level across significant voxel locations and both hemispheres was calculated. Then, using the lm function to generate linear models, we examined the relationship between average metabolite levels in BD subjects and YMRS scores and CDRS scores. Significance levels for all analyses were set at 0.05.
Results
Demographic and clinical characteristics of our sample are detailed in Table 1. There were no significant differences between BDs and HCs in age, gender, handedness, and years of education. Regarding the quality of the 1H MRS, mean SNR for the BD group was 15.3 ± 4.8 and 14.0 ± 4.4 for HCs. Mean FWHM was 6.2 ± 2.2 Hz (3.5–17.6 Hz) for BDs and 6.1 ± 2.1 Hz (3.5–14.7 Hz) for HCs. Mean, SD, and range of CRLB values for GPC + PC, PCr + Cr, NAA, MI, and Glu across all AWM voxels were 3.6 ± 1.4% (2–9%) and 3.7 ± 1.6% (2–12%), 3.4 ± 1.6% (2–11%) and 3.7 ± 1.6% (2–13%), 3.9 ± 1.5% (2–10%) and 4.2 ± 2.1% (1–16%), 9.1 ± 3.4% (5–19%) and 10.5 ± 3.4% (5–19%), and 9.8 ± 3.0% (5–19%) and 10.7 ± 3.1% (6–19%) for BDs and HCs, respectively.
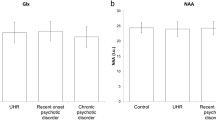
Compared to HC youth, BDs had lower levels of NAA (Wald χ2 = 3.78, p = 0.05, pcorrected = 0.13) and GPC + PC (Wald χ2 = 6.50, p = 0.01, pcorrected = 0.05) (Fig. 2). NAA differences did not survive FDR correction. GEE models that included a group X voxel location term did not find any significant interactions between the two variables for any of the metabolites. As we were unable to ascertain which voxels were driving the statistical significance using the group X voxel location model, Table 2 lists the mean metabolite differences between BDs and HCs along with a 95% confidence for each voxel. Supplementary Table 1 includes mean, SD, and SE values of metabolites for HCs and BDs. Linear models examining the relationship between average metabolite levels and YMRS and CDRS scores in BD subjects were not significant.
Metabolites marked with ★ were significantly different across all voxels and survived FDR correction. HC healthy controls; BD unmedicated bipolar disorder; BD-M medicated bipolar disorder; Glu glutamate; GPC + PC glycerophosphocholine plus phosphocholine; myo-Ins myo-inositol; NAA N-acetylaspartate; NAAG N-acetyl-aspartyl-glutamate; PCr + Cr phosphocreatine plus creatine.
Half of the subjects in our BD (12 out of 24) group were medicated at the time of scanning. Medicated and unmedicated BD subjects did not differ in age, gender, YMRS, and CDRS scores. ANOVAs evaluating the test and null GEE models for each metabolite indicated significant group differences in NAA (Wald χ2 = 14.0, p = 9.0 × 10−4, pcorrected = 3.8 × 10−3), GPC + PC (Wald χ2 = 978, p = 7.5 × 10−3, pcorrected = 0.01), PCr + Cr (Wald χ2 = 6.07, p = 0.05, pcorrected = 0.06), and MI (Wald χ2 = 12.9, p = 1.6 × 10−3, pcorrected = 3.8 × 10−3) levels. Evaluation of the test GEE indicated that medicated BDs had lower levels of NAA, GPC + PC, PCr + Cr, and MI than HCs and unmedicated BDs (Table 2). HCs and unmedicated BDs were not significantly different for any of the metabolites. GEE models that included a medication group X voxel location term did not find any significant interactions between the two variables for any of the metabolites. Mean differences and 95% confidence intervals between HCs and medicated BDs, HCs and unmedicated BDs, and medicated and unmedicated BDs for AWM 1–4 are shown in Table 2.
Comorbidity data are presented in Supplementary Table 2. All our medicated BD subjects had ADHD comorbidities while 4 out of the 12 unmedicated BD subjects had an ADHD diagnosis (χ2 = 8.18, p = 4.24 × 10−3). No other comorbidity differed between medicated and unmedicated BD groups.
Discussion
Our study found lower levels of NAA and GPC + PC within the AWM of BD youth. Further analysis indicated that the observed patterns were mostly driven by BD subjects who were medicated at the time of scanning.
NAA is a metabolite synthesized in neural mitochondria and has been implicated in a vast array of processes, including myelin and lipid formation, neuronal protection from osmotic stress, and mitochondrial energy production [44,45,46]. In a postmortem study examining hippocampal tissues in BD brains, mitochondrial proteins necessary for energy production through oxidative phosphorylation and proteasomal degradation processes were underexpressed [47]. MRS studies investigating frontal white matter regions in adults have also detected lower NAA levels [26, 27]. Hence, the observed decreases in NAA may signal early disruption in mitochondrial processing in the AWM of BD children remains present in adulthood within the frontal regions, basal ganglia, and hippocampus [21, 22, 48, 49].
Along with decreased NAA levels, we also detected lower GPC + PC levels in our BD subjects. The GPC + PC signal is a measure of free choline compounds, which are the primary building blocks of the neuronal cell membranes and are integral to the generation of the neurotransmitter acetylcholine [50, 51]. However, due to the identical spectral profile of GPC and PC in 1H MRS, these two compounds are classified under one choline peak. As such, it is not possible to pinpoint whether the observed decrease in GPC + PC is due to decreases in membrane turnover, which would be prompted by decreases in GPC, or decreases in de novo phosphocholine formation, which is necessary for the building of cellular membranes [52]. Furthermore, the literature regarding the role of GPC + PC in BD is mixed with certain adult studies reporting adult BD subjects exhibiting increased levels of GPC + PC than HCs in the anterior cingulate cortex and basal ganglia [41, 53, 54], while some adult and pediatric studies focusing on the prefrontal cortex have reported lower GPC + PC levels in BD subjects [49, 55, 56]. Nevertheless, there appears to be a dysregulation in the pathways responsible for cellular membrane integrity in BD. Markedly, a study investigating the effects of administering choline bitartrate to lithium treated BD adults on their metabolite levels and behavioral profiles found a decrease in the severity of manic and mood disorder symptoms accompanied the treated subjects’ elevated choline levels, suggesting a therapeutic effect of choline and a possible target for future treatments [57]. Of the two 1H MRS studies on the AWM in BD subjects, one reported decreases in GPC + PC in older BD adults while the other found no significant differences in GPC + PC [26, 27, 49]. Therefore, with such a scant body of the literature, future MRS studies should investigate the AWM of BD subjects to further characterize the metabolic profile of BD and to determine whether these signatures are related with white matter structural integrity.
We did not find any differences in Glu. As our PRESS sequence is optimized for Glu detection with TE = 80 ms at 3T, we do not believe this null finding to be due to a lack of signal sensitivity, and a power analysis based on effect sizes from prior publications indicated that our study had a power of 91% [58]. Although abnormal Glu activity is a hallmark of BD [59], these differences are largely reported in GM areas [60]. Studies reporting on the neurochemistry in the AWM of BD patients, however, are scant with only one study reporting significant alterations of Glu + Glutamine in AWM areas in BD young adults [27]. A meta-analysis examining the role of Glu in BD reported a lack of significant differences in both Glu and Glu + Glutamine levels in pediatric BD vs HCs in brain areas that included the anterior cingulate cortex, and gray matter in frontal cortices [60]. Therefore, more studies are needed in order to better characterize the role of Glu in pediatric BD neuropathology.
Due to the cross-sectional nature and the limited sample size of our study, we are unable to attribute the observed differences in medicated BD subjects to medication effects or other extraneous factors, including comorbidities, mood states, and illness duration. Thus, interpretations of our results and conclusions derived from them must bear in mind these study limitations. We did determine, however, that the medicated and unmedicated BD subjects did not differ in age, gender, mood status, handedness, YMRS, and CDRS score distributions. In addition, we have included the results of a GEE analysis performed on the metabolite levels of only right-handed and euthymic subjects in Supplementary Table 3. These findings were largely in accordance with the results from the full sample, thus suggesting that handedness and mood state are not main drivers of the patterns observed. We also examined whether the proportions of BD I, II, and NOS differed between medicated and unmedicated groups and found that while this trended toward significance, both groups had BD NOS subjects as the majority. We were unable to find studies that assessed metabolite level differences between all three BD subtypes, and studies that included just BD I and II subjects have reported conflicting findings [61, 62]. As the BD literature tends to focus more on BD I and BD II subtypes, the large proportion of BD NOS subjects in our sample may limit the generalizability of our results. Nevertheless, studies have shown BD NOS youth to have similar symptomologies, comorbidities, medications, functional impairments, and demographic profiles as the other BD subtypes [63,64,65]. In addition, 45% of BD NOS subjects have been shown to convert to either BD I or BD II within 5 years, which further highlights the overlap between the BD subtypes [1, 66]. All our medicated BD subjects had ADHD comorbidities while only 4 out of the 12 unmedicated BD subjects had an ADHD diagnosis. One must also note that of the 12 medicated subjects, 9 were taking stimulants. In solely ADHD populations, stimulants tended to increase NAA and decrease GPC + PC levels [67,68,69]. Given that ADHD has also been associated with decreased NAA, GPC + PC, PCr + Cr, and MI metabolite levels [70], it is possible that the counterintuitive decrease in NAA in medicated subjects may be due to concurrent ADHD and BD interacting to alter the metabolite profile of patients as well as the neurochemical effects of stimulants. In turn, while MRS studies have investigated frontal and prefrontal cortical regions, this is the first 1H MRS study to target the AWM in pediatric BD. Hence, more studies investigating the AWM in BD populations are needed to better conceptualize our findings. As our sample spanned a crucial period of frontal lobe neurodevelopment [71], we assured there were no age differences between groups and that age was a covariate in all analyses. Examining the covariate terms of the GEEs, however, indicated that both NAA and PCr + Cr levels had a significant relationship with age. When age was included as an interaction term, there were no significant group X age interactions and thus the group differences observed were not due to age effects (Supplementary Fig. 1). Future longitudinal studies would be better equipped to answer neurometabolic development and should seek to characterize the effects of psychoactive medications on the neurometabolites of BD youth and how this affects behavior. Larger studies should also further characterize the relationship between ADHD and BD and the phenotypes associated with comorbidity.
This is the first multivoxel 1H MRS study to examine the neurochemistry of the AWM in BD youth, an area responsible for the neuronal communication that is necessary for the facilitation of complex cognitive processes. The neurochemical changes observed in the AWM of our BD group, specifically the decreases in NAA and GPC + PC, were consistent with the literature detailing similar patterns in BD adults [53]. GPC + PC and NAA are metabolites involved in phospholipid metabolism and neuronal viability [72]. Thus, finding decreases in both metabolites during childhood and adolescence serves as preliminary evidence of an altered neurodevelopmental trajectory in BD youth that appears to extend into adulthood. Future studies may seek to characterize BD metabolites across the lifespan using longitudinal designs, assess how deviations in metabolite levels affect white and gray matter structural development, and investigate how medication may alter the biochemical profile of BD.
References
Goldstein BI, Birmaher B. Prevalence, clinical presentation and differential diagnosis of pediatric bipolar disorder. Isr J Psychiatry Relat Sci. 2012;49:3–14.
Nasrallah HA. Neurodevelopmental aspects of bipolar affective disorder. Biol Psychiatry. 1991;29:1–2.
Goldberg ME. Cortical function: the prefrontal cortex. Science. 1989;246:681–2.
Rimol LM, Hartberg CB, Nesvåg R, Fennema-Notestine C, Hagler DJ Jr, Pung CJ, et al. Cortical thickness and subcortical volumes in schizophrenia and bipolar disorder. Biol Psychiatry. 2010;68:41–50.
Dickstein DP, Milham MP, Nugent AC, Drevets WC, Charney DS, Pine DS, et al. Frontotemporal alterations in pediatric bipolar disorder: results of a voxel-based morphometry study. Arch Gen Psychiatry. 2005;62:734–41.
Yurgelun-Todd DA, Silveri MM, Gruber SA, Rohan ML, Pimentel PJ. White matter abnormalities observed in bipolar disorder: a diffusion tensor imaging study. Bipolar Disord. 2007;9:504–12.
Frazier JA, Breeze JL, Papadimitriou G, Kennedy DN, Hodge SM, Moore CM, et al. White matter abnormalities in children with and at risk for bipolar disorder. Bipolar Disord. 2007;9:799–809.
Miller EK, Cohen JD. An integrative theory of prefrontal cortex function. Annu Rev Neurosci. 2001;24:167–202.
Dixon ML, Thiruchselvam R, Todd R, Christoff K. Emotion and the prefrontal cortex: an integrative review. Psychol Bull. 2017;143:1033–81.
Stanley JA, Pettegrew JW, Keshavan MS. Magnetic resonance spectroscopy in schizophrenia: methodological issues and findings-part I. Biol Psychiatry. 2000;48:357–68.
Stanley JA, Raz N. Functional magnetic resonance spectroscopy: the ‘New’ MRS for cognitive neuroscience and psychiatry research. Front Psychiatry. 2018;9:76.
Ross B, Bluml S. Magnetic resonance spectroscopy of the human brain. Anat Rec. 2001;265:54–84.
Stanley JA, Vemulapalli M, Nutche J, Montrose DM, Sweeney JA, Pettegrew JW, et al. Reduced N-acetyl-aspartate levels in schizophrenia patients with a younger onset age: a single-voxel 1H spectroscopy study. Schizophr Res. 2007;93:23–32.
Yüksel C, Öngür D. Magnetic resonance spectroscopy studies of glutamate-related abnormalities in mood disorders. Biol Psychiatry. 2010;68:785–94.
Silverstone PH, McGrath BM, Kim H. Bipolar disorder and myo-inositol: a review of the magnetic resonance spectroscopy findings. Bipolar Disord. 2005;7:1–10.
Brand A, Richter-Landsberg C, Leibfritz D. Multinuclear NMR studies on the energy metabolism of glial and neuronal cells. Dev Neurosci. 1993;15:289–98.
Sartorius A, Lugenbiel P, Mahlstedt MM, Ende G, Schloss P, Vollmayr B. Proton magnetic resonance spectroscopic creatine correlates with creatine transporter protein density in rat brain. J Neurosci Methods. 2008;172:215–9.
Brambilla P, Stanley JA, Nicoletti MA, Sassi RB, Mallinger AG, Frank E, et al. 1H magnetic resonance spectroscopy investigation of the dorsolateral prefrontal cortex in bipolar disorder patients. J Affect Disord. 2005;86:61–7.
Galińska-Skok B, Małus A, Konarzewska B, Rogowska-Zach A, Milewski R, Tarasów E, et al. Choline compounds of the frontal lobe and temporal glutamatergic system in bipolar and schizophrenia proton magnetic resonance spectroscopy study. Dis Markers. 2018;2018:3654894.
Senaratne R, Milne AM, MacQueen GM, Hall GBC. Increased choline-containing compounds in the orbitofrontal cortex and hippocampus in euthymic patients with bipolar disorder: a proton magnetic resonance spectroscopy study. Psychiatry Res. 2009;172:205–9.
Winsberg ME, Sachs N, Tate DL, Adalsteinsson E, Spielman D, Ketter TA. Decreased dorsolateral prefrontal N-acetyl aspartate in bipolar disorder. Biol Psychiatry. 2000;47:475–81.
Frye MA, Thomas MA, Yue K, Binesh N, Davanzo P, Ventura J, et al. Reduced concentrations of N-acetylaspartate (NAA) and the NAA-creatine ratio in the basal ganglia in bipolar disorder: a study using 3-Tesla proton magnetic resonance spectroscopy. Psychiatry Res. 2007;154:259–65.
Cecil KM, DelBello MP, Sellars MC, Strakowski SM. Proton magnetic resonance spectroscopy of the frontal lobe and cerebellar vermis in children with a mood disorder and a familial risk for bipolar disorders. J Child Adolesc Psychopharmacol. 2003;13:545–55.
Olvera RL, Caetano SC, Fonseca M, Nicoletti M, Stanley JA, Chen HH, et al. Low levels of N-acetyl aspartate in the left dorsolateral prefrontal cortex of pediatric bipolar patients. J Child Adolesc Psychopharmacol. 2007;17:461–73.
Caetano SC, Olvera RL, Hatch JP, Sanches M, Chen HH, Nicoletti M, et al. Lower N-acetyl-aspartate levels in prefrontal cortices in pediatric bipolar disorder: a 1H magnetic resonance spectroscopy study. J Am Acad Child Adolesc Psychiatry. 2011;50:85–94.
Zhong S, Wang Y, Zhao G, Xiang Q, Ling X, Liu S, et al. Similarities of biochemical abnormalities between major depressive disorder and bipolar depression: a proton magnetic resonance spectroscopy study. J Affect Disord. 2014;168:380–6.
Bustillo JR, Jones T, Qualls C, Chavez L, Lin D, Lenroot RK, et al. Proton magnetic resonance spectroscopic imaging of gray and white matter in bipolar-I and schizophrenia. J Affect Disord. 2019;246:745–53.
Bhangoo RK, Lowe CH, Myers FS, Treland J, Curran J, Towbin KE, et al. Medication use in children and adolescents treated in the community for bipolar disorder. J Child Adolesc Psychopharmacol. 2003;13:515–22.
Brambilla P, Stanley JA, Sassi RB, Nicoletti MA, Mallinger AG, Keshavan MS, et al. 1H MRS study of dorsolateral prefrontal cortex in healthy individuals before and after lithium administration. Neuropsychopharmacology. 2004;29:1918–24.
Friedman SD, Dager SR, Parow A, Hirashima F, Demopulos C, Stoll AL, et al. Lithium and valproic acid treatment effects on brain chemistry in bipolar disorder. Biol Psychiatry. 2004;56:340–8.
Hajek T, Bauer M, Pfennig A, Cullis J, Ploch J, O’Donovan C, et al. Large positive effect of lithium on prefrontal cortex N-acetylaspartate in patients with bipolar disorder: 2-centre study. J Psychiatry Neurosci. 2012;37:185–92.
Moore CM, Frazier JA, Glod CA, Breeze JL, Dieterich M, Finn CT, et al. Glutamine and glutamate levels in children and adolescents with bipolar disorder: a 4.0-T proton magnetic resonance spectroscopy study of the anterior cingulate cortex. J Am Acad Child Adolesc Psychiatry. 2007;46:524–34.
Spencer TJ, Biederman J, Wozniak J, Faraone SV, Wilens TE, Mick E. Parsing pediatric bipolar disorder from its associated comorbidity with the disruptive behavior disorders. Biol Psychiatry. 2001;49:1062–70.
Kaufman J, Birmaher B, Brent D, Rao U, Flynn C, Moreci P, et al. Schedule for affective disorders and schizophrenia for school-age children-present and lifetime version (K-SADS-PL): initial reliability and validity data. J Am Acad Child Adolesc Psychiatry. 1997;36:980–8.
Poznanski E. Depression in children and adolescents: an overview. Psychiatr Ann. 1985;15:365–7.
Young RC, Biggs JT, Ziegler VE, Meyer DA. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry. 1978;133:429–35.
Schubert F, Gallinat J, Seifert F, Rinneberg H. Glutamate concentrations in human brain using single voxel proton magnetic resonance spectroscopy at 3 Tesla. Neuroimage. 2004;21:1762–71.
Haase A, Frahm J, Hänicke W, Matthaei D. 1H NMR chemical shift selective (CHESS) imaging. Phys Med Biol. 1985;30:341–4.
Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. 1999;9:179–94.
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TEJ, Johansen-Berg H, et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage. 2004;23 Suppl 1:S208–19.
Cao B, Stanley JA, Passos IC, Mwangi B, Selvaraj S, Zunta-Soares GB, et al. Elevated choline-containing compound levels in rapid cycling bipolar disorder. Neuropsychopharmacology. 2017;42:2252–8.
Provencher SW. Automatic quantitation of localized in vivo 1H spectra with LCModel. NMR Biomed. 2001;14:260–4.
Kreis R, Ernst T, Ross BD. Absolute quantitation of water and metabolites in the human brain. II. Metabolite concentrations. J Magn Reson Ser B. 1993;102:9–19.
Baslow MH, Baslow MH, Burlina AP. N-acetylaspartate metabolism underlays the structural and functional units of the vertebrate brain: a bioenergetic rationale for clinical observations of changes in the neuronal biomarker ‘NAA’ in many human brain disorders. Bioenerg: Open Access. 2012;01:1–10.
Stork C, Renshaw PF. Mitochondrial dysfunction in bipolar disorder: evidence from magnetic resonance spectroscopy research. Mol Psychiatry. 2005;10:900–19.
Madhavarao CN, Chinopoulos C, Chandrasekaran K, Namboodiri MAA. Characterization of the N-acetylaspartate biosynthetic enzyme from rat brain. J Neurochem. 2003;86:824–35.
Konradi C, Eaton M, MacDonald ML, Walsh J, Benes FM, Heckers S. Molecular evidence for mitochondrial dysfunction in bipolar disorder. Arch Gen Psychiatry. 2004;61:300.
Atmaca M, Yildirim H, Ozdemir H, Poyraz AK, Tezcan E, Ogur E. Hippocampal 1H MRS in first-episode bipolar I patients. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30:1235–9.
Cecil KM, DelBello MP, Morey R, Strakowski SM. Frontal lobe differences in bipolar disorder as determined by proton MR spectroscopy. Bipolar Disord. 2002;4:357–65.
Ansell GB, Spanner S. The source of choline for acetylcholine synthesis. Cholinergic Mech Psychopharmacol. 1978:431–45.
Sanders LM, Zeisel SH. Choline: dietary requirements and role in brain development. Nutr Today. 2007;42:181–6.
Stagg C, Rothman DL. Magnetic resonance spectroscopy: Tools for Neuroscience Research and Emerging Clinical Applications. Academic Press. 2013:105–6.
Cao B, Stanley JA, Selvaraj S, Mwangi B, Passos IC, Zunta-Soares GB, et al. Evidence of altered membrane phospholipid metabolism in the anterior cingulate cortex and striatum of patients with bipolar disorder I: a multi-voxel (1)H MRS study. J Psychiatr Res. 2016;81:48–55.
Wu HE, Melicher T, Cao B, Sanches M, Stanley JA, Zunta-Soares G, et al. MR spectroscopy findings of the Basal Ganglia in bipolar disorders: a systematic review. Curr Psychiatry Rev. 2018;14:99–104.
Frey BN, Stanley JA, Nicoletti MA, Hatch JP, Soares JC. Corrected values of brain metabolites for the article: ‘Abnormal cellular energy and phospholipid metabolism in the left dorsolateral prefrontal cortex of medication-free individuals with bipolar disorder: an in vivo1H MRS study’. Bipolar Disord. 2008;10:849.
Lacerda ALT, Caetano D, Keshavan MS. Urinary pH in panic disorder. Psychiatry Res. 2005;134:199–203.
Stoll AL, Sachs GS, Cohen BM, Lafer B, Christensen JD, Renshaw PF. Choline in the treatment of rapid-cycling bipolar disorder: clinical and neurochemical findings in lithium-treated patients. Biol Psychiatry. 1996;40:382–8.
Yildiz-Yesiloglu A, Ankerst DP. Neurochemical alterations of the brain in bipolar disorder and their implications for pathophysiology: a systematic review of the in vivo proton magnetic resonance spectroscopy findings. Prog Neuropsychopharmacol Biol Psychiatry. 2006;30:969–95.
Wilson GM, Flibotte S, Chopra V, Melnyk BL, Honer WG, Holt RA. DNA copy-number analysis in bipolar disorder and schizophrenia reveals aberrations in genes involved in glutamate signaling. Hum Mol Genet. 2006;15:743–9.
Gigante AD, Bond DJ, Lafer B, Lam RW, Young LT, Yatham LN. Brain glutamate levels measured by magnetic resonance spectroscopy in patients with bipolar disorder: a meta-analysis. Bipolar Disord. 2012;14:478–87.
Atagün Mİ, Şıkoğlu EM, Can SS, Uğurlu GK, Kaymak SU, Çayköylü A, et al. Neurochemical differences between bipolar disorder type I and II in superior temporal cortices: a proton magnetic resonance spectroscopy study. J Affect Disord. 2018;235:15–9.
Ketter TA, Kimbrell TA, George MS, Dunn RT, Speer AM, Benson BE, et al. Effects of mood and subtype on cerebral glucose metabolism in treatment-resistant bipolar disorder. Biol Psychiatry. 2001;49:97–109.
Axelson D, Birmaher B, Strober M, Gill MK, Valeri S, Chiappetta L, et al. Phenomenology of children and adolescents with bipolar spectrum disorders. Arch Gen Psychiatry. 2006;63:1139.
Birmaher B, Axelson D, Strober M, Gill MK, Valeri S, Chiappetta L, et al. Clinical course of children and adolescents with bipolar spectrum disorders. Arch Gen Psychiatry. 2006;63:175–83.
Danner S, Fristad MA, Arnold LE, Youngstrom EA, Birmaher B, Horwitz SM, et al. Early-onset bipolar spectrum disorders: diagnostic issues. Clin Child Fam Psychol Rev. 2009;12:271–93.
Axelson DA, Birmaher B, Strober MA, Goldstein BI, Ha W, Gill MK, et al. Course of subthreshold bipolar disorder in youth: diagnostic progression from bipolar disorder not otherwise specified. J Am Acad Child Adolesc Psychiatry. 2011;50:1001. e3.
Kronenberg G, Ende G, Alm B, Deuschle M, Heuser I, Colla M, et al. and reduced choline levels in the anterior cingulum following chronic methylphenidate. A spectroscopic test-retest study in adult ADHD. Eur Arch Psychiatry Clin Neurosci. 2008;258:446–50.
Wiguna T, Guerrero APS, Wibisono S, Sastroasmoro S. Effect of 12-week administration of 20-mg long-acting methylphenidate on Glu/Cr, NAA/Cr, Cho/Cr, and mI/Cr ratios in the prefrontal cortices of school-age children in Indonesia: a study using 1H magnetic resonance spectroscopy (MRS). Clin Neuropharmacol. 2012;35:81–5.
Spencer TJ, Brown A, Seidman LJ, Valera EM, Makris N, Lomedico A, et al. Effect of psychostimulants on brain structure and function in ADHD: a qualitative literature review of magnetic resonance imaging-based neuroimaging studies. J Clin Psychiatry. 2013;74:902–17.
Tafazoli S, O’Neill J, Bejjani A, Ly R, Salamon N, McCracken JT, et al. 1H MRSI of middle frontal gyrus in pediatric ADHD. J Psychiatr Res. 2013;47:505–12.
Spear LP. Adolescent neurodevelopment. J Adolesc Health. 2013;52:S7–13.
De Stefano N, Filippi M, Miller D, Pouwels PJ, Rovira A, Gass A, et al. Guidelines for using proton MR spectroscopy in multicenter clinical MS studies. Neurology.2007;69:1942–52.
Acknowledgements
The research reported was supported in part by NIMH grant R01 085667, the Dunn Foundation, and the Pat Rutherford, Jr. Endowed Chair in Psychiatry to JCS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
JCS has received grants/research support from BMS, Forrest, J&J, Merck, Stanley Medical Research Institute, NIH and has been a speaker for Pfizer and Abbott. No other authors have conflicts to declare.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Tannous, J., Cao, B., Stanley, J.A. et al. Altered neurochemistry in the anterior white matter of bipolar children and adolescents: a multivoxel 1H MRS study. Mol Psychiatry 26, 4117–4126 (2021). https://doi.org/10.1038/s41380-020-00927-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41380-020-00927-9
- Springer Nature Limited