Abstract
Background/Objective
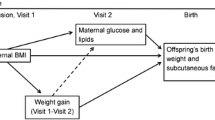
One potential mechanism by which maternal obesity impacts fetal growth is through hyperglycemia below the threshold for gestational diabetes. Data regarding which measures of maternal glucose metabolism mediate this association is sparse. The objectives of this study were to (i) quantify the associations of maternal pre-pregnancy body mass index (BMI) with neonatal size and adiposity and (ii) examine the role of markers of maternal glucose metabolism as mediators in these associations.
Subjects/Methods
This is a secondary analysis of 6,379 mother-infant dyads from the Hyperglycemia and Adverse Pregnancy Outcome cohort. Markers of glucose metabolism, including plasma glucose and c-peptide values, Stumvoll first-phase estimate, modified Matsuda index, and oral disposition index were measured and calculated from an oral glucose tolerance test (OGTT) between 24- and 32-weeks’ gestation. We calculated the direct effect of maternal BMI category, measured at the time of the OGTT and regressed to estimate pre-pregnancy BMI, on neonatal (1) birth weight (BW), (2) fat mass (FM), (3) % body fat (BF%), and (4) sum of skinfold thickness (sSFT). We then calculated the indirect effect of BMI category on these measures through markers of glucose metabolism.
Results
Maternal BMI category was positively associated with neonatal BW, FM, BF%, and sSFT. Additionally, mothers who were overweight or obese had higher odds of delivering an infant with BW, FM, BF%, or sSFT >90th percentile. Fasting glucose and c-peptide values were the strongest mediators in the linear associations between maternal BMI category and neonatal size and adiposity.
Conclusions
Maternal overweight and obesity were associated with higher odds of neonatal BW and adiposity >90th percentile. Fasting measures of glucose metabolism were the strongest mediators of these associations, suggesting that future studies should investigate whether incorporation of these markers in pregnant women with obesity may improve prediction of neonatal size and adiposity.
Similar content being viewed by others
Introduction
The prevalence of pre-pregnancy overweight and obesity in reproductive age women in the United States is currently over 50% [1, 2]. Maternal obesity, independent of gestational diabetes mellitus (GDM), is associated with excessive fetal growth [3, 4]. Fetal macrosomia and large for gestational age (LGA) at birth increase risk of delivery complications and also serve as predictors of childhood overweight and obesity later in life [5,6,7].
There is biologic plausibility that the association between maternal obesity and offspring growth is mediated in part by maternal glucose metabolism. Insulin sensitivity decreases over the course of a healthy pregnancy and women with overweight and obesity have decreased insulin sensitivity compared to average weight women, particularly in the third trimester [8]. Moreover, obesity during pregnancy is associated with hyperinsulinemia even without glucose dysregulation [8]. The “gold standard” test of insulin sensitivity is the euglycemic-hyperinsulinemic clamp, however this test is not practical for large-scale application [9]. Thus, alternative measures of insulin secretion and sensitivity, including the Stumvoll first-phase estimate and Matsuda index, respectively, have been described [10,11,12]. Additionally, the disposition index is a measure of beta cell dysfunction that is thought to precede overt glucose intolerance, or GDM, when inadequate [13, 14].
In current clinical practice in the U.S., a two-step approach is taken to diagnose GDM. All pregnant women undergo a glucose loading test at 24–28 weeks’ gestation. Blood glucose is measured once, 1 h after ingestion of a 50-g glucose drink. Depending on institutional policy, if the blood glucose concentration is greater than 130–140 mg/dL, then a 3-h 100-g oral glucose tolerance test (OGTT) is performed for GDM diagnosis [15]. Despite findings from the Hyperglycemia and Adverse Pregnancy Outcome (HAPO) Study and other studies suggesting that glycemia is continuously associated with maternal and infant outcomes [16], both glucose loading test and OGTT results continue to be viewed as binary in clinical practice. Internationally, GDM may be diagnosed using the International Association of Diabetes and Pregnancy Study Group (IADPSG) or World Health Organization criteria, both of which use a 75-g 2-h OGTT to diagnose GDM [17, 18].
Maternal overweight and obesity substantially increase the risk of developing GDM and it is estimated that the prevalence of GDM in women with overweight and obesity is 6.5% and 10.5%, respectively [19, 20]. Thus, the vast majority of women with overweight and obesity are considered normoglycemic. However, they and their neonates remain at risk for adverse outcomes. Use of alternative markers of maternal glucose metabolism in pregnancies affected by overweight and obesity could allow for improved risk stratification of neonatal and childhood size and adiposity.
The objectives of this study were to quantify the associations of maternal body mass index (BMI), which was measured at 24–32 weeks’ gestation and regressed to estimate pre-pregnancy BMI, with neonatal size and adiposity measures and to determine the role of several markers of maternal glucose metabolism as mediators in these associations. We hypothesized that higher maternal BMI would be associated with larger infant size and higher adiposity and that markers of maternal glucose metabolism would be significant mediators of these associations.
Methods
This was a secondary analysis of the blinded epidemiologic HAPO study. The HAPO study was a 5-year prospective observational study conducted to examine the associations between maternal glycemia and adverse pregnancy outcomes [16]. Participants did not have clinically diagnosed glucose intolerance or preexisting diabetes. All participants provided written informed consent for the parent study. The HAPO protocol was approved by the Institutional Review Board in all 15 field centers. This secondary analysis of the HAPO data was approved by the Brigham and Women’s Hospital Institutional Review Board.
Detailed methods for the HAPO study, including inclusion and exclusion criteria, have been previously described [16]. Briefly, the HAPO study recruited approximately 25,500 normoglycemic pregnant women from 15 centers in nine countries. Women underwent a blinded 75-g 2-h OGTT between 24- and 32-weeks’ gestation. Participants results were unblinded and their data not included in further analyses if the OGTT fasting plasma glucose was >105 mg/dl (5.8 mmol/l) and/or the 2-h plasma glucose was >200 mg/dl (11.1 mmol/l) and/or if a random plasma glucose between 34 and 37 weeks was >160 mg/dl (8.9 mmol/l) or if any plasma glucose was <45 mg/dl (2.5 mmol/l). Neonatal outcomes, including anthropometrics, were measured. The present analysis included data from five of the 15 centers, namely: Bellflower, CA, USA; Cleveland, OH, USA; Brisbane, Australia; Newcastle, Australia; and Hong Kong, China. Only dyads with available maternal BMI at OGTT and at least one infant outcome were included in the analysis, resulting in a final analytic sample of 6,379 mother-infant dyads.
Maternal measurements
Maternal height and weight were measured at the time of the OGTT and were used to calculate BMI. Category limits for BMI at the OGTT that could be considered comparable with nonpregnant World Health Organization BMI categories were obtained from a regression of OGTT BMI on pre-pregnancy BMI and gestational age (GA) at the OGTT [21]. BMI categories were defined as: underweight <22.5 kg/m2; normal weight ≥22.5 and <28.5 kg/m2; overweight ≥28.5 and <33 kg/m2; obese I ≥ 33 and <37.5 kg/m2; obese II ≥ 37.5 and <42 kg/m2; and obese III ≥ 42 kg/m2 for all ethnic groups. The Pearson’s correlation coefficient between pre-pregnancy BMI (available for 6,116 participants) and BMI at the OGTT was 0.94 (p < 0.01).
During the OGTT, mothers’ hemoglobin A1C (HbA1c), fasting venous plasma glucose, and c-peptide were measured prior to consuming the glucose solution. Plasma glucose was measured again 1-h and 2-h and c-peptide was measured 1-h after consumption [22].
The Stumvoll first-phase estimate, a modified Matsuda index, and an oral disposition index (DIO) were calculated using plasma glucose and c-peptide results from the OGTT [11, 12, 23]. Measures of insulin were not available; therefore, c-peptide was substituted for insulin in these calculations after conversion as it is produced in equimolar amounts to endogenous insulin [24]. C-peptide was assayed in µg/L and transformed to pmol/L by multiplying by 331.1 and then converted to insulin by dividing by six. Stumvoll first-phase estimate was calculated as 1194 + 4.724 x fasting insulin (pmol/L) − 117 x 1-h plasma glucose (mmol/L) + 1.414 x 1-h insulin (pmol/L) (ref. [10, 11]). A previously validated modified Matsuda index was calculated as 500,000/√[fasting plasma glucose (mg/dL) x fasting c-peptide (pmol/L) x average plasma glucose (mg/dL) (0–120 min) x average c-peptide (pmol/L) (0–60 min)] (ref. [12, 25]). This modified Matsuda index differs from the equation validated by Matsuda et al. which uses insulin instead of c-peptide, as well as fasting, 30-, 60-, 90-, and 120-min measures to calculate average glucose and insulin [12]. The DIO was calculated as [(1-h insulin (pmol/L) − fasting insulin (pmol/L))/(1-h plasma glucose (mmol/L) − fasting plasma glucose (mmol/L))] * (1/fasting insulin (pmol/L)) (ref. [23]).
Neonatal measurements
Neonatal anthropometric measurements, including birth weight (BW), length, and skinfold thickness (SFT), were obtained within 72 h of delivery. Rigorous training and certification procedures were established for study research nurses and midwives to ensure accuracy and reliability of measurements and consistency across centers [26]. The coefficient of variation among the examiners has previously been reported as <3% for the various anthropometric measurements and <7% for the SFT measurements [27]. Detailed information on anthropometric measurement procedures, as well as training and certification procedures, are available as supplementary information.
Neonatal fat mass (FM) was calculated from BW, length, and flank SFT using a previously validated equation [27]. Body fat percentage (BF%) was calculated as 100 x FM/BW [28]. The sum of SFTs (sSFT) was calculated by summing the flank, subscapular, and triceps SFT measurements.
Birthweight was considered >90th percentile if the measurement was >90th percentile for the infant’s sex, GA, maternal ethnicity, parity, and center for neonates ≥ 30 weeks’ GA. Body fat percentage and sSFT were considered >90th percentile if the measurement was >90th percentile for the infant’s sex, GA, maternal ethnicity, parity, and center for neonates ≥ 36 weeks’ GA [21]. Fat mass was considered >90th percentile if the measurement was >90th percentile for neonates ≥36 weeks’ GA. All analyses with FM > 90th percentile were additionally adjusted for infant sex, GA at delivery, maternal ethnicity, parity, and center since these factors were not accounted for in generating the percentile.
Statistical analyses
All analyses were performed using Stata/IC 15.1. Characteristics of the cohort were described as mean ± standard deviation (SD) or n (%) for continuous and categorical variables, respectively. Differences in characteristic variables across BMI categories were compared using one-way analysis of variance for continuous variables and Pearson’s Chi-squared test for categorical variables. Logistic regression analysis was then used to examine the associations between maternal BMI category and neonatal BW, FM, BF%, and sSFT >90th percentile, with normal BMI as the reference group. Maternal ethnicity, smoking status, parity, and gestational weight gain (GWG) (calculated as the difference between the first and last recorded prenatal weight) were considered as confounders as these are factors that differed between maternal BMI categories and are known to influence neonatal size and adiposity.
Maternal markers of glucose metabolism that were examined as potential mediators in the associations between maternal obesity and neonatal size and adiposity included HbA1C, fasting plasma glucose and c-peptide, 1-h plasma glucose and c-peptide, 2-h plasma glucose, Stumvoll first-phase estimate, modified Matsuda index, and DIO. Pearson’s correlations between potential mediators were assessed to identify collinearity between markers.
Multiple mediator models for each outcome were then created to assess the total and direct effects of BMI category on continuous neonatal size and adiposity measures (BW, FM, BF%, sSFT) and the indirect effects of markers of maternal glucose metabolism in each of the associations [29, 30]. For each outcome, a regression model was used to determine the direct effect of BMI. To determine the indirect effect of each potential mediator and the total indirect effect of BMI through all the mediators, the nonlinear combination command was used. The total effect of BMI on the outcome was then calculated by summing the direct effect and the total indirect effect of BMI. Percent mediation for significant indirect effects were then calculated by dividing the indirect effect of each mediator by the total effect (direct and total indirect) of BMI.
Women who met the IADPSG criteria for GDM and infants of all GAs were included in these analyses [17]. Sensitivity analyses were performed excluding (i) women with GDM by IADPSG criteria and (ii) preterm infants to determine whether exclusion of these groups would affect the primary associations between maternal BMI and neonatal size and adiposity outcomes.
For all analyses, the significance threshold was designated a priori as p < 0.05.
Results
Maternal and infant characteristics
Maternal and infant characteristics are presented in Table 1. Of the 6,379 women included in this analysis, 12% were underweight, 49% were normal weight, 22% were overweight, and 17% were obese. Age, race, smoking status, parity, and GWG all differed significantly among BMI categories. Mean ± SD GA at OGTT was 27.6 ± 1.7 weeks. Seventeen percent of women in this analysis met the IADPSG criteria for GDM. Compared to normal weight women, significantly more women with overweight or obesity were classified as having GDM by IASDPG criteria: 13% of normal weight women, 21% of women with overweight and 27%, 34%, and 40% of women with class I, II, and III obesity, respectively (p < 0.001).
There was a greater proportion of preterm births for women in higher BMI categories (p = 0.046). Infants born to women in higher BMI categories generally had greater BW, FM, BF%, and sSFT. The percent of infants who had BW, FM, BF%, and sSFT >90th percentile generally increased as BMI category increased (all p < 0.001).
Markers of glucose metabolism
Descriptive statistics of markers of maternal glucose metabolism by BMI category are also presented in Table 1. Women with overweight or obesity generally had higher levels of HbA1c, fasting, 1-h, and 2-h plasma glucose, and fasting and 1-h c-peptide (all p < 0.001). The mean Stumvoll first-phase estimate was 1569.0 and values increased as BMI category increased (p < 0.001), indicating greater insulin secretion. The mean modified Matsuda index was 5.6 and decreased as BMI category increased (p < 0.001), indicating lower insulin sensitivity. The mean DIO was 1.9 and values generally decreased as BMI category increased (p < 0.001), indicating greater loss of beta cell function relative to increased glycemia.
Figure 1 depicts the correlation matrix of individual maternal markers of glucose metabolism. Strong positive correlations were identified between the Stumvoll first-phase estimate and fasting c-peptide (r = 0.82, p < 0.01) and 1-h c-peptide (r = 0.79, p < 0.01). Strong negative correlations were also identified between the modified Matsuda index and fasting c-peptide (r = −0.79, p < 0.01) and 1-h c-peptide (r = −0.72, p < 0.01). Therefore, the Stumvoll first-phase estimate and the modified Matsuda index were not assessed as mediators because of the high degree of collinearity.
Maternal obesity and neonatal size and adiposity
The adjusted odds ratios of infant BW, FM, BF%, and sSFT >90th percentile are shown in Fig. 2. Compared to infants born to mothers of normal weight, infants of mothers who were overweight had significantly higher odds of BW (aOR 1.6, 95% CI 1.3, 2.0), FM (aOR 1.8, 95% CI 1.4, 2.4), BF% (aOR 1.8, 95% CI 1.4, 2.3), and sSFT (aOR 1.9, 95% CI 1.5, 2.4) >90th percentile. Mothers with obesity also had significantly higher odds of BW (obese class I: aOR 2.0, 95% CI 1.6, 2.7; obese class II: aOR 2.2, 95% CI 1.5, 3.2; obese class III: aOR 2.4, 95% CI 1.5, 3.9), FM (obese class I: aOR 2.2, 95% CI 1.6, 3.0; obese class II: aOR 2.8, 95% CI 1.9, 4.1; obese class III: aOR 2.2, 95% CI 1.3, 3.8), BF% (obese class I: aOR 1.8, 95% CI 1.3, 2.5; obese class II: aOR 2.9, 95% CI 1.9, 4.3; obese class III: aOR 2.6, 95% CI 1.5, 4.4), and sSFT (obese class I: aOR 2.6, 95% CI 1.9, 3.6; obese class II: aOR 2.5, 95% CI 1.6, 3.9; obese class III: aOR 3.0, 95% CI 1.7, 5.1) >90th percentile compared to mothers of normal weight.
*Normal weight BMI category as reference category. All logistic regressions were adjusted for maternal ethnicity, smoking status, parity, and GWG. Logistic regression of FM > 90th percentile was additionally adjusted for center, infant sex, and infant GA at delivery. n = 6301 (BW > 90th); 5078 (FM > 90th); 5078 (BF% > 90th); 5123 (sSFT > 90th). BW birthweight, FM fat mass, BF% body fat percent, sSFT sum of skinfold thickness, BMI body mass index, OGTT oral glucose tolerance test, GWG gestational weight gain, GA gestational age.
Results from the sensitivity analyses excluding (i) women with GDM by IADPSG criteria and (ii) preterm infants showed that the associations between maternal BMI category and neonatal size and adiposity remained unchanged within these populations (Supplementary Tables 1 and 2).
Mediation analysis
The direct and indirect effects of maternal BMI category on continuous neonatal size and adiposity measures through maternal metabolic markers of glucose metabolism are depicted in Table 2. The direct effect of BMI was significant for all four neonatal outcomes, including BW, FM, BF%, and sSFT.
Significant mediators of the association between BMI category and BW included fasting plasma glucose, 1-h plasma glucose, and 1-h c-peptide. The percent of total effect mediated by each of these markers of glucose metabolism is depicted in Fig. 3a. The strongest mediators of this association were fasting plasma glucose and 1-h c-peptide (13.9% and −13.4%, respectively). One-hour c-peptide was a negative mediator of this association, indicating that it attenuated the strength of the association between maternal BMI and BW. One-hour plasma glucose mediated 8.8% of the association.
Significant mediators of the association between BMI category and neonatal FM included fasting plasma glucose and c-peptide and 1-h plasma glucose and c-peptide. The percent of total effect mediated by each of these markers of glucose metabolism is depicted in Fig. 3b. The strongest mediator of this association was fasting c-peptide (15.6%), followed by fasting plasma glucose (11.6%), and 1-h plasma glucose (8.4%). Similar to the mediation of BW, 1-h c-peptide (−8.2%) was a negative mediator of this association, indicating that it reduced the association between maternal BMI and neonatal FM.
Significant mediators of the association between BMI category and neonatal BF% included fasting plasma glucose and c-peptide, 1-h plasma glucose, and 1-h c-peptide. The percent of total effect mediated by each of these markers of glucose metabolism is depicted in Fig. 3c. The strongest mediator of this association was fasting c-peptide (13.8%), followed by fasting plasma glucose (12.4%), 1-h plasma glucose (8.2%), and 1-h c-peptide (−6.6%), a negative mediator of the association.
Significant mediators of the association between BMI category and neonatal sSFT included fasting plasma glucose and c-peptide, 1-h plasma glucose, and HbA1c percent. The percent of total effect mediated by each of these markers of glucose metabolism is depicted in Fig. 3d. The strongest mediator of this association was fasting plasma glucose (20.7%) followed by fasting c-peptide (13.9%), and 1-h plasma glucose (6.9%). Hemoglobin A1c percent (−3.8%) was a negative mediator of the association between maternal BMI and neonatal sSFT.
Two-hour plasma glucose and DIO were not significant mediatiors of any of the associations examined. The Stumvoll first-phase estimate and modified Matsuda index were not assessed as mediators due to the high degree of collinearity with fasting and 1-h c-peptide.
Discussion
Here we report, from a secondary analysis of a sub-cohort of women from the HAPO study, that higher maternal BMI category was associated with higher odds of neonatal BW and adiposity >90th percentile. We also report that fasting measures of maternal glucose metabolism (fasting plasma glucose and c-peptide) at the time of the OGTT at 24–32 weeks’ gestation were the strongest mediators of the association between maternal BMI and newborn size and adiposity.
These findings are consistent with a body of evidence that maternal overweight and obesity, even in the absence of overt GDM, are associated with greater infant size [3, 4, 16, 31, 32]. Yu et al. reported that maternal pre-pregnancy overweight and obesity increased the risk for LGA, high BW, and macrosomia when compared to mothers of normal weight [32]. Retnakaran et al. reported that each 1 kg/m2 increase in pre-pregnancy BMI was associated with a 1.16 increased odds of having an LGA infant [31].
Our finding that maternal overweight and obesity are associated with increased neonatal adiposity is also consistent with previous studies of women with normal glucose tolerance [33,34,35]. Starling et al. found that each 1 kg/m2 increase in maternal BMI was associated with a 5.2-g, 7.7-g, and 0.12% increase in neonatal fat mass, fat-free mass, and percentage body fat, respectively [33]. Waters et al. reported that maternal pre-pregnancy BMI was the largest contributor to newborn percentage of body fat and total fat mass relative to GWG, smoking, infant gender, maternal age, maternal race, and GA at delivery [34]. Sewell et al. reported that maternal overweight and obesity are associated with increased birth weight, percent body fat, and fat mass, but not lean body mass [35].
This analysis was unique in that it assessed the mediating effect of a variety of markers of maternal glucose metabolism in the associations between maternal BMI and neonatal size and adiposity. We found that the significant mediators of the associations between maternal BMI and neonatal size and adiposity were fasting plasma glucose and c-peptide concentrations and to a lesser extent, 1-h plasma glucose, 1-h c-peptide, and HbA1c. Uvena-Celebrezze et al. reported that in women with GDM, fasting blood glucose was more strongly correlated with neonatal fat mass than pre-prandial, 2 h post-prandial or bedtime blood glucose [36]. Disse et al. reported that fasting glucose was significantly associated with increased odds of LGA regardless of weight gain during pregnancy and 2-h OGTT blood glucose [37]. Law et al. reported continuous glucose monitoring-derived mean glucose was significantly higher in women who delivered LGA infants compared to women who delivered an appropriate for GA infant. This difference was driven by a significantly higher overnight glucose [38]. These data and our findings strongly suggest that fasting markers of maternal glucose metabolism, which are experienced by the woman and fetus for a longer cumulative time-period during pregnancy, are stronger drivers of fetal growth than stimulated (post-prandial) measures and their derived indices. However, our current screening practices uniformly measure blood glucose only at 1-h post-glucose load, which may not be the strongest predictor of LGA.
One-hour plasma glucose also mediated each of the four primary associations, although to a lesser extent than fasting glucose. This highlights the clinical importance of understanding the role of glycemic control in pregnant women with impaired glucose tolerance that does not reach the threshold of GDM [39, 40]. Previous studies have found that women who failed a glucose challenge test but passed a 3-h glucose tolerance test were more likely to deliver an infant with macrosomia compared to women with normal screening values [39, 40]. These findings suggest that more stringent criteria for diagnosis of glucose intolerance during pregnancy (i.e., the IADPSG criteria) may be particularly relevant in identifying women with overweight and obesity who are at risk of delivering macrosomic infants. Adoption of the IADPSG criteria has led to a three-fold increase in GDM diagnosis and improved perinatal outcomes [41]. This benefit would likely be magnified in women with overweight and obesity, given the higher rates of adverse outcomes in these women [3, 4]. Our findings also suggest that more sensitive and comprehensive glycemic assessment techniques, such as continuous glucose monitoring, might be investigated as a modality to improve glycemic control and prediction and prevention of fetal overgrowth.
Hemoglobin A1c was a negative mediator of the association between maternal BMI and neonatal sSFT. We hypothesize that this finding is likely due to the unreliable nature of HbA1c levels during pregnancy. Hemoglobin A1c levels decrease in most women during the first trimester and it is likely that our findings reflect that HbA1c is an unreliable marker of glycemia in pregnancy that is influenced by non-glycemic factors [42, 43].
A unique strength of this analysis is that it simultaneously evaluated the mediating effect of multiple markers of maternal glucose metabolism to discern which markers exhibit the greatest individual mediating effect. We assessed both overall infant size and fat mass as these outcomes have different implications later in life. Additionally, the sub-cohort analyzed was large, international, and multi-ethnic, furthering the generalizability of our findings. We performed sensitivity analyses excluding women who met IADPSG criteria for GDM and preterm infants which also strengthens the generalizability of our results.
One limitation of our analysis is that we were unable to examine other potential metabolic mediators of the association between maternal obesity and offspring size and adiposity. Given our finding that the indirect effects of markers of glucose metabolism do not completely account for the association between maternal obesity and newborn size and adiposity, investigation of additional pathways (e.g., dyslipidemia and inflammation) should be undertaken to optimize screening of pregnant women with obesity. Another limitation of our analysis is that we did not have data on pre-pregnancy BMI. Early differences in BMI prior to the OGTT could have influenced the results. To address this limitation, we calculated an estimate of GWG. Additionally, to maintain uniformity amongst other reported findings from this cohort, we utilized the established, regressed BMI cutoffs for pregnancy within the HAPO cohort, which do not take into consideration differences in GWG across BMI categories. The regressions used also do not account for the lower BMI cut-offs for Asian women. Thus, we may have partly overestimated the effect of obesity on infant size and adiposity in this population. However, the high correlation between regressed pre-pregnancy BMI and BMI at OGTT suggests that these categories accurately reflect the ranking of participant’s pre-pregnancy BMI. Another inherent limitation of our study is that no women received treatment for GDM and therefore our findings cannot be applied to populations of women who were treated for GDM. We did, however, conduct a sensitivity analysis without women classified with GDM by the IADPSG criteria and this did not impact results.
With this analysis, we provide data examining how specific markers of maternal glucose metabolism mediate the association between maternal BMI and neonatal size and adiposity in normoglycemic pregnancies. Our finding that fasting measures of glucose metabolism were the strongest mediators of newborn size and adiposity suggests that future studies should evaluate whether including fasting glycemic screening measures in pregnant women with obesity can improve prediction of newborn weight and body composition.
References
Branum AM, Kirmeyer SE, Gregory ECW. Prepregnancy body mass index by maternal characteristics and state: data from the birth certificate, 2014. Natl Vital Stat Rep. 2016;65:1–11.
Deputy NP, Kim SY, Conrey EJ, Bullard KM. Prevalence and changes in preexisting diabetes and gestational diabetes among women who had a live birth — United States, 2012–2016. MMWR Morb Mortal Wkly Rep. 2018;67:1201–7.
Owens LA, O’Sullivan EP, Kirwan B, Avalos G, Gaffney G, Dunne F, et al. ATLANTIC DIP: the impact of obesity on pregnancy outcome in glucose-tolerant women. Diabetes Care. 2010;33:577–9.
Yang Z, Phung H, Freebairn L, Sexton R, Raulli A, Kelly P. Contribution of maternal overweight and obesity to the occurrence of adverse pregnancy outcomes. Aust N Z J Obstet Gynaecol. 2019;59:367–74.
Jolly MC, Sebire NJ, Harris JP, Regan L, Robinson S. Risk factors for macrosomia and its clinical consequences: a study of 350,311 pregnancies. Eur J Obstet Gyencol Reprod Biol. 2003;111:9–14.
Sparano S, Ahrens W, De Henauw S, Marild S, Molnar D, Moreno LA, et al. Being macrosomic at birth is an independent predictor of overweight in children: results from the IDEFICS study. Matern Child Health J. 2013;17:1373–81.
Langer O. Fetal macrosomia: etiologic factors. Clin Obstet Gyencol. 2000;43:283–97.
Ramsay JE, Ferrell WR, Crawford L, Wallace AM, Greer IA, Sattar N. Maternal obesity is associated with dysregulation of metabolic, vascular, and inflammatory pathways. J Clin Endocrinol Metab. 2002;87:4231–7.
DeFronzo RA, Tobin JD, Andres R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol Endocrinol Metab. 1979;237:E214.
Stumvoll M, Mitrakou A, Pimenta W, Jenssen T, Yki-Järvinen H, Van Haeften T, et al. Use of the oral glucose tolerance test to assess insulin release and insulin sensitivity. Diabetes Care. 2000;23:295–301.
Stumvoll M, Van Haeften T, Fritsche A, Gerich J. Oral glucose tolerance test indexes for insulin sensitivity and secretion based on various availabilities of sampling times. Diabetes Care. 2001;24:796–7.
Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care. 1999;22:1462–70.
Genova MP, Todorova-Ananieva K, Atanasova B, Tzatchev K. Assessment of beta-cell function during pregnancy and after delivery. Acta Medica Bulgarica. 2014;41:5–12.
Lowe WL, Scholtens DM, Kuang A, Linder B, Lawrence JM, Lebenthal Y, et al. Hyperglycemia and adverse pregnancy outcome follow-up study (HAPO FUS): maternal gestational diabetes and childhood glucose metabolism. Diabetes Care. 2019;42:372–80.
Committee on Practice Bulletins - Obstetrics. ACOG practice bulletin no. 190: gestational diabetes mellitus. Obstet Gynecol. 2018;131:e49–64.
The HAPO Study Cooperative Research Group. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med. 2008;358:1991–2002.
International Association of Diabetes and Pregnancy Study Groups Consensus Panel, Metzger BE, Gabbe SG, Persson B, Buchanan TA, Catalano PA, et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33:676–82.
WHO Reproductive Health Library. WHO recommendation on the diagnosis of gestational diabetes in pregnancy. The WHO Reproductive Health Library; Geneva: World Health Organization; 2016. https://extranet.who.int/rhl/topics/preconception-pregnancy-childbirth-and-postpartum-care/antenatal-care/who-recommendation-diagnosis-gestational-diabetes-pregnancy-0.
Chu SY, Callaghan WM, Kim SY, Schmid CH, Lau J, England LJ, et al. Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care. 2007;30:2070–6.
Casagrande SS, Linder B, Cowie CC. Prevalence of gestational diabetes and subsequent Type 2 diabetes among U.S. women. Diabetes Res Clin Pract. 2018;141:200–8.
Catalano PM, McIntyre HD, Cruickshank JK, McCance DR, Dyer AR, Metzger BE, et al. The hyperglycemia and adverse pregnancy outcome study: associations of GDM and obesity with pregnancy outcomes. Diabetes Care. 2012;35:780–6.
HAPO Study Cooperative Research Group. The hyperglycemia and adverse pregnancy outcome (HAPO) study. Int J Gynaecol Obstet. 2002;78:69–77.
Utzschneider KM, Prigeon RL, Faulenbach MV, Tong J, Carr DB, Boyko EJ, et al. Oral disposition index predicts the development of future diabetes above and beyond fasting and 2-h glucose levels. Diabetes Care. 2009;32:335–41.
Leighton E, Sainsbury CA, Jones GC. A practical review of c-peptide testing in diabetes. Diabetes Therapy. 2017;8:475–87.
Radaelli T, Farrell KA, Huston-Presley L, Amini SB, Kirwan JP, McIntyre HD. et al. Estimates of insulin sensitivity using glucose and C-Peptide from the hyperglycemia and adverse pregnancy outcome glucose tolerance test. Diabetes Care. 2010;33:490–4.
HAPO Study Cooperative Research Group. Hyperglycemia and adverse pregnancy outcome (HAPO) study - associations with neonatal anthropometrics. Diabetes. 2009;58:453–9.
Catalano PM, Thomas AJ, Avallone DA, Amini SB. Anthropometric estimation of neonatal body composition. Am J Obstet Gynecol. 1995;173:1176–81.
HAPO Study Cooperative Research Group. Hyperglycaemia and adverse pregnancy outcome (HAPO) study: associations with maternal body mass index. BJOG. 2010;117:575–84.
Preacher KJ, Hayes AF. Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behav Res Methods. 2008;40:879–91.
How can I analyze multiple mediators in Stata?. UCLA: Statistical Consulting Group. https://stats.idre.ucla.edu/stata/faq/how-can-i-analyze-multiple-mediators-in-stata/.
Retnakaran R, Ye C, Hanley AJG, Connelly PW, Sermer M, Zinman B, et al. Effect of maternal weight, adipokines, glucose intolerance and lipids on infant birth weight among women without gestational diabetes mellitus. CMAJ. 2012;184:1353–60.
Yu Z, Han S, Zhu J, Sun X, Ji C, Guo X. Pre-pregnancy body mass index in relation to infant birth weight and offspring overweight/obesity: a systematicreview and meta-analysis. PLoS ONE. 2013;8:e61627.
Starling AP, Brinton JT, Glueck DH, Shapiro AL, Harrod CS, Lynch AM, et al. Associations of maternal BMI and gestational weight gain with neonatal adiposity in the Healthy Start study. Am J Clin Nutr. 2015;101:302–9.
Waters TP, Huston-Presley L, Catalano PM. Neonatal body composition according to the revised institute of medicine recommendations for maternal weight gain. J Clin Endocrinol Metab. 2012;97:3648–54.
Sewell MF, Huston-Presley L, Super DM, Catalano P. Increased neonatal fat mass, not lean body mass, is associated with maternal obesity. Am J Obstet Gynecol. 2006;195:1100–3.
Uvena-Celebrezze J, Fung C, Thomas AJ, Hoty A, Huston-Presley L, Amini SB, et al. Relationship of neonatal body composition to maternal glucose control in women with gestational diabetes mellitus. J Matern-Fetal Neonatal Med. 2002;12:396–401.
Disse E, Graeppi-Dulac J, Joncour-Mills G, Dupuis O, Thivolet C. Heterogeneity of pregnancy outcomes and risk of LGA neonates in Caucasian females according to IADPSG criteria for gestational diabetes mellitus. Diabetes Metab. 2013;39:132–8.
Law GR, Alnaji A, Alrefaii L, Endersby D, Cartland SJ, Gilbey SG, et al. Suboptimal nocturnal glucose control is associated with large for gestational age in treated gestational diabetes mellitus. Diabetes Care. 2019;42:810–5.
Stamilio M, Olsen M, Ratcliffe A, Sehdev A, Macones A. False-positive 1-hour glucose challenge test and adverse perinatal outcomes. Obstet Gynecol. 2004;103:148–56.
Leikin EL, Jenkins JH, Pomerantz GA, Klein L. Abnormal glucose screening tests in pregnancy: a risk factor for fetal macrosomia. Obstet Gynecol. 1987;69:570–3.
Duran A, Sáenz S, Torrejón MJ, Bordiú E, Del Valle L, Galindo M, et al. Introduction of IADPSG criteria for the screening and diagnosis of gestational diabetes mellitus results in improved pregnancy outcomes at a lower cost in a large cohort of pregnant women: the St. Carlos Gestational Diabetes Study. Diabetes Care. 2014;37:2442–50.
Hughes RCE, Rowan J, Florkowski CM. Is there a role for HbA1c in pregnancy? Curr Diab Rep. 2016;16:5. Jan 6
Worth R, Potter JM, Drury J, Fraser RB, Cullen DR. Glycosylated haemoglobin in normal pregnancy: a longitudinal study with two independent methods. Diabetologia. 1985;28:76–9. Feb
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Andrews, C., Monthé-Drèze, C., Sacks, D.A. et al. Role of maternal glucose metabolism in the association between maternal BMI and neonatal size and adiposity. Int J Obes 45, 515–524 (2021). https://doi.org/10.1038/s41366-020-00705-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-020-00705-1
- Springer Nature Limited
This article is cited by
-
Mediation effect of maternal triglyceride and fasting glucose level on the relationship between maternal overweight/ obesity and fetal growth: a prospective cohort study
BMC Pregnancy and Childbirth (2023)
-
Umbilical cord blood concentration of connecting peptide (C-peptide) and pregnancy outcomes
BMC Pregnancy and Childbirth (2022)