Abstract
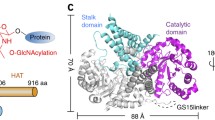
O-GlcNAc transferase (OGT) glycosylates a diverse range of intracellular proteins with O-linked N-acetylglucosamine (O-GlcNAc), an essential and dynamic post-translational modification in metazoans. Although this enzyme modifies hundreds of proteins with O-GlcNAc, it is not understood how OGT achieves substrate specificity. In this study, we describe the application of a high-throughput OGT assay to a library of peptides. We mapped sites of O-GlcNAc modification by electron transfer dissociation MS and found that they correlate with previously detected O-GlcNAc sites. Crystal structures of four acceptor peptides in complex with Homo sapiens OGT suggest that a combination of size and conformational restriction defines sequence specificity in the −3 to +2 subsites. This work reveals that although the N-terminal TPR repeats of OGT may have roles in substrate recognition, the sequence restriction imposed by the peptide-binding site makes a substantial contribution to O-GlcNAc site specificity.




Similar content being viewed by others
Change history
03 March 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41594-023-00945-5
References
Lubas, W.A., Frank, D.W., Krause, M. & Hanover, J.A. O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats. J. Biol. Chem. 272, 9316–9324 (1997).
O'Donnell, N., Zachara, N.E., Hart, G.W. & Marth, J.D. Ogt-dependent X-chromosome-linked protein glycosylation is a requisite modification in somatic cell function and embryo viability. Mol. Cell. Biol. 24, 1680–1690 (2004).
Hart, G.W., Housley, M.P. & Slawson, C. Cycling of O-linked β-N-acetylglucosamine on nucleocytoplasmic proteins. Nature 446, 1017–1022 (2007).
Hart, G.W. et al. O-GlcNAcylation of key nuclear and cytoskeletal proteins: reciprocity with O-phosphorylation and putative roles in protein multimerization. Glycobiology 6, 711–716 (1996).
Hanover, J.A., Krause, M.W. & Love, D.C. Post-translational modifications: bittersweet memories: linking metabolism to epigenetics through O-GlcNAcylation. Nat. Rev. Mol. Cell Biol. 13, 312–321 (2012).
Sinclair, D.A. et al. Drosophila O-GlcNAc transferase (OGT) is encoded by the Polycomb group (PcG) gene, super sex combs (sxc). Proc. Natl. Acad. Sci. USA 106, 13427–13432 (2009).
Daou, S. et al. Crosstalk between O-GlcNAcylation and proteolytic cleavage regulates the host cell factor-1 maturation pathway. Proc. Natl. Acad. Sci. USA 108, 2747–2752 (2010).
Durgan, D.J. et al. O-GlcNAcylation, novel post-translational modification linking myocardial metabolism and cardiomyocyte circadian clock. J. Biol. Chem. 286, 44606–44619 (2011).
Kim, E.Y. et al. A role for O-GlcNAcylation in setting circadian clock speed. Genes Dev. 26, 490–502 (2012).
Geng, F., Zhu, W., Anderson, R.A., Leber, B. & Andrews, D.W. Multiple post-translational modifications regulate E-cadherin transport during apoptosis. J. Cell Sci. 125, 2615–2625 (2012).
Lefebvre, T. et al. Evidence of a balance between phosphorylation and O-GlcNAc glycosylation of Tau proteins–a role in nuclear localization. Biochim. Biophys. Acta 1619, 167–176 (2003).
Yang, X. et al. Phosphoinositide signalling links O-GlcNAc transferase to insulin resistance. Nature 451, 964–969 (2008).
Pathak, S. et al. O-GlcNAcylation of TAB1 modulates TAK1-mediated cytokine release. EMBO J. 31, 1394–1404 (2012).
Rotty, J.D., Hart, G.W. & Coulombe, P.A. Stressing the role of O-GlcNAc: linking cell survival to keratin modification. Nat. Cell Biol. 12, 847–849 (2010).
Manning, G., Whyte, D.B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934 (2002).
Haltiwanger, R.S., Holt, G.D. & Hart, G.W. Enzymatic addition of O-GlcNAc to nuclear and cytoplasmic proteins: identification of a uridine diphospho-N-acetylglucosamine:peptide beta-N-acetylglucosaminyltransferase. J. Biol. Chem. 265, 2563–2568 (1990).
Kreppel, L.K., Blomberg, M.A. & Hart, G.W. Dynamic glycosylation of nuclear and cytosolic proteins: cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats. J. Biol. Chem. 272, 9308–9315 (1997).
Dong, D.L. & Hart, G.W. Purification and characterization of an O-GlcNAc selective N-acetyl-beta-D-glucosaminidase from rat spleen cytosol. J. Biol. Chem. 269, 19321–19330 (1994).
Gao, Y., Wells, L., Comer, F.I., Parker, G.J. & Hart, G.W. Dynamic O-glycosylation of nuclear and cytosolic proteins: cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain. J. Biol. Chem. 276, 9838–9845 (2001).
Jínek, M. et al. The superhelical TPR-repeat domain of O-linked GlcNAc transferase exhibits structural similarities to importin α. Nat. Struct. Mol. Biol. 11, 1001–1007 (2004).
Clarke, A.J. et al. Structural insights into mechanism and specificity of O-GlcNAc transferase. EMBO J. 27, 2780–2788 (2008).
Martinez-Fleites, C. et al. Structure of an O-GlcNAc transferase homolog provides insight into intracellular glycosylation. Nat. Struct. Mol. Biol. 15, 764–765 (2008).
Lazarus, M.B., Nam, Y., Jiang, J., Sliz, P. & Walker, S. Structure of human O-GlcNAc transferase and its complex with a peptide substrate. Nature 469, 564–567 (2011).
Iyer, S.P. & Hart, G.W. Roles of the tetratricopeptide repeat domain in O-GlcNAc transferase targeting and protein substrate specificity. J. Biol. Chem. 278, 24608–24616 (2003).
Kreppel, L.K. & Hart, G.W. Regulation of a cytosolic and nuclear O-GlcNAc transferase: role of the tetratricopeptide repeats. J. Biol. Chem. 274, 32015–32022 (1999).
Schimpl, M. et al. O-GlcNAc transferase invokes nucleotide sugar pyrophosphate participation in catalysis. Nat. Chem. Biol. 8, 969–974 (2012).
Lazarus, M.B. et al. HCF-1 is cleaved in the active site of O-GlcNAc transferase. Science 342, 1235–1239 (2013).
Vosseller, K. et al. O-linked N-acetylglucosamine proteomics of postsynaptic density preparations using lectin weak affinity chromatography and mass spectrometry. Mol. Cell. Proteomics 5, 923–934 (2006).
Cole, R.N. & Hart, G.W. Cytosolic O-glycosylation is abundant in nerve terminals. J. Neurochem. 79, 1080–1089 (2001).
Wells, L. et al. Mapping sites of O-GlcNAc modification using affinity tags for serine and threonine post-translational modifications. Mol. Cell. Proteomics 1, 791–804 (2002).
Khidekel, N. et al. Probing the dynamics of O-GlcNAc glycosylation in the brain using quantitative proteomics. Nat. Chem. Biol. 3, 339–348 (2007).
Chalkley, R.J., Thalhammer, A., Schoepfer, R. & Burlingame, A.L. Identification of protein O-GlcNAcylation sites using electron transfer dissociation mass spectrometry on native peptides. Proc. Natl. Acad. Sci. USA 106, 8894–8899 (2009).
Hahne, H. et al. Proteome wide purification and identification of O-GlcNAc-modified proteins using click chemistry and mass spectrometry. J. Proteome Res. 12, 927–936 (2013).
Trinidad, J.C. et al. Global identification and characterization of both O-GlcNAcylation and phosphorylation at the murine synapse. Mol. Cell. Proteomics 11, 215–229 (2012).
Hahne, H., Gholami, A.M. & Kuster, B. Discovery of O-GlcNAc-modified proteins in published large-scale proteome data. Mol. Cell. Proteomics 11, 843–850 (2012).
Alfaro, J.F. et al. Tandem mass spectrometry identifies many mouse brain O-GlcNAcylated proteins including EGF domain-specific O-GlcNAc transferase targets. Proc. Natl. Acad. Sci. USA 109, 7280–7285 (2012).
Schutkowski, M. et al. High-content peptide microarrays for deciphering kinase specificity and biology. Angew. Chem. Int. Edn. Engl. 43, 2671–2674 (2004).
Roquemore, E.P. et al. Vertebrate lens alpha-crystallins are modified by O-linked N-acetylglucosamine. J. Biol. Chem. 267, 555–563 (1992).
Housley, M.P. et al. A PGC-1alpha-O-GlcNAc transferase complex regulates FoxO transcription factor activity in response to glucose. J. Biol. Chem. 284, 5148–5157 (2009).
Ball, L.E., Berkaw, M.N. & Buse, M.G. Identification of the major site of O-linked beta-N-acetylglucosamine modification in the C terminus of insulin receptor substrate-1. Mol. Cell. Proteomics 5, 313–323 (2006).
Klein, A.L., Berkaw, M.N., Buse, M.G. & Ball, L.E. O-linked N-acetylglucosamine modification of insulin receptor substrate-1 occurs in close proximity to multiple SH2 domain binding motifs. Mol. Cell. Proteomics 8, 2733–2745 (2009).
Yuzwa, S.A. et al. Mapping O-GlcNAc modification sites on tau and generation of a site-specific O-GlcNAc tau antibody. Amino Acids 40, 857–868 (2011).
Wells, L., Slawson, C. & Hart, G.W. The E2F–1 associated retinoblastoma-susceptibility gene product is modified by O-GlcNAc. Amino Acids 40, 877–883 (2011).
Guo, K. et al. Translocation of HSP27 into liver cancer cell nucleus may be associated with phosphorylation and O-GlcNAc glycosylation. Oncol. Rep. 28, 494–500 (2012).
Khidekel, N., Ficarro, S.B., Peters, E.C. & Hsieh-Wilson, L.C. Exploring the O-GlcNAc proteome: direct identification of O-GlcNAc-modified proteins from the brain. Proc. Natl. Acad. Sci. USA 101, 13132–13137 (2004).
Maury, J.J., Ng, D., Bi, X., Bardor, M. & Choo, A.B. Multiple reaction monitoring mass spectrometry for the discovery and quantification of O-GlcNAc-modified proteins. Anal. Chem. 86, 395–402 (2014).
Gloster, T.M. et al. Hijacking a biosynthetic pathway yields a glycosyltransferase inhibitor within cells. Nat. Chem. Biol. 7, 174–181 (2011).
Lazarus, M.B. et al. Structural snapshots of the reaction coordinate for O-GlcNAc transferase. Nat. Chem. Biol. 8, 966–968 (2012).
Tvaroška, I., Kozmon, S., Wimmerova, M. & Koca, J. Substrate-assisted catalytic mechanism of O-GlcNAc transferase discovered by quantum mechanics/molecular mechanics investigation. J. Am. Chem. Soc. 134, 15563–15571 (2012).
Crooks, G.E., Hon, G., Chandonia, J.M. & Brenner, S.E. WebLogo: a sequence logo generator. Genome Res. 14, 1188–1190 (2004).
Gay, L.M., Zheng, X. & van Aalten, D.M. Molecular recognition: O-GlcNAc transfer: size matters. Nat. Chem. Biol. 7, 134–135 (2011).
Lubas, W.A. & Hanover, J.A. Functional expression of O-linked GlcNAc transferase: domain structure and substrate specificity. J. Biol. Chem. 275, 10983–10988 (2000).
Fujiki, R. et al. GlcNAcylation of histone H2B facilitates its monoubiquitination. Nature 480, 557–560 (2011).
Roquemore, E.P., Chevrier, M.R., Cotter, R.J. & Hart, G.W. Dynamic O-GlcNAcylation of the small heat shock protein alpha B-crystallin. Biochemistry 35, 3578–3586 (1996).
Zhu, Y. et al. O-GlcNAc occurs cotranslationally to stabilize nascent polypeptide chains. Nat. Chem. Biol. 11, 319–325 (2015).
Leavy, T.M. & Bertozzi, C.R. A high-throughput assay for O-GlcNAc transferase detects primary sequence preferences in peptide substrates. Bioorg. Med. Chem. Lett. 17, 3851–3854 (2007).
Liu, X. et al. A peptide panel investigation reveals the acceptor specificity of O-GlcNAc transferase. FASEB J. 28, 3362–3372 (2014).
Gupta, R. & Brunak, S. Prediction of glycosylation across the human proteome and the correlation to protein function. Pac. Symp. Biocomput. 310–322 (2002).
Wang, J., Torii, M., Liu, H., Hart, G.W. & Hu, Z.Z. dbOGAP: an integrated bioinformatics resource for protein O-GlcNAcylation. BMC Bioinformatics 12, 91 (2011).
Jia, C.Z., Liu, T. & Wang, Z.P. O-GlcNAcPRED: a sensitive predictor to capture protein O-GlcNAcylation sites. Mol. Biosyst. 9, 2909–2913 (2013).
Ojida, A., Takashima, I., Kohira, T., Nonaka, H. & Hamachi, I. Turn-on fluorescence sensing of nucleoside polyphosphates using a xanthene-based Zn(II) complex chemosensor. J. Am. Chem. Soc. 130, 12095–12101 (2008).
Lee, H.S. & Thorson, J.S. Development of a universal glycosyltransferase assay amenable to high-throughput formats. Anal. Biochem. 418, 85–88 (2011).
Battye, T.G., Kontogiannis, L., Johnson, O., Powell, H.R. & Leslie, A.G. iMOSFLM: a new graphical interface for diffraction-image processing with MOSFLM. Acta Crystallogr. D Biol. Crystallogr. 67, 271–281 (2011).
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Schüttelkopf, A.W. & van Aalten, D.M.F. PRODRG: a tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr. D Biol. Crystallogr. 60, 1355–1363 (2004).
Acknowledgements
We thank the European Synchrotron Radiation Facility (ESRF) and Diamond Light Source for beam time and assistance. This work was funded by a Wellcome Trust Senior Research Fellowship (WT087590MA) to D.M.F.v.A.
Author information
Authors and Affiliations
Contributions
S.P. and D.M.F.v.A. conceived the study; S.P., D.E.B. and K.R. performed the peptide assays; M.S., K.R. and D.M.F.v.A. performed structural biology; V.S.B. performed peptide synthesis; J.A. and O.A. performed MS studies; S.P., M.S., A.W.S. and D.M.F.v.A. interpreted the data; S.P., M.S. and D.M.F.v.A. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 OGT modifies specific sites on peptide substrates.
Peptides emerging as hits from the screen, ranked by OGT activity. The O-GlcNAc serine or threonine residue in each peptide is highlighted in red. Green shading marks peptides in which the site identified from the screen matches previous reports. Peptides derived from proteins that have been published in the literature as O-GlcNAcylated proteins (with unknown modification sites) are highlighted in blue.
Supplementary Figure 2 Individual structures for all OGT–substrate complexes including electron density maps.
Unbiased Fo-Fc difference electron density for ligands (UDP-5S-GlcNAc and peptide) contoured at 2.25 σ (a, b) or contoured at 3.5 σ after NCS averaging (c, d). E, f Previously reported OGT substrate complexes (PDB ID 4AY6, Schimpl, M. et al., Nat.Chem.Biol. 8: 969, 2012; and PDB ID 4GYY, Lazarus, M.B. et al., Nat.Chem.Biol. 8: 966-968, 2012). The entire sequence of the peptides used in the study is given; underlined residues are represented in the final model, and the amino acid in bold is the O-GlcNAc-modified serine or threonine.
Supplementary Figure 3 Tolerance of different OGT isoforms for single amino acid substitutions.
OGT activity on the reference peptide KKVPVSRA was measured with two different constructs of the enzyme possessing a different number of TPR repeats. Nucleocytoplasmic OGT (ncOGT) is the longest (full length) natural OGT isoform, whereas the truncated construct, OGT (312—1031), was used for crystallographic studies and library screening due to its increased stability. The reference peptide KKVPVSRA represents the optimal OGT hexapeptide sequon except for position −3, where Val was used in order to avoid a potential second O-GlcNAc acceptor. Two N-terminal Lys residues were added, in order to aid peptide solubility. Assay details are given in the Online Methods section. The average of three measurements is shown, with error bars depicting the s.e.m. Activity for each enzyme isoform was normalized to the reference peptide. Full-length human OGT was expressed and purified as described for the truncated construct in Schimpl, M. et al., Nat.Chem.Biol. 8: 969, 2012.
Supplementary Figure 4 List of 32 hexapeptides derived from the peptide-library hits used to generate the sequon for Figure 4.
Experimentally determined O-GlcNAc sites were analyzed for sequence similarities. Peptides were aligned by the modified serine or threonine, and the sequence truncated to include only 3 residues to the N-terminus of the GlcNAc site (“−3”) and 2 residues to the C-terminus of the site (“+2”) as dictated by the ordered binding of peptides in only these subsites from the structural data. The hexapeptides highlighted in yellow were used for the generation of the sequence logo shown in Fig. 4b.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 478 kb)
Supplementary Table 1
Peptide library sequences (XLS 134 kb)
Supplementary Data Set 1
Mass spectra for O-GlcNAc site determination of 26 peptides (PDF 3745 kb)
Rights and permissions
About this article
Cite this article
Pathak, S., Alonso, J., Schimpl, M. et al. The active site of O-GlcNAc transferase imposes constraints on substrate sequence. Nat Struct Mol Biol 22, 744–750 (2015). https://doi.org/10.1038/nsmb.3063
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3063
- Springer Nature America, Inc.
This article is cited by
-
Optimizing the binding of OGT and a peptidic substrate towards pseudo-substrate inhibitors via molecular dynamic simulations
Systems Microbiology and Biomanufacturing (2024)
-
O-GlcNAcylation determines the translational regulation and phase separation of YTHDF proteins
Nature Cell Biology (2023)
-
Glycosyltransferases as targets for therapeutic intervention in cancer and inflammation: molecular modeling insights
Chemical Papers (2022)
-
The human O-GlcNAcome database and meta-analysis
Scientific Data (2021)
-
O-GlcNAcylation: the “stress and nutrition receptor” in cell stress response
Cell Stress and Chaperones (2021)