Abstract
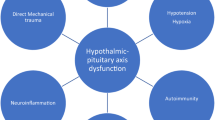
Traumatic brain injury (TBI) is a major public health problem with potentially debilitating consequences for the individual. Hypopituitarism after TBI has received increasing attention over the past decade; development of the condition as a consequence of TBI was previously hardly mentioned in textbooks on the subject. Hypopituitarism has been reported in more than 25% of patients with TBI and is now thought to be one of the most important causes of treatable morbidity in TBI survivors. However, most clinicians dealing with neuroendocrine diseases and TBI generally do not see such a high incidence of hypopituitarism. This disproportion is not clearly explained, but recent data indicate that diagnostic testing, which is designed for high-risk populations and not for a cohort of patients with, for example, de novo isolated growth hormone deficiency (the predominant finding in TBI), might have overestimated the true risk and disease burden of hypopituitarism. In this Opinion article, we discuss current recommendations for post-traumatic hypopituitarism in light of recent evidence.

Similar content being viewed by others
References
Roozenbeek, B., Maas, A. I. & Menon, D. K. Changing patterns in the epidemiology of traumatic brain injury. Nat. Rev. Neurol. 9, 231–236 (2013).
Schneider, H. J., Kreitschmann-Andermahr, I., Ghigo, E., Stalla, G. K. & Agha, A. Hypothalamopituitary dysfunction following traumatic brain injury and aneurysmal subarachnoid hemorrhage: a systematic review. JAMA 298, 1429–1438 (2007).
Molitch, M. E., Clemmons, D. R., Malozowski, S., Merriam, G. R. & Vance, M. L. Evaluation and treatment of adult growth hormone deficiency: an Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 96, 1587–1609 (2011).
Ho, K. K. Consensus guidelines for the diagnosis and treatment of adults with GH deficiency II: a statement of the GH Research Society in association with the European Society for Pediatric Endocrinology, Lawson Wilkins Society, European Society of Endocrinology, Japan Endocrine Society, and Endocrine Society of Australia. Eur. J. Endocrinol. 157, 695–700 (2007).
Benvenga, S., Campenni, A., Ruggeri, R. M. & Trimarchi, F. Hypopituitarism secondary to head trauma. J. Clin. Endocrinol. Metab. 85, 1353–1361 (2000).
Garrahy, A., Sherlock, M. & Thompson, C. J. Management of endocrine disease: Neuroendocrine surveillance and management of neurosurgical patients. Eur. J. Endocrinol. 176, R217–R233 (2017).
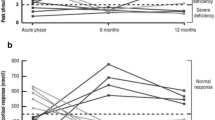
Marina, D., Klose, M., Nordenbo, A., Liebach, A. & Feldt-Rasmussen, U. Early endocrine alterations reflect prolonged stress and relate to 1-year functional outcome in patients with severe brain injury. Eur. J. Endocrinol. 172, 813–822 (2015).
Klose, M. et al. Acute and long-term pituitary insufficiency in traumatic brain injury: a prospective single-centre study. Clin. Endocrinol. (Oxf.) 67, 598–606 (2007).
Hannon, M. J. et al. Acute glucocorticoid deficiency and diabetes insipidus are common after acute traumatic brain injury and predict mortality. J. Clin. Endocrinol. Metab. 98, 3229–3237 (2013).
Agha, A. et al. Anterior pituitary dysfunction in survivors of traumatic brain injury. J. Clin. Endocrinol. Metab. 89, 4929–4936 (2004).
Leal-Cerro, A. et al. Prevalence of hypopituitarism and growth hormone deficiency in adults long-term after severe traumatic brain injury. Clin. Endocrinol. (Oxf.) 62, 525–532 (2005).
Klose, M. et al. Prevalence of posttraumatic growth hormone deficiency is highly dependent on the diagnostic set-up: results from The Danish National Study on Posttraumatic Hypopituitarism. J. Clin. Endocrinol. Metab. 99, 101–110 (2014).
Kelly, D. F. et al. Hypopituitarism following traumatic brain injury and aneurysmal subarachnoid hemorrhage: a preliminary report. J. Neurosurg. 93, 743–752 (2000).
Klose, M. et al. Prevalence and predictive factors of post-traumatic hypopituitarism. Clin. Endocrinol. (Oxf.) 67, 193–201 (2007).
Schneider, M. et al. Predictors of anterior pituitary insufficiency after traumatic brain injury. Clin. Endocrinol. (Oxf.) 68, 206–212 (2008).
Payne, T. H. & Desai, B. R. Examination of medication clinical decision support using Bayes' theorem. Am. J. Health Syst. Pharm. 73, 1876–1878 (2016).
Rasmussen, M. H. et al. Massive weight loss restores 24-hour growth hormone release profiles and serum insulin-like growth factor-I levels in obese subjects. J. Clin. Endocrinol. Metab. 80, 1407–1415 (1995).
Corneli, G. et al. The cut-off limits of the GH response to GH-releasing hormone–arginine test related to body mass index. Eur. J. Endocrinol. 153, 257–264 (2005).
Yuen, K. C. et al. Clinical characteristics, timing of peak responses and safety aspects of two dosing regimens of the glucagon stimulation test in evaluating growth hormone and cortisol secretion in adults. Pituitary 16, 220–230 (2013).
Silva, P. P. et al. Predictors of hypopituitarism in patients with traumatic brain injury. J. Neurotrauma 32, 1789–1795 (2015).
Bhasin, S. et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 95, 2536–2559 (2010).
Morley, J. E. The endocrinology of the opiates and opioid peptides. Metabolism 30, 195–209 (1981).
Persani, L. Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J. Clin. Endocrinol. Metab. 97, 3068–3078 (2012).
Persani, L., Ferretti, E., Borgato, S., Faglia, G. & Beck-Peccoz, P. Circulating thyrotropin bioactivity in sporadic central hypothyroidism. J. Clin. Endocrinol. Metab. 85, 3631–3635 (2000).
Feldt-Rasmussen, U., Hyltoft, P. P., Blaabjerg, O. & Horder, M. Long-term variability in serum thyroglobulin and thyroid related hormones in healthy subjects. Acta Endocrinol. (Copenh.) 95, 328–334 (1980).
Welsh, K. J. & Soldin, S. J. Diagnosis of endocrine disease: How reliable are free thyroid and total T3 hormone assays? Eur. J. Endocrinol. 175, R255–R263 (2016).
Clemmons, D. R. Consensus statement on the standardization and evaluation of growth hormone and insulin-like growth factor assays. Clin. Chem. 57, 555–559 (2011).
Kline, G. A., Buse, J. & Krause, R. D. Clinical implications for biochemical diagnostic thresholds of adrenal sufficiency using a highly specific cortisol immunoassay. Clin. Biochem. 50, 475–480 (2017).
El-Farhan, N., Rees, D. A. & Evans, C. Measuring cortisol in serum, urine and saliva — are our assays good enough? Ann. Clin. Biochem. 54, 308–322 (2017).
Vestergaard, P., Hoeck, H. C., Jakobsen, P. E. & Laurberg, P. Reproducibility of growth hormone and cortisol responses to the insulin tolerance test and the short ACTH test in normal adults. Horm. Metab. Res. 29, 106–110 (1997).
Yuen, K. C., Tritos, N. A., Samson, S. L., Hoffman, A. R. & Katznelson, L. American Associaton of Clinical Endocrinologists and American College of Endocrinology Disease State Clinical Review: Update on growth hormone stimulation testing and proposed revised cut-point for the glucagon stimulation test in the diagnosis of adult growth hormone deficiency. Endocr. Pract. 22, 1235–1244 (2016).
Klose, M. et al. Patient reported outcome in posttraumatic pituitary deficiency: results from The Danish National Study on posttraumatic hypopituitarism. Eur. J. Endocrinol. 172, 753–762 (2015).
Klose, M., Watt, T., Brennum, J. & Feldt-Rasmussen, U. Posttraumatic hypopituitarism is associated with an unfavorable body composition and lipid profile, and decreased quality of life 12 months after injury. J. Clin. Endocrinol. Metab. 92, 3861–3868 (2017).
Mossberg, K. A., Masel, B. E., Gilkison, C. R. & Urban, R. J. Aerobic capacity and growth hormone deficiency after traumatic brain injury. J. Clin. Endocrinol. Metab. 93, 2581–2587 (2008).
Heather, N. L. et al. Permanent hypopituitarism is rare after structural traumatic brain injury in early childhood. J. Clin. Endocrinol. Metab. 97, 599–604 (2012).
Gardner, C. J. et al. GH deficiency after traumatic brain injury: improvement in quality of life with GH therapy: analysis of the KIMS database. Eur. J. Endocrinol. 172, 371–381 (2015).
Maric, N. P. et al. Psychiatric and neuropsychological changes in growth hormone-deficient patients after traumatic brain injury in response to growth hormone therapy. J. Endocrinol. Invest. 33, 770–775 (2010).
Neyeloff, J. L., Fuchs, S. C. & Moreira, L. B. Meta-analyses and Forest plots using a microsoft excel spreadsheet: step-by-step guide focusing on descriptive data analysis. BMC Res. Notes 5, 52 (2012).
Tanriverdi, F. et al. High risk of hypopituitarism after traumatic brain injury: a prospective investigation of anterior pituitary function in the acute phase and 12 months after trauma. J. Clin. Endocrinol. Metab. 91, 2105–2111 (2006).
Dimopoulou, I. et al. Endocrine abnormalities in critical care patients with moderate-to-severe head trauma: incidence, pattern and predisposing factors. Intensive Care Med. 30, 1051–1057 (2004).
van der Eerden, A. W. et al. Should anterior pituitary function be tested during follow-up of all patients presenting at the emergency department because of traumatic brain injury? Eur. J. Endocrinol. 162, 19–28 (2010).
Kokshoorn, N. E. et al. Low prevalence of hypopituitarism after traumatic brain injury: a multicenter study. Eur. J. Endocrinol. 165, 225–231 (2011).
Krahulik, D., Zapletalova, J., Frysak, Z. & Vaverka, M. Dysfunction of hypothalamic–hypophysial axis after traumatic brain injury in adults. J. Neurosurg. 113, 581–584 (2010).
Bavisetty, S. et al. Chronic hypopituitarism after traumatic brain injury: risk assessment and relationship to outcome. Neurosurgery 62, 1080–1093 (2008).
Aimaretti, G. et al. Residual pituitary function after brain injury-induced hypopituitarism: a prospective 12-month study. J. Clin. Endocrinol. Metab. 90, 6075–6092 (2005).
Herrmann, B. L. et al. Hypopituitarism following severe traumatic brain injury. Exp. Clin. Endocrinol. Diabetes 114, 316–321 (2006).
Wachter, D., Gundling, K., Oertel, M. F., Stracke, H. & Boker, D. K. Pituitary insufficiency after traumatic brain injury. J. Clin. Neurosci. 16, 202–208 (2009).
Ulfarsson, T. et al. Pituitary function and functional outcome in adults after severe traumatic brain injury: the long-term perspective. J. Neurotrauma 30, 271–280 (2013).
Prodam, F. et al. Metabolic alterations in patients who develop traumatic brain injury (TBI)-induced hypopituitarism. Growth Horm. IGF Res. 23, 109–113 (2013).
Cuesta, M. et al. Symptoms of gonadal dysfunction are more predictive of hypopituitarism than nonspecific symptoms in screening for pituitary dysfunction following moderate or severe traumatic brain injury. Clin. Endocrinol. (Oxf.) 84, 92–98 (2016).
Popovic, V. et al. Hypopituitarism as a consequence of traumatic brain injury (TBI) and its possible relation with cognitive disabilities and mental distress. J. Endocrinol. Invest. 27, 1048–1054 (2004).
Schneider, H. J. et al. Structured assessment of hypopituitarism after traumatic brain injury and aneurysmal subarachnoid hemorrhage in 1242 patients: the German interdisciplinary database. J. Neurotrauma 28, 1693–1698 (2011).
Schneider, H. J. et al. Prevalence of anterior pituitary insufficiency 3 and 12 months after traumatic brain injury. Eur. J. Endocrinol. 154, 259–265 (2006).
Bondanelli, M. et al. Occurrence of pituitary dysfunction following traumatic brain injury. J. Neurotrauma 21, 685–696 (2004).
Lieberman, S. A., Oberoi, A. L., Gilkison, C. R., Masel, B. E. & Urban, R. J. Prevalence of neuroendocrine dysfunction in patients recovering from traumatic brain injury. J. Clin. Endocrinol. Metab. 86, 2752–2756 (2001).
Acknowledgements
The research salary of U.F.-R. is sponsored by an unrestricted research grant from the Novo Nordisk Foundation.
Author information
Authors and Affiliations
Contributions
M.K. and U.F.-R. researched data for the article, made substantial contributions to discussion of content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Klose, M., Feldt-Rasmussen, U. Chronic endocrine consequences of traumatic brain injury — what is the evidence?. Nat Rev Endocrinol 14, 57–62 (2018). https://doi.org/10.1038/nrendo.2017.103
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2017.103
- Springer Nature Limited
This article is cited by
-
The left–right side-specific endocrine signaling in the effects of brain lesions: questioning of the neurological dogma
Cellular and Molecular Life Sciences (2022)
-
Xenon treatment after severe traumatic brain injury improves locomotor outcome, reduces acute neuronal loss and enhances early beneficial neuroinflammation: a randomized, blinded, controlled animal study
Critical Care (2020)
-
Interactions between hypothalamic pituitary thyroid axis and other pituitary dysfunctions
Endocrine (2018)