Key Points
-
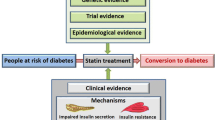
Meta-analyses of clinical trial data reveal a 10–12% increased risk of new-onset diabetes mellitus (NODM) associated with statin therapy; the risk is further increased with intensive treatment regimens
-
Mendelian randomization studies suggest that the effect of statins on glucose homeostasis reflect reduced activity of the target of statin therapy (HMG-CoA reductase)
-
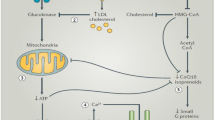
In vitro and in vivo data suggest that statins reduce synthesis of mevalonate pathway products and increase cholesterol loading, which leads to impaired β-cell function and decreased insulin sensitivity
-
The benefits of statins in reducing cardiovascular disease (CVD) events far outweigh the harm of NODM; therefore, the use of statin therapy should be governed by individual patient risk
-
Statin use before a diagnosis of NODM does not increase the prevalence of microvascular disease; furthermore, effects on glycaemic control are small among individuals with diabetes mellitus
-
Statin-treated patients (particularly those with risk factors for NODM) should be monitored for the development of dysglycaemia and given appropriate diet and lifestyle advice to prevent development of NODM
Abstract
Treatment with statins has transformed primary and secondary prevention of cardiovascular disease (CVD), including thrombotic stroke. Evidence-based data demonstrate the benefits and safety of statin therapy and help to guide clinicians in the management of populations at high risk of CVD. Nevertheless, clinical trials, meta-analyses and observational studies highlight a 10–12% increase in new-onset diabetes mellitus (NODM) among patients receiving statins. The risk further increases with intensive therapy and among individuals with known risk factors for NODM. Mechanisms underpinning this effect are not yet fully understood; however, Mendelian randomization studies suggest that they are related to lowered activity of HMG-CoA reductase, the target of statin therapy. In vitro research indicates that statins potentially impair β-cell function and decrease insulin sensitivity but how these findings relate to patients is unknown. In the clinic, statins should be prescribed on the basis of CVD risk and individual patient characteristics. In addition, diet and lifestyle interventions should be emphasized to help mitigate the risk of NODM. Individuals who develop NODM while taking statins do not exhibit increased microvascular disease, which is reassuring. In diabetes mellitus of long duration, the effect of statins on glycaemic control is small and unlikely to be clinically important.


Similar content being viewed by others
References
Endo, A. The discovery and development of HMG-CoA reductase inhibitors. J. Lipid Res. 33, 1569–1582 (1992).
Goldstein, J. L. & Brown, M. S. The LDL receptor. Arterioscler. Thromb. Vasc. Biol. 29, 431–438 (2009).
Brown, M. S. & Goldstein, J. L. The SREBP pathway: regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell 89, 331–340 (1997).
Hunninghake, D. B. HMG-CoA reductase inhibitors. Curr. Opin. Lipidol. 3, 22–28 (1992).
Slater, E. E. & MacDonald, J. S. Mechanism of action and biological profile of HMG CoA reductase inhibitors. A new therapeutic alternative. Drugs 36 (Suppl. 3), 72–82 (1988).
[No authors listed] Randomised trial of cholesterol lowering in 4444 patients with coronary heart disease: the Scandinavian Simvastatin Survival Study (4S). Lancet 344, 1383–1389 (1994).
Baigent, C. et al. Efficacy and safety of cholesterol-lowering treatment; prospective meta-analysis of data from 90,056 participants in 14 randomized trials of statins. Lancet 366, 1276–1278 (2005).
Cholesterol Treatment Trialists' (CTT) Collaborators. Efficacy of cholesterol lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: a meta-analysis. Lancet 371, 117–125 (2008).
Cholesterol Treatment Trialists' (CTT) Collaboration. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomized trials. Lancet 376, 1670–1681 (2010).
Cholesterol Treatment Trialists' (CTT) Collaboration. The effects of lowering LDL-cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomized trials. Lancet 380, 581–590 (2012).
Taylor, F. et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD004816. http://dx.doi.org/10.1002/14651858.CD004816.pub5.
Armitage, J. The safety of statins in clinical practice. Lancet 370, 1781–1790 (2007).
Ridker, P. M. et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N. Engl. J. Med. 359, 2195–2207 (2008).
FDA. FDA drug safety communication: important safety label changes to cholesterol-lowering statin drugs. FDA [online], (2012).
Freeman, D. J. et al. Pravastatin and the development of diabetes mellitus: evidence for a protective treatment effect in the West of Scotland Coronary Prevention Study. Circulation 103, 357–362 (2001).
Coleman, C. I., Reinhart, K., Kluger, J. & White, C.M. The effect of statins on the development of new-onset type 2 diabetes: a meta-analysis of randomized controlled trials. Curr. Med. Res. Opin. 24, 1359–1362 (2008).
Sattar, N. et al. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials. Lancet 375, 735–742 (2010).
Shepherd, J. et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomized controlled trial. Lancet 360, 1623–1630 (2002).
Preiss, D. et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy: a meta-analysis. JAMA 305, 2556–2564 (2011).
Waters, D. D. et al. Cardiovascular event reduction versus new-onset diabetes during atorvastatin therapy: effect of baseline risk factors for diabetes. J. Am. Coll. Cardiol. 61, 148–152 (2013).
Rajpathak, S. N. et al. Statin therapy and risk of developing type 2 diabetes: a meta-analysis. Diabetes Care 32, 1924–1929 (2009).
Dormuth, C. R. et al. Higher potency statins and the risk of new diabetes: multicentre, observational study of administrative databases. BMJ 348, g3244 (2014).
Mora, S. et al. Statins for the primary prevention of cardiovascular events in women with elevated high-sensitivity C-reactive protein or dyslipidemia: results from the Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) and meta-analysis of women from primary prevention trials. Circulation 121, 1069–1077 (2010).
Chan, D. C. & Watts, G. F. Dyslipidemia in the metabolic syndrome and type 2 diabetes: pathogenesis, priorities, pharmacotherapies. Expert Opin. Pharmacother. 12, 13–30 (2011).
Culver, A. L. et al. Statin use and risk of diabetes mellitus in postmenopausal women in the Women's Health Initiative. Arch. Intern. Med. 172, 144–152 (2012).
Ruscica, M., Macchi, C., Morlotti, B., Sirtori, C.R. & Magni, P. Statin therapy and related risk of new-onset type 2 diabetes mellitus. Eur. J. Intern. Med. 25, 401–406 (2014).
Sattar, N. A. et al. The use of statins in people at risk of developing diabetes mellitus: evidence and guidance for clinical practice. Atheroscler. Suppl. 15, 1–15 (2014).
Sekhar, M. S. & Unnikrishnan, M. K. South-Asian population has a higher likelihood for diabetes risk for statins regardless of potency. Med. Hypotheses 84, 283–284 (2015).
Waters, D. D. et al. Predictors of new-onset diabetes in patients treated with atorvastatin: results from 3 large randomized trials. J. Am. Coll. Cardiol. 57, 1535–1545 (2011).
Ridker, P. M. et al. Cardiovascular benefits and diabetes risk of statin therapy in primary prevention: an analysis from the JUPITER trial. Lancet 380, 565–571 (2012).
Arnaboldi, L. & Corsini, A. Could changes in adiponectin drive the effect of statins on the risk of new-onset diabetes? The case of pitavastatin. Atheroscler. Suppl. 16, 1–27 (2015).
Chan, D. C., Pang, J. & Watts, G. F. Pathogenesis and management of the diabetogenic effect of statins: a role for adiponectin and coenzyme Q10. Curr. Atheroscler. Rep. 17, 472 (2015).
Sabatine, M. S. et al. High-dose atorvastatin associated with worse glycemic control: a PROVE-IT TIMI 22 sub study [abstract]. Circulation 110, S834 (2004).
Sasaki, J., Iwashita, M. & Kono, M. Statins: beneficial or adverse for glucose metabolism. J. Atheroscler. Thromb. 13, 123–129 (2006).
Chapman, M. J. et al. Effect of high-dose pitavastatin on glucose homeostasis in patients at elevated risk of new-onset diabetes: insights from the CAPTAIN and PREVAIL-US studies. Curr. Med. Res. Opin. 30, 775–784 (2014).
Cederberg, H. et al. Increased risk of diabetes with statin treatment is associated with impaired insulin sensitivity and insulin secretion: a 6 year follow-up study of the METSIM cohort. Diabetologia 58, 1109–1117 (2015).
Yamakawa, T. et al. Influence of pitavastatin on glucose tolerance in patients with type 2 diabetes mellitus. J. Atheroscler. Thromb. 15, 269–275 (2008).
Teramoto, T. et al. New evidence on pitavastatin: efficacy and safety in clinical studies. Expert Opin. Pharmacother. 11, 817–828 (2010).
Odawara, M. et al. Pitavastatin for the delay or prevention of diabetes development in individuals with impaired glucose tolerance. American Diabetes Association [online], (2013).
Cho, Y. et al. Risk of diabetes in patients treated with HMG-CoA reductase inhibitors. Metabolism 64, 482–488 (2015).
Vallejo-Vaz, A. J. et al. Effect of pitavastatin on glucose, HbA1c and incident diabetes: a meta-analysis of randomized controlled clinical trials in individuals without diabetes. Atherosclerosis 241, 409–418 (2015).
Erqou, S., Lee, C. C. & Adler, A. I. Statins and glycemic control in individuals with diabetes: a systematic review and meta-analysis. Diabetologia 57, 2444–2452 (2014).
Ray, K. K. et al. Effect of intensive control of glucose on cardiovascular outcomes and death in patients with diabetes mellitus: a meta-analysis of randomized controlled trials. Lancet 373, 1765–1772 (2009).
Colhoun, H. M. et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicenter randomized placebo-controlled trial. Lancet 364, 685–696 (2004).
Swerdlow, D. I. & Sattar, N. A dysglycaemic effect of statins in diabetes: relevance to clinical practice? Diabetologia 57, 2433–2435 (2014).
van de Woestijne, A. P., van der Graaf, Y., Westerink, J., Nathoe, H. M. & Visseren, F. L. Effect of statin therapy on incident type 2 diabetes mellitus in patients with clinically manifest vascular disease. Am. J. Cardiol. 115, 441–446 (2015).
Skoumas, J. et al. Statin therapy and risk of diabetes in patients with heterozygous familial hypercholesterolemia or familial combined hyperlipidemia. Atherosclerosis 237, 140–145 (2014).
Panz, V., Immelman, A., Paiker, J., Pilcher, G. & Raal, F. High-dose statin therapy does not induce insulin resistance in patients with familial hypercholesterolemia. Metab. Syndr. Relat. Disord. 10, 351–357 (2012).
Besseling, J., Kastelein, J. J., Defesche, J. C., Hutton, B. A. & Hovingh, G. K. Association between familial hypercholesterolaemia and prevalence of type 2 diabetes mellitus. JAMA 313, 1029–1036 (2015).
Swerdlow, D. J. et al. HMG-coenzyme A reductase inhibition, type 2 diabetes, and bodyweight: evidence from genetic analysis and randomised trials. Lancet 385, 351–361 (2015).
Czech, M. P. Cellular basis of insulin insensitivity in large rat adipocytes. J. Clin. Invest. 57, 1523–1532 (1976).
Olefsky, J. M. Mechanisms of decreased insulin responsiveness of large adipocytes. Endocrinology 100, 1169–1177 (1977).
Friedman, J. M. Obesity in the new millennium. Nature 404, 632–634 (2000).
Kovanen, P. T., Nikkila, E. A. & Miettinen, T. A. Regulation of cholesterol synthesis and storage in fat cells. J. Lipid Res. 16, 211–223 (1975).
Krause, B. R. & Hartman, A. D. Adipose tissue and cholesterol metabolism. J. Lipid Res. 25, 97–110 (1984).
Van Harmelen, V. et al. Increased adipose angiotensinogen gene expression in human obesity. Obes. Res. 8, 337–341 (2000).
Hotamisligil, G. S., Shargill, N. S. & Spiegelman, B. M. Adipose expression of tumour necrosis factor-α: a direct role in obesity-linked insulin resistance. Science 259, 87–91 (1993).
Fried, S. K., Bunkin, D. A. & Greenberg, S. A. Omental and subcutaneous adipose tissues of obese subjects release interleukin-6, depot difference and regulation by glucocorticoid. J. Clin. Endocrinol. Metab. 83, 847–850 (1998).
Le Lay, S. et al. Cholesterol, a cell size-dependent signal that regulates glucose metabolism and gene expression in adipocytes. J. Biol. Chem. 276, 16904–16910 (2001).
Parpal, S., Karlsson, M., Thorn, H. & Strålfors, P. Cholesterol depletion disrupts caveolae and insulin receptor signalling for metabolic control via insulin receptor substrate-1 but not for mitogen-activated protein kinase control. J. Biol. Chem. 276, 9670–9678 (2001).
Gustavasson, J. et al. Localization of the insulin receptor in caveolae of adipocyte plasma membrane. FASEB J. 13, 1961–1971 (1999).
Chamberlain, L. H. Inhibition of isoprenoid biosynthesis causes insulin resistance in 3T3-L1 adipocytes. FEBS Lett. 507, 357–361 (2001).
Takaguri, A., Sotoh, K., Itagaki, M., Tokumitsu, Y. & Ichihara, K. Effects of atorvastatin and pravastatin on signal transduction related to glucose uptake in 3T3L-1 adipocytes. J. Pharmacol. Sci. 107, 80–89 (2008).
Ganesan, S. & Ito, M. K. Coenzyme Q10 ameliorates the reduction in GLUT4 transporter expression induced by simvastatin in 3T3-L1 adipocytes. Metab. Syndr. Relat. Disord. 11, 251–255 (2013).
Donath, M. Y. & Shoelson, S. E. Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 11, 98–107 (2011).
Shoelson, S. E., Lee, J. & Goldfine, A. B. Inflammation and insulin resistance. J. Clin. Invest. 116, 1793–1801 (2006).
Jager, J., Grémeaux, T., Cormont, M., Le Marchand-Brustel, Y. & Tanti, J. F. Interleukin-β-induced insulin resistance in adipocytes through down-regulation of insulin receptor substrate-1 expression. Endocrinology 148, 241–251 (2007).
Lagathu, C. Long term treatment with interleukin-1β induces insulin resistance in murine and human adipocytes. Diabetologia 49, 2162–2173 (2006).
Vandanmagsar, B. et al. The NLRP3 inflammasone instigates obesity-induced inflammation and insulin resistance. Nat. Med. 17, 179–188 (2011).
Stienstra, R. et al. The inflammasone-mediated caspase-1 activation controls adipocyte differentiation and insulin sensitivity. Cell Metab. 12, 593–605 (2010).
Lee H-M. et al. Upregulated NLRP3 inflammasome in patients with type 2 diabetes. Diabetes 62, 194–204 (2013).
Henriskbo, B. D. et al. Fluvastatin causes NLRP3 inflammasome-mediated adipose insulin resistance. Diabetes 63, 3742–3747 (2014).
Baker, W. L., Talati, R., White, C. M. & Coleman, C. I. Differing effects on insulin sensitivity in non-diabetics: a systematic review and meta-analysis. Diabetes Res. Clin. Pract. 87, 98–107 (2010).
Kadowaki, T. et al. Adiponectin and adiponectin receptors in insulin resistance, diabetes and the metabolic syndrome. J. Clin. Invest. 116, 1784–1792 (2006).
Li, S., Shin, H. J., Ding, E. L. & van Dam, R. M. Adiponectin levels and risk of type 2 diabetes: a systematic review and meta-analysis. JAMA 302, 179–188 (2009).
Hara, K. et al. Genetic variation in the gene encoding adiponectin is associated with an increased risk of type 2 diabetes in the Japanese population. Diabetes 51, 536–540 (2002).
Kondo, H. et al. Association of adiponectin mutation with type 2 diabetes: a candidate gene for the insulin resistance syndrome. Diabetes 51, 2325–2328 (2002).
Strumvoll, M. et al. Association of the T-G polymorphism in adiponectin (exon 20 with obesity and insulin sensitivity: interaction with family history of type 2 diabetes. Diabetes 51, 37–41 (2002).
Roehrich, M. E. et al. Insulin-secreting β-cell dysfunction induced by human lipoproteins. J. Biol. Chem. 278, 18368–18375 (2003).
Hao, M., Head, W. S., Gunawardeana, S. C., Hasty, A. H. & Piston, D. W. Direct effect of cholesterol on insulin secretion: a novel mechanism for pancreatic β-cell dysfunction. Diabetes 56, 2328–2338 (2007).
Rütti, S. et al. Low and high density lipoproteins modulate function, apoptosis and proliferation of primary human and murine pancreatic β-cells. Endocrinology 150, 4521–4530 (2009).
Kruit, J. K., Brunham, L. R., Verchere, C. B. & Hayden, M. R. HDL and LDL cholesterol significantly influence β-cell function in type 2 diabetes mellitus. Curr. Opin. Lipidol. 21, 178–185 (2010).
Brunham, L. R. et al. β-cell ABCA1 influences insulin secretion, glucose homeostasis and response to thiazolidinedione treatment. Nat. Med. 13, 340–347 (2007).
Fryirs, M. A. et al. Effects of high-density lipoproteins on pancreatic β-cell insulin secretion. Arterioscler. Thromb. Vasc. Biol. 30, 1642–1648 (2010).
Sturek, J. M. et al. An intracellular role for ABCG1-mediated cholesterol transport in the regulated secretory pathway of mouse pancreatic β cells. J. Clin. Invest. 120, 2575–2589 (2010).
Bogan, J. S., Xu, Y. & Hao, M. Cholesterol accumulation increases insulin granule size and impairs membrane trafficking. Traffic 13, 1466–1480 (2012).
Metz, S. A., Rabaglia, M. E., Stock, J. B. & Kowluru, A. Modulation of insulin secretion from normal rat islets by inhibitors of the post-translational modifications of GTP-binding proteins. Biochem. J. 295, 31–40 (1993).
Li, G., Regazzi, R., Roche, E. & Wollheim, C. B. Blockade of mevalonate production by lovastatin attenuates bombesin and vasopressin potentiation of nutrient-induced insulin secretion in HIT-T15 cells. Biochem. J. 289, 379–385 (1993).
Yada, T., Nakata, M., Shiraishi, T. & Kakei, M. Inhibition by simvastatin, but not pravastatin, of glucose-induced cytosolic Ca2+ signalling and insulin secretion due to blockade of L-type Ca2+ channels in rat islet β-cells. Br. J. Pharmacol. 126, 1205–1213 (1999).
Zhou, J. et al. Effect of glucose metabolism in mouse MIN6 cells. J. Diabetes Res. 2014, 376570 (2014).
Chen, Z. et al. Atorvastatin helps preserve pancreatic β cell function in obese C57BL/6 J mice and the effect is related to increased pancreas proliferation and amelioration of endoplasmic-reticulum stress. Lipids Health Dis. 13, 98 (2014).
Mita, T. et al. Preferable effect of pravastatin compared to atorvastatin on β cell function in Japanese early-state type 2 diabetes with hypercholesterolemia. Endocr. J. 54, 441–447 (2007).
Stumvoll, M. et al. Use of the oral glucose tolerance test to assess insulin release and insulin sensitivity. Diabetes Care 23, 295–301 (2000).
Matsuda, M. & DeFronzo, R. A. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycaemic insulin clamp. Diabetes Care 22, 1462–1470 (1999).
Larsen, S. et al. Simvastatin effects on skeletal muscle: relation to decreased mitochondrial function and glucose intolerance. JAMA 61, 44–53 (2013).
Banach, M. et al. Statin therapy and new-onset diabetes: molecular mechanisms and clinical relevance. Curr. Pharm. Des. 19, 4904–4912 (2013).
Barylski, M. et al. Statins and new-onset diabetes. Curr. Pharm. Des. 20, 3657–3664 (2014).
Sattar, N. & Taskinen, M. R. Statins are diabetogenic—myth or reality? Atheroscler. Suppl. 13, 1–10 (2012).
Lindström, J. et al. The Finnish Diabetes Prevention Study (DPS): lifestyle intervention and 3 year results on diet and physical activity. Diabetes Care 26, 3230–3236 (2003).
Lindström, J. et al. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: follow-up of the Finnish Diabetes Prevention Study. Lancet 368, 1673–1679 (2006).
Knowler, W. C. et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 346, 393–403 (2002).
Diabetes Prevention Program Research Group. The 10 year cost-effectiveness of lifestyle and metformin for diabetes prevention: an intent-to-treat analysis of the DPP/DPPOS. Diabetes Care 35, 723–730 (2012).
Fradkin, J. E., Roberts, B. T. & Rodgers, G. P. What's preventing us from preventing type 2 diabetes? N. Engl. J. Med. 367, 1177–1179 (2012).
Nielsen, S. F. & Nordestgaard, B. G. Statin use before diabetes diagnosis and risk of microvascular disease: a nationwide nested matched study. Lancet Diabetes Endocrinol. 2, 894–900 (2014).
Chen, S. C & Tseng, C. H. Dyslipidaemia, kidney disease and cardiovascular disease in diabetic patients. Rev. Diabet. Stud. 10, 88–100 (2013).
Zafrir, B. & Jain, M. Lipid-lowering therapies, glucose control and incident diabetes: evidence, mechanisms and clinical implications. Cardiovasc. Drugs Ther. 28, 361–377 (2014).
Tennebaum, A. & Fisman, E. Z. Balanced pan-PPAR activator bezafibrate in combination with statin: comprehensive lipids control and diabetes prevention? Cardiovasc. Diabetol. 11, 140–149 (2012).
Author information
Authors and Affiliations
Contributions
D.J.B. and R.C. contributed equally to researching the data for the article, discussion of the content, writing the article and to reviewing and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Betteridge, D., Carmena, R. The diabetogenic action of statins — mechanisms and clinical implications. Nat Rev Endocrinol 12, 99–110 (2016). https://doi.org/10.1038/nrendo.2015.194
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2015.194
- Springer Nature Limited
This article is cited by
-
Long-term statins administration exacerbates diabetic nephropathy via ectopic fat deposition in diabetic mice
Nature Communications (2023)
-
Impact of pitavastatin on new-onset diabetes mellitus compared to atorvastatin and rosuvastatin: a distributed network analysis of 10 real-world databases
Cardiovascular Diabetology (2022)
-
New onset diabetes mellitus and cardiovascular events in Korean patients with acute myocardial infarction receiving high-intensity statins
BMC Pharmacology and Toxicology (2021)
-
Diabetogenic effect of pravastatin is associated with insulin resistance and myotoxicity in hypercholesterolemic mice
Journal of Translational Medicine (2019)
-
TAZ couples Hippo/Wnt signalling and insulin sensitivity through Irs1 expression
Nature Communications (2019)