Abstract
Background/Objectives:
Exercise is considered a protective factor in the prevention of type 2 diabetes, although its role as a sole treatment for pre-diabetes remains unknown. The present meta-analysis compared the effect of exercise-only with exercise-diet interventions on plasma glucose levels among a pre-diabetic population.
Subjects/Methods:
A literature search was conducted using PubMed, EMBASE and Cochrane databases. The Cochrane Collaboration tool was used to assess the quality of each trial. Two reviewers independently performed quality assessment of all included articles. A random effects model was used to calculate the pooled effect.
Results:
A total of 4021 participants from 12 studies were included in this meta-analysis, 2045 of them were in the intervention group and 1976 were in the control group. Compared with the exercise-only interventions, the exercise-diet interventions showed a significant effect on decreasing fasting plasma glucose (FPG) levels, with a weighted mean difference (WMD) =−0.22 mmol/l, 95% confidence interval (CI): −0.25, −0.18 (Z=12.06, P<0.05). The subgroup effect of exercise-only interventions did not produce a statistically significant result (WMD=–0.09 mmol/l, 95% CI: −0.18, 0.00, Z=1.91, P>0.05). According to the intervention periods, the pooled effect in the ⩾2-year group was the highest, and its WMD (95% CI) was −0.24 mmol/l (−0.43,−0.05). The pooled effects were statistically significant among the elderly and those of American and European descent, with WMD (95% CI) being −0.19 mmol/l (95% CI: −0.22, −0.15), −0.17 mmol/l (−0.21,−0.12) and −0.22 mmol/l (−0.27, −0.17), respectively.
Conclusions:
Evidence from published trials indicates that exercise-diet interventions showed a significant effect on decreasing FPG levels.
Similar content being viewed by others
Introduction
Pre-diabetes mellitus is defined as a status between normal individuals and diabetes and involves impaired glucose tolerance (IGT) or impaired fasting glucose (IFG).1, 2, 3, 4 Individuals with pre-diabetes mellitus typically develop type 2 diabetes within 10 years as well as a high risk for cardiovascular disease;1 and approximately 5–10% of individuals with pre-diabetes progress to diabetes each year. Prevalence of pre-diabetes is increasing worldwide, and experts estimate that >470 million people will become pre-diabetic by 2030.5 In 2010, the American Centers for Disease Control and Prevention (CDC) reported that approximately one in three US adults aged ⩾20 years (an estimated 79 million persons) had pre-diabetes mellitus.6 In 2013, Xu et al.7 reported that 493.4 million Chinese adults were pre-diabetic. Interventions during the pre-diabetes stage are crucial to avoid development of full-blown diabetes and other adverse cardio-metabolic risk factors.8
Many studies have examined medication efficacy or lifestyle interventions among type 2 diabetes patients,9,10,11although few have reported on physical activity or exercise-diet interventions among a pre-diabetic population.12, 13 The effect of lifestyle interventions among a pre-diabetic population remains unclear, particularly for exercise-only interventions. To address this research gap, we conducted a systematic review and a meta-analysis of published randomized controlled trials (RCTs) and non-RCTs to examine the efficacy of exercise-only and exercise-diet interventions in the prevention of type 2 diabetes.
Materials and methods
Search strategy
A literature search was conducted for English articles in PubMed from January 1980 to December 2013, EMBASE from January 1988 to December 2013 and Cochrane databases from January 1995 to December 2013. Articles pertaining to dietary or exercise interventions for pre-diabetic individuals were identified.
Key search words included the following: pre-diabetes/IGT/IFT, intervention (lifestyle, exercise, sport and physical activity) and RCT/non-RCT (see Supplementary Appendix 1). Data extraction was performed independently by two investigators (J Wu and L Zheng) using a predefined form (Table 1).
Inclusion and exclusion criteria
Inclusion criteria were as follows: (1) the study population was diagnosed as pre-diabetic and over 40-year old; (2) the study design included RCT or non-RCT; and (3) interventions included exercise-only or exercise-diet. Exclusion criteria were as follows: (1) those who had a history of type2 diabetes mellitus; (2) those who used diabetes medication as part of the intervention.
Subjects
Pre-diabetic patients were defined as IGT or IFG using one of the established criteria from the WHO14 or the American Diabetes Association.15 Studies that defined IGT or IFG using other criteria were included for review if the mean value of the individuals’ plasma glucose levels fell within the range of IGT or IFG as defined by the WHO or American Diabetes Association criteria.14, 15 We also included studies whose participants had a fasting blood glucose of <7.8 mmol/l and their 2-h plasma glucose was between 7.8 and 12.5 mmol/l; or their fasting blood glucose was between 5.3 and 6.9 mmol/l.
Assessment scale
Two researchers (J Wu and L Zheng) independently conducted quality assessment of all the included articles using the Cochrane Collaboration tool.16, 17 If there were discrepancies, a third researcher (GW) took part in the discussion to make the final assessment decision. The Cochrane Collaboration tool assessed the risk of bias in the following domains: selection bias, performance bias, detection bias, attrition bias, reporting bias and other bias. Each domain was classified as ‘low risk,’’unclear’ or ‘high risk.’
Statistical methods
The primary outcome of this meta-analysis was mean change in fasting plasma glucose (FPG) levels. Calculation of mean change in FPG between baseline and follow-up visits was conducted. Heterogeneity between studies was analyzed by means of I2=[(Q–df)/Q] × 100%, where Q is the χ2 heterogeneity statistic and df is the degrees of freedom.18, 19 Meta-regression20 was undertaken to explore the impact of risk of bias for the included studies, such as intervention style and intervention period. A random effects meta-analysis was used to estimate the overall mean changes. I2 >0.5 indicated substantial heterogeneity (statistical heterogeneity).21 A forest plot was conducted to explore the relationship between interventions (exercise-only or exercise-diet) and mean change of FPG in the pre-diabetes population. Publication bias was examined using a funnel plot (Begg’s test).22 All statistical analyses were conducted using the statistical software package Stata12.0 (Stata Corp., College Station, TX, USA).23
Results
Search results
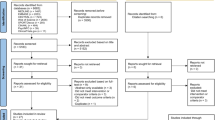
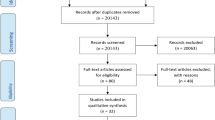
Overall, 206 studies were indexed in the primary screen and search, 12 of which were duplicates. On the basis of our criteria, 13 studies were eligible for a full-text review. Exclusion of 193 articles is shown in Figure 1. One article12 was excluded because the s.d. value of FPG was missing. A total of 12 studies 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35 were included in the present meta-analysis.
Baseline characteristics of participants
Eleven of the 12 studies were RCT. The number of participants in each study ranged from 8 to 256. For the intervention and control groups, there were 2045 and 1976 participants, respectively. Three studies reported the mean change of FPG for exercise-only. The remaining nine studies reported the pooled effect of exercise-dietary interventions (Table 1). Duration of follow-up ranged from 12 weeks to 6 years for the intervention arms.
Quality assessment
Using the Cochrane Collaboration Tool, detailed information of quality assessment is shown in Table 2. Eight27, 28, 29, 31, 32, 33, 34, 35 of the included studies have more than five domains classified as a low risk of bias, whereas four studies24, 25, 26, 30 written by Katula,24 Oldroyd,25 Lu HY26 and Steven30 were deemed unclear in the ‘detection bias’ and other bias domain. Allocation concealment was not mentioned in the studies with the exception of Oldroyd’s article.25 In fact, all of the included studies were open-label trials, whose performance bias was defined as ‘high risk’. Several studies 22, 30, 33 indicated that FPG outcome assessment was blinded to the laboratory technicians.
A meta-regression was carried out to examine reasons for heterogeneity. Regression results revealed that intervention style was the main reason for heterogeneity. Heterogeneity remained present despite the creation of two subgroups for intervention style, and overall I2 was 94.5% (P<0.05). Therefore, random effects models were used to calculate the total effect and subgroup effect. Exercise-diet interventions showed a significant effect on decreasing FPG levels (weighted mean difference (WMD)=−0.22 mmol/l, 95% confidence interval (CI): −0.25, −0.18, Z=12.06, P<0.05). However, the exercise-only intervention did not demonstrate a significant effect (WMD=−0.09 mmol/l, 95% CI: −0.18, 0.00, Z=1.91P>0.05). The overall WMD and its 95% CI were −0.19 mmol/l and (−0.22, −0.15) (Figure 2).
According to four different intervention periods, the shortest period intervention (<1 year) did not display a significant effect for glucose control (WMD=−0.12 mmol/l, 95% CI: −0.29, 0.05, Z=1.35, P>0.05). There was a significant effect (WMD=−0.20 mmol/l, 95% CI: −0.25, −0.14, Z=7.19, P<0.05) in the 1-year subgroup. The longer the intervention period, the higher the subtotal effect (Figure 3). The pooled effect in the ⩾2-year subgroup was the highest with WMD (95% CI)=−0.24 mmol/l (−0.43, −0.05).
As for different regions, significant heterogeneity existed among studies conducted in America (I2=98.30%) and China (I2=94.50%) but not in Europe (I2=47.00%). The subtotal effect was varied in different regions. The studies conducted among the European population displayed a higher subtotal effect, and its WMD was −0.22 mmol/l and its 95% CI was (−0.27, −0.17) with Z=8.01 and P<0.05. WMD (95% CI) in the American population was −0.17 mmol/l (−0.21, −0.12).
When participants were divided into two age groups (40–55 years and ⩾55 years), no significant subgroup pooled effects were found in the younger subgroup (WMD=−0.27 mmol/l, 95% CI: −0.60, 0.05, Z=1.65, P>0.05). However, in the elderly subgroup, there was a significant effect (WMD=−0.19 mmol/l, 95% CI: −0.22, −0.15, Z=10.31, P<0.05). Among the two groups, the subtotal effects were −0.27(−0.60, 0.05) and −0.19 (−0.22, −0.15), respectively. The Begg funnel plot showed an asymmetrical distribution and a Kendall score of 7 (P=0.865), indicating no statistical evidence of publication bias in the present meta-analysis (Figure 4).
Discussion
The current meta-analysis calculated the pooled effect of exercise-only and exercise-diet interventions on FPG control. Meta-regression analysis demonstrated significant heterogeneity among the included studies. However, the clinical heterogeneity among articles was available as the effect of each study had the same direction. Subgroup analysis and random effects models were used to examine the pooled effect based on different intervention style, period, region and age group.
Results reported from a Da Qing study displayed that the exercise-only intervention may decrease the risk of diabetes.28 However, the findings of our current analysis showed that the change in FPG levels was not significant in the exercise-only interventions. Exercise-diet interventions had more significant subtotal effects in decreasing FPG. One possible explanation for these findings could be due to the status of weight loss, which had a strong relationship with the FPG level.36, 37, 38 It was clear that exercise-only had a weaker effect compared with the exercise-diet interventions in decreasing body weight.39 Furthermore, there was limited data available for exercise-only interventions, thereby decreasing reliability. Among the included studies, the effect of exercise-only intervention was inconsistent. In addition, some of the studies reported on exercise time, whereas others reported exercise intensity, causing difficultly in calculating a standard value of exercise quantity. Therefore, more studies focusing on exercise-only interventions are needed to explore the relationship of FPG changes in a pre-diabetic population.
To our knowledge, this is the first meta-analysis assessing intervention length, as it relates to change in FPG levels. According to findings reported by Lindstrom et al.,40 the lifestyle intervention (exercise-diet) group had a significant change in FPG levels during the first year. There was almost no significant effect after follow-up from 3 to 4 years. However, our present meta-analysis findings are to the contrary, indicating that the longer the intervention the greater the intervention effect (including exercise-only and exercise-diet), with no significant effect when the intervention period was <1 year.
The subgroup analysis indicated that the subtotal effect was significant in the 55-year age group but not among the 40- to 55-year age group. These results showed that the intervention effect (exercise-only and exercise-diet intervention) may be stronger in the elderly population. Our findings indicated that age was also correlated with glucose metabolism and were consistent with several previous studies conducted in human 41, 42 and animal models.43 The current results also showed that the intervention group had a better outcome among the European and the American population than in the Asian population. Research by Danaei et al.44 found that glucose metabolism varied by nations and regions.
The strength of our investigation was that the majority of included studies were RCTs, with the exception of one article written by Steven et al.30 In addition, large numbers of participants and varying geographic locations contributed to a diverse and a comprehensive set of data. The present analysis also had several limitations as studies included in this meta-analysis were limited to articles published in English. In addition, 12 of the studies were heterogeneous, and despite subgroup analysis most of the statistical heterogeneities remained. Therefore, further rigorous studies are needed to confirm this finding. Furthermore, the intervention period range varied from 12 weeks to 6 years, which may add to the heterogeneity in our analysis. Finally, the quantitative information of exercise-diet was not assessed between studies according to the same standard, which could potentially modify the association between intervention effect and the FPG level.
Both exercise-only and exercise-diet interventions have displayed effects on decreasing FPG, with better results in the later group. The current data also showed that the pooled effect was significant in longer intervention periods (⩾1 year), the elderly population (⩾55 years age) and Europeans/Americans. More studies are needed to assess the efficacy of exercise-only interventions among a pre-diabetic population.
References
Pour OR, Dagogo-Jack S . Prediabetes as a therapeutic target. Clin Chem 2011; 57: 215–220.
Hopper I, Billah B, Skiba M, Krum H . Prevention of diabetes and reduction in major cardiovascular events in studies of subjects with prediabetes: meta-analysis of randomised controlled clinical trials. Eur J Cardiovasc Prev Rehabil 2011; 18: 813–823.
Association AD. Diagnosis and classification of diabetes mellitus. Diabetes Care 2010; 33: S62–S69.
Yilmaz Y, Senates E, Yesil A, Ergelen R, Colak Y . Not only type 2 diabetes but also prediabetes is associated with portal inflammation and fibrosis in patients with non-alcoholic fatty liver disease. J Diabetes Complications 2014; 28: 328–331.
Tabak AG, Herder C, Rathmann W, Brunner EJ, Kivimaki M . Prediabetes: a high-risk state for diabetes development. Lancet 2012; 379: 2279–2290.
Centers for Disease Control and Prevention (CDC). Awareness of prediabetes—United States, 2005–2010. MMWR Morb Mortal Wkly Rep 2013; 62: 209–212.
Xu Y, Wang L, He J, Bi Y, Li M, Wang T et al. Prevalence and control of diabetes in Chinese adults. JAMA 2013; 310: 948–959.
Li G, Zhang P, Wang J, Gregg EW, Yang W, Gong Q et al. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention Study: a 20-year follow-up study. Lancet 2008; 371: 1783–1789.
Cefalu WT, Leiter LA, Yoon KH, Arias P, Niskanen L, Xie J et al. Efficacy and safety of canagliflozin versus glimepiride in patients with type 2 diabetes inadequately controlled with metformin (CANTATA-SU): 52 week results from a randomised, double-blind, phase 3 non-inferiority trial. Lancet 2013; 382: 941–950.
Charbonnel B, Steinberg H, Eymard E, Xu L, Thakkar P, Prabhu V et al. Efficacy and safety over 26 weeks of an oral treatment strategy including sitagliptin compared with an injectable treatment strategy with liraglutide in patients with type 2 diabetes mellitus inadequately controlled on metformin: a randomised clinical trial. Diabetologia 2013; 56: 1503–1511.
Linmans JJ, Spigt MG, Deneer L, Lucas AE, de Bakker M, Gidding LG et al. Effect of lifestyle intervention for people with diabetes or prediabetes in real-world primary care: propensity score analysis. BMC. Fam Pract 2011; 12: 95.
Lindahl B, Nilsson TK, Jansson JH, Asplund K, Hallmans G . Improved fibrinolysis by intense lifestyle intervention. A randomized trial in subjects with impaired glucose tolerance. J Intern Med 1999; 246: 105–112.
Zhou QP, Oh KM . Comparison of lifestyle behaviors and related factors between Asian American and white adults with prediabetes. Nurs Health Sci 2012; 14: 58–66.
Alberti KG, Zimmet PZ . Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 1998; 15: 539–553.
Genuth S, Alberti KG, Bennett P, Buse J, Defronzo R, Kahn R et al. Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 2003; 26: 3160–3167.
Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011; 343: d5928.
Zhang D, Chen L, Peng K, Xing F, Wang H, Xiang Z . Effectiveness and safety of the posterior approach with soft tissue repair for primary total hip arthroplasty: a meta-analysis. Orthop Traumatol Surg Res 2015; 101: 39–44.
Al-Mallah M, Khawaja O, Sinno M, Alzohaili O, Samra AB . Do angiotensin converting enzyme inhibitors or angiotensin receptor blockers prevent diabetes mellitus? A meta-analysis. Cardiol J 2010; 17: 448–456.
Higgins JP, Thompson SG, Deeks JJ, Altman DG . Measuring inconsistency in meta-analyses. BMJ 2003; 327: 557–560.
Hayashino Y, Jackson JL, Hirata T, Fukumori N, Nakamura F, Fukuhara S et al. Effects of exercise on C-reactive protein, inflammatory cytokine and adipokine in patients with type 2 diabetes: a meta-analysis of randomized controlled trials. Metabolism 2014; 63: 431–440.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 2010; 8: 336–341.
Mesfin YM, Hailemariam D, Biadglign S, Kibret KT . Association between HIV/AIDS and multi-drug resistance tuberculosis: a systematic review and meta-analysis. PLoS One 2014; 9: e82235.
Luo C, Zhang Y, Ding Y, Shan Z, Chen S, Yu M et al. Nut consumption and risk of type 2 diabetes, cardiovascular disease, and all-cause mortality: a systematic review and meta-analysis. Am J Clin Nutr 2014; 100: 256–269.
Katula JA, Vitolins MZ, Morgan TM, Lawlor MS, Blackwell CS, Isom SP et al. The Healthy Living Partnerships to Prevent Diabetes study: 2-year outcomes of a randomized controlled trial. Am J Prev Med 2013; 44: S324–S332.
Oldroyd JC, Unwin NC, White M, Mathers JC, Alberti KG . Randomised controlled trial evaluating lifestyle interventions in people with impaired glucose tolerance. Diabetes Res Clin Pract 2006; 72: 117–127.
Lu YH, Lu JM, Wang SY, Li CL, Zheng RP, Tian H et al. Outcome of intensive integrated intervention in participants with impaired glucose regulation in China. Adv Ther 2011; 28: 511–519.
Lindstrom J, Louheranta A, Mannelin M, Rastas M, Salminen V, Eriksson J et al. The Finnish Diabetes Prevention Study (DPS): lifestyle intervention and 3-year results on diet and physical activity. Diabetes Care 2003; 26: 3230–3236.
Pan XR, Li GW, Hu YH, Wang JX, Yang WY, An ZX et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study. Diabetes Care 1997; 20: 537–544.
Carr DB, Utzschneider KM, Boyko EJ, Asberry PJ, Hull RL, Kodama K et al. A reduced-fat diet and aerobic exercise in Japanese Americans with impaired glucose tolerance decreases intra-abdominal fat and improves insulin sensitivity but not beta-cell function. Diabetes 2005; 54: 340–347.
Malin SK, Gerber R, Chipkin SR, Braun B . Independent and combined effects of exercise training and metformin on insulin sensitivity in individuals with prediabetes. Diabetes Care 2012; 35: 131–136.
Eriksson J, Lindstrom J, Valle T, Aunola S, Hamalainen H, Ilanne-Parikka P et al. Prevention of type II diabetes in subjects with impaired glucose tolerance: the Diabetes Prevention Study (DPS) in Finland. Study design and 1-year interim report on the feasibility of the lifestyle intervention programme. Diabetologia 1999; 42: 793–801.
Tuomilehto J, Lindstrom J, Eriksson JG, Valle TT, Hamalainen H, Ilanne-Parikka P et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001; 344: 1343–1350.
Uusitupa M, Lindi V, Louheranta A, Salopuro T, Lindstrom J, Tuomilehto J . Long-term improvement in insulin sensitivity by changing lifestyles of people with impaired glucose tolerance: 4-year results from the Finnish Diabetes Prevention Study. Diabetes 2003; 52: 2532–2538.
Roumen C, Corpeleijn E, Feskens EJ, Mensink M, Saris WH, Blaak EE . Impact of 3-year lifestyle intervention on postprandial glucose metabolism: the SLIM study. Diabet Med 2008; 25: 597–605.
Mensink M, Blaak EE, Corpeleijn E, Saris WH, de Bruin TW, Feskens EJ . Lifestyle intervention according to general recommendations improves glucose tolerance. Obes Res 2003; 11: 1588–1596.
Heilbronn LK, Noakes M, Clifton PM . Effect of energy restriction, weight loss, and diet composition on plasma lipids and glucose in patients with type 2 diabetes. Diabetes Care 1999; 22: 889–895.
Kim MJ, Hur KY . Short-term outcomes of laparoscopic single anastomosis gastric bypass (LSAGB) for the treatment of type 2 diabetes in lower BMI (<30 kg/m) patients. Obes Surg 2014; 24: 1044–1051.
Kahleova H, Mari A, Nofrate V, Matoulek M, Kazdova L, Hill M et al. Improvement in beta-cell function after diet-induced weight loss is associated with decrease in pancreatic polypeptide in subjects with type 2 diabetes. J Diabetes Complications 2012; 26: 442–449.
Beavers DP, Beavers KM, Loeser RF, Walton NR, Lyles MF, Nicklas BJ et al. The independent and combined effects of intensive weight loss and exercise training on bone mineral density in overweight and obese, older adults with osteoarthritis. Osteoarthritis Cartilage 2014; 22: 726–733.
Lindstrom J, Peltonen M, Eriksson JG, Ilanne-Parikka P, Aunola S, Keinanen-Kiukaanniemi S et al. Improved lifestyle and decreased diabetes risk over 13 years: long-term follow-up of the randomised Finnish Diabetes Prevention Study (DPS). Diabetologia 2013; 56: 284–293.
Tessari P . Changes in protein, carbohydrate, and fat metabolism with aging: possible role of insulin. Nutr Rev 2000; 58: 11–19.
Noordam R, Gunn DA, Tomlin CC, Maier AB, Mooijaart SP, Slagboom PE et al. High serum glucose levels are associated with a higher perceived age. Age (Dordr) 2013; 35: 189–195.
Higgins J, Proctor D, Denyer G . Aging changes tissue-specific glucose metabolism in rats. Metabolism 1999; 48: 1445–1449.
Danaei G, Finucane MM, Lu Y, Singh GM, Cowan MJ, Paciorek CJ et al. National, regional, and global trends in fasting plasma glucose and diabetes prevalence since 1980: systematic analysis of health examination surveys and epidemiological studies with 370 country-years and 2.7 million participants. Lancet 2011; 378: 31–40.
Acknowledgements
This study was supported by grants from the National Nature Science Foundation of China (no. 81170325), Foundation of Shanghai Municipal Key Discipline (ZK2012A27) and Foundation of Key Disciplines Group Project of Pudong Health Bureau of Shanghai (PWZxq2014-01).
Author contributions
JL, HF, L Zheng and J Wu designed and wrote the manuscript; GW was a third reviewer to aid in the decision; YM, GP, HF and GW gave the suggestions not only on detail information but also in language. L Zou, L Zhang, MZ, ZL and J Wang took part in the discussion about the manuscript. We are grateful to all individuals who took part in for searching references. We are also indebted to Yanfei Li and Jing Wu for their theoretical support on exercise and diet.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Clinical Nutrition website
Supplementary information
Rights and permissions
About this article
Cite this article
Zheng, L., Wu, J., Wang, G. et al. Comparison of control fasting plasma glucose of exercise-only versus exercise-diet among a pre-diabetic population: a meta-analysis. Eur J Clin Nutr 70, 424–430 (2016). https://doi.org/10.1038/ejcn.2015.128
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2015.128
- Springer Nature Limited