Abstract
Background/Objectives:
Diets high in saturated and trans fat and low in unsaturated fat may increase type 2 diabetes (T2D) risk, but studies on foods high in fat per unit weight are sparse. We assessed whether the intake of vegetable oil, butter, margarine, nuts and seeds and cakes and cookies is related to incident T2D.
Subjects/Methods:
A case-cohort study was conducted, nested within eight countries of the European Prospective Investigation into Cancer (EPIC), with 12 403 incident T2D cases and a subcohort of 16 835 people, identified from a cohort of 340 234 people. Diet was assessed at baseline (1991–1999) by country-specific questionnaires. Country-specific hazard ratios (HRs) across four categories of fatty foods (nonconsumers and tertiles among consumers) were combined with random-effects meta-analysis.
Results:
After adjustment not including body mass index (BMI), nonconsumers of butter, nuts and seeds and cakes and cookies were at higher T2D risk compared with the middle tertile of consumption. Among consumers, cakes and cookies were inversely related to T2D (HRs across increasing tertiles 1.14, 1.00 and 0.92, respectively; P-trend <0.0001). All these associations attenuated upon adjustment for BMI, except the higher risk of nonconsumers of cakes and cookies (HR 1.57). Higher consumption of margarine became positively associated after BMI adjustment (HRs across increasing consumption tertiles: 0.93, 1.00 and 1.12; P-trend 0.03). Within consumers, vegetable oil, butter and nuts and seeds were unrelated to T2D.
Conclusions:
Fatty foods were generally not associated with T2D, apart from weak positive association for margarine. The higher risk among nonconsumers of cakes and cookies needs further explanation.
Similar content being viewed by others
Introduction
Diet is considered to be a crucial factor in the development of type 2 diabetes (T2D) and there has been a considerable interest in the role of the fat composition of the diet.1, 2, 3 The amount of total fat in the diet may increase the risk of T2D by stimulating weight gain, whereas the type of dietary fat may influence risk of T2D independent of obesity. For example, increasing intakes of saturated fat but not of polyunsaturated fat induces insulin resistance in short-term studies.4 Also, isocalorically replacing long-chain polyunsaturated fat for saturated fat improves insulin sensitivity in people with normal to moderately increased body weights5 and vice versa.6 In obese people with the metabolic syndrome, however, reducing saturated fat intake, by substitution with monounsaturated fat or as part of a diet low in fat and high in complex carbohydrates, did not improve insulin resistance.7 Trans-fatty acids, finally, may also increase T2D risk by inducing insulin resistance.3, 8
Findings from long-term prospective studies on estimated habitual intake of specific dietary fats and incident T2D have been less clear. The intake of saturated fatty acids has generally not been related to incident T2D,9 whereas high habitual intakes of trans-fatty acids have been related to higher risk.10 Finally, high intake of polyunsaturated fatty acids10 or high ratios of polyunsaturated to saturated fatty acids11 have been related to lower diabetes risk, but not all studies are consistent.12, 13
Prospective studies on the consumption of foods rich in fat and risk of T2D have mainly focused on meat,14, 15 fatty fish,16, 17 high-fat dairy18, 19, 20 and nuts and seeds.21 Other fatty foods like edible oils, butter, margarine and also cakes and cookies, which contain up to 30% fat-by-weight,22 have only sparingly been investigated. Consumption of vegetable oils and nuts and seeds may lower T2D risk, at least in part, because they are high in unsaturated fatty acids. Butter, on the other hand, may increase T2D risk because of its high content of saturated fatty acids (~50% saturated fat-by-weight). Margarine may also increase T2D risk, because of its high concentration of industrial trans-fatty acids, at least until the mid-1990s.
In the current study, we investigated the associations between five foods that have a high fat-by-weight content—vegetable oil, butter, margarine, nuts and seeds and cakes and cookies—and incident T2D in a large prospective case-cohort study nested within the European Prospective Investigation into Cancer and Nutrition (EPIC), the EPIC-InterAct Study. The large variability in the intake of fatty acids and their food sources in the EPIC study23 provided a unique opportunity to assess the associations between fatty food intakes and the risk of T2D. Results on intakes of dairy including cheese,24 fatty fish17 and meat25 have been reported separately for EPIC-InterAct.
Materials and methods
Study design and study population
The EPIC-InterAct Study is a case-cohort study nested within EPIC,26 as previously described.27 Briefly, InterAct consortium partners ascertained and verified incident cases of type 2 diabetes occurring in EPIC cohorts between 1991 and 2007 from 8 of the 10 EPIC countries (France, Italy, Spain, the UK, the Netherlands, Germany, Denmark and Sweden; n=455 680), excluding those without stored blood (n=109 625) or reported diabetes status (n=5821). Among this sample of 340 234 participants with 3.99 million years of follow-up, a subcohort of 16 835 individuals was randomly selected from those with available stored blood and buffy coat, stratified by the centre. After exclusion of 681 individuals with either prevalent diabetes, lack of information on diabetes status, or with post-censoring diabetes, the subcohort had 16 154 participants. Because of random selection, this subcohort also included 778 individuals who had developed incident of T2D during follow-up.
Case ascertainment and verification
Incident diabetes cases were ascertained and verified by each participating EPIC centre, as previously described.27 New cases occurring up to until 31 December 2007 were ascertained using multiple data sources: self-report of doctor-diagnosed diabetes or diabetes medication, linkage to primary or secondary care registers, linkage to pharmacy database and hospital admissions data or mortality data. Verification of incident diabetes was undertaken, for participants with <two independent sources of information, by individual medical record checking or confirmation from another independent source of information. Follow-up was censored at the date of diagnosis, 31 December 2007 or date of death, whichever came first. In total, 12 403 incident diabetes cases were identified, including the 778 cases in the subcohort. Participants in the subcohort were similar to all EPIC participants eligible for inclusion in EPIC-InterAct. They were followed for a median (inter-quartile range) of 12.3 (11.0, 13.4) years. Incident cases of T2D were followed for a median (inter-quartile range) of 6.8 (4.4, 9.5) years.
Study sample for current analysis
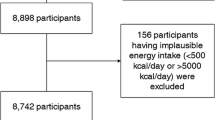
From a total sample of 27 779 participants, we made exclusions because of the following: incomplete dietary information (n=117; 70 cases, 47 nondiabetic subcohort participants); in the lowest or highest 1% of the ratio of energy intake to energy expenditure in the original EPIC centres (n=619; 339 cases, 280 nondiabetic subcohort participants); missing information on important covariates (n=955; 435 cases, 520 nondiabetic subcohort participants); and prevalent myocardial infarction or stroke (n=781; 500 cases, 281 nondiabetic subcohort participants). Thus, the current analysis included 25 307 participants, among whom 11 059 were cases and 14 939 were subcohort participants (including 691 cases).
Assessment of diet
Habitual diet of the previous year was assessed at recruitment between 1991 and 1999 by country-specific questionnaires.28 Self-administered quantitative dietary questionnaires were used in Florence, Turin and Varese (all Italy), the Netherlands and Germany. A similar questionnaire, but structured by eating occasions, was used in France. Centres in Spain and Ragusa (Italy) conducted face-to-face interviews using computerized questionnaires. Semi-quantitative questionnaires with standard portion sizes were used in the UK, Denmark, Naples (Italy) and in Sweden. Intake (g/day) of similar individual foods were aggregated into food groups. Intake of energy, total fat and alcohol were calculated by using a standardized nutrient database;29 detailed information on trans-fatty acids was not available.
In addition, diet was also assessed with a standardized 24-h recall method in a stratified subsample of ~8% (n=36 900) of EPIC study participants, of whom 2161 participants were included in the current analysis. On the basis of this sample, ecological correlation coefficients for the paired study centres’ mean intakes from the dietary questionnaires with the study centres’ mean intakes from the 24-h recall were moderate to high for the fatty foods under investigation, except for nuts and seeds in women (r=0.10) (see Table 1).
Other measures
Standardized questionnaires were used to collect information on lifestyle at baseline including questions on highest achieved education level and cigarette smoking. Physical activity was classified as ‘inactive,’ ‘moderately inactive,’ ‘moderately active’ and ‘active’ (Cambridge index) on the basis of questions about sports, cycling and occupational activity.30
Height, body weight and waist circumference were measured by trained staff using standardized protocols, except in Oxford (UK) and France, where these variables were self-reported3 and in Umea (Sweden), where waist circumference was not measured. Basal metabolic rate was calculated by using Schofield equations.32
Information on prevalent chronic conditions—that is, hypertension, hyperlipidemia, heart disease and stroke—was assessed in most centres, except for hypertension in Sweden. Information on family history of T2D among first-degree relatives was collected for all participants, except those in Italy, Spain, Heidelberg (Germany) and Oxford (UK).
Evaluation of energy intake
Accuracy of reported energy intake was evaluated by the ratio of reported energy to estimated energy expenditure (EI/EE) under the assumption of energy balance. Cut-points for under-reporting and over-reporting were calculated from the 95% confidence limits of agreement between EI and EE, with a coefficient of variation for energy expenditure of 15% as recommended by Black.33 The lower confidence limit was 0.7 and the upper limit 1.3. EE was estimated on the basis of the sum of resting energy expenditure (as estimated by basal metabolic rate), physical activity energy expenditure and a component for the thermic effect of food (assumed to be 10% of total EE). Physical activity energy expenditure was objectively estimated with a heart rate and movement sensor in another sample of adults in the 10 EPIC countries (n~200 per centre).34 Each sample was ~200 people large and had the same age and sex distribution as the original cohort at recruitment. Mean values of physical activity energy expenditure (kcal/kg/day) by sex and by reported physical activity categories (Cambridge index) from that study were used to estimate physical activity energy expenditure in the current study. The number of participants with EI/EEs >1.3 (n=1130) was too small to perform a meaningful separate analysis for this group.
Statistics
The association between intake of fatty foods and incident T2D was analyzed by Prentice-weighted Cox proportional-hazards regression models with age as the underlying timescale.35 Each fatty food was analyzed in categories. The nonconsumers were separately analyzed and the consumers were divided into tertiles of intake on the basis of the distributions in the entire subcohort. The middle tertile among consumers served as the referent because all countries were sufficiently represented in this category for all five fatty foods. When a country contributed fewer compared with 20 subcohort members to a particular category, that country was omitted from the meta-analysis of that category (see Supplementary Table 1). Hazard ratios (HRs) of incident type 2 diabetes across categories were calculated within each country separately and then combined using random-effects meta-analysis. Heterogeneity between countries was quantified by the I2 measure.
We controlled for confounding in three regression models: model A included centre and sex; model B further adjusted for education, energy intake, smoking, alcohol intake and physical activity; and model C assessed the effect of adjustment for body mass index (BMI).
In sensitivity analyses, we assessed potential deviations from linearity in the associations more rigorously by fitting intakes of each fatty food as fractional polynomials while adjusting for model C covariates. Because some fatty foods had a sizeable number of nonconsumers, we used a procedure that allows for a proportion unexposed.36 Briefly, in a two-stage approach, we assessed whether the best-fitting fractional polynomial or the indicator variable for nonconsumption or both were essential for a good fit to the data by omitting one at a time from the model.36 This procedure was conducted for all countries combined while allowing each centre to have its own baseline hazard function. Further, we checked the robustness of our findings to adjustment of the consumption of vegetables, fruit, red meat, processed meat and coffee. We also stratified the analysis by BMI <25 versus ⩾25 kg/m2. Interaction by BMI (in kg/m2) was formally tested by pooling country-specific interaction terms in random-effects meta-analysis. The effect of potential misreporting of energy intake was tested by adjusting for the EI/EE ratio and by modelling product terms with fatty food intake. To account for latency, we omitted the first 2 years of follow-up. Finally, we repeated our analysis after excluding participants with hypertension or hyperlipidemia.
The potential for measurement error in fatty food consumption was evaluated by regressing intake values from the 24-h recall on those from the diet questionnaire in a mixed linear model while adjusting for age, sex, season and day of 24-h recall, energy intake and BMI. A random intercept and a random slope were fitted, allowing the association between 24-h recalls and dietary questionnaires to differ across countries. For each country, the obtained regression dilution ratio was used to correct the HRs in model C by exp(log(HR)/regression dilution ratio). Standard errors of the corrected HR were calculated by the delta method.37 Corrected country-specific HRs were then combined with random-effects meta-analysis. Analyses were performed with SAS Enterprise Guide (version 4.3) and Stata SE (version 12).
Results
Consumption of fatty foods in the subcohort
The percentage of nonconsumers was lowest for cakes and cookies (8.5%) and the highest for butter (48.3%). Median intakes (5th, 95th percentiles) in g/day of fatty foods from the dietary questionnaires in the subcohort ranged from 0.1 (0.0, 20.1) for butter to 30.0 (0.0, 125.0) for cakes and cookies (Table 1). Intakes of most fatty foods differed markedly between the European cohorts (Supplementary Tables 1 and 2). In both men and women, Spain and Italy had the highest intake of vegetable oil, whereas this was the lowest in Denmark and Sweden. Butter intake was generally low in all countries, with the highest intakes seen in the Netherlands and Germany. Margarine intake was the highest in Sweden and the lowest in France, Italy and Spain. The Netherlands had the highest intake of nuts and seeds. Finally, cake and cookie intake in men was the highest in the UK and in women the highest in Germany. Intakes from the diet questionnaires with those from the 24-h dietary recall correlated reasonably well for fatty foods other than butter (men and women) and nuts and seeds (men).
Characteristics associated with fatty food consumption in the subcohort
Categories of consumption of each fatty food were different in many characteristics (Supplementary Tables 3). This was most notable when nonconsumers were compared with consumers of any amount. For example, nonconsumers of butter, margarine, nuts and seeds and cakes and cookies were less educated, had higher BMIs and waist circumferences and engaged less in physical activity than consumers. Nonconsumers of butter and in particular of margarine and cakes and cookies also consumed more vegetables and fruit.
Differences between consumers were also seen. For example, participants with higher intakes of vegetable consumption had higher BMIs and waist circumferences, were less physically active and had higher intakes of vegetables and fruit and lower intakes of soft drinks and coffee compared with participants with lower intakes. Participants with higher intakes of butter were more educated, ate less fruit and drank more coffee. For margarine, those with higher intakes consumed less fruit and vegetables and more processed meat, soft drinks and coffee. Higher consumption of nuts and seeds was associated with higher intakes of fruits and vegetables. Finally, participants with higher intakes of cakes and cookies were more likely to have never smoked and drank less coffee. Across tertiles of all fatty foods, except margarine, the reported energy intake and the ratio of energy intake to basal metabolic rate increased.
Associations between consumption of fatty foods and incident T2D
HRs of T2D diabetes according to the consumption of fatty foods are shown in Table 2. After adjustment for age, sex, education, energy intake and lifestyle factors (model B), nonconsumption of butter (HR 1.22, I2 for between-country heterogeneity=0%, based on seven countries), nuts and seeds (HR 1.11, I2=0%, eight countries) and cakes and cookies (HR 1.61, I2=70.2%, five countries) was associated with higher T2D risk as compared with the middle tertile of consumption. Adjustment for BMI (model C) partly explained the higher risk among the nonconsumers of butter and completely explained it among the nonconsumers of nuts and seeds. Nonconsumption of cakes and cookies related to higher T2D independently of BMI (model C HR 1.57, based on five countries). Although this HR differed between countries (I2=64.9%), nonconsumption of cakes and cookies was associated with higher risk in all five countries, ranging from 1.12 in Spain to 3.10 in the United Kingdom. Nonconsumption of margarine tended to be also positively related to T2D, but this only became apparent after adjustment for BMI (model C HR 1.26, based on four countries, I2=22.7%).
Among tertiles of consumption of fatty foods, the following results were seen. Vegetable oil appeared linearly positively associated with T2D after adjustment for model B covariates (P for linear trend 0.002), although this was not confirmed by the HRs across the tertiles. Margarine consumption was positively associated with T2D in the least and most adjusted models, with the most completely adjusted HRs across tertiles of increasing intake being 0.93 (based on eight countries), 1.00 (referent, eight countries) and 1.12 (six countries). The corresponding I2 value for between-country heterogeneity was ~50% for both the lowest and highest tertile. Cake and cookie consumption was inversely related to T2D in models before but not after adjustment for BMI. Between-country heterogeneity was low to moderate when adjusted for model C covariates, with I2 values of 53.5 and 34.6% for comparing the lowest and the highest tertile with the middle tertile of cakes and cookies, respectively. Nut and seed consumption was not related to T2D, with no heterogeneity for comparing the lowest with the middle tertile (I2=0%, seven countries included) and moderate heterogeneity for comparing the highest with the middle tertile (I2=49.0%, eight countries). Modelling a quadratic term for fatty foods indicated a U-shaped association among consumers of margarine (P=0.03) and possibly of butter (P=0.09) in the multiple-adjusted model C.
Sensitivity analyses
We applied fractional polynomial analysis to the entire case-cohort to detect more subtle nonlinear associations across the entire range of intake, including nonconsumption. After adjustment for model C covariates, this analysis confirmed the lack of association for vegetable oil (P for difference between best-fitting second-degree fractional polynomial along with separate term for nonconsumers versus the null model=0.11; Supplementary Table 8). For the other fatty foods, significant nonlinear associations were seen that agreed with what could be expected from the categorical analysis (Supplementary Figure 1). For nuts and seeds, margarine and cakes and cookies, modelling the nonconsumers as a separate term significantly contributed to the fit of the data.
Further, we adjusted the categorical analysis for the consumption of vegetables, fruit, red meat, processed meat and coffee, but this did not change the results appreciably. Omitting the first 2 years of follow-up did not change the HRs materially either. Stratification for sex and BMI also did not result in a consistent pattern of effect modification. For example, the HR comparing nonconsumption of cakes and cookies with that of the middle tertile of consumption did not differ much in those with BMIs <25 (HR 1.45, 95% confidence interval 1.01–2.06, I2=0%) and ⩾25 kg/m2 (HR 1.56, 95% confidence interval 1.09–2.22, I2=60.9%). None of the other prespecified sensitivity analysis had an impact on the primary results, including the correction of the HRs for measurement error in the assessment of intake of fatty foods by the dietary questionnaires.
Discussion
In this prospective study in European populations, the consumption of fatty was generally not associated with risk of T2D. In primary analyses, we found that nonconsumption of nuts and seeds related to a higher diabetes risk, which was completely explained by higher BMIs in nonconsumers compared with consumers. No consistent trend was seen among consumers of nuts and seeds. Further, higher consumption of margarine was weakly positively associated with diabetes risk, but this association was not completely consistent across countries. Finally, higher consumption of cakes and cookies was unexpectedly associated with lower diabetes risk before but not after adjustment for BMI. Interestingly, nonconsumers of cakes and cookies in some but not all countries were at greatest T2D risk.
Limitations of the current study need to be addressed. First, not all countries were represented in all categories of fatty food consumption because of substantial differences in consumption. This may hamper a fair comparison across categories. On the other hand, we divided our study sample in categories of fatty foods on the basis of the same cutoff across countries, thereby increasing comparability. Second, even though our sample size was large, it may be possible that we could not detect an association because of a small range in intake. This is perhaps the most evident for nuts and seeds. Third, we measured dietary intake only once, at baseline, and, therefore, we could not take any changes in diet over time into account. Fourth, the intake of fatty foods was assessed with country-specific dietary questionnaires, which themselves are likely to have different degrees of measurement error. However, our primary findings are based on a categorical analysis, which should be less sensitive to misclassification arising from measurement error. Finally, we adjusted for a wide range of factors to account for differences between participants with high and low intakes of fatty foods, but any measurement error in these variables may have led to incomplete adjustments of the hazard ratios.
We found no evidence for an inverse relation of vegetable oils with T2D risk, which is in line with the finding that habitual olive oil consumption is not associated with T2D risk in a Spanish cohort study (relative risk highest versus lowest quintile 1.11, 95% confidence interval 0.45, 2.78).38 Four studies in US populations, however, have reported inverse associations between vegetable fat intake and diabetes risk,10, 12, 39, 40 with relative risks for comparing the top with the bottom quintile ranging from 0.6010 to 0.89.39 Thus, evidence from prospective cohort studies is conflicting, with North American but not European studies suggesting an inverse association. Randomized trials on vegetable oils and incident diabetes may provide further insight. In the PREvención con DIeta MEDiterránea trial, the consumption of olive oil (1 l/wk) was related to a lower incidence of types diabetes compared with a low-fat control diet.41 Of note, the people whose diet was supplemented with nuts also improved, to some extent, other aspects of diet. This may also have contributed to the lower diabetes risk.
Higher consumption of margarine was weakly related to higher T2D risk in our study. However, although a clear positive association was seen in the Netherlands, less consistent associations were found in other countries where margarine is consumed in meaningful amounts, including Denmark and Sweden. One explanation for this inconsistency may be the different fatty acid composition of margarines in European countries during the period our participants underwent dietary assessment.42 In the mid-1990s, trans-fatty acids in margarines from hydrogenated oils were replaced by other fatty acids,42 which adds complexity to the interpretation of our results. Ongoing research on plasma fatty acid profiles and incident T2D in EPIC-InterAct may help clarify this complex association.
There is considerable interest in the potential of nuts and seeds to lower T2D risk. Nuts and seeds are high in unsaturated fat, fibre and magnesium and low in carbohydrates (and, hence, low in glycaemic value). They are able to blunt the glycaemic response when eaten in combination with carbohydrate-rich foods.43, 44, 45 Nuts may also help control body weight,46, 47, 48 despite their high-energy content. In a meta-analysis of four prospective cohort studies, Luo et al.21 reported a pooled relative risk of type 2 diabetes of 0.88 for each additional serving in habitual nut consumption. This inverse association was, however, completely explained by lower BMIs among those eating more nuts.21 The PREvención con DIeta MEDiterránea trial showed that a Mediterranean diet supplemented with mixed nuts (30 g/day) resulted in a marginally lower T2D incidence compared with a low-fat diet.41 It is difficult, however, to separate the effect nuts versus the other smaller dietary changes in that trial. Taken together, the observational evidence of nut consumption and incident diabetes is mixed and that of randomized trials is restricted to one study.
Cakes and cookies are recommended to be eaten with discretion because of their high content of fat and sugar. There are, however, surprisingly few studies reported on the health effects of cakes and cookies. Contrary to what can be expected on the basis of the nutrient composition of cakes and cookies, a study in Sweden found that the mean intake of cakes and biscuits was nonsignificantly lower in women who later developed diabetes compared with women who did not.49 Cake consumption was also inversely related to incident diabetes in the Nurses’ Health Study,50 but relative risks were not reported. Our current findings show that participants with very low intakes of cakes and cookies, particularly nonconsumers, were at a higher T2D risk, whereas no meaningful relation was seen across the remaining range of intake after adjustment for BMI. Compared with consumers, nonconsumers had higher BMIs and waist circumferences, reported fewer calories and more vegetables and had a lower ratio of EI/EE. There were also some nonconsumers in the questionnaires who reported consumption of cakes and cookies in the 24-h recall. Although this may indicate that nonconsumers were more likely to have modified their diet to control body weight or to have underreported energy, we were not able to demonstrate this in our data. However, the higher risk among nonconsumers may be an artefact and the precise reason for this phenomenon requires further attention.
In conclusion, consumption of vegetable oil and nuts and seeds is unlikely to modify T2D risk under isocaloric conditions and independent from BMI according to our data. The exact reason for the higher T2D risk among those with low intakes of cakes and cookies, in particular the nonconsumers, as well as the apparent positive association between margarine consumption and T2D risk in some countries need further understanding.
References
Riserus U, Willett WC, Hu FB . Dietary fats and prevention of type 2 diabetes. Prog Lipid Res 2009; 48: 44–51.
Melanson EL, Astrup A, Donahoo WT . The relationship between dietary fat and fatty acid intake and body weight, diabetes, and the metabolic syndrome. Ann Nutr Metab 2009; 55: 229–243.
Bray GA, Lovejoy JC, Smith SR, DeLany JP, Lefevre M, Hwang D et al. The influence of different fats and fatty acids on obesity, insulin resistance and inflammation. J Nutr 2002; 132: 2488–2491.
Xiao C, Giacca A, Carpentier A, Lewis GF . Differential effects of monounsaturated, polyunsaturated and saturated fat ingestion on glucose-stimulated insulin secretion, sensitivity and clearance in overweight and obese, non-diabetic humans. Diabetologia 2006; 49: 1371–1379.
Summers LK, Fielding BA, Bradshaw HA, Ilic V, Beysen C, Clark ML et al. Substituting dietary saturated fat with polyunsaturated fat changes abdominal fat distribution and improves insulin sensitivity. Diabetologia 2002; 45: 369–377.
Vessby B, Unsitupa M, Hermansen K, Riccardi G, Rivellese AA, Tapsell LC et al. Substituting dietary saturated for monounsaturated fat impairs insulin sensitivity in healthy men and women: The KANWU Study. Diabetologia 2001; 44: 312–319.
Tierney AC, McMonagle J, Shaw DI, Gulseth HL, Helal O, Saris WH et al. Effects of dietary fat modification on insulin sensitivity and on other risk factors of the metabolic syndrome–LIPGENE: a European randomized dietary intervention study. Int J Obes (Lond) 2011; 35: 800–809.
Mozaffarian D, Aro A, Willett WC . Health effects of trans-fatty acids: experimental and observational evidence. Eur J Clin Nutr 2009; 63: S5–S21.
Micha R, Mozaffarian D . Saturated fat and cardiometabolic risk factors, coronary heart disease, stroke, and diabetes: a fresh look at the evidence. Lipids 2010; 45: 893–905.
Salmeron J, Hu FB, Manson JE, Stampfer MJ, Colditz GA, Rimm EB et al. Dietary fat intake and risk of type 2 diabetes in women. Am J Clin Nutr 2001; 73: 1019–1026.
Harding AH, Day NE, Khaw KT, Bingham S, Luben R, Welsh A et al. Dietary fat and the risk of clinical type 2 diabetes: the European prospective investigation of Cancer-Norfolk study. Am J Epidemiol 2004; 159: 73–82.
Meyer KA, Kushi LH, Jacobs Jr DR, Folsom AR . Dietary fat and incidence of type 2 diabetes in older Iowa women. Diabetes Care 2001; 24: 1528–1535.
Kroger J, Zietemann V, Enzenbach C, Weikert C, Jansen EH, Doring F et al. Erythrocyte membrane phospholipid fatty acids, desaturase activity, and dietary fatty acids in relation to risk of type 2 diabetes in the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Am J Clin Nutr 2011; 93: 127–142.
Micha R, Wallace SK, Mozaffarian D . Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus: a systematic review and meta-analysis. Circulation 2010; 121: 2271–2283.
Pan A, Sun Q, Bernstein AM, Schulze MB, Manson JE, Willett WC et al. Red meat consumption and risk of type 2 diabetes: 3 cohorts of US adults and an updated meta-analysis. Am J Clin Nutr 2011; 94: 1088–1096.
van Woudenbergh GJ, van Ballegooijen AJ, Kuijsten A, Sijbrands EJ, van Rooij FJ, Geleijnse JM et al. Eating fish and risk of type 2 diabetes: A population-based, prospective follow-up study. Diabetes Care 2009; 32: 2021–2026.
Patel PS, Forouhi NG, Kuijsten A, Schulze MB, van Woudenbergh GJ, Ardanaz E et al. The prospective association between total and type of fish intake and type 2 diabetes in 8 European countries: EPIC-InterAct Study. Am J Clin Nutr 2012; 95: 1445–1453.
Choi HK, Willett WC, Stampfer MJ, Rimm E, Hu FB . Dairy consumption and risk of type 2 diabetes mellitus in men: a prospective study. Arch Intern Med 2005; 165: 997–1003.
Liu S, Choi HK, Ford E, Song Y, Klevak A, Buring JE et al. A prospective study of dairy intake and the risk of type 2 diabetes in women. Diabetes Care 2006; 29: 1579–1584.
van Dam RM, Hu FB, Rosenberg L, Krishnan S, Palmer JR . Dietary calcium and magnesium, major food sources, and risk of type 2 diabetes in U.S. black women. Diabetes Care 2006; 29: 2238–2243.
Luo C, Zhang Y, Ding Y, Shan Z, Chen S, Yu M et al. Nut consumption and risk of type 2 diabetes, cardiovascular disease, and all-cause mortality: a systematic review and meta-analysis. Am J Clin Nutr 2014; 100: 256–269.
RIVM. Dutch food-composition database. NEVO-online version 2011/3.0. 2011, Available from: http://nevo-online.rivm.nl/ (accessed on 18 December 2013).
Linseisen J, Welch AA, Ocke M, Amiano P, Agnoli C, Ferrari P et al. Dietary fat intake in the European Prospective Investigation into Cancer and Nutrition: results from the 24-h dietary recalls. Eur J Clin Nutr 2009; 63: S61–S80.
Sluijs I, Forouhi NG, Beulens JW, van der Schouw YT, Agnoli C, Arriola L et al. The amount and type of dairy product intake and incident type 2 diabetes: results from the EPIC-InterAct Study. Am J Clin Nutr 2012; 96: 382–390.
The Interact Cosortium. Association between dietary meat consumption and incident type 2 diabetes: the EPIC-InterAct study. Diabetologia 2013; 56: 47–59.
Riboli E, Hunt KJ, Slimani N, Ferrari P, Norat T, Fahey M et al. European Prospective Investigation into Cancer and Nutrition (EPIC): study populations and data collection. Public Health Nutr 2002; 5: 1113–1124.
The InterAct Consortium. Design and cohort description of the InterAct Project: an examination of the interaction of genetic and lifestyle factors on the incidence of type 2 diabetes in the EPIC Study. Diabetologia 2011; 54: 2272–2282.
Kaaks R, Riboli E . Validation and calibration of dietary intake measurements in the EPIC project: methodological considerations. European Prospective Investigation into Cancer and Nutrition. Int J Epidemiol 1997; 26: S15–S25.
Slimani N, Deharveng G, Unwin I, Southgate DA, Vignat J, Skeie G et al. The EPIC nutrient database project (ENDB): a first attempt to standardize nutrient databases across the 10 European countries participating in the EPIC study. Eur J Clin Nutr 2007; 61: 1037–1056.
Wareham NJ, Jakes RW, Rennie KL, Schuit J, Mitchell J, Hennings S et al. Validity and repeatability of a simple index derived from the short physical activity questionnaire used in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Public Health Nutr 2003; 6: 407–413.
Haftenberger M, Lahmann PH, Panico S, Gonzalez CA, Seidell JC, Boeing H et al. Overweight, obesity and fat distribution in 50- to 64-year-old participants in the European Prospective Investigation into Cancer and Nutrition (EPIC). Public Health Nutr 2002; 5: 1147–1162.
Food and Agriculture Organization/World Health Organization/United Nations University Energy and protein requirements. WHO: Geneva, Switzerland, 1985, (WHO Technical Report Series #724).
Black AE . Critical evaluation of energy intake using the Goldberg cut-off for energy intake:basal metabolic rate. A practical guide to its calculation, use and limitations. Int J Obes Relat Metab Disord 2000; 24: 1119–1130.
InterAct Consortium. Validity of a short questionnaire to assess physical activity in 10 European countries. Eur J Epidemiol 2012; 27: 15–25.
Prentice RL . A case-cohort design for epidemiologic cohort studies and disease prevention trials. Biometrika 1986; 73: 1–11.
Royston P, Sauerbrei W, Becher H . Modelling continuous exposures with a 'spike' at zero: a new procedure based on fractional polynomials. Stat Med 2010; 29: 1219–1227.
Rosner B, Willett WC, Spiegelman D . Correction of logistic regression relative risk estimates and confidence intervals for systematic within-person measurement error. Stat Med 1989; 8: 1051–1069.
Mari-Sanchis A, Beunza JJ, Bes-Rastrollo M, Toledo E, Basterra Gortariz FJ, Serrano-Martinez M et al. [Olive oil consumption and incidence of diabetes mellitus, in the Spanish sun cohort]. Nutr Hosp 2011; 26: 137–143.
van Dam RM, Willett WC, Rimm EB, Stampfer MJ, Hu FB . Dietary fat and meat intake in relation to risk of type 2 diabetes in men. Diabetes Care 2002; 25: 417–424.
Song Y, Manson JE, Buring JE, Liu S . A prospective study of red meat consumption and type 2 diabetes in middle-aged and elderly women: the women's health study. Diabetes Care 2004; 27: 2108–2115.
Salas-Salvado J, Bullo M, Estruch R, Ros E, Covas MI, Ibarrola-Jurado N et al. Prevention of diabetes with Mediterranean diets: a subgroup analysis of a randomized trial. Ann Intern Med 2014; 160: 1–10.
Aro A, van Amersfoort J, Becker W, vE-B MA, Kafatos A, Leth T et al. Trans fatty acids in dietary fats and oils from 14 European countries: the Transfair Study. J Food Comp Anal 1998; 11: 137–149.
Jenkins DJ, Kendall CW, Josse AR, Salvatore S, Brighenti F, Augustin LS et al. Almonds decrease postprandial glycemia, insulinemia, and oxidative damage in healthy individuals. J Nutr 2006; 136: 2987–2992.
Josse AR, Kendall CW, Augustin LS, Ellis PR, Jenkins DJ . Almonds and postprandial glycemia—a dose-response study. Metabolism 2007; 56: 400–404.
Kendall CWC, Josse AR, Esfahani A, Jenkins DJA . The impact of pistachio intake alone or in combination with high-carbohydrate foods on post-prandial glycemia. Eur J Clin Nutr 2011; 65: 696–702.
Bes-Rastrollo M, Sabate J, Gomez-Gracia E, Alonso A, Martinez JA, Martinez-Gonzalez MA . Nut consumption and weight gain in a Mediterranean cohort: The SUN study. Obesity (Silver Spring) 2007; 15: 107–116.
Bes-Rastrollo M, Wedick NM, Martinez-Gonzalez MA, Li TY, Sampson L, Hu FB . Prospective study of nut consumption, long-term weight change, and obesity risk in women. Am J Clin Nutr 2009; 89: 1913–1919.
Brennan AM, Sweeney LL, Liu X, Mantzoros CS . Walnut Consumption Increases Satiation but Has No Effect on Insulin Resistance or the Metabolic Profile Over a 4-day Period. Obesity 2010; 18: 1176–1182.
Lundgren H, Bengtsson C, Blohme G, Isaksson B, Lapidus L, Lenner R et al. Dietary habits and incidence of noninsulin-dependent diabetes mellitus in a population study of women in Gothenburg, Sweden. Am J Clin Nutr 1989; 49: 708–712.
Colditz GA, Manson JE, Stampfer MJ, Rosner B, Willett WC, Speizer FE . Diet and risk of clinical diabetes in women. Am J Clin Nutr 1992; 55: 1018–1023.
Acknowledgements
We thank all EPIC participants and staff for their contributions to the study. We thank Nicola Kerrison (MRC Epidemiology Unit, Cambridge) for managing the data for the InterAct Project.
Author Contributions
Author contributions were as follows: BB had access to all data for this study, analyzed the data, wrote the manuscript and takes primary responsibility for the final content. All authors have contributed to the conception of the design and to the interpretation of data, revising the article critically for important intellectual content and final approval of the version to be published.
Funding
Funding for the InterAct project was provided by the EU FP6 programme (grant number LSHM-CT-2006-037197). In addition, InterAct investigators acknowledge funding from the following agencies: IS, JWJB and YTvdS: Verification of diabetes cases was additionally funded by NL Agency grant IGE05012 and an Incentive Grant from the Board of the UMC Utrecht (The Netherlands); HBBdM, AMWS and DLvdA: Dutch Ministry of Public Health, Welfare and Sports (VWS), Netherlands Cancer Registry (NKR), LK Research Funds, Dutch Prevention Funds, Dutch ZON (Zorg Onderzoek Nederland), World Cancer Research Fund (WCRF), Statistics Netherlands (The Netherlands); FLC: Cancer Research UK; PWF: Swedish Research Council, Novo nordisk, Swedish Heart Lung Foundation, Swedish Diabetes Association; JH, KO and AT: Danish Cancer Society; RK: Deutsche Krebshilfe; SP: Associazione Italiana per la Ricerca sul Cancro; JRQ: Asturias Regional Government; MT: Health Research Fund (FIS) of the Spanish Ministry of Health; the CIBER en Epidemiología y Salud Pública (CIBERESP), Spain; Murcia Regional Government (Nº 6236); RT: AIRE-ONLUS Ragusa, AVIS-Ragusa, Sicilian Regional Government.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The California Walnut Commission covered travel expenses for both BB and HB to attend an expert meeting on the health effects of nuts in 2012. None of the other authors declared a conflict of interest.
Additional information
Supplementary Information accompanies this paper on European Journal of Clinical Nutrition website
Supplementary information
Rights and permissions
About this article
Cite this article
Buijsse, B., Boeing, H., Drogan, D. et al. Consumption of fatty foods and incident type 2 diabetes in populations from eight European countries. Eur J Clin Nutr 69, 455–461 (2015). https://doi.org/10.1038/ejcn.2014.249
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2014.249
- Springer Nature Limited
This article is cited by
-
Trends in energy and macronutrient intake among Taiwanese older adults in 1999–2000, 2005–2008 and 2013–2016 periods
BMC Public Health (2023)
-
Substitution of animal-based with plant-based foods on cardiometabolic health and all-cause mortality: a systematic review and meta-analysis of prospective studies
BMC Medicine (2023)
-
Ultra-processed food and incident type 2 diabetes: studying the underlying consumption patterns to unravel the health effects of this heterogeneous food category in the prospective Lifelines cohort
BMC Medicine (2022)
-
Cooking oil/fat consumption and deaths from cardiometabolic diseases and other causes: prospective analysis of 521,120 individuals
BMC Medicine (2021)
-
The influence of adjustment for energy misreporting on relations of cake and cookie intake with cardiometabolic disease risk factors
European Journal of Clinical Nutrition (2016)