Abstract
Purpose
Few studies have evaluated the effects of processed red meat (PRM) consumption on chronic disease risk, especially among low average PRM consumption populations. This study examined association between PRM intake and type 2 diabetes mellitus (T2DM) and cardiovascular disease (CVD) incidences in Korea.
Methods
Participants (n = 10,030; aged 40–69 years) were recruited from the Ansan–Ansung cohort study, a subset of Korean Genome Epidemiology Study. Validated 103-item semi-quantitative food frequency questionnaire was used to assess dietary information, at baseline (2001–2002) and at second follow-up (2005–2006). T2DM and CVD incidences were identified using biennial questionnaire-based interview during a 10-year follow-up period. Cox proportional hazards regression analysis was used to calculate multivariable-adjusted hazard ratios (HRs) and 95% confidence intervals (CIs).
Results
Over the 10-year follow-up period, 668 and 493 incident cases of T2DM (62,130 person-years) and CVD (63,150 person-years), respectively, were documented. In crude models, comparing the highest and lowest levels of PRM intake, no significant association occurred with incident T2DM [HR, 95% CI (0.94, 0.76–1.17)], while significant association occurred with incident CVD (0.67, 0.51–0.88). However, in adjusted models, this association was no longer significant, showing HRs (95% CI) of PRM intake for T2DM and CVD of 1.07 (0.85–1.35) and 1.14 (0.85–1.55), respectively.
Conclusions
PRM intake did not affect T2DM and CVD incidences among Korean middle-aged adults, although extremely low average consumption of PRM may explain the finding. Therefore, limiting PRM intake may not be a top priority for the prevention of chronic diseases in Korean population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Type 2 diabetes mellitus (T2DM) and cardiovascular diseases (CVD) are the leading causes of mortality worldwide. According to the World Health Organization (WHO), 17.7 million people died from CVD in 2015, accounting for approximately 31% of all deaths worldwide [1]. In addition, a total of 415 million people had DM, a major risk factor for CVD; and the medical cost from DM accounted for 5–20% of the total health expenditure in a majority of countries in 2015 [2]. Asian populations tend to have lower obesity rates, but the prevalence of metabolic syndrome has rapidly increased in many Asian countries over the past decades [3, 4]. In particular, the 2014 Korea National Health and Nutrition Examination Survey (KNHANES) found that more than half of Korean adults had at least one metabolic risk factor, such as obesity, hypertension, and hypercholesterinemia [5], that could eventually develop into DM or CVD.
Recent research suggests that processed red meat (PRM) consumption is clearly associated with increased chronic disease risk, while the health effects of unprocessed red meat consumption remain unclear [6,7,8,9,10,11]. Recent meta-analysis showed that PRM intake increased the relative risk of coronary heart disease (CHD) [9,10,11] and T2DM [11], whereas unprocessed red meat intake seemed to increase the risk of T2DM [11], and the association with CHD incidence seemed inconsistent [9,10,11].
According to the KNHANES 2007–2009, average individual consumption of unprocessed red meat in Korea was 60 g/day [12], lower than that in the USA or European countries (USA, 78 g/day; UK, 85 g/day; Denmark, 134 g/day) [13, 14], but similar to China (66 g/day) [15]. Average PRM consumption among Koreans (5.9–7.2 g/day) was much lower than unprocessed red meat consumption, and substantially lower than the average PRM consumption in other countries (range: 21.4–33.2 g/day) [13, 16, 17].
Recently, WHO designated PRM as a group 1 carcinogen [18]. Thus, a number of dietary guidelines that restrict PRM intake to prevent chronic diseases in adults have been published [18,19,20,21]. Further, the Ministry of Food and Drug Safety of South Korea has declared the necessity of such dietary guidelines for Koreans [17]. However, few studies have examined the association between PRM intake and chronic diseases in Koreans, and the study results or guidelines from other countries where people consume relatively high levels of PRM should not direct the development of dietary guidelines for Koreans.
Therefore, this study was conducted to evaluate PRM intake levels in Korean adults, and to investigate the prospective association between PRM intake levels and incidence of T2DM and CVD.
Methods
Study population
The Korean Genome Epidemiology Study (KoGES) was conducted to examine and identify genetic and environmental factors related to the incidence of chronic diseases, such as hypertension, T2DM, CVD, and cancer, among Koreans [22]. The Ansan–Ansung cohort is a community-based ongoing prospective cohort study that is one of six cohorts in the KoGES. Details regarding these data were described previously [23]. Briefly, men and women aged 40–69 years old in Ansan (urban) and Ansung (rural) areas in South Korea were recruited using two-stage cluster sampling based on geographic and demographic information. A total of 4758 men and 5272 women participated in the baseline examination (2001–2002), and data on demographics, lifestyle, dietary habits, and medical history were collected using a questionnaire-based interview. In addition, health professionals trained with standardized protocols performed comprehensive physical examination. Follow-up examinations were repeated biennially, and 10-year follow-up data were analyzed.
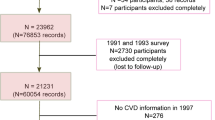
Participants with a history of T2DM, CVD, or cancer at baseline were excluded from this analysis. In addition, those with implausible levels of energy intake (< 500 or > 5000 kcal/day) [24] or no information on PRM intake were excluded. Finally, a total of 8618 participants (86%) were included in the analysis (Fig. 1).
The present study was approved by the Institutional Review Board (IRB) of the Korea Centers for Disease Control and Prevention (IRB number: KU-IRB-15-EX-256-A-1). Written informed consent was obtained from all participants. The research procedure for the second analysis was approved by the Yeungnam University (IRB number: 7002016-E-2016-003).
Assessment of dietary intake
Dietary intake was assessed using a 103-item Semi-Quantitative Food Frequency Questionnaire (SQFFQ) with proven validity and reproducibility using 12-day dietary records [25], and dietary data were collected at baseline (2001–2002) and at the second follow-up period (2005–2006). The average servings of food intake (servings/week) were calculated by multiplying intake frequency per week by one of the serving size options, such as 0.5 for smaller than, 1 for equal to, or 1.5 for larger than a standard serving size [26]. One standard serving portion of PRM was set to 50 g, which corresponds to five Vienna sausages or one piece of ham, in the SQFFQ. Weekly amounts of PRM consumption (g) were calculated using the weekly frequency intake and corresponding serving size of PRM. Missing values for dietary data at the second follow-up period were imputed with retest values (baseline data) [27].
In order to estimate the total intake levels of fruits and vegetables, we included all fruit and vegetable items listed on SQFFQ in this cohort study, except salted vegetables. The total fruit intake level was calculated by summating the overall weekly servings of 11 types of fruits, including strawberries, persimmons, mandarins, oranges, muskmelon/melons, watermelons, pears, apples, bananas, peaches/plums, and grapes. Similarly, the total vegetable intake level was calculated by summating the overall weekly servings of 19 types of vegetables. These include green peppers, pepper leaves/chamnamuls/seasoned asters, spinaches, lettuces, perilla leaves, crown daisies/leeks/water dropworts, deoducks/dorajis (kinds of balloon flower roots), onions, Chinese cabbages, cucumbers, soybean sprouts/mung bean sprouts, carrots, pumpkins, zucchinis, brackens/sweet potato stems/taro stems, tomatoes, other vegetables, oyster mushrooms, and other mushrooms. The daily nutrient intake levels were calculated using the food composition database created by the Rural Development Administration of Korea [28].
Ascertainment of T2DM and CVD
The identifications of T2DM and CVD were according to self-reported answers to the biennial questionnaires, and all reported cases were confirmed by trained staff during personal interviews. The incidence of T2DM was defined as at least one positive response to T2DM-related questionnaire items, including diagnosis by a physician, current treatment, or use of insulin or oral medication for T2DM. The incidence of CVD (included myocardial infarction, angina, and stroke) was defined as at least one positive response to CVD-related questionnaire items, including diagnosis by a physician, treatment, or use of medications.
Assessment of demographic, lifestyle, and health-related information
Educational level was categorized into four groups: elementary school graduation or less, middle school graduation, high school graduation, and college graduation or higher. Monthly household income was also classified into four groups: < 1,000,000; 1,000,000–1,999,999; 2,000,000–3,999,999; and ≥ 4,000,000 Korean republic won (KRW). Current drinking status and daily alcohol intake were estimated. Current smoking status was classified into current smoker and non-smoker. Cumulative smoking amount (pack-year) was calculated by multiplying the number of total smoking years by the number of packs after converting the daily mean number of cigarettes smoked to pack units. To calculate physical activity level, metabolic equivalent tasks (METs) were calculated [29], and physical activity level was then divided into low (< 20 METs-h/week), moderate (20–39 METs-h/week), and high (≥ 40 METs-h/week). Anthropometric measurements, such as height, body weight, and waist circumference, were obtained through a standardized procedure by trained experts. Body mass index (BMI) was categorized into underweight (< 18.5 kg/m2), normal weight (18.5–22.9 kg/m2), overweight (23–24.9 kg/m2), and obese (≥ 25 kg/m2) according to BMI cut-offs for Asians by the WHO [30]. Systolic and diastolic blood pressures were repeatedly measured using a mercury sphygmomanometer in a sitting position after at least 5 min of rest. The average value of two blood pressure readings was used for analysis. A fasting blood sample was drawn and collected in ethylenediaminetetraacetic acid (EDTA)-containing tubes. Fasting blood glucose, triglycerides, and high-density lipoprotein cholesterol were measured at the Seoul Clinical Laboratories (Seoul, Korea). Methodological information on biochemical assays has been described in a previous study [23].
Statistical analysis
Approximately 68% of participants (n = 5845) reported that they never consumed PRM. The participants were divided into three groups based on PRM intake distribution: 0 g/week (67.8%), 0.1–11.5 g/week (16.8%), and > 11.5 g/week (15.4%). The energy-adjusted mean levels of food and nutrient intake and the age- and sex-adjusted mean values of health indicators, such as waist circumference, fasting blood glucose, and serum lipid levels, were calculated using the standard multivariate method. For the incidence of T2DM and CVD according to PRM intake, hazard ratios (HR) and 95% confidence intervals (CI) were calculated using Cox proportional hazards regression models. To determine potential confounding factors, related literature was reviewed [31, 32], and the preliminary analysis was performed. Based on this information, we evaluated three covariate models. Model 1 was adjusted for age, sex, educational level, monthly household income, and residential area. Model 2 was adjusted for model 1, and additionally adjusted for smoking, physical activity, BMI, alcohol intake, energy intake, consumption levels of dietary fat, crude fiber, sodium, and fruit and vegetable. Model 3 was adjusted for model 2, and additionally adjusted for current use of antihypertensive and antihyperlipidemic medications. To test the linear trend (P for trend) of the association, the median value of each PRM intake group was used as a continuous variable. In addition, stratified analysis was performed for variables that could affect the relationship between PRM intake and T2DM and CVD, and P for interaction was calculated using a multiplicative term in the regression analysis model. Kaplan–Meier technique was used to construct survival curves, and unadjusted survival rates from incident T2DM and CVD were calculated. Statistical analyses were performed using Statistical Analysis System version 9.4 (SAS Institute, Cary, NC, USA). P values were subjected to two-sided testing, and statistical significance was determined based on α = 0.05.
Results
Study characteristics by levels of PRM intake
General baseline characteristics of the cohorts by PRM intake levels are presented in Table 1. Approximately 67.8% of participants rarely consumed PRM. Study participants were middle-aged, with 53.1, 49.6, and 48.0 years in age, in the lowest, mid, and highest groups of PRM intakes, respectively. The proportion of overweight and obese participants were almost two-thirds of all participants, including 41.3%, 43.4%, and 44.4% of obese participants in the lowest, mid, and highest groups of PRM intakes, respectively. Approximately, half of participants were current alcohol drinkers (46.2%, 51.3%, and 53.7% in the lowest, mid, highest groups of PRM intake, respectively) and less than a third of the participants were current smokers (25.4%, 26.9%, and 27.9% in the lowest, mid, highest groups of PRM intake, respectively).
Association between PRM intake and incident T2DM and CVD
Participants in the Ansan–Ansung cohort who participated at the baseline examination were followed up for 10 years; 7.8% (n = 668) and 5.7% (n = 493) of these participants had T2DM (62,130 person-years) and CVD (63,150 person-years), respectively. In a multivariable Cox proportional hazard regression model, there were no significant associations between PRM intake and incidence of T2DM in any model. Higher intake of PRM reduced the incidence of CVD in the crude model (P for trend = 0.01); however, this trend did not persist in the fully adjusted models (Table 2).
Even in a further stratified analysis by age, sex, alcohol intake, smoking status, residential area (rural/urban), antihypertensive medication use, and dietary intake (fruit, vegetable, fat, crude fiber, and sodium); intake of PRM was statistically associated with neither T2DM nor CVD risk in this population (Online Resource 1). The Kaplan–Meier plots showed that 10-year overall survival rates of T2DM with 0, > 0 and ≤ 11.5, and > 11.5 g/week of PRM were 89.7, 90.7, and 90.5%, respectively (P = 0.6). The 10-year survival rates of CVD with 0, > 0 and ≤ 11.5, and > 11.5 g/week of PRM were 91.7, 93.5, and 94.1%, respectively (P = 0.01, Online Resource 2).
Discussion
To investigate the effects of PRM intake on the incidence of CVD and T2DM, we analyzed data from a 10-year follow-up cohort comprising 8618 participants aged 40–69 years in Ansan and Ansung areas in South Korea. Only 32% of participants consumed PRM, and their mean intake was 8.8 g/week. PRM intake had no significant effects on the incidence of T2DM and CVD at this mean intake level.
A similar result from a cross-sectional analysis in a Japanese cohort study showed that PRM intake was not associated with T2DM risk [33]. In contrast, a number of cohort studies in the USA and Europe reported that higher intake of PRM led to an increased risk of developing T2DM [14, 34, 35] and CVD, as well as subsequent mortality [16, 36]. Similarly, a Chinese cohort study showed PRM intake raised the risk of T2DM [37]. Furthermore, recent meta-analyses presented that there was a 32% and 15% increase in the incidence of T2DM and the risk of CVD mortality per 50 g of PRM intake, respectively [10, 38]. In addition, the highest PRM intake group had 51% and 17% higher risks of these diseases compared to the lowest intake group [14, 39].
In previously published literature, higher PRM intake showed a stronger association with an increased risk of T2DM or CVD, while there were inconsistent results regarding unprocessed red meat [11]. This may be attributable to food ingredients or additives in PRM. For example, PRM contained ≥ 400% more sodium and ≥ 50% more nitrates per gram than unprocessed red meat [11]. High sodium intake altered renal vascular and adrenal responses to angiotensin-II, and subsequently led to increased blood pressure [40]. Nitrates and nitrites, mostly used as preservatives in PRM, have also been reported to cause intravascular oxidative damage and to lead to vascular disorders [41, 42]. Although these potential mechanisms provide an explanation for the elevated incidence of T2DM and CVD, further studies should be conducted.
Though the results of this study were not consistent with those of previous studies [9, 35], it does not indicate that PRM intake is harmless. Instead, the results may be explained by the extremely low intake of PRM in this Korean population [43]. In Japanese population whose PRM intake level is similar with those in our study, PRM intake was not associated with incident T2DM [33], rather, in another study, an inverse association was observed, showing that the higher category of PRM intake was associated with decreasing mortality from ischemic heart disease in men [7]. In general, the association between nutritional intake and disease risk shows a nonlinear U shape [43]. The range of PRM intake related to the increasing risk of T2DM and CVD were 2- to 10-fold higher in previous studies (10.5–56.2 g/day) than in this study (< 5.4 g/day) [14, 44]. Thus, the amount of PRM consumed in this study seems to be too low to have an impact on disease risk. In fact, according to the results of KNHANES 2010–2013, the mean intake of PRM among Koreans was 6.0 g/day [17], which corresponds to approximately 1/5 of the mean daily intake in the US (28.2 g) [45]. In addition, PRM intake varied considerably by age. According to KNHANES 2013, the mean intake of PRM was 14.9 g/day in individuals 10–20 years of age, and substantially decreased to 7.1 and ≤ 2.0 g/day in individuals in their 40 and 50 s, respectively [17]. In younger generations, PRM consumption increased due to westernized dietary habits [46], and frequent dining out [47], whereas these changes appeared to have less effect on the dietary habits of middle-aged adults. This may be due to the fact that dietary habits are usually formed at a young age and maintained throughout the lifespan [48].
To the best of our knowledge, this is the first study that investigated the prospective association of PRM with the incidence of T2DM and CVD among Koreans. Nevertheless, a few limitations were observed. First, although we adjusted for multiple potential confounding factors, there might be residual confounding due to the characteristics of an observational study. Second, the range for the highest PRM intake group was wide in this study population, consisted mainly of middle-aged adults. PRM intake varies considerably by age group, thus the findings of this study cannot be generalized to all age groups. Thus, this finding should be confirmed through future investigation with a long-term follow-up cohort study encompassing various age groups.
Conclusions
PRM intake did not affect the development of T2DM and CVD among Korean middle-aged adults. The current intake level of PRM is substantially low to induce the risk of chronic diseases in this population. Therefore, limiting PRM intake may not be a top priority when establishing dietary guidelines for the prevention of chronic diseases in the Korean population.
References
World Health Organization (2017) Cardiovascular diseases (CVDs). http://www.who.int/mediacentre/factsheets/fs317/en/. Accessed 8 Dec 2017
International Diabetes Federation (2015) IDF DIABETES ATLAS. http://www.diabetesatlas.org/. Accessed 13 Nov 2017
Deurenberg P, Deurenberg-Yap M, Guricci S (2002) Asians are different from Caucasians and from each other in their Body Mass Index/body fat per cent relationship. Obes Rev 3(3):141–146. https://doi.org/10.1046/j.1467-789X.2002.00065.x
Ram CVS, Farmer JA (2012) Metabolic syndrome in south Asians. J Clin Hypertens 14(8):561–565. https://doi.org/10.1111/j.1751-7176.2012.00652.x
Ministry of Health and Welfare (2015) The major results of national health nutrition survey 2014. http://www.mohw.go.kr/front_new/al/sal0301vw.jsp?PAR_MENU_ID=04&MENU_ID=0403&page=16&CONT_SEQ=326126. Accessed 27 Mar 2017
Wang D, Campos H, Baylin A (2017) Red meat intake is positively associated with non-fatal acute myocardial infarction in the Costa Rica Heart Study. Br J Nutr 118(4):303–311. https://doi.org/10.1017/S000711451700201X
Nagao M, Iso H, Yamagishi K, Date C, Tamakoshi A (2012) Meat consumption in relation to mortality from cardiovascular disease among Japanese men and women. Eur J Clin Nutr 66(6):687–693. https://doi.org/10.1038/ejcn.2012.6
Isanejad M, LaCroix AZ, Thomson CA, Tinker L, Larson JC, Qi Q, Qi L, Cooper-DeHoff RM, Phillips LS, Prentice RL, Beasley JM (2017) Branched-chain amino acid, meat intake and risk of type 2 diabetes in the Women’s Health Initiative. Br J Nutr 117(11):1523–1530. https://doi.org/10.1017/S0007114517001568
Kim K, Hyeon J, Lee SA, Kwon SO, Lee H, Keum N, Lee JK, Park SM (2017) Role of total, red, processed, and white meat consumption in stroke incidence and mortality: a systematic review and meta-analysis of prospective cohort studies. J Am Heart Assoc. https://doi.org/10.1161/jaha.117.005983
Wang X, Lin X, Ouyang YY, Liu J, Zhao G, Pan A, Hu FB (2016) Red and processed meat consumption and mortality: dose–response meta-analysis of prospective cohort studies. Public Health Nutr 19(5):893–905. https://doi.org/10.1017/s1368980015002062
Micha R, Michas G, Mozaffarian D (2012) Unprocessed red and processed meats and risk of coronary artery disease and type 2 diabetes—an updated review of the evidence. Curr Atheroscler Rep 14(6):515–524. https://doi.org/10.1007/s11883-012-0282-8
Park MK, Paik HY, Lee Y (2016) Intake trends of red meat, alcohol, and fruits and vegetables as cancer-related dietary factors from 1998 to 2009. Osong Public Health Res Perspect 7(3):180–189. https://doi.org/10.1016/j.phrp.2016.04.005
Micha R, Penalvo JL, Cudhea F, Imamura F, Rehm CD, Mozaffarian D (2017) Association between dietary factors and mortality from heart disease, stroke, and type 2 diabetes in the United States. JAMA 317(9):912–924. https://doi.org/10.1001/jama.2017.0947
Bendinelli B, Palli D, Masala G, Sharp SJ, Schulze MB, Guevara M, van der AD, Sera, Amiano F, Balkau P, Barricarte B, Boeing A, Crowe H, Dahm FL, Dalmeijer CC, de Lauzon-Guillain G, Egeberg B, Fagherazzi R, Franks G, Krogh PW, Huerta V, Jakszyn JM, Khaw P, Li KT, Mattiello K, Nilsson A, Overvad PM, Ricceri K, Rolandsson F, Sanchez O, Slimani MJ, Sluijs N, Spijkerman I, Teucher AM, Tjonneland B, Tumino A, van den Berg R, Forouhi SW, Langeberg NG, Feskens C, Riboli EJ, Wareham E NJ (2013) Association between dietary meat consumption and incident type 2 diabetes: the EPIC-InterAct study. Diabetologia 56(1):47–59. https://doi.org/10.1007/s00125-012-2718-7
He Y, Yang X, Xia J, Zhao L, Yang Y (2016) Consumption of meat and dairy products in China: a review. Proc Nutr Soc 75(3):385–391. https://doi.org/10.1017/s0029665116000641
Rohrmann S, Overvad K, Bueno-de-Mesquita HB, Jakobsen MU, Egeberg R, Tjonneland A, Nailler L, Boutron-Ruault MC, Clavel-Chapelon F, Krogh V, Palli D, Panico S, Tumino R, Ricceri F, Bergmann MM, Boeing H, Li K, Kaaks R, Khaw KT, Wareham NJ, Crowe FL, Key TJ, Naska A, Trichopoulou A, Trichopoulos D, Leenders M, Peeters PH, Engeset D, Parr CL, Skeie G, Jakszyn P, Sanchez MJ, Huerta JM, Redondo ML, Barricarte A, Amiano P, Drake I, Sonestedt E, Hallmans G, Johansson I, Fedirko V, Romieux I, Ferrari P, Norat T, Vergnaud AC, Riboli E, Linseisen J (2013) Meat consumption and mortality—results from the European prospective investigation into cancer and nutrition. BMC Med 11:63. https://doi.org/10.1186/1741-7015-11-63
Ministry of Food and Drug Safety (2015) The level of red meat and processed meat intake were not worrying level for Koreans. http://www.mfds.go.kr/index.do?searchkey=title:contents&mid=675&searchword=%C0%C7%BE%E0%C7%B0&pageNo=19&seq=29262&cmd=v. Accessed 27 Mar 2017
World Health Organization (2015) IARC monographs evaluate consumption of red meat and processed meat. http://www.iarc.fr/en/media-centre/pr/2015/pdfs/pr240_E.pdf. Accessed 27 Mar 2017
U.S. Department of Health and Human Services, U.S. Department of Agriculture (2015) Dietary guidelines for Americans 2015–2020. https://health.gov/dietaryguidelines/2015/guidelines/. Accessed 27 Mar 2017
Public Health England (2014) A quick guide to the government’s healthy eating recommendations. http://elearning.ymca.co.uk/ymcafit/pluginfile.php/11442/mod_resource/content/1/A%20Quick%20Guide%20to%20Government%20Healthy%20Eating.pdf. Accessed 27 Mar 2017
National Health and Medical Research Council, Department of Health and Ageing (2013) Australian dietary guidelines. https://www.nhmrc.gov.au/guidelines-publications/n55. Accessed 27 Mar 2017
Kim Y, Han BG (2017) Cohort profile: the Korean Genome and Epidemiology Study (KoGES) Consortium. Int J Epidemiol 46(2):e20. https://doi.org/10.1093/ije/dyv316
Baik I, Kim J, Abbott RD, Joo S, Jung K, Lee S, Shim J, In K, Kang K, Yoo S, Shin C (2008) Association of snoring with chronic bronchitis. Arch Intern Med 168(2):167–173. https://doi.org/10.1001/archinternmed.2007.8
Willett W (2013) Nutritional epidemiology, 3rd edn. Oxford University Press, New York
Ahn Y, Kwon E, Shim JE, Park MK, Joo Y, Kimm K, Park C, Kim DH (2007) Validation and reproducibility of food frequency questionnaire for Korean genome epidemiologic study. Eur J Clin Nutr 61(12):1435–1441. https://doi.org/10.1038/sj.ejcn.1602657
Baik I, Lee M, Jun NR, Lee JY, Shin C (2013) A healthy dietary pattern consisting of a variety of food choices is inversely associated with the development of metabolic syndrome. Nutr Res Pract 7(3):233–241. https://doi.org/10.4162/nrp.2013.7.3.233
Parr CL, Hjartaker A, Scheel I, Lund E, Laake P, Veierod MB (2008) Comparing methods for handling missing values in food-frequency questionnaires and proposing k nearest neighbours imputation: effects on dietary intake in the Norwegian Women and Cancer study (NOWAC). Public Health Nutr 11(4):361–370. https://doi.org/10.1017/s1368980007000365
National Rural Living Science Institute (1996) Food composition table, 5th edn. Rural Development Administration, Suwon Korea
Pate RR, Pratt M, Blair SN, Haskell WL, Macera CA, Bouchard C, Buchner D, Ettinger W, Heath GW, King AC (1995) Physical activity and public health: a recommendation from the Centers for Disease Control and Prevention and the American College of Sports Medicine. JAMA 273(5):402–407. https://doi.org/10.1001/jama.1995.03520290054029
World Health Organization (2000) The Asia–Pacific perspective: redefining obesity and its treatment. http://www.wpro.who.int/nutrition/documents/Redefining_obesity/en/. Accessed 8 Dec 2017
Chen GC, Lv DB, Pang Z, Liu QF (2013) Red and processed meat consumption and risk of stroke: a meta-analysis of prospective cohort studies. Eur J Clin Nutr 67(1):91–95. https://doi.org/10.1038/ejcn.2012.180
Fretts AM, Follis JL, Nettleton JA, Lemaitre RN, Ngwa JS, Wojczynski MK, Kalafati IP, Varga TV, Frazier-Wood AC, Houston DK, Lahti J, Ericson U, van den Hooven EH, Mikkila V, Kiefte-de Jong JC, Mozaffarian D, Rice K, Renstrom F, North KE, McKeown NM, Feitosa MF, Kanoni S, Smith CE, Garcia ME, Tiainen AM, Sonestedt E, Manichaikul A, van Rooij FJ, Dimitriou M, Raitakari O, Pankow JS, Djousse L, Province MA, Hu FB, Lai CQ, Keller MF, Perala MM, Rotter JI, Hofman A, Graff M, Kahonen M, Mukamal K, Johansson I, Ordovas JM, Liu Y, Mannisto S, Uitterlinden AG, Deloukas P, Seppala I, Psaty BM, Cupples LA, Borecki IB, Franks PW, Arnett DK, Nalls MA, Eriksson JG, Orho-Melander M, Franco OH, Lehtimaki T, Dedoussis GV, Meigs JB, Siscovick DS (2015) Consumption of meat is associated with higher fasting glucose and insulin concentrations regardless of glucose and insulin genetic risk scores: a meta-analysis of 50,345 Caucasians. Am J Clin Nutr 102(5):1266–1278. https://doi.org/10.3945/ajcn.114.101238
Kurotani K, Nanri A, Goto A, Mizoue T, Noda M, Oba S, Kato M, Matsushita Y, Inoue M, Tsugane S (2013) Red meat consumption is associated with the risk of type 2 diabetes in men but not in women: a Japan Public Health Center-Based Prospective Study. Br J Nutr 110(10):1910–1918. https://doi.org/10.1017/s0007114513001128
van Woudenbergh GJ, Kuijsten A, Tigcheler B, Sijbrands EJ, van Rooij FJ, Hofman A, Witteman JC, Feskens EJ (2012) Meat consumption and its association with C-reactive protein and incident type 2 diabetes: the Rotterdam Study. Diabetes Care 35(7):1499–1505. https://doi.org/10.2337/dc11-1899
Steinbrecher A, Erber E, Grandinetti A, Kolonel LN, Maskarinec G (2011) Meat consumption and risk of type 2 diabetes: the Multiethnic Cohort. Public Health Nutr 14(4):568–574. https://doi.org/10.1017/s1368980010002004
Bernstein AM, Pan A, Rexrode KM, Stampfer M, Hu FB, Mozaffarian D, Willett WC (2012) Dietary protein sources and the risk of stroke in men and women. Stroke 43(3):637–644. https://doi.org/10.1161/strokeaha.111.633404
Villegas R, Shu XO, Gao Y-T, Yang G, Cai H, Li H, Zheng W (2006) The association of meat intake and the risk of type 2 diabetes may be modified by body weight. Int J Med Sci 3(4):152
Feskens EJ, Sluik D, van Woudenbergh GJ (2013) Meat consumption, diabetes, and its complications. Curr Diabetes Rep 13(2):298–306. https://doi.org/10.1007/s11892-013-0365-0
Yang C, Pan L, Sun C, Xi Y, Wang L, Li D (2016) Red meat consumption and the risk of stroke: a dose–response meta-analysis of prospective cohort studies. J Stroke Cerebrovasc Dis Off J Natl Stroke Assoc 25(5):1177–1186. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.01.040
Garfinkle MA (2017) Salt and essential hypertension: pathophysiology and implications for treatment. J Am Soc Hypertens 11(6):385–391. https://doi.org/10.1016/j.jash.2017.04.006
Pacher P, Beckman JS, Liaudet L (2007) Nitric oxide and peroxynitrite in health and disease. Physiol Rev 87(1):315–424. https://doi.org/10.1152/physrev.00029.2006
Forstermann U (2008) Oxidative stress in vascular disease: causes, defense mechanisms and potential therapies. Nat Clin Pract Cardiovasc Med 5(6):338–349. https://doi.org/10.1038/ncpcardio1211
Heaney RP (2014) Guidelines for optimizing design and analysis of clinical studies of nutrient effects. Nutr Rev 72(1):48–54. https://doi.org/10.1111/nure.12090
Pan A, Sun Q, Bernstein AM, Schulze MB, Manson JE, Stampfer MJ, Willett WC, Hu FB (2012) Red meat consumption and mortality: results from 2 prospective cohort studies. Arch Intern Med 172(7):555–563. https://doi.org/10.1001/archinternmed.2011.2287
Daniel CR, Cross AJ, Koebnick C, Sinha R (2011) Trends in meat consumption in the USA. Public Health Nutr 14(4):575–583. https://doi.org/10.1017/s1368980010002077
Kang M, Joung H, Lim J, Lee Y, Song Y (2011) Secular trend in dietary patterns in a Korean adult population, using the 1998, 2001, and 2005 Korean National Health and Nutrition Examination Survey. (Secular Trend in Dietary Patterns in a Korean Adult Population, Using the 1998, 2001, and 2005 Korean National Health and Nutrition Examination Survey). Korean J Nutr 44(2):152–161. https://doi.org/10.4163/kjn.2011.44.2.152
Korea Rural Economic Institute (2015) Consumption trend of food processing products and consumption activation plan. Korea Rural Economic Institute. Available via Nuri media. http://www.dbpia.co.kr/Article/NODE06597572. Accessed 27 Mar 2017
Brown JE, Lechtenberg E, Splett PL (2016) Nutrition through the life cycle, 6th edn. Cengage Learning, Boston
Acknowledgements
Data in this study were from the Korean Genome and Epidemiology Study (KoGES; 4851-302), National Research Institute of Health, Centers for Disease Control and Prevention, Ministry for Health and Welfare, Republic of Korea. This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (Grant number: NRF-2017R1A1A3A04069759). The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Author information
Authors and Affiliations
Contributions
JS contributed to the writing original draft preparation, formal analysis, visualization, and software. JS, YL and KP contributed to the investigation, data curation, and writing the review and editing. KP contributed to the conceptualization, supervision, project administration, resources, funding acquisition, and validation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Institutional Review Board (IRB) of the Korea Centers for Disease Control and Prevention (IRB number: KU-IRB-15-EX-256-A-1), and the research procedure for the second analysis was approved by the Yeungnam University IRB (7002016-E-2016-003).
Informed consent
Written informed consent was obtained from all participants.
Electronic supplementary material
Below is the link to the electronic supplementary material.
394_2018_1799_MOESM1_ESM.docx
Online Resource 1: Hazard ratios for incidence type 2 diabetes mellitus and cardiovascular diseases by potential modifiers (DOCX 481 KB)
Rights and permissions
About this article
Cite this article
Son, J., Lee, Y. & Park, K. Effects of processed red meat consumption on the risk of type 2 diabetes and cardiovascular diseases among Korean adults: the Korean Genome and Epidemiology Study. Eur J Nutr 58, 2477–2484 (2019). https://doi.org/10.1007/s00394-018-1799-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-018-1799-6