Abstract
Pulmonary fibrosis may be partially the result of deregulated tissue repair in response to chronic hypoxia. In this study we explored the effects of hypoxia on lung fibroblasts and the effects of neotuberostemonine (NTS), a natural alkaloid isolated from Stemona tuberosa, on activation of fibroblasts in vitro and in vivo. PLFs (primary mouse lung fibroblasts) were activated and differentiated after exposure to 1% O2 or treatment with CoCl2 (100 μmol/L), evidenced by markedly increased protein or mRNA expression of HIF-1α, TGF-β, FGF2, α-SMA and Col-1α/3α, which was blocked after silencing HIF-1α, suggesting that the activation of fibroblasts was HIF-1α-dependent. NTS (0.1–10 μmol/L) dose-dependently suppressed hypoxia-induced activation and differentiation of PLFs, whereas the inhibitory effect of NTS was abolished by co-treatment with MG132, a proteasome inhibitor. Since prolyl hydroxylation is a critical step in initiation of HIF-1α degradation, we further showed that NTS treatment reversed hypoxia- or CoCl2-induced reduction in expression of prolyl hydroxylated-HIF-1α. With hypoxyprobe immunofiuorescence staining, we showed that NTS treatment directly reversed the lower oxygen tension in hypoxia-exposed PLFs. In a mouse model of lung fibrosis, oral administration of NTS (30 mg·kg-1·d-1, for 1 or 2 weeks) effectively attenuated bleomycin-induced pulmonary fibrosis by inhibiting the levels of HIF-1α and its downstream profibrotic factors (TGF-β, FGF2 and α-SMA). Taken together, these results demonstrate that NTS inhibits the protein expression of HIF-1α and its downstream factors TGF-β, FGF2 and α-SMA both in hypoxia-exposed fibroblasts and in lung tissues of BLM-treated mice. NTS with anti-HIF-1α activity may be a promising pharmacological agent for the treatment of pulmonary fibrosis.
Similar content being viewed by others
Introduction
Fibrosis is a common pathway to organ injury and failure. Like other organ fibrosis, pulmonary fibrosis is characterized by exaggerated, persistent, and usually irreversible accumulation of extracellular matrix (ECM)1. Aberrant accumulation of ECM replaces normally functioning parenchyma and distorts the architecture of adjacent tissue2. This fibrotic process often leads to rapid and severe abnormalities in lung mechanics and gas exchange properties3. Pulmonary fibrosis is the end stage of almost all advanced interstitial lung diseases with diverse etiologies and has attracted increasing attention in recent years due to its increasing occurrence and poor therapy4,5. Idiopathic pulmonary fibrosis (IPF), clinically characterized by progressive dyspnea, is the most common type of idiopathic interstitial pneumonia, has a poor prognosis and is unresponsiveness to traditional therapies6. Although nintedanib and pirfenidone are available to treat IPF, neither of them appears to be able to completely arrest disease progression, and long-term treatment is accompanied by serious adverse effects7,8. Therefore, it is necessary to provide more therapeutic options for patients.
The cellular and molecular mechanisms of IPF are still not well understood. However, it is well known that the key hallmark of pulmonary fibrosis is the excessive differentiation of fibroblasts into myofibroblasts (marked by α-smooth muscle actin, α-SMA) and the excessive deposition of ECM (eg, collagen). Under pathological conditions, abnormal wound healing will augment the transformation of fibroblasts into myofibroblasts and prolong the survival of these ECM-producing cells9. Transforming growth factor-β1 (TGF-β1) is a major profibrotic mediator that converts fibroblasts to myofibroblasts and regulates the turnover of collagen10. In addition, increasing evidence has revealed that hypoxia-inducible factor-1α (HIF-1α) is required for TGF-β1-stimulated collagen production in pulmonary or other fibrotic diseases11,12,13,14.
HIF-1 is a master regulator of hypoxic signaling by activating gene transcription to mediate cellular adaption to hypoxia15,16. It is a heterodimeric transcription factor consisting of a constitutive β-subunit and an oxygen-sensitive α-subunit. Under normoxic conditions, HIF-1α is subject to ubiquitination and proteosomal degradation. However, under hypoxic conditions, HIF-1α is stabilized and translocates into the nucleus, where it dimerizes with HIF-1β and cofactors to drive the transcription of various target genes in response to hypoxia17,18. Chronic hypoxia has been considered a critical event during the development of renal, lung and hepatic fibrosis19,20,21,22. Hypoxia could induce the differentiation of fibroblasts into myofibroblasts23,24,25. Therefore, targeting HIF-1α may be a new therapeutic strategy for the treatment of fibrotic diseases26.
The root of Stemona tuberosa Lour, known as Baibu in Chinese, is one of traditional Chinese medicines with good antitussive, expectorant and anti-asthmatic effects27,28. Recent studies showed that the extract of S tuberosa could inhibit subacute lung inflammation, attenuate alveolar airspace enlargement and bronchiole epithelial hyperplasia in a cigarette smoke-induced mouse model29. Neotuberostemonine (NTS) is one of the main alkaloids isolated from S tuberosa with significant antitussive activity that is comparable to codeine but does not involve opioid receptors28. Our previous study showed that NTS effectively ameliorated bleomycin-induced lung fibrosis by suppressing the recruitment and activation of macrophages30.
However, fibroblast activation, which may be related to the polarization of M2 macrophages31, is a key step in the development of organ fibrosis. As mentioned above, chronic hypoxia is a critical event for fibrosis development. Therefore, in the present study, we aimed to investigate the role of hypoxia in the activation of lung fibroblasts and the effect of NTS on hypoxia-exposed fibroblasts.
Materials and methods
Reagents
Neotuberostemonine (C22H33NO4, MW 375.24) (Figure 1A) was isolated from the root of S tuberosa in our lab32 with a purity above 98% by HPLC analysis. Bleomycin hydrochloride (BLM) was purchased from Nippon Kayaku (Tokyo, Japan). Prednisone acetate (PDN) and digoxin (Dig) were provided by Jiangsu Pingguang Pharmaceutical Co Ltd (Xuzhou, China) and Tianjin Shilan Technology Co Ltd (Tianjin, China), respectively. CoCl2 was obtained from Chengdu Kelong Chemical Reagent Factory (Chengdu, China), and MG132 was purchased from APEXBIO (Houston, TX, USA). The above drugs were dissolved in dimethylsulfoxide (DMSO) as a stock solution, and the final working concentration of DMSO was less than 0.1% (v/v). All other reagents, including DMSO, were of analytical purity and were commercially available.
NTS suppressed the activation of PLFs under hypoxia. (A) Structure of NTS; (B–D) PLFs were treated with NTS or digoxin (Dig) at indicated concentrations and then incubated under 1% O2 or with 100 μmol/L CoCl2 under normoxia for 24 h; (B, C) the protein expression of α-SMA was determined by Western blot (n=3); and (D) the mRNA expression of Col-1α, Col-3α and Lox (lysyl oxidase) was assayed by RT-PCR (n=4). Data were expressed as the mean±SD. * P<0.05 vs the 1% O2 or CoCl2 treatment.
Animals
Six- to eight-week-old ICR male mice weighing 20–25 g were purchased from the Comparative Medicine Center of Yangzhou University (Yangzhou, China). Mice were housed at a constant temperature of 25 °C with alternatively 12 h light and dark cycles and kept on a standard mouse chow and water ad libitum. All animal experiments were conducted in accordance with the National Institutes of Health Guide. Care and use of animals complied with the Provisions and General Recommendation of the Chinese Experimental Animals Administration Legislation. Mice were allowed to acclimate for one week before any experiment and fasted on the day prior to experiments being conducted.
Cell experiments
Isolation, cultivation and identification of fibroblasts
Primary lung fibroblasts (PLFs) were isolated from male ICR mice by combining trypsin digestion and tissue adherent methods33,34 with modifications. Briefly, mouse lungs were excised and removed of tracheas and bronchials. After heavily washing with phosphate-buffered saline (PBS), lungs were dissected into 1–2 cm2-sized pieces and digested with 2.5 mg/mL of trypsin (Biosharp, Hefei, China) in PBS at 25 °C for 15 min. Digested lung pieces were suspended in Dulbecco's minimum essential (Eagles's) medium (DMEM) (Gibco, NY, USA) supplemented with 10% (v/v) fetal bovine serum (FBS) (Bioind, Israel), 100 μg/mL streptomycin (Biosharp, Hefei, China) and 100 U/mL penicillin (Biosharp, Hefei, China) at 37 °C in 5% CO2 for 8 h before being placed on the bottom of the cell culture flask. The lung pieces placed in the flasks were cultured in the same medium (changed 3 times a week) to allow the PLFs to migrate and divide continuously to reach a confiuence of 70%–80%. Isolated PLFs were cultured and purified for three passages in DMEM supplemented with 10% FBS and streptomycin/penicillin at 37 °C in a 5% CO2 atmosphere. PLFs were identified using vimentin immunohistochemical staining. PLFs of passages 3–5 with a purity greater than 95% (checked by the trypan-blue exclusion assay) were used for the experiments.
Treatment of PLFs
PLFs were seeded at an initial density of 1×105 cells/mL in 6-well plates for 24 h. After 12 h of starvation, the cells were treated with drugs at the indicated concentrations for 30 min and then exposed to hypoxia (1% O2, 94% N2, 5% CO2) or treated with 100 μmol/L CoCl2 under normoxia. After 24 h of incubation, cells were harvested for the detection of relevant indicators.
Immunofluorescent assay
Hypoxyprobe™-1 (Hypoxyprobe Inc, Burlington, USA) is a substituted 2-nitroimidazole named pimonidazole. It will bind to cells if the p O2 levels are less than 10 mmHg, so it is often used as a probe for detecting cell hypoxia. PLFs were cultured to 80% confluence. After 12 h of starvation, PLFs were incubated with 200 μmol/L hypoxyprobe and 10 μmol/L NTS under normoxia, hypoxia (1% O2) or 100 μmol/L CoCl2 treatment at 37 °C for 12 h. PLFs were fixed and blocked with 3% bovine serum albumin (BSA), then treated with anti-hypoxyprobe and DAPI. The blank control was treated in the same manner. All the treated PLFs were examined under confocal scanning microscopy.
Transfection with HIF-1α siRNA
NIH-3T3 cells (Passage 9, Cell Bank of the Chinese Academy of Sciences, Shanghai, China) grown to 70%–80% confluence were transfected with mouse HIF-1α siRNA (Santa Cruz, CA, USA) according to the manufacturer's instructions. After 8 h of transfection, the cells were cultured in fresh medium for 48 h and exposed to hypoxia (1% O2, 94% N2, 5% CO2) or treated with 100 μmol/L CoCl2 under normoxia for 24 h and then collected for the indicated experiments.
Animal experiments
BLM-induced pulmonary fibrosis and drug treatment design
ICR male mice were randomly divided into 5 groups (n=20), ie, the sham, model, NTS, PND and Dig groups. After 12 h of fasting, mice were intratracheally injected with BLM (3.5 U/kg in 0.9% NaCl) or 0.9% NaCl (sham group) after anesthesia with 4% chloral hydrate (10 mL/kg). After 7 d of model formation, BLM-treated mice were orally administered NTS (30 mg/kg), PDN (6.5 mg/kg) and an equivalent volume of the same menstruum (sham and model group) or intraperitoneally injected with Dig (1 mg/kg) once a day for 7 or 14 consecutive days. The lungs were excised on d 15 and 22 after BLM treatment.
Histopathological examination
Lung specimens fixed in 4% paraformaldehyde and embedded in paraffin were cut into 5-μm-thick sections and stained with hematoxylin & eosin (H&E) or Masson trichrome. The slides were examined under an Olympus BX53TR microscope and photographed with a DP72 digital color camera (Olympus, Tokyo, Japan). The contents of absolute collagen were semi-quantified by determining the integrated optical density (IOD) of at least five sections each in 10 microscopic fields using Image-Pro Plus image analysis software (version 6.0, Media Cybernetics, USA)35,36.
Immunohistochemical analysis
For immunohistochemical analysis, 5-μm-thick sections as mentioned above were dewaxed and rehydrated, quenched with 3% hydrogen peroxide, incubated in 0.1% citric buffer for antigen retrieval and blocked with 3% BSA. After pretreatment, sections were incubated with rabbit anti-fibronectin (BS1644, Bioworld, St Louis, MO, USA) antibodies using reagents from an ElivisionTM super HRP IHC Kit (Maixin Biotech, Fuzhou, China) and developed with a DAB Kit (Maixin Biotech, Fuzhou, China).
Quantitative real-time polymerase chain reaction (RT-PCR)
Total RNA of PLFs or lung tissue was extracted using TRIzol Reagent (Kangweishiji, Beijing, China) and reverse-transcribed to cDNA templates using an RT-MasterMix Kit (Applied Biological Materials Inc, Vancouver, Canada). Quantitative RT-PCR was performed using SYBR Green Supermix (BIO-RAD, CA, USA). Samples were measured in at least triplicate with a CFX96 real-time system (BIO-RAD, CA, USA). The specific gene primers for Col (collagen)-1α, Col-3α, LOX (lysyl oxidase), HIF-1α and β-actin were designed using Primer5 software by PRIMER-E Ltd (Plymouth, Devon, UK) (Table 1). Mouse β-actin was used as an internal control. All sample data were quantified using the comparative Ct method and presented as the mean ratio to β-actin.
Western blot analysis
Immunoblot analysis of PLFs or lung tissue was performed on total lysates as previously described30 using primary rabbit anti-α-SMA (smooth muscle actin) (YH090708D, Epitomics, Burlingame, USA), rabbit anti-HIF-1α (ab51608, Abcam, Cambridge, UK), rabbit anti-TGF-β (transforming growth factor-β) (#3711, Cell Signaling Technology, Beverly, USA), rabbit anti-FGF-2 (fibroblast growth factor-2) (ab8880, Abcam, Cambridge, UK), rabbit anti-FGFR-2 (fibroblast growth factor receptor-2) (ab109372, Abcam, Cambridge, UK), rabbit anti-hydroxy-HIF-1α (Pro564) (#3434, Cell Signaling Technology, Beverly, USA) or mouse anti-β-actin (BS6007M, Bioworld Technology, St Louis, MO, USA) antibodies and then incubated with corresponding secondary antibodies. Membranes were developed with enhanced chemiluminescence (ECL) reagent (Beyotime, Shanghai, China). The band intensity was quantitatively analyzed with Image-Pro Plus (IPP) 6.0 software and presented as the mean ratio to β-actin.
Statistical analysis
Data were expressed as the mean±SD (standard deviation) of at least three independent experiments. Statistical analysis was performed using one-way ANOVA followed by Bonferroni's multiple comparison post hoc tests (GraphPad Prism 5, GraphPad Software, USA). P-values less than 0.05 were considered statistically significant.
Results
NTS suppresses the activation of hypoxia-exposed lung fibroblasts
To determine the effect of hypoxia on lung fibroblasts and the effect of NTS (Figure 1A) on hypoxia-exposed lung fibroblasts, PLFs were exposed to 1% O2 or 100 μmol/L CoCl2 (a model of chemical hypoxia) and treated with or without NTS and digoxin (positive control). Both 1% O2 and CoCl2 treatments significantly increased the expression of α-SMA, a marker of myofibroblasts, indicating that the PLFs had been activated (Figure 1B, 1C). NTS treatment significantly reduced the 1% O2-induced α-SMA levels at concentrations of 1.0 and 10 μmol/L (Figure 1B) and obviously reduced the CoCl2-induced α-SMA levels at concentrations of 0.1, 1.0 and 10 μmol/L (Figure 1C). Correspondingly, NTS decreased the hypoxia-induced mRNA expression of Col-1α, Col-3α and LOX (a critical enzyme in the covalent cross-linkages of collagen and elastin) at 1.0 and 10 μmol/L (Figure 1D). Expectedly, digoxin, a HIF-1α inhibitor, effectively reduced the protein expression of α-SMA and the mRNA expression of Col-1α, Col-3α and LOX after hypoxia/chemical hypoxia exposure. Taken together, these results demonstrated that hypoxia can directly activate fibroblasts and differentiate them into myofibroblasts that produce ECM, while NTS inhibits collagen synthesis in hypoxia-exposed PLFs by suppressing the activation and differentiation of fibroblasts.
NTS decreases HIF-1α expression in hypoxia-induced lung fibroblasts
HIF-1α is a master regulator that mediates cellular adaption to hypoxia. For the two models mentioned above, 1% O2 induces HIF-1α accumulation by lowering local oxygen tension, while CoCl2 stabilizes HIF-1α by inhibiting the activity of HIF-1 prolyl hydroxylases (not true hypoxia). Therefore, we first detected whether hypoxia was present in the cells of the above two models. PLFs were incubated with hypoxyprobe (pimonidazole) and stained using an anti-hypoxyprobe antibody. As expected, pimonidazole adducts were clearly observed in cells exposed to 1% O2 but were rarely found in cells treated with CoCl2 (Figure 2A). Surprisingly, a reduction of adducts was observed with NTS (10 μmol/L) treatment, indicating that NTS can protect cells from hypoxia via an unknown mechanism. Next, we examined the expression of HIF-1α by Western blot analysis. The HIF-1α level was significantly increased in cells exposed to 1% O2 or CoCl2 (100 μmol/L) compared with the control, but NTS reduced the expression of HIF-1α in a dose-dependent manner in both models (Figure 2B, 2C). Digoxin, a HIF-1α inhibitor, also displayed positive effects in both models (Figure 2B, 2C). The above results showed that NTS prevents the activation of hypoxia-exposed lung fibroblasts by improving hypoxic microenvironments and promoting the decomposition of HIF-1α.
NTS decreased HIF-1α levels by improving hypoxic microenvironments and promoting decomposition in hypoxia-exposed PLFs. (A) PLFs were treated with hypoxyprobe (200 μmol/L) and NTS (10 μmol/L), incubated under 1% O2 or with 100 μmol/L CoCl2 under normoxia for 12 h, then treated with anti-hypoxyprobe antibody and DAPI, observed under confocal scanning microscopy (×200). (B, C) PLFs were treated with NTS or digoxin (Dig) at the indicated concentrations and incubated under the same hypoxic conditions mentioned above for 24 h. The protein expression of HIF-1α was determined by Western blot (n=3). Data were expressed as the mean±SD. * P<0.05 vs the 1% O2 or CoCl2 treatment.
NTS decreases the expression of TGF-β and FGF2/FGFR2 in hypoxia-exposed lung fibroblasts
TGF-β, FGF2 and FGFR2 are involved in the activation of fibroblasts37,38; thus, we examined the activity of TGF-β and FGF2/FGFR2 in hypoxia-exposed PLFs and the effect of NTS on them. In fibroblasts challenged with 1% O2 (Figure 3A–3D), the protein expression of TGF-β, FGF2 and FGFR2 was increased significantly. However, NTS treatment effectively decreased the expression of TGF-β (at 1–10 μmol/L), FGF2 (at 0.1–10 μmol/L) and FGFR2 (at 1–10 μmol/L). Similarly, in fibroblasts stimulated with CoCl2 (Figure 3E–3H), the expression of TGF-β, FGF2 and FGFR2 was also significantly elevated, and this elevated expression was effectively inhibited by NTS at 10 μmol/L (for TGF-β) or 1–10 μmol/L (for both FGF2 and FGFR2). The above results demonstrated that hypoxia or chemical hypoxia induced increased expression of TGF-β, FGF2 and FGFR2 but that NTS could reverse the elevations in expression to suppress fibroblast activation. As an inhibitor of HIF-1α, digoxin also blunted 1% O2- or CoCl2-exposed changes in TGF-β, FGF2 and FGFR2 activity (Figure 3), indicating that the up-regulation of TGF-β, FGF2 and FGFR2 might be dependent, at least in part, on the accumulation of HIF-1α.
NTS decreased the expression of TGF-β, FGF2 and FGFR2 in hypoxia-exposed PLFs. PLFs were treated with NTS or digoxin (Dig) at the indicated concentrations, incubated under 1% O2 or with 100 μmol/L CoCl2 under normoxia for 24 h. The protein expression of TGF-β, FGF2 and FGFR2 in cells with (A–D) 1% O2 or (E–H) CoCl2 treatment was determined by Western blot (n=3). Data were expressed as the mean±SD. * P<0.05 vs the 1% O2 or CoCl2 treatment.
HIF-1α is essential for the induction of TGF-β and FGF2 under hypoxia
To further dissect the role of HIF-1α on the protein expression of TGF-β, FGF2 and α-SMA under hypoxia, NIH-3T3 cells, a mouse embryo fibroblast cell line, were transfected with HIF-1α siRNA plasmid to silence endogenous HIF-1α. HIF-1α induction responding to 1% O2 or CoCl2 was successfully counteracted by specific HIF-1α siRNA transfection in NIH-3T3 cells (Figure 4A, 4B). Correspondingly, the expression of α-SMA, TGF-β and FGF2 in cells transfected with HIF-1α siRNA plasmids (Figure 4A, 4C–4F) was not increased after exposure to 1% O2 or CoCl2, although their expression was significantly increased in cells transfected with scramble siRNA plasmids. These results demonstrated that HIF-1α is essential for the induction of TGF-β and FGF2 to activate and differentiate fibroblasts into myofibroblasts (marked by α-SMA).
HIF-1α was essential for the induction of TGF-β and FGF2 under hypoxic conditions. NIH-3T3 cells were transfected with HIF-1α or control scrambled siRNAs and then incubated under 1% O2 or with 100 μmol/L CoCl2 under normoxia for 24 h. (A–F) The protein expression of HIF-1α, α-SMA, TGF-β and FGF2 was determined by Western blot (n=3). Data were expressed as the mean±SD. # P<0.05 or * P<0.05 vs the indicated treatment.
NTS inhibits the activation of lung fibroblasts by promoting the degradation of HIF-1α
Given that HIF-1α is essential for fibroblast activation and NTS inhibited the induction of HIF-1α, TGF-β and FGF2 under hypoxia, we hypothesized that NTS inhibits the activation of lung fibroblasts in a HIF-1α-dependent manner. Therefore, the PLFs exposed to 1% O2 or CoCl2 were treated with or without NTS and MG132 (a proteasome inhibitor blocking the degradation of HIF-1α), and the protein expression of HIF-1α, α-SMA, TGF-β and FGF2 was analyzed by Western blot. As expected, the inhibitory effect of NTS (10 μmol/L) on HIF-1α accumulation (Figure 5A, 5B, 5F, 5G) was absolutely abolished in both hypoxic models when MG132 (10 μmol/L) was used. Accordingly, the inhibitory effect of NTS on the expression of α-SMA, TGF-β and FGF2 (Figure 5A, 5C–5E, 5F, 5H–5J) was also completely reversed by co-treatment with MG132 in both hypoxic models. This suggested that NTS suppresses the activation of fibroblasts under hypoxia in a HIF-1α-dependent manner.
Effect of NTS inhibiting the activation of fibroblasts was HIF-1α-dependent. PLFs were treated with NTS (10 μmol/L) and incubated with MG132 (10 μmol/L) (A–E) under 1% O2 or (F–J) with CoCl2 (100 μmol/L) for 24 h. The protein expression of HIF-1α, α-SMA, TGF-β and FGF2 was determined by Western blot (n=3); or (K) directly incubated under hypoxic conditions for 24 h, the protein expression of PHD (prolyl hydroxylated)-HIF-1α was determined by Western blot (n=3). Data were expressed as the mean±SD. # P<0.05 vs the blank; * P<0.05 vs the 1% O2 or CoCl2 treatment.
Moreover, it is well known that prolyl hydroxylation is a critical step in the initiation of HIF-1α degradation. Thus, the protein expression of PHD (prolyl hydroxylated)-HIF-1α in lung fibroblasts exposed to 1% O2 or CoCl2 was also examined. The level of PHD-HIF-1α decreased markedly under 1% O2 and CoCl2 conditions, but this decrease was effectively reversed by NTS (10 μmol/L) (Figure 5K). This result indicated that NTS promotes degradation of HIF-1α partially due to its improvement on HIF-1α prolyl hydroxylation.
NTS inhibits HIF-1α, TGF-β and FGF2 expression in the lungs of BLM-induced mice
IPF patients universally exhibit hypoxemia in association with increased levels of HIF-1α16. To determine whether hypoxia was present in BLM-induced mice, the protein and mRNA expression of HIF-1α in lung tissue was detected. Compared with sham mouse lungs (Figure 6A, 6B), both HIF-1α protein and mRNA were significantly increased in BLM-induced mouse lungs, especially mRNA expression after 14 days of bleomycin administration. Our previous study found that NTS had a weak inhibitory effect at 15 mg/kg and a strong inhibitory effect at 30 or 60 mg/kg on mouse lung fibrosis; however, the mortality of high-dose-treated animals was higher. Therefore, the medium dose (30 mg/kg) was chosen as the experimental dose. After 7 days of model formation, BLM-induced mice were administered with NTS and positive drugs for 7 or 14 consecutive days. After 7 and 14 days of treatment, NTS effectively reduced the protein and mRNA expression of HIF-1α (Figure 6A, 6B). Although PDN and digoxin also reduced the levels of HIF-1α, PDN destroyed the normal structure of lung tissue. Further, the changes in TGF-β and FGF2 in BLM-induced mouse lungs before and after drug administration were also analyzed by Western blotting. Compared to the control, the increased protein expression of FGF2 was significantly inhibited by NTS in both the 7- and 14-day treatments (Figure 6C). However, NTS reduced the expression of TGF-β only after a long treatment period (21 days = 7 days model formation + 14 days treatment) (Figure 6D).
NTS inhibited HIF-1α, TGF-β and FGF2 expression in BLM-induced pulmonary fibrosis mice. Mice were administered with NTS (30 mg/kg, orally) per day from days 8 to 14 or to 21 after BLM intratracheally administration (3.5 U/kg), taking PDN (prednisone, 6.5 mg/kg, orally) and Dig (digoxin, 1 mg/kg, ip) as the positive controls. (A, C, D) The protein expression of HIF-1α, FGF2 and TGF-β in lungs was determined by Western blot (n=3-4), and (B) the mRNA expression of HIF-1α was measured by RT-PCR (n=4). Data were expressed as the mean±SD. * P<0.05 vs the model.
NTS inhibits ECM accumulation in the lungs of BLM-induced mice
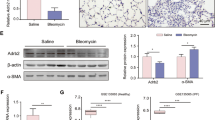
Our in vitro experiments demonstrated that NTS reduced the deposition of ECM by inhibiting the activation of fibroblasts, which needed to be confirmed by experiments in vivo. Thus, we investigated the effects of NTS on histopathological changes and ECM accumulation in the lungs of BLM-induced mice. Compared with the sham mouse lungs, BLM-injured lungs showed enhanced infiltration of infiammatory cells, the loss of normal alveolar structure, extensive thickening of the interalveolar septa, and extremely high Ashcroft scores (Figure 7A, 7C). NTS treatment for 7 or 14 days could effectively reduce BLM-induced histopathological changes and lower the Ashcroft scores in the lungs (Figure 7A, 7C). Masson trichrome staining and IOD values showed that BLM challenge led to substantial interstitial fibrosis in lung tissue with increased collagen deposition (indicated by blue staining) but treatment with NTS for 7 or 14 days significantly attenuated the collagen deposition in the lungs (Figure 7B, 7D). Moreover, the protein expression of α-SMA (Figure 7E) and the mRNA expression of Col-1α, Col-3α and LOX (Figure 7F, 7G) were also markedly elevated in the lungs of BLM-induced mice, and the administration of NTS for 7 or 14 days effectively reduced the levels of these factors, except that Col-1α was reduced only after a long (14-day) treatment.
NTS inhibited ECM accumulation in BLM-induced pulmonary fibrosis. Mice were administered with NTS (30 mg/kg, orally) per day from days 8 to 14 or to 21 after BLM intratracheally administration (3.5 U/kg), taking PDN (prednisone, 6.5 mg/kg, orally.) and Dig (digoxin, 1 mg/kg, ip) as the positive controls. (A, B) Lung tissue was stained with HE or Masson trichrome (collagen in blue color) and (C, D) semiquantitatively assessed by Ashcroft score for histopathological change (n=3) and integrated optical density (IOD) for collagen deposition (n=6). (E) The protein expression of α-SMA and (F-G) the mRNA expression of Col-1α, Col-3α and LOX (lysyl oxidase) were determined by Western blot (n=3) or qPCR (n=4). (H) The expression of fibronectin in lung tissue was examined by immunofluorescence staining. Data were expressed as the msean±SD. * P<0.05 vs the model.
Fibronectin, a high-molecular-weight glycoprotein of the extracellular matrix, is often secreted from fibroblasts. It assembles extracellular matrix components such as collagen, fibrin, and heparan sulfate proteoglycan. Thus, the protein expression of fibronectin in BLM-induced mice was detected by immunofluorescence assay. The green-colored fibronectin was strongly increased in the lungs of BLM-induced mice, while the over-expression of fibronectin was obviously inhibited by NTS treatment for 7 and 14 days (Figure 7H).
Taken together, the above results showed that NTS can effectively inhibit fibroblast activation and prevent ECM deposition in vivo.
Discussion
Although pirfenidone and nintedanib can slow lung function decline in IPF, lung transplantation remains the only curative treatment for this devastating disease. It is imperative to discover and develop more effective therapies and to provide more therapeutic options for patients. Our previous studies have demonstrated that NTS, an alkaloid isolated from S tuberosa, effectively ameliorated BLM-induced lung fibrosis in mice30. The present study investigated the effect and underlying mechanism of NTS on the activation of lung fibroblasts under hypoxia. In vitro experiments demonstrated that lung fibroblasts were activated and differentiated into myofibroblasts under hypoxic or chemical hypoxic conditions, whereas NTS blocked this activation and differentiation in a HIF-1α-dependent manner. Accordingly, in vivo experiments confirmed that NTS attenuates BLM-induced pulmonary fibrosis by inhibiting the levels of HIF-1α and related downstream cytokines such as TGF-β and FGF2.
As discussed earlier, fibrosis is increasingly regarded as the result of deregulated tissue repair in response to chronic hypoxia19,20,21,22, and severe chronic hypoxia is overt in involved tissues of patients with fibrotic disease26. Significant expression of HIF-1α was observed in the lung samples of IPF patients, especially of those with pulmonary hypertension16,39. It was also reported that hypoxia robustly stimulated the proliferation and differentiation of IPF fibroblasts40,41. Our experiment demonstrated that NTS could effectively alleviate the hypoxic conditions in pulmonary fibroblast cells.
Inhibition of the pathways regulated by pro-fibrotic cytokines, such as TGF-β, FGF, VEGF and PDGF, is thought to be an effective approach to the treatment of pulmonary fibrosis42. As a potent pro-fibrotic growth factor, TGF-β directly affects the proliferation and differentiation of fibroblasts43, but the underlying mechanism is still not completely established. TGF-β1 can up-regulate HIF-1α expression in MRC-5 cells (human fetal lung fibroblasts) at both the protein and mRNA levels41. Our in vitro results showed that the stabilized HIF-1α exposed to hypoxia or chemical hypoxia greatly up-regulated TGF-β proteins, supporting that HIF-1α and TGF-β may form a positive feedback loop during the development of pulmonary fibrosis14.
FGF2 is released by activated fibroblasts. It stimulates the proliferation of lung fibroblasts and the production of fibronectin38,44. Moreover, TGF-β1-induced proliferation of fibroblasts is regulated through the release of extracellular FGF238. Therefore, blocking the FGF2/FGFR2 signaling pathway may be required for intervention in the proliferation and differentiation of fibroblasts. Our data showed that the high expression of HIF-1α in hypoxia-exposed fibroblasts led to a great elevation of FGF2 and FGFR2 protein levels, together with significantly increased α-SMA, Col-1α and Col-3α expression.
However, all the elevation of TGF-β, FGF2 and α-SMA expression after exposure to 1% O2 or CoCl2 was reversed after the transfection of HIF-1α, suggesting that hypoxia-induced HIF-1α is the key upstream regulator of TGF-β and FGF2 signaling. Importantly, NTS exerts protective effects against experimental pulmonary fibrosis by regulating HIF-1α signaling. NTS significantly inhibited the protein expression of HIF-1α, TGF-β and FGF2 both in hypoxia-exposed lung fibroblasts and in BLM-induced lung tissue. The inhibitory effect of NTS on HIF-1α, TGF-β and FGF2 led to an inhibitory effect on ECM deposition. These results suggested that NTS ameliorates pulmonary fibrosis by at least partially targeting HIF-1α.
Molecular oxygen is a cofactor for prolyl hydroxylases that hydroxylate HIF-1α protein at positions P564 and P402. Hydroxylated HIF-1α protein is recognized by the VHL-E3 ligase, which transports HIF-1α to the proteasome for degradation45,46,47. Low oxygen levels inhibit the prolyl hydroxylation at positions P564 and P402 and allow HIF-1α to escape degradation and enter the nucleus to drive the expression of target genes48. Consistent with the above, PHD (prolyl hydroxylated)-HIF-1α was obviously inhibited under hypoxic/chemical hypoxic conditions, while NTS effectively elevated the PHD-HIF-1α levels. Proteasomes are the executor to degrade the protein of HIF-1α. It was reported that HIF-1α can be degraded by proteasomes through ubiquitin-dependent or ubiquitin-independent pathways49,50. Our experiments showed that the inhibitory effects of NTS on hypoxia-exposed HIF-1α could be abolished by co-treatment with proteasome inhibitors. Thus, we speculated that the effect of NTS on the promotion of HIF-1α degradation may be achieved by increasing the activity of both prolyl hydroxylases and proteasomes. This speculation requires further studies to draw conclusions.
Our results highlight that the activation of lung fibroblasts under hypoxia is HIF-1α dependent and HIF-1α will stimulate downstream profibrotic cytokines such as TGF-β and FGF2/FGFR2. NTS, a natural compound from S tuberosa, effectively inhibited the activation of lung fibroblasts and ameliorated mouse pulmonary fibrosis by alleviating hypoxic conditions and inhibiting HIF-1α signaling. This finding suggested that NTS or related small molecules, which can improve the cell hypoxic milieu and promote HIF-1α degradation, possibly representing a promising novel therapeutic approach for pulmonary fibrosis treatment26. However, the therapeutic mechanism of NTS for pulmonary fibrosis may not be limited to HIF-1α signaling. Moreover, the upstream factors of HIF-1α in BLM-induced mice and the manner in which HIF-1α induces profibrotic cytokines, especially TGF-β, require further investigation.
In conclusion, hypoxia, which stabilizes HIF-1α, is an early event for the activation of lung fibroblasts, and NTS, or related drugs with activities that improve hypoxic conditions and promote HIF-1α degradation, may be a novel pharmacological approach in the treatment of pulmonary fibrosis. However, there is still an unmet need for selective, effective and safe anti-lung fibrosis drugs.
Abbreviations
α-SMA, α-smooth muscle actin; BLM, bleomycin; BSA, bovine serum albumin; Col, collagen; Dig, digoxin; DMEM, Dulbecco's minimum essential medium; DMSO, dimethylsulfoxide; ECM, extracellular matrix; FBS, fetal bovine serum; FGF2, fibroblast growth factor 2; FGFR2, fibroblast growth factor receptor 2; HIF-1α, hypoxia inducible factor-1α; IOD, integrated optical density; IPF, idiopathic pulmonary fibrosis; NTS, neotuberostemonine; PDN, prednisone acetate; PLFs, primary mouse lung fibroblasts; RT-PCR, real-time polymerase chain reaction; TGF-β1, transforming growth factor-β1; VEGF, vascular endothelial growth factor.
Author contribution
Ming-dan LI, Bao-lin LIU and Mian ZHANG designed the study; Kang LIU, Chao-feng ZHANG and Xiang-hong XU participated in the experimental design; Xin-miao LV and Ming-dan LI conducted the experiments; Si CHENG participated in the animal experiments and analyzed experimental data; Chao-feng ZHANG and Xiang-hong XU conducted data analysis; Xin-miao LV and Ming-dan LI summarized the experimental data and drafted the paper; Mian ZHANG, Bao-lin LIU and Kang LIU revised the paper.
References
Rockey DC, Bell PD, Hill JA. Fibrosis - a common pathway to organ injury and failure. N Engl J Med 2015; 372: 1138–49.
odd NW, Luzina IG, Atamas SP. Molecular and cellular mechanisms of pulmonary fibrosis. Fibrogenesis Tissue Repair 2012; 5: 11–35.
Luzina IG, Todd NW, Sundararajan S, Atamas SP. The cytokines of pulmonary fibrosis: much learned, much more to learn. Cytokine 2015; 74: 88–100.
Steele MP, Schwartz DA. Molecular mechanisms in progressive idiopathic pulmonary fibrosis. Annu Rev Med 2013; 64: 265–76.
Rangarajan S, Locy ML, Luckhardt TR, Thannickal VJ. Targeted therapy for idiopathic pulmonary fibrosis: where to now? Drugs 2016; 76: 291–300.
Wolters PJ, Collard HR, Jones KD. Pathogenesis of idiopathic pulmonary fibrosis. Annu Rev Pathol Mech Dis 2014; 9: 157–79.
Richeldi L, du Bois RM, Raghu G, Azuma A, Brown KK, Costabel U, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med 2014; 29: 2071–82.
Noble PW, Albera C, Bradford WZ, Costabel U, Glassberg MK, Kardatzke D, et al. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomised trials. Lancet 2011; 377: 1760–9.
Kramann R, DiRocco DP, Humphreys BD. Understanding the origin, activation and regulation of matrix-producing myofibroblasts for treatment of fibrotic disease. J Pathol 2013; 231: 273–89.
Hinz B, Phan SH, Thannickal VJ, Prunotto M, Desmoulière A, Varga J, et al. Recent developments in myofibroblast biology paradigms for connective tissue remodeling. Am J Pathol 2012; 180: 1340–55.
Abdul-Hafez A, Shu R, Uhal BD. JunD and HIF-1α mediate transcriptional activation of angiotensinogen by TGF-β1 in human lung fibroblasts. FASEB J 2009; 23: 1655–62.
Ueno M, Maeno T, Nomura M, Aoyagi-Ikeda K, Matsui H, Hara K, et al. Hypoxia-inducible factor-1α mediates TGF-induced PAI-1 production in alveolar macrophages in pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol 2011; 300: L740–52.
Basu RK, Hubchak S, Hayashida T, Runyan CE, Schumacker PT, Schnaper HW. Interdependence of HIF-1α and TGF-β/Smad3 signaling in normoxic and hypoxic renal epithelial cell collagen expression. Am J Physiol Renal Physiol 2011; 300: F898–905.
Qian F, He M, Duan W, Mao L, Li Q, Yu Z, et al. Cross regulation between hypoxia-inducible transcription factor-1α (HIF-1α) and transforming growth factor (TGF)- β1 mediates nickel oxide nanoparticles (NiONPs)-induced pulmonary fibrosis. Am J Transl Res 2015; 7: 2364–78.
Semenza GL. Regulation of mammalian O2 homeostasis by hypoxia-inducible factor 1. Annu Rev Cell Dev Biol 1999; 15: 551–78.
Tzouvelekis A, Harokopos V, Paparountas T, Oikonomou N, Chatziioannou A, Vilaras G, et al. Comparative expression profiling in pulmonary fibrosis suggests a role of hypoxia-inducible factor-1α in disease pathogenesis. Am J Respir Crit Care Med 2007; 176: 1108–19.
Gőrlach A, Camenisch G, Kvietikova I, Vogt L, Wenger RH, Gassmann M. Effcient translation of mouse hypoxia-inducible factor-1α under normoxic and hypoxic conditions. Biochim Biophys Acta 2000; 1493: 125–34.
Shimoda LA, Semenza GL. HIF and the lung: role of hypoxia-inducible factors in pulmonary development and disease. Am J Respir Crit Care Med 2011; 183: 152–6.
Brauna RK, Broytmanc O, Brauna FM, Brinkmanc JA, Clitheroa A, Modic D, et al. Chronic intermittent hypoxia worsens bleomycin-induced lung fibrosis in rats. Respir Physiol Neurobiol 2017. pii: S1569-9048(16) 30325-1.
Zhan L, Huang C, Meng XM, Song Y, Wu XQ, Yang Y, et al. Hypoxia-inducible factor-1α in hepatic fibrosis: A promising therapeutic target. Biochimie 2015; 108: 1–7.
Higgins DF, Kimura K, Bernhardt WM, Shrimanker N, Akai Y, Hohenstein B, et al. Hypoxia promotes fibrogenesis in vivo via HIF-1 stimulation of epithelial-to-mesenchymal transition. J Clin Invest 2007; 117: 3810–20.
Liu M, Ning X, Li R, Yang Z, Yang X, Sun S, et al. Signalling pathways involved in hypoxia-induced renal fibrosis. J Cell Mol Med 2017; 21: 1248–59.
Norman JT, Clark IM, Garcia PL. Hypoxia promotes fibrogenesis in human renal fibroblasts. Kidney Int 2000; 58: 2351–66.
Short M, Nemenoff RA, Zawada WM, Stenmark KR, Das M. Hypoxia induces differentiation of pulmonary artery adventitial fibroblasts into myofibroblasts. Am J Physiol Cell Physiol 2004; 286: C416–25.
Zhong H, Belardinelli L, Maa T, Zeng D. Synergy between A2B adenosine receptors and hypoxia in activating human lung fibroblasts. Am J Respir Cell Mol Biol 2005; 32: 2–8.
Xiong A, Liu Y. Targeting hypoxia inducible factors-1a as a novel therapy in fibrosis. Front Pharmacol 2017; 8: 326–33.
Xua YT, Shaw PC, Jiang RW, Honc PM, Chana YM, But PP. Antitussive and central respiratory depressant effects of Stemona tuberose. J Ethnopharmacol 2010; 128: 679–84.
Chung HS, Hon PM, Lin G, But PP, Dong H. Antitussive activity of Stemona alkaloids from Stemona tuberose. Planta Med 2003; 69: 914–20.
Lee H, Jung KH, Park S, Kil YS, Chung EY, Jang YP, et al. Inhibitory effects of Stemona tuberosa on lung inflammation in a subacute cigarette smoke induced mouse model. BMC Complement Altern Med 2014; 14: 513–24.
Xiang J, Cheng S, Feng TL, Wu Y, Xie WN, Zhang M, et al. Neotuberostemonine attenuates bleomycin-induced pulmonary fibrosis by suppressing the recruitment and activation of macrophages. Int Immunopharmacol 2016; 36: 158–64.
Murray PJ, Wynn TA. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 2011; 11: 723–37.
Gao Y, Wang J, Zhang CF, Xu XH, Zhang M, Kong LY. Seven new alkaloids from the roots of stemona tuberose. Tetrahedron 2014; 70: 967–74.
Bruce MC, Honaker CE. Transcriptional regulation of tropoelastin expression in rat lung fibroblasts: changes with age and hyperoxia. Am J Physiol 1998; 274: 940–50.
Hao CF, Li XF, Yao W. Role of insulin-like growth factor II receptor in transdifferentiation of free silica-induced primary rat lung fibroblasts. Biomed Environ Sci 2013; 26: 979–85.
Ashcroft T, Simpson JM, Timbrell V. Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 1988; 41: 467–70.
Hübner RH, Gitter W, El Mokhtari NE, Mathiak M, Both M, Bolte H, et al. Standardized quantification of pulmonary fibrosis in histological samples. Biotechniques 2008; 44: 507–17.
Wang P, Wang Y, Nie X, Braïni C, Bai R, Chen CY. Multiwall carbon nanotubes directly promote fibroblast-myofibroblast and epithelial-mesenchymal transitions through the activation of the TGF-β/Smad signaling pathway. Small 2015; 11: 446–55.
Khalil N, Xu YD, O'Connor R, Duronio V. Proliferation of pulmonary interstitial fibroblasts is mediated by transforming growth factor-1-induced release of extracellular fibroblast growth factor-2 and phosphorylation of p38 MAPK and JNK. J Biol Chem 2005; 280: 43000–9.
Bryant AJ, Carrick RP, McConaha ME, Jones BR, Shay SD, Moore CS, et al. Endothelial HIF signaling regulates pulmonary fibrosis-associated pulmonary hypertension. Am J Physiol Lung Cell Mol Physiol 2016; 310: L249–62.
Bodempudi V, Hergert P, Smith K, Xia H, Herrera J, Peterson M, et al. MiR-210 promotes IPF fibroblast proliferation in response to hypoxia. Am J Physiol Lung Cell Mol Physiol 2014; 307: 283–94.
Xie N, Tan Z, Banerjee SM, Cui HC, Ge J, Liu RM, et al. Glycolytic reprogramming in myofibroblast differentiation and lung fibrosis. Am J Respir Crit Care Med 2015; 192: 1462–74.
Chaudhary NI, Roth GJ, Hilberg F, Müller-Quernheim J, Prasse A, Zissel G, et al. Inhibition of PDGF, VEGF and FGF signalling attenuates fibrosis. Eur Respir J 2007; 29: 976–85.
Aschner Y, Downey GP. Transforming growth factor-β: master regulator of the respiratory system in health and disease. Am J Respir Cell Mol Biol 2016; 54: 647–55.
Ge Z, Li B, Zhou X, Yang Y, Zhang J. Basic fibroblast growth factor activates β-catenin/RhoA signaling in pulmonary fibroblasts with chronic obstructive pulmonary disease in rats. Mol Cell Biochem 2016; 423: 165–74.
Masson N, Willam C, Maxwell PH, Pugh CW, Ratcliffe PJ. Independent function of two destruction domains in hypoxia-inducible factor-α chains activated by prolyl hydroxylation. EMBO J 2001; 20: 5197–206.
Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ, et al. Targeting of HIF-α to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 2001; 292: 468–72.
Ivan M, Kondo K, Yang HF, Kim W, Valiando J, Ohh M, et al. HIF-1α targeted for VHL-mediated destruction by proline hydroxylation: Implications for O2 sensing. Science 2001; 292: 464–8.
Pugh CW, Ratcliffe PJ. The von Hippel–Lindau tumor suppressor, hypoxia-inducible factor-1 (HIF-1) degradation, and cancer pathogenesis. Semin Cancer Biol 2003; 13: 83–9.
Uchida T, Rossignol F, Matthay MA, Mounier R, Couette S, Clottes E, et al. Prolonged hypoxia differentially regulates hypoxia-inducible factor (HIF)-1α and HIF-2α expression in lung epithelial cells. J Biol Chem 2004; 279: 14871–8.
Kong XG, Alvarez-Castelao B, Lin Z, Castaño JG, Caro J. Constitutive/hypoxic degradation of HIF-1α proteins by the proteasome is independent of von Hippel Lindau protein ubiquitylation and the transactivation activity of the protein. J Biol Chem 2007; 282: 15498–505.
Acknowledgements
This work was funded by the National New Drug Innovation Major Project of China (2011ZX09307-002-02).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lv, Xm., Li, Md., Cheng, S. et al. Neotuberostemonine inhibits the differentiation of lung fibroblasts into myofibroblasts in mice by regulating HIF-1α signaling. Acta Pharmacol Sin 39, 1501–1512 (2018). https://doi.org/10.1038/aps.2017.202
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/aps.2017.202
- Springer Nature Singapore Pte Ltd.
Keywords
This article is cited by
-
MKLN1-AS promotes pancreatic cancer progression as a crucial downstream mediator of HIF-1α through miR-185-5p/TEAD1 pathway
Cell Biology and Toxicology (2024)
-
A review of the botany, traditional uses, phytochemistry and pharmacology of Stemonae Radix
Phytochemistry Reviews (2022)