Abstract
Background
Acute myocardial infarction (AMI) causes irreversible myocardial damage and release of inflammatory mediators, including cytokines, chemokines and miRNAs. We aimed to investigate changes in the levels of cytokines (IL-6, TNF-α and IL-10), miRNAs profiles (miR-146 and miR-155) and distribution of different monocyte subsets (CD14++CD16-, CD14++CD16+, CD14+CD16++) in the acute and post-healing phases of AMI.
Methods
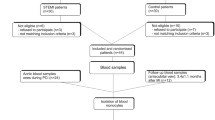
In eighteen consecutive AMI patients (mean age 56.78 ± 12.4 years, mean left ventricle ejection fraction — LVEF: 41.9 ± 9.8%), treated invasively, monocyte subsets frequencies were evaluated (flow cytometry), cytokine concentrations were analyzed (ELISA) as well as plasma miRNAs were isolated twice — on admission and after 19.2 ± 5.9 weeks of follow-up. Measurements were also performed among healthy volunteers.
Results
AMI patients presented significantly decreased frequencies of classical cells in comparison to healthy controls (median 71.22% [IQR: 64.4–79.04] vs. 84.35% [IQR: 81.2–86.7], p = 0.001) and higher percent of both intermediate and non-classical cells, yet without statistical significance (median 6.54% [IQR: 5.14–16.64] vs. 5.87% [IQR: 4.48–8.6], p = 0.37 and median 5.99% [IQR: 3.39–11.5] vs. 5.26% [IQR: 3.62–6.2], p = 0.42, respectively). In AMI patients both, analyzed plasma miRNA concentrations were higher than in healthy subjects (miR-146: median 5.48 [IQR: 2.4–11.27] vs. 1.84 [IQR: 0.87–2.53], p = 0.003; miR-155: median 25.35 [IQR: 8.17–43.15] vs. 8.4 [IQR: 0.08–16.9], p = 0.027, respectively), and returned back to the values found in the control group in follow-up. miR-155/miR-146 ratio correlated with the frequencies of classical monocytes (r=0.6, p = 0.01) and miR-155 correlated positively with the concentration of inflammatory cytokines − IL-6 and TNF-α.
Conclusions
These results may suggest cooperation of both pro-inflammatory and anti-inflammatory signals in AMI in order to promote appropriate healing of the infarcted myocardium.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AMI:
-
acute myocardial infarction
- CAD:
-
coronary artery disease
- CCL2:
-
chemokine (C-C motif) ligand 2
- CCR2:
-
C-C chemokine receptor type 2
- ESC:
-
European Society of Cardiology
- HSC:
-
hematopoietic stem cell
- IL:
-
interleukin
- Inpp5d:
-
Inositol Polyphosphate-5-Phosphatase D
- IRAK1:
-
interleukin-1 receptor-associated kinase 1
- LVEF:
-
left ventricle ejection fraction
- LPS:
-
lipopolysaccharide
- miRNA:
-
micro RNA
- pPCI:
-
primary percutaneous coronary intervention
- ROS:
-
reactive oxygen species
- Socs1:
-
Suppressor of cytokine signaling 1
- TNF-α:
-
tumour necrosis factor α
- TRAF6:
-
TNF receptor associated factor 6
- WMSI:
-
wall-motion score index
References
Christa P., Frangogiannis NG. Targetting inflammatory pathways in myocardial infarction. Eur J Clin Invest 2013;43:986–95.
Karpiński L, Płaksej R, Kosmala W, Witkowska M. Serum levels of interleukin-6, interleukin-10 and C-reactive protein in relation to left ventricular function in patients with myocardial infarction treated with primary angioplasty. Kardiol Pol 2008;66:1279–85.
Kehmeier ES, Lepper W, Kropp M, Heiss C, Hendgen-Cotta U, Balzer J, et al. TNF-α, myocardial perfusion and function in patients with ST-segment elevation myocardial infarction and primary percutaneous coronary intervention. Clin Res Cardiol 2012;101(10):815–27.
Dutta P, Nahrendorf M. Monocytes in myocardial infarction. Arterioscler Thromb Vasc Biol 2015;35:1066–70.
Swirski FK, Nahrendorf M, Etzrodt M, Wildgruber M, Cortez-Retamozo V, Panizzi P, et al. Identification of splenic Reservoir monocytes and their deployment to inflammatory sites. Science 2009;325:612–6.
Frangogiannis NG. Regulation of the inflammatory response in cardiac repair. Circ Res 2012;110:159–73.
van Amerongen MJ, Harmsen MC, van Rooijen N, Petersen AH, van Luyn MJ. Macrophage depletion impairs wound healing and increases left ventricular remodeling after myocardial injury in mice. Am J Pathol 2007;170:818–29.
Frantz S, Nahrendorf M. Cardiac macrophages and their role in ischaemic heart disease. Cardiovasc Res 2014;102:240–8.
Ziegler-Heitbrock L, Ancuta P, Crowe S, Dalod M, Grau V, Hart DN, et al. Nomenclature of monocytes and dendritic cells in blood. Blood 2010;116:e74–80.
Wong KL, Tai JJ, Wong WC, Han H, Sem X, Yeap WH, et al. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood 2011;118:e16–31.
Cros J, Cagnard N, Woollard K, Patey N, Zhang SY, Senechal B, et al. Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity 2010;33:375–86.
Zawada AM, Rogacev KS, Rotter B, Winter P, Marell RR, Fliser D, et al. SuperSAGE evidence for CD14++CD16+ monocytes as a third monocyte subset. Blood 2011;118:e50–61.
Skrzeczynska-Moncznik J, Bzowska M, Loseke S, Grage-Griebenow E, Zembala M, Pryjma J. Peripheral blood CD14high CD16 + monocytes are main producers of IL-10. Scand J Immunol 2008;67:152–9.
Shantsila E, Wrigley B, Tapp L, Apostolakis S, Montoro-Garcia S, Drayson MT, et al. Immunophenotypic characterization of human monocyte subsets: possible implications for cardiovascular disease pathophysiology. J Thromb Haemost 2011;9:1056–66.
Venneri MA, De Palma M, Ponzoni M, Pucci F, Scielzo C, Zonari E, et al. Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood 2007;109:5276–85.
Mukherjee R, Barman PK, Thatoi PK, Tripathy R, Das BK, Ravindrana B. Non-classical monocytes display inflammatory features: validation in sepsis and systemic lupus erythematous. Sci Rep 2015;5:13886.
Eljaszewicz A, Jankowski M, Gackowska L, Helmin-Basa A, Wiese M, Kubiszewska I, et al. Gastric cancer increase the percentage of intermediate (CD14++CD16+) and nonclassical (CD14+CD16+) monocytes. Cent Eur J Immunol 2012;37:355–61.
Nahrendorf M, Swirski FK, Aikawa E, Stangenberg L, Wurdinger T, Figueiredo JL, et al. The healing myocardium sequentially mobilizes two monocyte subsets with divergent and complementary functions. J Exp Med. 2007;204:3037–47.
Tsujioka H, Imanishi T, Ikejima H, Kuroi A, Takarada S, Tanimoto T, et al. Impact of heterogeneity of human peripheral blood monocyte subsets on myocardial salvage in patients with primary acute myocardial infarction. J Am Coll Cardiol. 2009;54(2):130–8.
Tapp LD, Shantsila E, Wrigley BJ, Pamukcu B, Lip GY. The CD14++CD16+ monocyte subset and monocyte-platelet interactions in patients with ST-elevation myocardial infarction. J Thromb Haemost 2012;10:1231–41.
Zeng S, Yan LF, Luo YW, Liu XL, Liu JX, Guo ZZ, et al. Trajectories of circulating monocyte subsets after ST-elevation myocardial infarction during hospitalization: latent class growth modeling for High-risk patient identification. J Cardiovasc Transl Res 2018;11(1):22–32.
Nazari-Jahantigh M, Wei Y, Noels H, Akhtar S, Zhou Z, Koenen RR, et al. MicroRNA-155 promotes atherosclerosis by repressing Bcl6 in macrophages. J Clin Invest 2012;122:4190–202.
Huang RS, Hu GQ, Lin B, Lin ZY, Sun CC. MicroRNA-155 silencing enhances inflammatory response and lipid uptake in oxidized low-density lipoprotein-stimulated human THP-1 macrophages. J Investig Med 2010;58:961–7.
Deddens JC, Colijn JM, Oerlemans MI, Pasterkamp G, Chamuleau SA, Doevendans PA, et al. Circulating microRNAs as novel biomarkers for the early diagnosis of acute coronary syndrome. J Cardiovasc Transl Res 2013;6:884–98.
Chen X, Zhang L, Su T, Li H, Huang Q, Wu D, et al. Kinetics of plasma microRNA-499 expression in acute myocardial infarction. J Thorac Dis 2015;7(5):890–6.
Zhang L, Chen X, Su T, Li H, Huang Q, Wu D, et al. Circulating miR-499 are novel and sensitive biomarker of acute myocardial infarction. J Thorac Dis 2015;7(3):303–8.
Diehl P, Fricke A, Sander L, Stamm J, Bassler N, Htun N, et al. Microparticles: major transport vehicles for distinct microRNAs in circulation. Cardiovasc Res 2012;93:633–44.
Tian FJ, An LN, Wang GK, Zhu QJ, Li Q, Zhang YY, et al. Elevated microRNA-155 promotes foam cell formation by targeting HBP1 in atherogenesis. Cardiovasc Res 2014;103:100–10.
Saba R, Sorensen DL, Booth SA. MicroRNA-146a: a dominant, negative regulator of the innate immune response. Front Immunol 2014;5:578.
Eigsti RL, Sudan B, Wilson ME, Graff JW. Regulation of activation-associated microRNA accumulation rates during monocyte-to-macrophage differentiation. J Biol Chem 2014;289:28433–47.
Zhou J, Chaudhry H, Zhong Y, Ali MM, Perkins LA, Owens WB, et al. Dysregulation in microRNA expression in peripheral blood mononuclear cells of sepsis patients is associated with immunopathology. Cytokine 2014;71:89–100.
Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD. The writing group on behalf of the joint ESC/ACCF/AHA/WHF task force for the universal definition of myocardial infarction. Third universal definition of myocardial infarction. Eur Heart J 2012;33:2551–67.
Kinney EL, Wright RJ. Echocardiographic score versus wall motion index for risk stratification after acute myocardial infarction. Angiology 1990;41:112–7.
Frangogiannis NG. The mechanistic basis of infarct healing. Antioxid Redox Signal 2006;8:1907–39.
Maekawa Y, Anzai T, Yoshikawa T, Asakura Y, Takahashi T, Ishikawa S, et al. Prognostic significance of peripheral monocytosis after reperfused acute myocardial infarction: a possible role for left ventricular remodeling. J Am Coll Cardiol 2002;39:241–6.
Takahashi T, Hiasa Y, Ohara Y, Miyazaki S, Ogura R, Suzuki N, et al. Relationship of admission neutrophil count to microvascular injury, left ventricular dilation, and long-term outcome in patients treated with primary angioplasty for acute myocardial infarction. Circ J 2008;72:867–72.
Dutta P, Nahrendorf M. Monocytes in myocardial infarction. Arterioscler Thromb Vasc Biol 2015;35:01066–70.
Zhou X, Liu XL, Ji WJ, Liu JX, Guo ZZ, Ren D, et al. The kinetics of circulating monocyte subsets and monocyte-platelet aggregates in the acute phase of ST-elevation myocardial infarction: associations with 2-year cardiovascular events. Medicine (Baltimore) 2016;95(18):e3466.
Costantini A, Viola N, Berretta A, Galeazzi R, Matacchione G, Sabbatinelli J, et al. Age-related M1/M2 phenotype changes in circulating monocytes from healthy/unhealthy individuals. Aging (Albany NY) 2018;10(6):1268–80.
Heeschen C, Dimmeler S, Hamm CW, Fichtlscherer S, Boersma E, Simoons ML, et al. Serum level of the anti-inflammatory cytokine interleukin-10 is an important prognostic determinant in patients with acute coronary syndromes. Circulation 2003;107:2109–14.
Huang M, Yang D, Xiang M, Wang J. Role of interleukin-6 in regulation of immune responses to remodeling after myocardial infarction. Heart Fail Rev 2015;20:25–38.
Ritschel VN, Seljeflot I, Arnesen H, Halvorsen S, Weiss T, Eritsland J, et al. IL-6 signalling in patients with acute ST-elevation myocardial infarction. Results Immunol 2013;4:8–13.
Mantovani A, Garlanda C, Locati M. Macrophage diversity and polarization in atherosclerosis: a question of balance. Arterioscler Thromb Vasc Biol 2009;29:1419–23.
Mostafa Mtairag E, Chollet-Martin S, Oudghiri M, Laquay N, Jacob MP, Michel JB, et al. Effects of interleukin-10 on monocyte/endothelial cell adhesion and MMP-9/TIMP-1 secretion. Cardiovasc Res 2011;49:882–90.
Lacraz S, Nicod LP, Chicheportiche R, Welgus HG, Dayer JM. IL-10 inhibits metalloproteinase and stimulates TIMP-1 production in human mononuclear phagocytes. J Clin Invest 1995;96:2304–10.
Pasqui AL, Di Renzo M, Maffei S, Pastorelli M, Pompella G, Auteri A, et al. Pro/Anti-inflammatory cytokine imbalance in postischemic left ventricular remodeling. Mediators Inflamm 2010;2010:974694.
Oemrawsingh RM, Lenderink T, Akkerhuis KM, Heeschen C, Baldus S, Fichtlscherer S, et al. Multimarker risk model containing troponin-T, interleukin 10, myeloperoxidase and placental growth factor predicts long-term cardiovascular risk after non-ST-segment elevation acute coronary syndrome. Heart 2011;97:1061–6.
Jetten N, Verbruggen S, Gijbels MJ, Post MJ, De Winther MP, Donners MM. Anti-inflammatory M2, but not pro-inflammatory M1 macro- phages promote angiogenesis in vivo. Angiogenesis 2014;17:109–18.
Lee CG, Homer RJ, Zhu Z, Lanone S, Wang X, Koteliansky V, et al. Interleukin-13 induces tissue fibrosis by selectively stimulating and activating transforming growth factor beta(1). J Exp Med 2001;194:809–21.
Sierra-Filardi E, Vega MA, Sanchez-Mateos P, Corbi AL, Puig-Kroger A. Heme oxygenase-1 expression in M-CSF-polarized M2 macrophages contributes to LPS-induced IL-10 release. Immunobiology 2010;215:788–95.
Pokorney SD, Rodriguez JF, Ortiz JT, Lee DC, Bonow RO, Wu E. Infarct healing is a dynamic process following acute myocardial infarction. J Cardiovasc Magn Reson 2012;2:62.
Siwik DA, Chang DL, Colucci WS. Interleukin-1beta and tumor necrosis factor-alpha decrease collagen synthesis and increase matrix metalloproteinase activity in cardiac fibroblasts in vitro. Circ Res 2000;86:1259–65.
Mitchell MD, Laird RE, Brown RD, Long CS. IL-1beta stimulates rat cardiac fibroblast migration via MAP kinase pathways. Am J Physiol Heart Circ Physiol 2007;292:H1139–1147.
Frank J, Born K, Barker JH, Marzi I. In vivo effect of tumor necrosis factor alpha on wound angiogenesis and epithelialization. Eur J Trauma 2003;29:208–19.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kazimierczyk, E., Eljaszewicz, A., Zembko, P. et al. The relationships among monocyte subsets, miRNAs and inflammatory cytokines in patients with acute myocardial infarction. Pharmacol. Rep 71, 73–81 (2019). https://doi.org/10.1016/j.pharep.2018.09.007
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.09.007