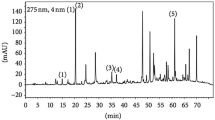
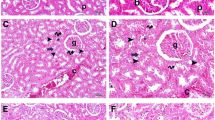
Abstract
Background
Aspalathin (Aspt) and nothofagin (Not) were reported to have antioxidant activity and are the two major active dihydrochalcones in green rooibos. This study was conducted to determine whether Asp and Not can modulate renal functional damage in a mouse model of sepsis and to elucidate the underlying mechanisms.
Methods
The potential of Aspt and Not treatment to reduce renal damage induced by cecal ligation and puncture (CLP) surgery in mice was measured by assessing blood urea nitrogen (BUN), serum creatinine, total urine protein, levels of lactate dehydrogenase (LDH), nitric oxide (NO), tumour necrosis factor (TNF)-α, interleukin (IL)-6, and myeloperoxidase (MPO), lipid peroxidation, total glutathione, glutathione peroxidase activity, catalase activity, and superoxide dismutase activity.
Results
Treatment with Aspt and Not decreased plasma levels of BUN, creatinine, urine protein, and LDH in mice with CLP-induced renal damage. Moreover, Aspt and Not inhibited nuclear factor (NF)-κB activation and reduced the induction of NO synthase and excessive production of nitric acid. Aspt and Not treatment also reduced the plasma levels of NO, TNF-α, IL-6, and MPO and reduced lethality due to CLP-induced sepsis, increased lipid peroxidation, and markedly enhanced the antioxidant defence system by restoring the levels of superoxide dismutase, glutathione peroxidase, and catalase in the kidney tissues.
Conclusion
Our results suggest that Aspt and Not protect mice against sepsis-triggered renal injury.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Russell J.A.. Management of sepsis. N Engl J Med 2006;355:1699–713.
Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016;315:801–10.
Chaudhry H, Zhou J, Zhong Y, Ali MM, McGuire F, Nagarkatti PS, et al. Role of cytokines as a double-edged sword in sepsis. In Vivo 2013;27:669–84.
Parratt JR. Nitric oxide in sepsis and endotoxaemia. J Antimicrob Chemother 1998;41(Suppl. A):31–9.
Symeonides S, Balk RA. Nitric oxide in the pathogenesis of sepsis. Infect Dis Clin North Am 1999;13:449–63 x.
Draisma A, Dorresteijn MJ, Bouw MP, van der Hoeven JG, Pickkers P. The role of cytokines and inducible nitric oxide synthase in endotoxemia-induced endothelial dysfunction. J Cardiovasc Pharmacol 2010;55:595–600.
Vincent JL, Zhang H, Szabo C, Preiser JC. Effects of nitric oxide in septic shock. Am J Respir Crit Care Med 2000;161:1781–5.
Cadenas S, Cadenas AM. Fighting the stranger-antioxidant protection against endotoxin toxicity. Toxicology 2002;180:45–63.
Prior RL, Cao G. Antioxidant capacity and polyphenolic components of teas: implications for altering in vivo antioxidant status. Proc Soc Exp Biol Med 1999;220:255–61.
Warren CP. Antioxidant effects of herbs. Lancet 1999;353:676.
McKay DL, Blumberg JB. A review of the bioactivity of South African herbal teas: rooibos (Aspalathus linearis) and honeybush (Cyclopia intermedia). Phytother Res 2007;21:1–16.
Kazuno S, Yanagida M, Shindo N, Murayama K. Mass spectrometric identification and quantification of glycosyl flavonoids, including dihydrochalcones with neutral loss scan mode. Anal Biochem 2005;347:182–92.
Jung B, Kang H, Lee W, Noh HJ, Kim YS, Han MS, et al. Anti-septic effects of dabrafenib on HMGB1-mediated inflammatory responses. BMB Rep 2016;49:214–9.
Wang H, Liao H, Ochani M, Justiniani M, Lin X, Yang L, et al. Cholinergic agonists inhibit HMGB1 release and improve survival in experimental sepsis. Nat Med 2004;10:1216–21.
Miranda KM, Espey MG, Wink DA. A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 2001;5:62–71.
Beutler E, Duron O, Kelly BM. Improved method for the determination of blood glutathione. J Lab Clin Med 1963;61:882–8.
Kim J, Bae JS. ROS homeostasis and metabolism: a critical liaison for cancer therapy. Exp Mol Med 2016;48:e269.
Mackman N, Brand K, Edgington TS. Lipopolysaccharide-mediated transcriptional activation of the human tissue factor gene in THP-1 monocytic cells requires both activator protein 1 and nuclear factor kappa B binding sites. J Exp Med 1991;174:1517–26.
Cohen J, Guyatt G, Bernard GR, Calandra T, Cook D, Elbourne D, et al. New strategies for clinical trials in patients with sepsis and septic shock. Crit Care Med 2001;29:880–6.
Fink MP, Heard SO. Laboratory models of sepsis and septic shock. J Surg Res 1990;49:186–96.
Nemzek JA, Hugunin KM, Opp MR. Modeling sepsis in the laboratory: merging sound science with animal well-being. Comp Med 2008;58:120–8.
Buras JA, Holzmann B, Sitkovsky M. Animal models of sepsis: setting the stage. Nat Rev Drug Discov 2005;4:854–65.
Deitch EA. Animal models of sepsis and shock: a review and lessons learned. Shock 1998;9:1–11.
Garrido AG, Poli de Figueiredo LF, Rocha e Silva M. Experimental models of sepsis and septic shock: an overview. Acta Cir Bras 2004;19:82–8.
Remick DG, Newcomb DE, Bolgos GL, Call DR. Comparison of the mortality and inflammatory response of two models of sepsis: lipopolysaccharide vs. cecal ligation and puncture. Shock 2000;13:110–6.
Rittirsch D, Hoesel LM, Ward PA. The disconnect between animal models of sepsis and human sepsis. J Leukoc Biol 2007;81:137–43.
Deitch EA. Rodent models of intra-abdominal infection. Shock 2005;24(Suppl. 1):19–23.
Stearns-Kurosawa DJ, Osuchowski MF, Valentine C, Kurosawa S, Remick DG. The pathogenesis of sepsis. Annu Rev Pathol 2011;6:19–48.
Huber-Lang M, Sarma VJ, Lu KT, McGuire SR, Padgaonkar VA, Guo RF, et al. Role of C5a in multiorgan failure during sepsis. J Immunol 2001;166:1193–9.
Bhargava R, Altmann CJ, Andres-Hernando A, Webb RG, Okamura K, Yang Y, et al. Acute lung injury and acute kidney injury are established by four hours in experimental sepsis and are improved with pre, but not post, sepsis administration of TNF-alpha antibodies. PLoS One 2013;8:e7903.
Guo RF, Ward PA. C5a, a therapeutic target in sepsis. Recent Pat Antiinfect Drug Discov 2006;1:57–65.
Oeckinghaus A, Ghosh S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol 2009;1:a000034.
Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O. Oxidative stress and antioxidant defense. World Allergy Organ J 2012;5:9–19.
Horton JW. Free radicals and lipid peroxidation mediated injury in burn trauma: the role of antioxidant therapy. Toxicology 2003;189:75–88.
Hatwalne MS. Free radical scavengers in anaesthesiology and critical care. Indian J Anaesth 2012;56:227–33.
Qiao Y, Bai XF, Du YG. Chitosan oligosaccharides protect mice from LPS challenge by attenuation of inflammation and oxidative stress. Int Immunopharmacol 2011;11:121–7.
Zhang J, Yu Y, Zhang Z, Ding Y, Dai X, Li Y. Effect of polysaccharide from cultured Cordyceps sinensis on immune function and anti-oxidation activity of mice exposed to 60Co. Int Immunopharmacol 2011;11:2251–7.
Xiao JH, Xiao DM, Chen DX, Xiao Y, Liang ZQ, Zhong JJ. Polysaccharides from the medicinal mushroom cordyceps taii show antioxidant and immunoenhancing activities in a D-galactose-induced aging mouse model. Evid-Based Compl Alt. 2012.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, S., Lee, C., Lee, BS. et al. Renal protective effects of aspalathin and nothofagin from rooibos (Aspalathus linearis) in a mouse model of sepsis. Pharmacol. Rep 70, 1195–1201 (2018). https://doi.org/10.1016/j.pharep.2018.07.004
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2018.07.004