Abstract
Background
A non-invasive phenotypic indicator of the rate of nicotine metabolism is nicotine metabolite ratio (NMR) defined as a ratio of two major metabolites of nicotine — trans-3′-hydroxycotinine/cotinine. The rate of nicotine metabolism has important clinical implications for the likelihood of successful quitting with nicotine replacement therapy (NRT). We conducted a study to measure NMR among Polish smokers.
Methods
In a cross-sectional study of 180 daily cigarette smokers (42% men; average age 34.6 ± 13.0), we collected spot urine samples and measured trans-3′-hydroxycotinine (3-HC) and cotinine levels with LC–MS/MS method. We calculated NMR (molar ratio) and analyzed variations in NMR among groups of smokers.
Results
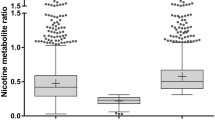
In the whole study group, an average NMR was 4.8 (IQR 3.4–7.3). The group of women below 51 years had significantly greater NMR compared to the rest of the population (6.4; IQR 4.1–8.8 vs. 4.3; IQR 2.8–6.4). No differences were found among group ages of male smokers.
Conclusions
This is a first study to describe variations in nicotine metabolism among Polish smokers. Our findings indicate that young women metabolize nicotine faster than the rest of population. This finding is consistent with the known effects of estrogen to induce CYP2A6 activity. Young women may require higher doses of NRT or non-nicotine medications for most effective smoking cessation treatment.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Benowitz N.L., Hukkanen J, Jacob 3rd P. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb Exp Pharmacol 2009;29–60.
Lerman C, Tyndale R, Patterson F, Wileyto EP, Shields PG, Pinto A, et al. Nicotine metabolite ratio predicts efficacy of transdermal nicotine for smoking cessation. Clin Pharmacol Ther 2006;79:600–8.
Malaiyandi V, Lerman C, Benowitz NL, Jepson C, Patterson F, Tyndale RF. Impact of CYP2A6 genotype on pretreatment smoking behaviour and nicotine levels from and usage of nicotine replacement therapy. Mol Psychiatry 2006;11:400–9.
Gu DF, Hinks LJ, Morton NE, Day IN. The use of long PCR to confirm three common alleles at the CYP2A6 locus and the relationship between genotype and smoking habit. Ann Hum Genet 2000;64:383–90.
Schnoll RA, Wileyto EP, Leone FT, Tyndale RF, Benowitz NL. High dose transdermal nicotine for fast metabolizers of nicotine: a proof of concept placebo-controlled trial. Nicotine Tob Res 2013;15:348–54.
Strasser AA, Benowitz NL, Pinto AG, Tang KZ, Hecht SS, Carmella SG, et al. Nicotine metabolite ratio predicts smoking topography and carcinogen biomarker level. Cancer Epidemiol Biomarkers Prev 2011;20:234–8.
Hukkanen J, Jacob 3rd P, Benowitz NL. Metabolism and disposition kinetics of nicotine. Pharmacol Rev 2005;57:79–115.
Dempsey D, Tutka P, Jacob 3rd P, Allen F, Schoedel K, Tyndale RF, et al. Nicotine metabolite ratio as an index of cytochrome P450 2A6 metabolic activity. Clin Pharmacol Ther 2004;76:64–72.
Jacob 3rd P, Yu L, Duan M, Ramos L, Yturralde O, Benowitz NL. Determination of the nicotine metabolites cotinine and trans-3′-hydroxycotinine in biologic fluids of smokers and non-smokers using liquid chromatography-tandem mass spectrometry: biomarkers for tobacco smoke exposure and for phenotyping cytochrome P450 2A6 activity. J Chromatogr B Analyt Technol Biomed Life Sci 2011;879:267–76.
Swan GE, Lessov-Schlaggar CN, Bergen AW, He Y, Tyndale RF, Benowitz NL. Genetic and environmental influences on the ratio of 3′hydroxycotinine to cotinine in plasma and urine. Pharmacogenet Genom 2009;19:388–98.
Schoedel KA, Hoffmann EB, Rao Y, Sellers EM, Tyndale RF. Ethnic variation in CYP2A6 and association of genetically slow nicotine metabolism and smoking in adult Caucasians. Pharmacogenetics 2004;14:615–26.
Benowitz NL, Swan SG, Lessov CN, Jacob 3rd. P. Oral contraceptives induce CYP2A6 activity and accelerate nicotine metabolism. Clin Pharmacol Ther 2004;75:36.
Zeman MV, Hiraki L, Sellers EM. Gender differences in tobacco smoking: higher relative exposure to smoke than nicotine in women. J Womens Health Gend Based Med 2002;11:147–53.
Dempsey D, Jacob 3rd P, Benowitz NL. Accelerated metabolism of nicotine and cotinine in pregnant smokers. J Pharmacol Exp Ther 2002;301:594 59-8.
Benowitz NL, Lessov-Schlaggar CN, Swan GE, Jacob P. 3rd: female sex and oral contraceptive use accelerate nicotine metabolism. Clin Pharmacol Ther 2006;79:480–8.
Research on the menopause. World Health Organ Tech Rep Ser 1981;670:1–120.
Kaczmarek M. The timing of natural menopause in Poland and associated factors. Maturitas 2007;57:139–53.
Goniewicz ML, Eisner MD, Lazcano-Ponce E, Zielinska-Danch W, Koszowski B, Sobczak A, et al. Comparison of urine cotinine and the tobacco-specific nitrosamine metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) and their ratio to discriminate active from passive smoking. Nicotine Tob Res 2011;13:202–8.
Benowitz NL. Nicotine addiction. N Engl J Med 2010;362(24):2295–303.
Lea RA, Dickson S, Benowitz NL. Within-subject variation of the salivary 3HC/COT ratio in regular daily smokers: prospects for estimating CYP2A6 enzyme activity in large-scale surveys of nicotine metabolic rate. J Anal Toxicol 2006;30:386–9.
Malaiyandi V, Goodz SD, Sellers EM, Tyndale RF. CYP2A6 genotype, phenotype, and the use of nicotine metabolites as biomarkers during ad libitum smoking. Cancer Epidemiol Biomarkers Prev 2006;15:1812–9.
Johnstone E, Benowitz N, Cargill A, Jacob R, Hinks L, Day I, et al. Determinants of the rate of nicotine metabolism and effects on smoking behavior. Clin Pharmacol Ther 2006;80:319–30.
Smith P, Weinberger A, Zhang J, Emme E, Mazure C, McKee S. Sex differences in smoking cessation pharmacotherapy comparative efficacy: a network meta-analysis. Nicotine Tob Res 2017;19:273–81.
Perkins KA, Karelitz JL. Sex differences in acute relief of abstinence-induced withdrawal and negative affect due to nicotine content in cigarettes. Nicotine Tob Res 2015;17(4):443–8.
Perkins KA, Gerlach D, Vender J, Grobe J, Meeker J, Hutchison S. Sex differences in the subjective and reinforcing effects of visual and olfactory cigarette smoke stimuli. Nicotine Tob Res 2001;3(2):141–50.
Perkins KA, Donny E, Caggiula AR. Sex differences in nicotine effects and self-administration: review of human and animal evidence. Nicotine Tob Res 1999;1(4):301–15.
Berlin I, Gasior MJ, Moolchan ET. Sex-based and hormonal contraception effects on the metabolism of nicotine among adolescent tobacco-dependent smokers. Nicotine Tob Res 2007;9:493–8.
Rubinstein ML, Shiffman S, Rait MA, Benowitz NL. Race, gender, and nicotine metabolism in adolescent smokers. Nicotine Tob Res 2013;15:1311–5.
Nakajima M, Yokoi T. Interindividual variability in nicotine metabolism: c-oxidation and glucuronidation. Drug Metab Pharmacokinet 2005;20:227–35.
Chen G, Giambrone NE, Lazarus P. Glucuronidation of trans-3′-hydroxycotinine by UGT2B17 and UGT2B10. Pharmacogenet Genom 2012;22(3):183–90.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kosmider, L., Delijewski, M., Koszowski, B. et al. Slower nicotine metabolism among postmenopausal Polish smokers. Pharmacol. Rep 70, 434–438 (2018). https://doi.org/10.1016/j.pharep.2017.11.009
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2017.11.009