Abstract
Background
Epigenetic histone modifications are considered as a promising avenue for cancer preventive and therapeutic strategies. The purpose of this study was to evaluate the antiproliferative and histone deacetylase (HDAC) inhibitory activity of selected peanut phenolics, including p-coumaric acid, ferulic acid, sinapinic acid and resveratrol, in MCF-7 and HeLa cells.
Methods
The cytotoxic and HDAC inhibitory activities were assessed by MTT assays, flow cytometric analyses of cell cycle arrest and apoptosis induction, and western blotting.
Results
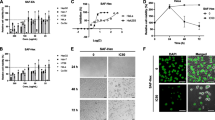
The results showed that all four phenolics inhibited proliferation of both MCF-7 and HeLa cells in a dose-dependent manner. Among the phenolics tested, resveratrol was the most effective in inhibiting growth of cancer cells. Treatment with all phenolics resulted in histone H3 hyperacetylation in both cell lines, indicating potential for HDAC inhibition. These phenolics induced apoptosis in both MCF-7 and HeLa cells in a concentration-dependent manner. Moreover, all phenolics induced G0/G1-phase arrest of the cell cycle in MCF-7 cells while p-coumaric and ferulic acids caused S-phase arrest in HeLa cells. Exposure to p-coumaric acid increased p53 and p21 expression but decreased CDK4 levels in both cell types, which could result in the observed G0/G1 arrest. Moreover, inhibition of ERK1/2 phosphorylation by ferulic acid and resveratrol contributed to cell growth inhibition.
Conclusion
Peanut phenolics appear to influence the extent of histone acetylation in MCF-7 and HeLa cells, and this activity modulates multiple pathways that are implicated in cancer prevention.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ATM:
-
ataxia telangiectasia mutated
- CDK:
-
cyclin-dependent kinase
- DMSO:
-
dimethyl sulfoxide
- HAT:
-
histone acetyltransferase
- HDAC:
-
histone deacetylase
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- NaB:
-
sodium butyrate
- PI:
-
propidium iodide
- PVDF:
-
polyvinylidene fluoride
- RNase A:
-
ribonuclease A
References
Hieda M, Matsuura N, Kimura H. Histone modifications associated with cancer cell migration and invasion. Methods Mol Biol 2015;1238:301–17.
Ropero S, Esteller M. The role of histone deacetylases (HDACs) in human cancer. Mol Oncol 2007;1(1):19–25.
Roberts CW, Leroux MM, Fleming MD, Orkin SH. Highy penetrant, rapid tumorigenesis through conditional inversion of the tumor suppressor gene Snf5. Cancer Cell 2002;2:415–25.
Robert C, Rassool FV. HDAC inhibitors: roles of DNA damage and repair. Adv Cancer Res 2012;116:87–129.
Luo J, Su F, Chen D, Shiloh A, Gu W. Deacetylation of p53 modulates its effect on cell growth and apoptosis. Nature 2000;408(6810):377–81.
Sarfstein R, Bruchim I, Fishman A, Werner H. The mechanism of action of the histone deacetylase inhibitor vorinostat involves interaction with the insulinlike growth factor signaling pathway. PLoS One 2011;6(9):e24468.
Marks PA, Richon VM, Rifkind RA. Histone deacetylase inhibitors: inducers of differentiation or apoptosis of transformed cells. J Natl Cancer Inst 2000;92(15):1210–6.
Rajendran P, Ho E, Williams DE, Dashwood RH. Dietary phytochemicals HDAC inhibition, and DNA damage/repair defects in cancer cells. Clin Epigenet 2011;3(1):4.
Kastan MB, Lim DS. The many substrates and functions of ATM. Nat Rev Mol Cell Biol 2000;1(3):179–86.
Brew CT, Aronchik I, Hsu JC, Sheen JH, Dickson RB, Bjeldanes LF, et al. Indole-3-carbinol activates the ATM signaling pathway independent of DNA damage to stabilize p53 and induce G1 arrest of human mammary epithelial cells. Int J Cancer 2006;118(4):857–68.
Lewandowska U, Gorlach S, Owczarek K, Hrabec E, Szewczyk K. Synergistic interactions between anticancer chemotherapeutics and phenolic compounds and anticancer synergy between polyphenols. Postepy Hig Med Dosw (Online) 2014;68:528–40.
Zhang HM, Zhao L, Li H, Xu H, Chen WW, Tao L. Research progress on the anticarcinogenic actions and mechanisms of ellagic acid. Cancer Biol Med 2014;11(2):92–100.
Nandi S, Vracko M, Bagchi MC. Anticancer activity of selected phenolic compounds: QSAR studies using ridge regression and neural networks. Chem Biol Drug Des 2007;70(5):424–36.
Jemal A, Center MM, DeSantis C, Ward EM. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiol Biomarkers Prev 2010;19(8):1893–907.
Jemal A, Simard EP, Dorell C, Noone AM, Markowitz LE, Kohler B, et al. Annual Report to the Nation on the Status of Cancer: 1975–2009, featuring the burden and trends in human papillomavirus (HPV)-associated cancers and HPV vaccination coverage levels. J Natl Cancer Inst 2013;105(3):175–201.
Khaopha S, Jogloy S, Patanothai A, Senawong T. Histone deacetylase inhibitory activity of peanut testa extracts against human cancer cell lines. J Food Biochem 2015;39(3):263–73.
Sobolev VS, Cole RJ. trans-resveratrol content in commercial peanuts and peanut products. J Agric Food Chem 1999;47(4):1435–9.
Khaopha S, Senawong T, Jogloy S, Patanothai A. Comparison of total phenolic content and composition of individual phenolic acids in testae and testaremoved kernels of 15 Valencia-type peanut (Arachis hypogaea L.) genotypes. Afr J Biotechnol 2012;11(92):15923–30.
Vanden Berghe W. Epigenetic impact of dietary polyphenols in cancer chemoprevention: lifelong remodeling of our epigenomes. Pharmacol Res 2012;65(6):565–76.
Conley BA, Wright JJ, Kummar S. Targeting epigenetic abnormalities with histone deacetylase inhibitors. Cancer 2006;107(4):832–40.
Bellucci L, Dalvai M, Kocanova S, Moutahir F, Bystricky K. Activation of p21 by HDAC inhibitors requires acetylation of H2A.Z. PLoS One 2013;8(1):e54102.
Gui CY, Ngo L, Xu WS, Richon VM, Marks PA. Histone deacetylase (HDAC) inhibitor activation of p21WAF1 involves changes in promoter- associated proteins, including HDAC1. Proc Natl Acad Sci U S A 2004;101(15):1241–6.
Eot-Houllier G, Fulcrand G, Magnaghi-Jaulin L, Jaulin C. Histone deacetylase inhibitors and genomic instability. Cancer Lett 2009;274(2):169–76.
Rogakou EP, Pilch DR, Orr AH, Ivanova VS. Bonner WM: DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 1998;273(10):5858–68.
Jang ER, Choi JD, Park MA, Jeong G, Cho H, Lee JS. ATM modulates transcription in response to histone deacetylase inhibition as part of its DNA damage response. Exp Mol Med 2010;42(3):195–204.
Jafary H, Ahmadian S, Soleimani M. The enhanced apoptosis and antiproliferative response to combined treatment with valproate and nicotinamide in MCF-7 breast cancer cells. Tumor Biol 2014;35:2701–10.
Han BR, You BR, Park WH. Valproic acid inhibits the growth of HeLa cervical cancer cells via caspase-dependent apoptosis. Oncol Rep 2013;30:2999–3005.
Pal-Bhadra M, Ramaiah MJ, Reddy TL, Krishnan A, Pushpavalli SN, Babu KS, et al. Plant HDAC inhibitor chrysin arrest cell growth and induce p21WAF1 by altering chromatin of STAT response element in A375 cells. BMC Cancer 2012;12:180.
Ahn MY, Jung JH, Na YJ, Kim HS. A natural histone deacetylase inhibitor, Psammaplin A, induces cell cycle arrest and apoptosis in human endometrial cancer cells. Gynecol Oncol 2008;108(1):27–33.
Fan X, Liu Y, Chen JJ. Down-regulation of p21 contributes to apoptosis induced by HPV E6 in human mammary epithelial cells. Apoptosis 2005;10(1):63–73.
Dhandapani KM, Mahesh VB, Brann DW. Curcumin suppresses growth and chemoresistance of human glioblastoma cells via AP-1 and NFkappaB transcription factors. J Neurochem 2007;102(2):522–38.
Skommer J, Brittain T, Raychaudhuri S. Bcl-2 inhibits apoptosis by increasing the time-to-death and intrinsic cell-to-cell variations in the mitochondrial pathway of cell death. Apoptosis 2010;15(10):1223–33.
Li L, Dai HJ, Ye M, Wang SL, Xiao XJ, Zheng J, et al. Lycorine induces cell-cycle arrest in the G0/G1 phase in K562 cells via HDAC inhibition. Cancer Cell Int 2012;12(1):49.
Agrawal S, Agarwal ML, Chatterjee-Kishore M, Stark GR. Chisolm GM. Stat1-dependent, p53-independent expression of p21(waf1) modulates oxysterol-induced apoptosis. Mol Cell Biol 2002;22:1981–92.
Joe AK, Liu H, Suzui M, Vural ME, Xiao D, Weinstein IB. Resveratrol induces growth inhibition, S-phase arrest, apoptosis, and changes in biomarker expression in several human cancer cell lines. Clin Cancer Res 2002;8:893–903.
Roy M, Chakraborty S, Siddiqi M, Bhattacharya RK. Induction of Apoptosis in Tumor Cells by Natural Phenolic Compounds. Asian Pac J Cancer Prev 2002;3:61–7.
Lakshminarasimhan M, Rauh D, Schutkowski M, Steegborn C. Sirt1 activation by resveratrol is substrate sequence-selective. AGING 2013;5(3):151–4.
Lu Z, Xu S. ERK1/2 MAP kinases in cell survival and apoptosis. IUBMB Life 2006;58(11):621–31.
Tan BJ, Chiu GN. Role of oxidative stress: endoplasmic reticulum stress and ERK activation in triptolide-induced apoptosis. Int J Oncol 2013;42(5):1605–12.
Tang D, Wu D, Hirao A, Lahti JM, Liu L, Mazza B, et al. ERK activation mediates cell cycle arrest and apoptosis after DNA damage independently of p53. J Biol Chem 2002;277(15):12710–7.
Polier G, Neumann J, Thuaud F, Ribeiro N, Gelhaus C, Schmidt H, et al. The natural anticancer compounds rocaglamides inhibit the Raf-MEK-ERK pathway by targeting prohibitin 1 and 2. Chem Biol 2012;19(19):1093–1104.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saenglee, S., Jogloy, S., Patanothai, A. et al. Cytotoxic effects of peanut phenolics possessing histone deacetylase inhibitory activity in breast and cervical cancer cell lines. Pharmacol. Rep 68, 1102–1110 (2016). https://doi.org/10.1016/j.pharep.2016.06.017
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2016.06.017