Abstract
Background
In the vascular system, ATP-sensitive K+-channels are a target for H2 S. Recent evidence suggests that H2 S may also modulate Na+ — and Ca2+-permeable channels and intracellular Ca2+ stores, but the influence of H2S on endothelial Ca2+ dynamics and Ca2+-dependent activation of endothelial nitric oxide synthase (eNOS) is unclear. In this study, we investigated the effects of H2 S on Ca2+ signaling in endothelial and smooth muscle cells with special emphasis given to the role of H2 S in modulating endothelial NO formation.
Methods
Experiments were performed with endothelial cells from porcine aorta, the human endothelial cell line HMEC-1, and smooth muscle cells from rat aorta and trachea. Mobilization of intracellular Ca2+ and Ca2+ entry was monitored with Fura-2. Activity of eNOS was determined as conversion of incorporated l-[3H]arginine into l-[3H]citrulline.
Results
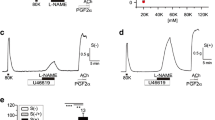
Incubation of endothelial cells with the H2 S donors sodium hydrogen sulfide (NaHS) and GYY4137 blocked activation of eNOS by the receptor agonist ATP but not by the Ca2+ ionophore A23187. Data revealed that H2 S inhibited ATP-induced release of Ca2+ from intracellular stores indicating that H2 S attenuates eNOS activity by blocking capacitative Ca2+ entry. A similar inhibitory effect of H2 S on ATP-induced Ca2+ release and Ca2+ entry was also observed in human microvascular endothelial cells and smooth muscle cells.
Conclusions
H2 S antagonized Ca2+ mobilization by receptor agonists and store-operated Ca2+ entry thereby limiting eNOS activation and NO formation. The effect of H2 S on Ca2+ stores was not restricted to endothelial cells but was also observed in vascular and tracheal smooth muscle cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- eNOSk:
-
endothelial nitric oxide synthase
- IP3:
-
inositol (1,4,5)-trisphosphate
- H2S:
-
hydrogen sulfide
- HUVECs:
-
human umbilical vein endothelial cells
- NaHS:
-
sodium hydrogen sulfide
- PAECs:
-
porcine aortic endothelial cells
- SMCs:
-
smooth muscle cells
References
Yang G, Wu L, Jiang B, Yang W, Qi J, Cao K, et al. H2S as a physiologic vasorelaxant: hypertension in mice with deletion of cystathionine gammalyase. Science 2008;322:587–90.
Li L, Rose P, Moore PK. Hydrogen sulfide and cell signaling. Annu Rev Pharmacol Toxicol 2011;51:169–87.
Wang R. Physiological implications of hydrogen sulfide: a whiff exploration that blossomed. Physiol Rev 2012;92:791–896.
Vandiver M, Snyder SH. Hydrogen sulfide: a gasotransmitter of clinical relevance. J Mol Med (Berl) 2012;90:255–63.
Liu YH, Lu M, Hu FL, Wong PT, Webb GD, Bian JS. Hydrogen sulfide in the mammalian vascular system. Antioxid Redox Signal 2012;17:141–85.
Cheng Y, Ndisang JF, Tang G, Cao K, Wang R. Hydrogen sulfide-induced relaxation of resistance mesenteric artery beds of rats. Am J Physiol 2004;287:H2316–23.
Zhao W, Zhang J, Lu Y, Wang R. The vasorelaxant effect of H2S as a novel endogenous gaseous K(ATP) channel opener. EMBO J 2001;20:6008–16.
Zhao W, Wang R. H2S-induced vasorelaxation and underlying cellular and molecular mechanisms. Am J Physiol Heart Circ Physiol 2002;283:H474–80.
Kiss L, Deitch EA, Szabo C. Hydrogen sulfide decreases adenosine triphosphate levels in aortic rings and leads to vasorelaxation via metabolic inhibition. Life Sci 2008;83:589–94.
Cheang WS, Wong WT, Shen B, Lau CW, Tian XY, Tsang SY, et al. 4-Amino-pyridine-sensitive K+ channels contributes to NaHS-induced membrane hyperpolarization and relaxation in the rat coronary artery. Vascul Pharmacol 2010;53:94–8.
Coletta C, Papapetropoulos A, Erdelyi K, Olah G, Modis K, Panopoulos P, et al. Hydrogen sulfide and nitric oxide are mutually dependent in the regulation of angiogenesis and endothelium-dependent relaxation. Proc Natl Acad Sci USA 2012;109:9161–6.
Altaany Z, Yang G, Wang R. Crosstalk between hydrogen sulfide and nitric oxide in endothelial cells. J Cell Mol Med 2013;17:879–88.
Predmore BL, Julian D, Cardounel AJ. Hydrogen sulfide increases nitric oxide production from endothelial cells by an Akt-dependent mechanism. Front Physiol 2011;2:104.
Kida M, Sugiyama T, Yoshimoto T, Ogawa Y. Hydrogen sulfide increases nitric oxide production with calcium-dependent activation of endothelial nitric oxide synthase in endothelial cells. Eur J Pharm Sci 2013;48:211–5.
Geng B, Cui Y, Zhao J, Yu F, Zhu Y, Xu G, et al. Hydrogen sulfide downregulates the aortic l-arginine/nitric oxide pathway in rats. Am J Physiol Regul Integr Comp Physiol 2007;293:R1608–18.
Kubo S, Kurokawa Y, Doe I, Masuko T, Sekiguchi F, Kawabata A. Hydrogen sulfide inhibits activity of three isoforms of recombinant nitric oxide synthase. Toxicology 2007;241:92–7.
Kubo S, Doe I, Kurokawa Y, Nishikawa H, Kawabata A. Direct inhibition of endothelial nitric oxide synthase by hydrogen sulfide: contribution to dual modulation of vascular tension. Toxicology 2007;232:138–46.
Bauer CC, Boyle JP, Porter KE, Peers C. Modulation of Ca2+ signalling in human vascular endothelial cells by hydrogen sulfide. Atherosclerosis 2010;209: 374–80.
Moccia F, Bertoni G, Pla AF, Dragoni S, Pupo E, Merlino A, et al. Hydrogen sulfide regulates intracellular Ca2+ concentration in endothelial cells from excised rat aorta. Curr Pharm Biotechnol 2011;12:1416–26.
Pupo E, Pla AF, Avanzato D, Moccia F, Cruz JE, Tanzi F, et al. Hydrogen sulfide promotes calcium signals and migration in tumor-derived endothelial cells. Free Radic Biol Med 2011;51:1765–73.
Wagner S, Groschner K, Mayer B, Schmidt K. Desensitization of endothelial nitric oxide synthase by receptor agonists. Biochem J 2002;364:863–8.
Lin S, Fagan KA, Li KX, Shaul PW, Cooper DM, Rodman DM. Sustained endothelial nitric-oxide synthase activation requires capacitative Ca2+ entry. J Biol Chem 2000;275:17979–85.
Iouzalen L, Lantoine F, Pernollet MG, Millanvoye-Van Brussel E, Devynk MA, David-Dufilho M. SK&F inhibits intracellular Ca2+ pumps and raises cytosolic Ca2+ concentration without production of nitric oxide and von Willebrand factor. Cell Calcium 1996;20:501–8.
Klatt P, Pfeiffer S, List BM, Lehner D, Glatter O, Bächinger HP, et al. Characterization of heme-deficient neuronal nitric-oxide synthase reveals a role for heme in subunit dimerization and binding of the amino acid substrate and tetrahydrobiopterin. J Biol Chem 1996;271:7336–42.
Leber A, Hemmens B, Klösch B, Goessler W, Raber G, Mayer B, et al. Characterization of recombinant human endothelial nitric-oxide synthase purified from the yeast Pichia pastoris. J Biol Chem 1999;274:37658–64.
Ades EW, Candal FJ, Swerlick RA, George VG, Summers S, Bosse DC, et al. HMEC: establishment of an immortalized human microvascular endothelial cell line. J Invest Dermatol 1992;99:683–90.
Schmidt K, Werner-Felmayer G, Mayer B, Werner ER. Preferential inhibition of inducible nitric oxide synthase in intact cells by the 4-amino analogue of tetrahydrobiopterin. Eur J Biochem 1999;259:25–31.
Mayer B, Klatt P, Werner ER, Schmidt K. Molecular mechanisms of inhibition of porcine brain nitric oxide synthase by the antinociceptive drug 7-nitroindazole. Neuropharmacology 1994;33:1254–9.
Grynkiewicz G, Poenie M, Tsien RY. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 1985;260:3440–50.
Gorren AC, Schrammel A, Schmidt K, Mayer B. Thiols and neuronal nitric oxide synthase: complex formation, competitive inhibition, and enzyme stablilization. Biochemistry 1997;36:4360–6.
Teubl M, Groschner K, Kohlwein SD, Mayer B, Schmidt K. Na+/Ca+ exchange facilitates Ca2+-dependent activation of endothelial nitric-oxide synthase. J Biol Chem 1999;41:29529–35.
Castro-Piedras I, Perez-Zoghbi JF. Hydrogen sulphide inhibits Ca2+ release through InsP3 receptors and relaxes airway smooth muscle. J Physiol 2013;591:5999–6015.
Alderton WK, Cooper CE, Knowles RG. Nitric oxide synthases: structure, function and inhibition. Biochem J 2001;357:593–615.
Parekh AB, Putney JW. Store-operated calcium channels. Physiol Rev 2005;85:757–810.
Putney JW, Phospholipase TT. C signaling and calcium influx. Adv Biol Regul 2012;52:152–64.
Munaron L, Avanzato D, Moccia F, Mancardi D. Hydrogen sulfide as a regulator of calcium channels. Cell Calcium 2013;53:77–84.
Chen Y, Zhao J, Du J, Xu G, Tang C, Geng B. Hydrogen sulfide regulates cardium sarcoplasmic reticulum Ca(2+) uptake via K(ATP) channel and PI3K/Akt pathway. Life Sci 2012;91:271–8.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Kloesch, B., Steiner, G., Mayer, B. et al. Hydrogen sulfide inhibits endothelial nitric oxide formation and receptor ligand-mediated Ca2+ release in endothelial and smooth muscle cells. Pharmacol. Rep 68, 37–43 (2016). https://doi.org/10.1016/j.pharep.2015.05.026
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.pharep.2015.05.026