Abstract
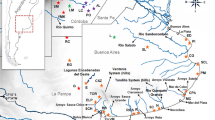
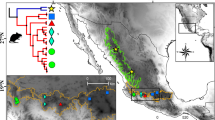
Eligmodontia is a genus of phyllotine rodents adapted to arid environments with seven recognized species. The sister species E. puerulus and E. moreni are distributed in the adjacent highland Puna and lowland Monte deserts respectively, and show remarkable morphological and chromosomal differences. However, analyses of the cytochrome b gene showed important variability, without reciprocal monophyly between them. In order to study the evolutionary processes involved in the diversification of both taxa, we analyzed 1161 bp of the mitochondrial control region and flanking sequences (N = 60), as well as 759 bp of the first exon of the nuclear gene IRBP (N = 14). Individuals of both species from Jujuy, Catamarca and Mendoza Provinces of Argentina were previously karyotyped. Results showed that the mitochondrial sequences present high haplotype and nucleotide diversity within all population, and no haplotype was shared between both species. FST indicated that populations of both species were moderately structured. The network was constituted by two major haplogroups, one composed by E. puerulus samples from Jujuy, and the other composed of sequences of all studied populations. The Bayesian analysis showed three clusters, matching the network. Phylogenetic analysis recovered two clades with high support, in coincidence with the network groups. There was only one close join between sequences of both species, corresponding to samples from Catamarca. Thus, mitochondrial data suggested hybridization between both species in Catamarca, with asymmetric introgression. The IRBP showed low variability and, in the phylogenetic analysis, the sequences of E. puerulus form a monophyletic group with intermediate support, whereas those of E. moreni collapse into a basal polytomy. Our data indicated a recent divergence and absence of introgression in the nuclear genomes. The results at the population level with mitochondrial sequences, together with integrative taxonomy at the species level in a biogeographic context, suggest that climatic and geologic changes could have had an important role in the determination of genetic variability patterns observed in these rodents.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abramson, N.I., Rodchenkova, E.N., Kostygov, A.Yu., 2009. Genetic variation and phylogeography of the bank vole (Clethrionomys glareolus, Arvicolinae, Rodentia) in Russia with special reference to the introgression of the mtDNA of a closely related species, red-backed vole (Cl. rutilus). Russ. J. Genet. 45, 533–545.
Allmendinger, R.W., Jordan, T.E., Kay, S.M., Isacks, B.L., 1997. The evolution of the Altiplano-Puna plateau of the Central Andes. Annu. Rev. Earth Planet. Sci. 25, 139–174.
Baker, R.J., Bradley, R.D., 2006. Speciation in mammals and the genetic species concept. J. Mammal. 87, 643–662.
Bandelt, H.J., Forster, P., Röhl, A., 1999. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 16, 37–48.
Beysard, M., Heckel, G., 2014. Structure and dynamics of hybrid zones at different stages of speciation in the common vole (Microtus arvalis). Mol. Ecol. 23, 673–687.
Braun, J.K., 1993. Systematic Relationships of the Tribe Phyllotini (Muridae: Sigmodontinae) of South American. Spec. Publ., Norman: Oklahoma Mus. Nat. Hist.
Corander, J., Marttinen, P., Mäntyniemi, S., 2006. Bayesian identification of stock mixtures from molecular markerdata. Fish. Bull. 104, 550–558.
Cornuet, J.M., Pudlo, P., Veyssier, J., Dehne-Garcia, A., Gautier, M., Leblois, R., Marin, J.M., Estoup, A., 2014. DIYABC v2.0: a software to make Approximate Bayesian Computation inferences about population history using Single Nucleotide Polymorphism, DNA sequence and microsatellite data. Bioinformatics 30, 1187–1189.
Coyne, JA, Orr, H.A., 2004. Speciation. Sinauer Associates, Inc. Pub., Sunderland, Massachusetts USA.
Coyner, B.S., Murphy, P.J., Matocq, M.D., 2015. Hybridization and asymmetric introgression across a narrow zone of contact between Neotoma fuscipes and N. macrotis (Rodentia: Cricetidae). Biol. J. Linn. Soc. 115, 162–172.
Díaz, G.B., Ojeda, R.A., 1999. Kidney structure of Argentine desert rodents. J. Arid Environ. 41, 453–461.
Elder, F.F., 1980. Tandem fusion, centric fusion, and chromosomal evolution in the cotton rats, genus Sigmodon. Cytogenet. Cell Genet. 26, 199–210.
Excoffier, L, Lischer, H.E.L., 2010. Arlequin suite ver3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Res. 10, 564–567.
Excoffier, L, Smouse, P., Quattro, J., 1992. Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131, 479–491.
Funk, D.J., Omland, K.E., 2003. Species-level paraphyly and polyphyly frequency, causes, and consequences, with insights from animal mitochondrial DNA. Annu. Rev. Evol. Syst. 34, 397–423.
Goloboff, P., Catalano, S., 2016. TNT, version 1.5, with a full implementation of phylogenetic morphometrics. Cladistics 32, 221–238.
González-Ittig, R.E., Theiler, G.R., Gardenal, C.N., 2002. A contribution to the subgeneric systematics of Oligoryzomys (Rodentia, Muridae) from Argentina by means ofPCR-RFLP patterns of mitochondrial DNA. Biochem. Syst. Ecol. 30, 23–33.
Hershkovitz, P., 1962. Evolution of Neotropical Cricetine Rodents (Muridae) with special reference to the Phyllotine group. Fieldiana Zool., 46.
Jansa, S.A., Voss, R.S., 2000. Phylogenetic studies on Didelphid marsupials I. Introduction and preliminary results from nuclear IRBPgene sequences.J. Mamm. Evol. 7, 43–77.
King, M., 1993. Species Evolution. The Role of Chromosome Change. Cambridge University Press, Cambridge, UK.
Lanzone, C, Ojeda, R.A., Gallardo, M.H., 2007. Integrative taxonomy, systematics and distribution of the genus Eligmodontia (Rodentia, Cricetidae, Sigmodontinae) in the temperate Monte Desert of Argentina. Z. Säugetierk. 72, 299–312.
Lanzone, C, Ojeda, A.A., Ojeda, R.A., Albanese, S., Rodríguez, D., Dacar, M.A., 2011. Integrated analyses of chromosome, molecular and morphological variability in the andean mice Eligmodontia puerulus and E. moreni (Rodentia, Cricetidae, Sigmodontinae). Mamm. Biol. 76, 555–562.
Lanzone, C, Suárez, S.N., Rodríguez, D., Ojeda, A., Albanese, S., Ojeda, R.A., 2014. Chromosomal variability and morphological notes in Graomys griseoflavus (Rodentia, Cricetidae, Sigmodontinae), from Catamarca and Mendoza provinces, Argentina. Mastozool. Neotrop. 21, 47–58.
Lanzone, C, Cardozo, D., Sánchez, D.M., Martí, D.A., Ojeda, R.A., 2016. Chromosomal variability and evolution in the tribe Phyllotini (Rodentia, Cricetidae, Sigmodontinae). Mamm. Res. 61, 373–382.
Librado, P., Rozas, J., 2009. DnaSP v5: a software forcomprehensive analysis of DNA polymorphism data. Bioinformatics 25, 1451–1452.
Maddison, W.P., 1997. Gene trees in species trees. Syst. Biol. 46, 523–536.
Mares, M.A., Braun, J.K., Coyner, B.S., Van Den Bussche, R.A., 2008. Phylogenetic and biogeographic relationships of gerbil mice Eligmodontia (Rodentia, Cricetidae) in South America, with a description of a new specie. Zootaxa 1753, 1–33.
Martínez, J.J., González-Ittig, R.E., Theiler, G.R., Ojeda, R.A., Lanzone, C, Ojeda, A., Gardenal, C.N., 2010. Patterns of speciation intwo sibling species of Graomys (Rodentia, Cricetidae) based on mtDNA sequences.J. Zool. Syst. Evol. Res. 48, 159–166.
Melnikova, E.N., Kshnyasev, I.A., Bodrov, S.Yu., Mukhacheva, S.V., Davydova, YuA, Abramson, N.I., 2012. Sympatric area of Myodes glareolus andM. rutilus (Rodentia, Cricetidae): historic and recent hybridization. Proc. Zool. Inst. RAS 316, 307–323.
Melo-Ferreira, J., Boursot, P., Carneiro, M., Esteves, P.J., Farelo, L., Alves, P.C., 2012. Recurrent introgression of mitochondrial DNA among hares (Lepus spp.) revealed by species-tree inference and coalescent simulations. Syst. Biol. 61, 367–381.
Ortiz-Jaureguizar, E., Cladera, G.A., 2006. Paleoenvironmental evolution of souther South America during Cenozoic. J. Arid. Environ. 66, 489–532.
Parada, A., Pardiñas, U.F.J., Salazar-Bravo, J., D’Elía, G., Palma, R.E., 2013. Dating an impressive Neotropical radiation: molecular time estimates for the Sigmodontinae (Rodentia) provide insights into its historical biogeography. Mol. Phylogenet. Evol. 66, 960–968.
Parinas, U.F.J., Lessa, G., Teta, P., Salazar-Bravo, J., Câmara, E.M.V.C., 2014. A new genus of sigmodontine rodent from eastern Brazil and the origin of the tribe Phyllotini. J. Mammal. 95, 201–215.
Patton, J.L., Smith, M.F., 1994. Paraphyly, polyphyly and the nature of species boundaries in pocket gophers (Genus Thomomys). Syst. Biol. 43, 11–26.
Patton, J.L., Pardiñas, U.F.J., DíElía, G., 2015. Mammals of South America. Rodents, vol. 2. University of Chicago Press, Chicago.
Pearson, O., Martin, S., Bellati, J., 1987. Demography and reproduction of the silky desert mouse (Eligmodontia) in Argentina. Fieldiana 39, 433–446.
Spotorno, A.E., Sufan-Catalan, J., Walker, L.I., 1994. Cytogenetic diversity and evolution of Andean species of Eligmodontia (Rodentia, Muridae). Z. Säugetierkd. 59, 299–308.
Spotorno, A.E., Walker, L.I., Flores, S.V., Yevenes, M., Marín, J.C., Zuleta, C, 2001. Evolución de los filotinos (Rodentia, Muridae) en los Andes del Sur. Rev. Chil. His. Nat. 74, 151–166.
Spotorno, A.E., Zuleta, C.R., Walker, L.I., Manriquez, G.S., Valladares, P.F., Marin, J.C., 2013. A small, new gerbil-mouse Eligmodontia (Rodentia: Cricetidae) from dunes at the coasts and deserts of north-central Chile: molecular, chromosomic, and morphological analyses. Zootaxa 3683, 377–394.
Steppan, S.J., Ramirez, O., Banbury,J., Huchon, D., Pacheco, V., Walker, L.I., Spotorno, A.E., 2007. A molecular reappraisal of the systematics of the leaf-eared mice Phyllotis and their relatives. In: Kelt, D.A., Lessa, E.P., Salazar-Bravo, J.A., Patton, J.L. (Eds.), The Quintessential Naturalist: Honoringthe Life and Legacy of Oliver P. Pearson. Univ. California Pub. Zool., USA, pp. 799-826 (134).
Steppan, S.J., 1995. Revision of the Tribe Phyllotini (Rodentia: Sigmodontinae), with a phylogenetic hypothesis forthe Sigmodontinae. Fieldiana Zool. 80, 1–112.
Tamura, K., Stecher, G., Peterson, D., Filipski, A., Kumar, S., 2013. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 30, 2725–2729.
Taylor, P.J., 2000. Patterns of chromosomal variation in southern African rodents. J. Mammal. 81, 317–331.
Theiler, G.R., Gardenal, C.N., Blanco, A., 1999. Patterns of evolution in Graomys griseoflavus (Rodentia, Muridae) IV. A case of rapid speciation. J. Evol. Biol. 12, 970–979.
Wright, S., 1978. Evolution and the genetics of populations. Variability Within and Among Natural Populations, vol. 4. University of Chicago press, Chicago.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sierra, A.B.A., Castillo, E.R., Labaroni, C. et al. Genetic studies in the recently divergent Eligmodontia puerulus and E. moreni (Rodentia, Cricetidae, Sigmodontinae) from Puna and Monte deserts of South America. Mamm Biol 87, 93–100 (2017). https://doi.org/10.1016/j.mambio.2017.06.001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.mambio.2017.06.001