Abstract
Background
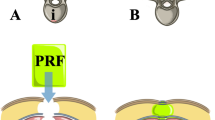
Decorticated bone is a significant source of blood loss in scoliosis surgery. Current hemostatic methods include packed gauze (GS), physical barriers such as bone wax, and xenograft collagen-based materials. We assessed the safety and efficacy of a novel fibrin dressing (dextran-thrombin-fibrinogen [DTF]) compared to GS. This dressing comprises lyophilized thrombin and fibrinogen embedded in an elastic electrospun nanofiber dextran matrix.
Purpose
The study tests the hypothesis that DTF is more efficacious than GS in control of bleeding from cancellous bone.
Study Design
A preclinical Good Laboratory Practices (GLP) study.
Methods
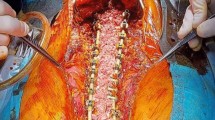
We enrolled 10 goats that were followed for 28 ± 1 days. Each animal was randomly assigned to the test or control group. Both test and control animals had 4 cancellous bone injuries. Test animal injuries were treated with DTF, whereas standard GS was used to control bleeding in the control animals. Bleeding at the bone injury site was characterized as either none, oozing, flowing, or pulsatile and was assessed at 4 and 8 minutes after dressing application. Goats were survived 28 ± 1 days and then necropsied.
Results
Application of the fibrin dressing to bleeding cancellous bone, both posterior spinal lamina, and iliac crest graft sites, resulted in control of bleeding within 4 minutes at all injury sites. Eighty percent of control injury sites continued to bleed after 8 minutes and required application of bone wax to control bleeding. There were no differences in prothrombin time, partial thromboplastin time, or fibrinogen levels between test and control animals at 1 or 28 days. We observed no adverse histologic reactions at 28 days.
Conclusion
The fibrin dressing is an efficacious and safe method of controlling blood loss from cancellous bone in a spine surgery model.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
McNeill TW, DeWald RL, Kuo KN, et al. Controlled hypotensive anesthesia in scoliosis surgery. J Bone Joint Surg Am 1974;56:1167–72.
Cheriyan T, Maier SP, Bianco K, et al. Efficacy of tranexamic acid on surgical bleeding in spine surgery: a meta-analysis. Spine J 2015;15:752–61.
Abbott EM, Nandyala SV, Schwend RM. Does a kaolin-impregnated hemostatic dressing reduce intraoperative blood loss and blood transfusions in pediatric spinal deformity surgery? Spine 2014;39:E1174–80.
Price JS, Tackett S, Patel V. Observational evaluation of outcomes and resource utilization from hemostatic matrices in spine surgery. J Med Econ 2015:1-10.
Wu J, Jin Y, Zhang J, et al. Hemostatic techniques following multilevel posterior lumbar spine surgery: a randomized control trial. J Spinal Disord Tech 2014;27:442–6.
Epstein NE. Tisseel does not reduce postoperative drainage, length of stay, and transfusion requirements for lumbar laminectomy with non-instrumented fusion versus laminectomy alone. Surg Neurol Int 2015;6:S172–6.
Colombo GL, Bettoni D, Di Matteo S, et al. Economic and outcomes consequences of Tachosil®: a systematic review. Vasc Health Risk Manage 2014;10:569–76.
Rothwell SW, Reid TJ, Dorsey J, et al. A salmon thrombin-fibrin bandage controls arterial bleeding in a swine aortotomy model. J Trauma 2005;59:143–9.
Floyd CT, Rothwell SW, Martin R, et al. A salmon thrombin-fibrinogen dressing controls hemorrhage in a swine model compared to standard kaolin-coated gauze. J Spec Oper Med 2012;12:49–55.
Floyd CT, Rothwell SW, Risdahl J, et al. N. Salmon thrombin-fibrinogen dressing allows greater survival and preserves distal blood flow compared to standard kaolin gauze in coagulopathic swine with a standardized lethal femoral artery injury. J Spec Oper Med 2012;12:16–26.
Padua RA, Olson CE, Rothwell RS, Floyd CT. Performance of a dextran-only dressing versus SurgiClot to achieve hemostasis in a swine injury model of cancellous bone bleeding. Paper presented at: Orthopaedic Research Society Annual Meeting 2016.
Bayne K. Revised Guide for the Care and Use of Laboratory Animals available. American Physiological Society. Physiologist 1996;39:199.208-11.
US Food and Drug Administration web site, http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRsearch.cfm?CFRPart=58. Accessed June 17, 2015.
Virzi R. Refining the test phase of usability evaluation: how many subjects is enough? Hum Factors 1992;34:457–68.
Rothwell SW, Sawyer E, Dorsey J, et al. Wound healing and the immune response in swine treated with a hemostatic bandage composed of salmon thrombin and fibrinogen. J Mater Sci Mater Med 2009;20:2155–66.
Rothwell SW, Settle T, Wallace S, et al. The long term immunological response of swine after two exposures to a salmon thrombin and fibrinogen hemostatic bandage. Biologicals 2010;38:619–28.
Padua RA, Olson CE, Floyd CT. Resorption characteristics of a novel fibrin hemostatic dressing. Paper presented at: Accepted, Orthopaedic Research Society Annual Meeting 2016.
Kheirabadi BS, Mace JE, Terrazas IB, et al. Clot-inducing minerals versus plasma protein dressing for topical treatment of external bleeding in the presence of coagulopathy. J Trauma 2010;69:1062–72.
Kim DH, Chun YS, Vaccaro AR, et al. Complications of iliac crest: bone graft harvesting. In: An HS, Jenis LG, editors. Complications of spine surgery: treatment and prevention. Philadelphia, PA: Lippincott Williams & Wilkins; 2006. p. 143–52.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author disclosures
CTF (other from St. Teresa Medical, Inc., during the conduct of the study; personal fees from St. Teresa Medical, Inc., outside the submitted work); RAP (other from St. Teresa Medical, Inc, outside the submitted work); CEO (other from St. Teresa Medical, Inc, outside the submitted work).
Rights and permissions
About this article
Cite this article
Floyd, C.T., Padua, R.A. & Olson, C.E. Hemostasis and Safety of a Novel Fibrin Dressing Versus Standard Gauze in Bleeding Cancellous Bone in a Caprine Spine Surgery Model. Spine Deform 5, 310–313 (2017). https://doi.org/10.1016/j.jspd.2017.03.003
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.jspd.2017.03.003