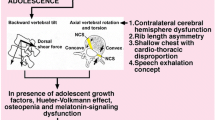
Abstract
Purpose
There is great controversy about the etiologic origin of adolescent idiopathic scoliosis. Multiple theories have been suggested, including metabolic aspects, endocrine dysfunction, neurological central abnormalities, genetic predisposition and epigenetic factors involved in the development of scoliosis. However, there has always been speculations based on human biomechanical behavior.
Methods
In this article, we performed a literature review on the biomechanical traits of human posture, and the proposed theories that explain the special characteristics present in idiopathic scoliosis.
Results
The current theory on the etiopathogeneis of AIS suggests that dorsally directed shear loads acting on a preexisting axial plane rotation, in a posteriorly inclined sagittal plane of a growing patient, together with disc maturation, collagen quality at this phase of development and immaturity of proprioception, is the perfect scenario to spark rotational instability and create the three-dimensional deformity that defines idiopathic scoliosis.
Conclusion
The unique spinal alignment of human bipedalism, gravity and muscle forces acting straight above the pelvis to preserve an upright balance, and the instability of the soft tissue in a period of growth development, is an appealing cocktail to try to explain the genesis of this condition in humans.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although a lot of research has been done in the field [1], there is still no current agreed theory demonstrating the exact etiopathogenesis of adolescent idiopathic scoliosis (AIS). The term idiopathic cannot be erased, as this condition has yet to be proven to be linked to a pathological state. One of the main difficulties we encounter in this endeavor is that we are searching for answers analyzing an already established deformity. We have not been able yet to create an experimental model that truly resembles human scoliosis. One of the lines of thought imputes gravity acting in bipedalism as a potential driver for scoliosis. Thus, some authors have tried to convert quadrupeds into bipeds, mostly in chicken, rabbits and rats [2]. However, all required drastic surgical or systemic interventions, and they are far from representing the status of human bipedalism. Nevertheless, other non-bipedal species (fish for example) are also known to develop scoliosis (water stream resistance could the driving force in these cases) [3]. Thus, we do not have an insight on scoliosis origin at its early stages, it is impossible to understand its cause if we only have access to the end product, which is the resulting deformity.
Review of the proposed etiologies of AIS
Melatonin deficiency after pineal gland resections in chickens led to scoliosis development, and some authors have detected decreased serum levels of melatonin in patients with progressive deformities [4]. It seems that the absence of this hormone could interfere with skeletal growth and symmetrical development of their proprioceptive system [5]. On the other hand, some authors have failed to confirm this theory in primates (rhesus monkeys) [6] or unbalance on serum levels in AIS patients. So experiments in lower animal models are different from primates, and findings in lower animals cannot necessarily be extrapolated to human beings [7].
Other metabolic dysregulations could interfere with growth. Higher levels of Growth Hormone (GH) and somatomedin have been found in AIS patients, but this is based on small trials that are neither conclusive nor updated [8]. Calmodulin regulates contractile properties of muscles interacting with myosin and actin. Asymmetric distributions of calmodulin have been detected in the paraspinal muscles of AIS patients, and an increased concentration of calmodulin in the platelets of AIS was associated with curve progression [9, 10]. Low leptin levels have been spotted in these patients, and this protein contributes to body weight, body mass index and bone mineral density, eventually interacting with body growth. In fact, selective hypothalamic dysfunction with upregulation sensitivity to circulating leptin creates skeletal asymmetries [11, 12].
Abnormal bone mineralization in combination with increased bone growth during the growth spurt can disturb bone microarchitecture, affect mechanical bone strength, and contribute to the vicious circle of the deformity [13, 14]. Therefore, osteoporosis and lower levels of serum vitamin D have been associated with the development and progression of AIS [15].
All these metabolic hypotheses have been extensively reviewed, and the predictive values of all these parameters for curve progression are still limited and based in low levels of evidence [16].
The vestibulo-ocular-spinal axis is related to balance and proprioception, so central disorders may be incriminated in the development of AIS. Visuo-oculomotor dysfunction [17], hidden neurologic impairment [18], brain interhemispheric asymmetry function [19]; all of them can contribute to asymmetric muscle activation of the paraspinal muscles and are proposed as neuromuscular subclinical etiology of AIS. There are findings supporting the hypothesis that a sensorimotor integration disorder underlies the pathogenesis of idiopathic scoliosis [20]. Surface electromyographic analysis detected visible disturbances and asymmetries in the erector spinae frequency (activity and tension) correlating with curve side [21] and the convexity of the curve [22].
A review was conducted to assess how strong the current evidence is in favor of all the different anomalies associated with idiopathic scoliosis [23] and an overall weak evidence was found for any consistent pattern of co-occurrence of AIS and any abnormality.
Familial clustering and studies in twins have shown a genetic predisposition for scoliosis [24], multiple genes have been associated with idiopathic scoliosis [25, 26], the most important have been recently summarized in a review article [27]. However, the exact mode of inheritance is not yet established, partially due to the genetic heterogeneity of scoliosis [28]. The ScoliScore project was launched in 2010 as the first genetic DNA-based test for predicting AIS progression using an algorithm incorporating results from 53 single nucleotide variants (SNVs) and the Cobb angle of Caucasian US teens [29]. Unfortunately, several separate independent research teams were unable to reproduce these findings in subsequent Japanese [30], French Canadian [31] or Han Chinese populations [32], and the use of the test was progressively abandoned.
The development of efficient gene editing methods and high throughput sequencing technology is promising for future research. Zebrafish are increasingly being used as a model thanks to their high genetic homology with humans [3].
Recently epigenetics and specific biomarkers are been studied to clarify the etiology and potential progression of idiopathic scoliosis [33]. There is a hypothesis that supports epigenetic internal and external environmental factors that affect vertebral growth during childhood in a pre-existing heritable genetic variation [34].
Apart from the above-mentioned theories, biomechanical hypotheses have always been debated. In this paper, we will focus on the current biomechanical approach to scoliosis.
The biomechanical approach to scoliosis
Scoliosis is a 3D deformity of the human trunk. Classical anatomists like Adams 1864, Meyer 1866, or Nicoladoni 1904 were already aware of the 3D nature of scoliosis, and were able to describe the anatomical changes of discs and vertebrae in scoliotic specimens. These anatomic abnormalities have been further studied with modern technology (EOS low-dose radiograph system, magnetic resonance imaging, and computed tomography scan) [35,36,37,38] (Fig. 1). However, idiopathic scoliosis does not occur naturally in other mammals apart from humans [2], and strong forces need to be applied in experimental studies in order to biomechanically force spines to curve. The unique spinal alignment of human bipedalism, gravity and muscle forces acting to preserve an upright balance, and the instability of the soft tissue in a period of growth development, is an appealing cocktail to try to explain the genesis of this condition in humans.
Biomechanics of the upright human spine
Humans are unique in their spinal alignment. Apes became bipedal in great amount thanks to changes occurring in the pelvis. The pelvis became wider (pelvic incidence increased), more horizontal, and lordotic within the ischio-iliac angle relationship [39]. These facts led to the development of lumbar lordosis (to accommodate to the upright position) and to a change in the orientation of the hips allowing hip and knee extension (that enabled humans to walk efficiently with the minimal use of energy) [40, 41]. With this structure, the center of gravity lies straight above the pelvis instead of in front of the pelvis compared to other vertebrates, changing completely spinal biomechanics (Fig. 2).
All deformities of the skeleton are originated by a disturbance of the relation between the forces applied and the counteracting structures. In quadrupeds, the main forces of the spine (muscle and gravity wise) are in an axial compression as well as in a ventral direction, parallel to gravity, sustaining an equilibrium [42]. These anterior load vectors create stability in the vertebrae of the spine. The mentioned changes of the upright position made anterior shear loads diminish, directing the loads (gravity and muscle wise again) in vectors that act in opposite directions. Certain vertebrae of the spine are located in a place in space where anterior vectors are no longer existent. These forces are then replaced by dorsally directed shear loads that drive the vertebra away from the anterior gravity line (Fig. 3), leading to rotational instability of the exposed human vertebrae [43].
The unique human sagittal profile
The sagittal profile of the spine plays an important role in determining the rotational stability of certain areas of the spine. During growth, the spine moves from a complete kyphosis, at intra-uterine and early neonatal stage, to an S-shaped sagittal profile in adulthood creating kyphotic and lordotic segments [44]. The axis of rotation of normal dorsal vertebrae lies in front in the vertebral body, so in an anteriorly inclined environment, shear loads fall anteriorly, increasing vertebral stability against rotation [45] (Fig. 4A). However, if the vertebrae display a posterior inclination, the axis of rotation shifts posteriorly, and posterior shear loads further move the center of rotation closer to the facets, then the vertebrae become unstable against rotation [43] (Fig. 4B). For this reason, if a sector of the spine shows backward inclined segments, it becomes more vulnerable to fail with posterior forces, and posterior directed shear forces may lead to vertebral rotation and scoliosis (Fig. 4F).
The center of the image shows a preexisting axial plane rotation. A Segments of the spine with anterior load vectors, anterior (stable) axis of rotation. B Posterior load vectors shift the axis of rotation posteriorly (unstable). C Anterior spinal overgrowth seems to occur mostly around the apex and especially in the intervertebral discs. D Changes in the structure and properties of the disc during growth may impair its resistance to the different loading vectors especially rotation. E The acceleration phase of rapid growth coincides in females with a period in which the spine is more backwardly inclined. F Dorsally directed shear loads drive the vertebrae away from the anterior gravity line
Spine development during growth
Spine evolution differs between age and sexes [46]. Females in general mature earlier than males, and the acceleration phase of rapid growth coincides with a period in which the spine is more backwardly inclined [46, 47] (Fig. 4E). This posterior inclination (usually located in the lower thoracic and upper lumbar segments, Fig. 3) further shift spinal loads posteriorly, leaving that segment with less rotational stability (Fig. 5). This may explain why initiation and progression of AIS are more prevalent in girls around puberty than in boys, who are already in a more kyphotic and anteriorly inclined phase at that stage (in whom Scheuermann kyphosis is more predominant).
Spine evolution during growth also affects the development of the discs and their mechanical properties. The spine grows predominantly through the vertebral bodies. The disc matures by ossifying its insertion in the vertebra through the Sharpey fibers into the initially cartilaginous ring apophyses, eventually fusing itself to the vertebral body. Thoracic disc height increases initially and then the transverse surface area increases decreasing disc slenderness [48]. By these segmental attachments to the vertebral body, the disc represents the most important stabilizer of the spine, and in the apical segment of a deformity, the intervertebral disc seems to be an important contributory factor in curve progression [49]. Changes in the structure and properties of the disc during growth or during degeneration in adulthood may impair its resistance to the different loading vectors especially rotation [50] (Fig. 4D), disturbing the natural equilibrium and predisposing to alignment failures. Apparently, deformity could start in the disc area and metabolic development characteristics can impact soft tissue (disc and ligaments) and bone quality. Actually, idiopathic scoliosis initially increases through disc wedging during the rapid growth spurt with progressive vertebral wedging occurring later [51].
Rotational instability
Scoliosis demonstrate changes in all three planes in space: axial, coronal and sagittal [52, 53]. Up to now, it has been very difficult to elucidate which plane is the initiator of scoliosis. However, the axial plane seems to be special [54, 55]. No quadrupeds have shown spontaneous rotation of the axial skeleton, but humans do. Non-scoliotic spines show a spontaneous thoracic rotatory pattern to the right of small magnitude related to the asymmetrical distribution of the internal organs [56]. As we have seen, the human spine’s sagittal shape carrying the center of gravity straight above the pelvis makes it a rotationally unstable construct [42]. Thus, it appears that once the spine decompensates, it tends to follow that built-in rotational pattern that the spine exhibits at the time of onset [57] (Fig. 4).
One of the mechanical theories of scoliosis origin is the excess of the anterior length of the spine [58], producing a Relative Anterior Spinal Overgrowth (RASO). Anterior lengthening of the spine (where the spine is longer anteriorly than posteriorly) has been found in AIS patients [59]. This has been attributed to an asymmetrical growth between the anterior body endochondral ossification and the membranous ossification occurring in posterior structures [60]. However, there is some controversy in this topic, as some authors describe that scoliosis vertebrae have a wedged kyphotic morphology, and anterior lengthening seems to occur mostly around the apex and especially in the intervertebral disc (Fig. 4C), suggesting an adaptation to altered loading and not a primary growth disturbance [61]. Furthermore, anterior growth is not unique for idiopathic scoliosis, it also occurs in scoliosis with a known origin (neuromuscular, and congenital and traumatic compensatory curves), suggesting that it is part of the mechanism, rather than its cause.
Current biomechanical theory
Dickson [62] in the 80’s, with his theory of biplanar asymmetry [63], speculated that thoracic lordosis developed in children with a coronal plane asymmetry and a previously rotated spine in the transverse plane [57] triggered a spinning moment that further increased spinal rotation [58]. In his theory, a flattening of the kyphosis was essential.
Nowadays, Castelein proposes a theory [43] (Fig. 4) supporting that a preexisting axial plane rotation [64], together with a rotational instability probably linked to failure constrains in the immaturity period [57], most probably coming from immature intervertebral discs [50], couples with a posteriorly inclined sagittal plane [46] that shifts the axis of vertebral rotation posteriorly, displacing the loads from an anterior to a posterior position, and this dorsal directed shear loads initiate scoliosis deformity.
Once the three dimensional deformity starts, it progressively enters in the vicious cycle described by Stokes [65], both for the discs but also the bone. The progression of the deformity increases coronal vertebral body wedging as a result of asymmetric muscle activation and loading, eventually affecting vertebral body growth by disturbing the endplate physes.
We need, however, to remember that these biomechanical theories coexist with other mentioned hypothesis of AIS etiologies that include: genetics, epigenetics, and neurological, endocrine and metabolic dysregulations, which are also areas that merit further investigation.
Conclusion
In this article, we review the biomechanical traits of human posture, and the proposed theories that explain the special characteristics present in idiopathic scoliosis. The current biomechanical theory supports that dorsally directed shear loads acting on a preexisting axial plane rotation, in a posteriorly inclined sagittal plane of a growing patient, together with disc maturation, collagen quality at this phase of development and immaturity of proprioception, is the perfect scenario to spark rotational instability and create the three-dimensional deformity that defines idiopathic scoliosis.
Data availability
It is a literature review article, data shown here comes from the references compiled in the text. No patient raw data was used.
References
Wang WJ, Yeung HY, Chu WC-W et al (2011) Top theories for the etiopathogenesis of adolescent idiopathic scoliosis. J Pediatr Orthop 31:S14-27. https://doi.org/10.1097/BPO.0b013e3181f73c12
Janssen MMA, de Wilde RF, Kouwenhoven J-WM, Castelein RM (2011) Experimental animal models in scoliosis research: a review of the literature. Spine J 11:347–358. https://doi.org/10.1016/j.spinee.2011.03.010
Xie H, Li M, Kang Y et al (2022) Zebrafish: an important model for understanding scoliosis. Cell Mol Life Sci 79:506–516. https://doi.org/10.1007/s00018-022-04534-5
Machida M, Dubousset J, Yamada T, Kimura J (2009) Serum melatonin levels in adolescent idiopathic scoliosis prediction and prevention for curve progression–a prospective study. J Pineal Res 46:344–348. https://doi.org/10.1111/j.1600-079X.2009.00669.x
Machida M, Dubousset J, Imamura Y et al (1996) Melatonin. A possible role in pathogenesis of adolescent idiopathic scoliosis. Spine 21:1147–1152. https://doi.org/10.1097/00007632-199605150-00005
Cheung KMC, Wang T, Poon AMS et al (2005) The effect of pinealectomy on scoliosis development in young nonhuman primates. Spine 30:2009–2013. https://doi.org/10.1097/01.brs.0000179087.38730.5d
Gargano G, Oliva F, Migliorini F, Maffulli N (2022) Melatonin and adolescent idiopathic scoliosis: the present evidence. Surgeon 20:e315–e321. https://doi.org/10.1016/j.surge.2021.07.008
Willner S, Nilsson KO, Kastrup K, Bergstrand CG (1976) Growth hormone and somatomedin A in girls with adolescent idiopathic scoliosis. Acta Paediatr Scand 65:547–552. https://doi.org/10.1111/j.1651-2227.1976.tb04930.x
Acaroglu E, Akel I, Alanay A et al (2009) Comparison of the melatonin and calmodulin in paravertebral muscle and platelets of patients with or without adolescent idiopathic scoliosis. Spine 34:E659–E663. https://doi.org/10.1097/BRS.0b013e3181a3c7a2
Lowe TG, Burwell RG, Dangerfield PH (2004) Platelet calmodulin levels in adolescent idiopathic scoliosis (AIS): can they predict curve progression and severity? Summary of an electronic focus group debate of the IBSE. Eur Spine J 13:257–265. https://doi.org/10.1007/s00586-003-0655-3
Burwell RG, Dangerfield PH, Moulton A, Anderson SI (2008) Etiologic theories of idiopathic scoliosis: autonomic nervous system and the leptin-sympathetic nervous system concept for the pathogenesis of adolescent idiopathic scoliosis. Stud Health Technol Inform 140:197–207
Wang Q, Wang C, Hu W et al (2020) Disordered leptin and ghrelin bioactivity in adolescent idiopathic scoliosis (AIS): a systematic review and meta-analysis. J Orthop Surg Res 15:502. https://doi.org/10.1186/s13018-020-01988-w
Cheuk KY, Hu Y, Tam EMS et al (2019) Bone measurements at multiple skeletal sites in adolescent idiopathic scoliosis-an in vivo correlation study using DXA, HR-pQCT and QCT. Arch Osteoporos 14:70–10. https://doi.org/10.1007/s11657-019-0621-2
Yang KG, Lee WYW, Hung ALH et al (2022) Decreased cortical bone density and mechanical strength with associated elevated bone turnover markers at peri-pubertal peak height velocity: a cross-sectional and longitudinal cohort study of 396 girls with adolescent idiopathic scoliosis. Osteoporos Int 33:725–735. https://doi.org/10.1007/s00198-021-06200-1
Alsiddiky A, Alfadhil R, Al-Aqel M et al (2020) Assessment of serum vitamin D levels in surgical adolescent idiopathic scoliosis patients. BMC Pediatr 20:202–205. https://doi.org/10.1186/s12887-020-02114-9
Noshchenko A, Hoffecker L, Lindley EM et al (2015) Predictors of spine deformity progression in adolescent idiopathic scoliosis: A systematic review with meta-analysis. World J Orthop 6:537–558. https://doi.org/10.5312/wjo.v6.i7.537
Lion A, Haumont T, Gauchard GC et al (2013) Visuo-oculomotor deficiency at early-stage idiopathic scoliosis in adolescent girls. Spine 38:238–244. https://doi.org/10.1097/BRS.0b013e31826a3b05
Woods LA, Haller RJ, Hansen PD et al (1995) Decreased incidence of scoliosis in hearing-impaired children. Implications for a neurologic basis for idiopathic scoliosis. Spine 20:776–780. https://doi.org/10.1097/00007632-199504000-00006. (Discussion 781)
Doménech J, Tormos JM, Barrios C, Pascual-Leone A (2010) Motor cortical hyperexcitability in idiopathic scoliosis: could focal dystonia be a subclinical etiological factor? Eur Spine J 19(2):223–230. https://doi.org/10.1007/s00586-009-1243-y. (Epub 2009 Dec 24)
Domenech J, García-Martí G, Martí-Bonmatí L et al (2011) Abnormal activation of the motor cortical network in idiopathic scoliosis demonstrated by functional MRI. Eur Spine J 20:1069–1078. https://doi.org/10.1007/s00586-011-1776-8
Wilczyński J, Karolak P (2021) relationship between electromyographic frequency of the erector spinae and location, direction, and number of spinal curvatures in children with scoliotic changes. Risk Manag Healthc Policy 14:1881–1896. https://doi.org/10.2147/RMHP.S302360
Wilczyński J (2021) Relationship between muscle tone of the erector spinae and the concave and convex sides of spinal curvature in low-grade scoliosis among children. Children (Basel) 8:1168. https://doi.org/10.3390/children8121168
Schlösser TPC, van der Heijden GJMG, Versteeg AL, Castelein RM (2014) How “idiopathic” is adolescent idiopathic scoliosis? A systematic review on associated abnormalities. PLoS ONE 9:e97461. https://doi.org/10.1371/journal.pone.0097461
Andersen MO, Thomsen K, Kyvik KO (2007) Adolescent idiopathic scoliosis in twins: a population-based survey. Spine 32:927–930. https://doi.org/10.1097/01.brs.0000259865.08984.00
Zhang Z, Zhang Z, Shu L et al (2023) A genetic variant of the ROBO3 gene is associated with adolescent idiopathic Scoliosis in the Chinese population. Spine 48:E20–E24. https://doi.org/10.1097/BRS.0000000000004484
Liu B, Zhao S, Liu L et al (2022) Aberrant interaction between mutated ADAMTSL2 and LTBP4 is associated with adolescent idiopathic scoliosis. Gene 814:146126. https://doi.org/10.1016/j.gene.2021.146126
Marya S, Tambe AD, Millner PA, Tsirikos AI (2022) Adolescent idiopathic scoliosis : a review of aetiological theories of a multifactorial disease. Bone Joint J 104-B(8):915–921. https://doi.org/10.1302/0301-620X.104B8.BJJ-2021-1638.R1
Terhune EA, Monley AM, Cuevas MT et al (2022) Genetic animal modeling for idiopathic scoliosis research: history and considerations. Spine Deform 10:1003–1016. https://doi.org/10.1007/s43390-022-00488-7
Ward K, Ogilvie JW, Singleton MV et al (2010) Validation of DNA-based prognostic testing to predict spinal curve progression in adolescent idiopathic scoliosis. Spine 35:E1455–E1464. https://doi.org/10.1097/BRS.0b013e3181ed2de1
Ogura Y, Takahashi Y, Kou I et al (2013) A replication study for association of 53 single nucleotide polymorphisms in a scoliosis prognostic test with progression of adolescent idiopathic scoliosis in Japanese. Spine 38:1375–1379. https://doi.org/10.1097/BRS.0b013e3182947d21
Tang QL, Julien C, Eveleigh R et al (2015) A replication study for association of 53 single nucleotide polymorphisms in ScoliScore test with adolescent idiopathic scoliosis in French-Canadian population. Spine 40:537–543. https://doi.org/10.1097/BRS.0000000000000807
Xu L, Qin X, Sun W et al (2016) Replication of association Between 53 single-nucleotide polymorphisms in a DNA-based diagnostic test and AIS progression in Chinese Han population. Spine 41:306–310. https://doi.org/10.1097/BRS.0000000000001203
Pérez-Machado G, Berenguer-Pascual E, Bovea-Marco M et al (2020) From genetics to epigenetics to unravel the etiology of adolescent idiopathic scoliosis. Bone 140:115563. https://doi.org/10.1016/j.bone.2020.115563
Burwell RG, Clark EM, Dangerfield PH, Moulton A (2016) Adolescent idiopathic scoliosis (AIS): a multifactorial cascade concept for pathogenesis and embryonic origin. Scoliosis Spinal Disord. https://doi.org/10.1186/s13013-016-0063-1
Newton PO, Fujimori T, Doan J et al (2015) Defining the “three-dimensional sagittal plane” in thoracic adolescent idiopathic scoliosis. J Bone Joint Surg Am 97:1694–1701. https://doi.org/10.2106/JBJS.O.00148
Hayashi K, Upasani VV, Pawelek JB et al (2009) Three-dimensional analysis of thoracic apical sagittal alignment in adolescent idiopathic scoliosis. Spine 34:792–797. https://doi.org/10.1097/BRS.0b013e31818e2c36
Sangole AP, Aubin C-E, Labelle H et al (2009) Three-dimensional classification of thoracic scoliotic curves. Spine 34:91–99. https://doi.org/10.1097/BRS.0b013e3181877bbb
Dubousset J (1994) Three-dimensional analysis of the scoliotic deformity. In: Weinstein SL (ed) The pediatric spine: principles and practice. Raven Press Ltd, New York, pp. 479–496
Schlösser TPC, Janssen MMA, Hogervorst T et al (2017) The odyssey of sagittal pelvic morphology during human evolution: a perspective on different Hominoidae. Spine J 17:1202–1206. https://doi.org/10.1016/j.spinee.2017.03.016
Roussouly P, Pinheiro-Franco JL (2011) Sagittal parameters of the spine: biomechanical approach. Eur Spine J 20:578–585. https://doi.org/10.1007/s00586-011-1924-1
Le Huec JC, Saddiki R, Franke J et al (2011) Equilibrium of the human body and the gravity line: the basics. Eur Spine J 20(Suppl 5):558–563. https://doi.org/10.1007/s00586-011-1939-7
Kouwenhoven J-WM, Smit TH, van der Veen AJ et al (2007) Effects of dorsal versus ventral shear loads on the rotational stability of the thoracic spine: a biomechanical porcine and human cadaveric study. Spine 32:2545–2550. https://doi.org/10.1097/BRS.0b013e318158cd86
Castelein RM, van Dieën JH, Smit TH (2005) The role of dorsal shear forces in the pathogenesis of adolescent idiopathic scoliosis–a hypothesis. Med Hypotheses 65:501–508. https://doi.org/10.1016/j.mehy.2005.03.025
Giglio CA, Volpon JB (2007) Development and evaluation of thoracic kyphosis and lumbar lordosis during growth. J Child Orthop 1:187–193. https://doi.org/10.1007/s11832-007-0033-5
White AA, Panjabi MM (1978) The basic kinematics of the human spine. A review of past and current knowledge. Spine 3:12–20. https://doi.org/10.1097/00007632-197803000-00003
Schlösser TP, Vincken KL, Rogers K, Castelein RM, Shah SA (2015) Natural sagittal spino-pelvic alignment in boys and girls before, at and after the adolescent growth spurt. Eur Spine J 24:1158–1167. https://doi.org/10.1007/s00586-014-3536-z
Schlösser TP, Shah SA, Reichard SJ et al (2014) Differences in early sagittal plane alignment between thoracic and lumbar adolescent idiopathic scoliosis. Spine J 14:282–290. https://doi.org/10.1016/j.spinee.2013.08.059
de Reuver S, Costa L, van Rheenen H et al (2022) Disc and vertebral body morphology from birth to adulthood. Spine 47:E312–E318. https://doi.org/10.1097/BRS.0000000000004278
Grivas TB, Vasiliadis E, Malakasis M et al (2006) Intervertebral disc biomechanics in the pathogenesis of idiopathic scoliosis. Stud Health Technol Inform 123:80–83
Homminga J, Lehr AM, Meijer GJM et al (2013) Posteriorly directed shear loads and disc degeneration affect the torsional stiffness of spinal motion segments: a biomechanical modeling study. Spine 38:E1313–E1319. https://doi.org/10.1097/BRS.0b013e3182a0d5fa
Will RE, Stokes IA, Qiu X et al (2009) Cobb angle progression in adolescent scoliosis begins at the intervertebral disc. Spine 34:2782–2786. https://doi.org/10.1097/BRS.0b013e3181c11853
Stokes IAF, Sangole AP, Aubin C-E (2009) Classification of scoliosis deformity three-dimensional spinal shape by cluster analysis. Spine 34:584–590. https://doi.org/10.1097/BRS.0b013e318190b914
Pasha S (2019) 3D Deformation patterns of s shaped elastic rods as a pathogenesis model for spinal deformity in adolescent idiopathic scoliosis. Sci Rep. https://doi.org/10.1038/s41598-019-53068-7
Stokes IA (1989) Axial rotation component of thoracic scoliosis. J Orthop Res 7:702–708. https://doi.org/10.1002/jor.1100070511
Illés T, Somoskeöy S (2013) Comparison of scoliosis measurements based on three-dimensional vertebra vectors and conventional two-dimensional measurements: advantages in evaluation of prognosis and surgical results. Eur Spine J 22(6):1255–1263. https://doi.org/10.1007/s00586-012-2651-y
Schlösser TPC, Semple T, Carr SB et al (2017) Scoliosis convexity and organ anatomy are related. Eur Spine J 26:1595–1599. https://doi.org/10.1007/s00586-017-4970-5
Janssen MMA, Kouwenhoven J-WM, Schlösser TPC et al (2011) Analysis of preexistent vertebral rotation in the normal infantile, juvenile, and adolescent spine. Spine 36:E486–E491. https://doi.org/10.1097/BRS.0b013e3181f468cc
Roaf R (1960) Vertebral growth and its mechanical control. J Bone Joint Surg Br 42-B(1):40–59
Schlösser TPC, van Stralen M, Chu WCW et al (2016) Anterior overgrowth in primary curves, compensatory curves and junctional segments in adolescent idiopathic scoliosis. PLoS ONE 11:e0160267. https://doi.org/10.1371/journal.pone.0160267
Guo X, Chau WW, Chan YL, Cheng JC (2003) Relative anterior spinal overgrowth in adolescent idiopathic scoliosis. Results of disproportionate endochondralmembranous bone growth. J Bone Joint Surg Br 85:1026–1031
Brink RC, Schlösser TPC, Colo D et al (2017) Anterior spinal overgrowth is the result of the scoliotic mechanism and is located in the disc. Spine 42:818–822. https://doi.org/10.1097/BRS.0000000000001919
Dickson RA (1985) Aetiology of idiopathic spinal deformities. Arch Dis Child 60:508–511
Dickson RA, Lawton JO, Archer IA, Butt WP (1984) The pathogenesis of idiopathic scoliosis. Biplanar spinal asymmetry. J Bone Joint Surg Br 66:8–15
Kouwenhoven J-WM, Bartels LW, Vincken KL et al (2007) The relation between organ anatomy and pre-existent vertebral rotation in the normal spine: magnetic resonance imaging study in humans with situs inversus totalis. Spine 32:1123–1128. https://doi.org/10.1097/01.brs.0000261563.75469.b0
Stokes IAF, Burwell RG, Dangerfield PH, IBSE (2006) Biomechanical spinal growth modulation and progressive adolescent scoliosis–a test of the “vicious cycle” pathogenetic hypothesis: summary of an electronic focus group debate of the IBSE. Scoliosis. https://doi.org/10.1186/1748-7161-1-16
Acknowledgements
No acknowledgements to include.
Funding
This research received no external funding. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Author information
Authors and Affiliations
Contributions
JP, D-GC, S-IS and EI have done: Substantial contributions to the conception and design of the work, acquisition, analysis, or interpretation of data. Drafting the work and revising it critically for important intellectual content. Final approval of the version to be published and JP, D-GC, S-IS and EI agree to be accountable for the author’s own contributions and for ensuring that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and documented in the literature.
Corresponding author
Ethics declarations
Conflict of interest
Javier Pizones declares that he has no conflict of interest. Dong-Gune Chang declares that he has no conflict of interest. Se-Il Suk declares that he has no conflict of interest. Enrique Izquierdo declares that he has no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Institutional review board statement
As it is a review article, it did not need Institutional Review Board Statement, it was not applicable.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pizones, J., Chang, DG., Suk, SI. et al. Current biomechanical theories on the etiopathogenesis of idiopathic scoliosis. Spine Deform 12, 247–255 (2024). https://doi.org/10.1007/s43390-023-00787-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43390-023-00787-7