Abstract
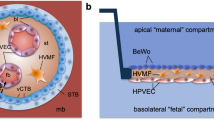
Objective: Fetal membranes, a vital component that helps maintain pregnancy and contribute to parturition signaling, are often studied in segments due to its structural complexity. Transwells are traditionally used to study cell interactions; however, their usefulness is limited. To overcome these difficulties, a fetal membrane-organ-on-chip (FM-OO-C) was created to study interactive properties of amnion epithelial cells (AECs) and decidual cells compared to transwell systems. Methods: Primary AECs and decidual cells from term, nonlaboring fetal membranes were cultured in a 2-chamber (AEC/decidual cell) FM-OO-C device and sandwiched between a semipermeable membrane. Cells were treated with cigarette smoke extract (CSE) or dioxin, and membrane permeability and cellular senescence were measured after 48 hours. The same experiments were conducted in transwells for comparisons. Results: Compared to transwell cultures, FM-OO-C model produced better membrane permeability readings regardless of the side of treatment or time point. Membrane permeabilization was higher in AECs directly treated with CSE (1.6 fold) compared to similar treatment on the decidual side (1.2 fold). In FM-OO-C, treatments forced changes between cellular layers. This was evident when CSE and dioxin-induced senescence on one side of the chamber produced similar changes on the opposite side. This effect was minimal in the transwell system. Conclusion: The controlled environment of an FM-OO-C allows for improved signal propagation between cells by minimizing noise and highlighting the small changes between treatments that cannot be seen in conventional transwell devices. Fetal membrane-organ-on-chip provides a better interaction between cell types that can be used to study fetal–maternal signaling during pregnancy in future studies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Behnia F, Taylor BD, Woodson M, et al. Chorioamniotic membrane senescence: a signal for parturition? Am J Obstet Gynecol. 2015;213(3):359.e1-416.e1.
Bredeson S, Papaconstantinou J, Deford JH, et al. HMGB1 promotes a p38MAPK associated non-infectious inflammatory response pathway in human fetal membranes. PLoS One. 2014; 9(12):e113799.
Dixon CL, Richardson L, Sheller-Miller S, Saade G, Menon R. A distinct mechanism of senescence activation in amnion epithelial cells by infection, inflammation, and oxidative stress. Am J Reprod Immunol. 2017;79(3).
Menon R, Boldogh I, Urrabaz-Garza R, et al. Senescence of primary amniotic cells via oxidative DNA damage. PLoS One. 2013;8(12):e83416.
Menon R, Boldogh I, Hawkins HK, et al. Histological evidence of oxidative stress and premature senescence in preterm premature rupture of the human fetal membranes recapitulated in vitro. Am J Pathol. 2014;184(6):1740–51.
Menon R, Bonney EA, Condon J, Mesiano S, Taylor RN. Novel concepts on pregnancy clocks and alarms: redundancy and synergy in human parturition. Hum Reprod Update. 2016;22(5):535–60.
Gnecco J, Osteen K. Compartmentalized culture of perivascular stroma and endothelial cells. Biomedical Engineering Society. 2017;45(7):1758–69.
Duval K, Grover H, Han LH, et al. Modeling physiological events in 2D vs. 3D cell culture. Physiology (Bethesda). 2017;32(4):266–77.
Katt ME, Placone AL, Wong AD, Xu ZS, Searson PC. In vitro tumor models: advantages, disadvantages, variables, and selecting the right platform. Front Bioeng Biotechnol. 2016;4:12.
Sances S, Ho R, Vatine G, et al. Svendsen, human iPSC-derived endothelial cells and microengineered organ-chip enhance neuronal development. Stem Cell Reports. 2018;10(4):1222–36.
van der Helm MW, van der Meer AD, Eijkel JC, van den Berg A, Segerink LI. Microfluidic organ-on-chip technology for bloodbrain barrier research. Tissue Barriers. 2016;4(1):e1142493.
Huh DD. A human breathing lung-on-a-chip. Ann Am Thorac Soc. 2015;12(suppl 1):S42-S44.
Konar D, Devarasetty M, Yildiz DV, Atala A, Murphy SV. Lungon- a-chip technologies for disease modeling and drug development. Biomed Eng Comput Biol. 2016;7(suppl 1):17-27.
Zhang YS, Aleman J, Arneri A, et al. From cardiac tissue engineering to heart-on-a-chip: beating challenges. Biomed Mater. 2015;10(3):034006.
Materne EM, Maschmeyer I, Lorenz AK, et al. The multi-organ chip—a microfluidic platform for long-term multi-tissue coculture. J Vis Exp. 2015;(98):e52526.
Gnecco JS, Anders AP, Cliffel D, et al. Instrumenting a fetal membrane on a chip as emerging technology for preterm birth research. Curr Pharm Des. 2017;23(40):6115–24.
Bruner-Tran KL, Gnecco J, Ding T, Glore DR, Pensabene V, Osteen KG. Exposure to the environmental endocrine disruptor TCDD and human reproductive dysfunction: translating lessons from murine models. Reprod Toxicol. 2017;68:59–71.
Anders AP, Gaddy JA, Doster RS, Aronoff DM. Current concepts in maternal-fetal immunology: recognition and response to microbial pathogens by decidual stromal cells. Am J Reprod Immunol. 2017;77(3).
Richardson L, Vargas G, Brown T, et al. Redefining 3Dimensional placental membrane microarchitecture using multiphoton microscopy and optical clearing. 2017;53:66–75.
Rogers L, Anjali A, Doster R, et al. Decidual stromal cell-derived PGE2 regulates macrophage responses to microbial threat. Am J Reprod Immunol. 2018;80(4):e13032.
Menon R, Fortunato SJ, Yu J, et al. Cigarette smoke induces oxidative stress and apoptosis in normal term fetal membranes. Placenta. 2011;32(4):317–22.
Sheller-Miller S, Urrabaz-Garza R, Saade G, Menon R. Damageassociated molecular pattern markers HMGB1 and cell-free fetal telomere fragments in oxidative-stressed amnion epithelial cellderived exosomes. J Reprod Immunol. 2017;123:3–11.
Abe Y, Sinozaki H, Takagi T, et al. Identification of 2,3,7,8- tetrachlorodibenzo-p-dioxin (TCDD)-inducible genes in human amniotic epithelial cells. Reprod Biol Endocrinol. 2006;4:27.
Jin J, Richardson L, Sheller-Miller S, Zhong N, Menon R. Oxidative stress induces p38MAPK-dependent senescence in the feto-maternal interface cells. Placenta. 2018;67:15–23.
Richardson L, Menon R. Proliferative, migratory, and transition properties reveal metastate of human amnion cells. Am J Pathol. 2018;188(9):2004–15.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Richardson, L., Gnecco, J., Ding, T. et al. Fetal Membrane Organ-On-Chip: An Innovative Approach to Study Cellular Interactions. Reprod. Sci. 27, 1562–1569 (2020). https://doi.org/10.1007/s43032-020-00184-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00184-9