Abstract
The Water Framework Directive (WFD) suggests establishing reference conditions to help evaluate the ecological status of rivers in a specific ecoregion. Here we used eighteen diatom indices to estimate the ecological status of 44 candidate rivers for reference conditions in Türkiye's Western Mediterranean Basin. We collected data on epilithic diatoms and environmental variables three times (spring, summer, and fall) in 2017. Based on the redundancy analysis (RDA), seven environmental parameters significantly affected the distribution of abundant and indicator diatom species. Indicator species highly correlated with nitrate–nitrogen (NO3–N) and total nitrogen (TN). These two environmental parameters also showed significant correlations with the Pampean Diatom Index (IDP), the Eutrophication/Pollution Index (EPI-D), the Trophic Diatom Index (TDI), and the Rott's Saprobic Index (ROTTs). Therefore, here we focused on the ecological quality status indication by these four indices. The TDI index was more consistent for estimating our streams' ecological status and revealed high ecological status for fifteen sampling sites. We conclude that areas with high ecological status will help set up a reference condition for future studies in the Western Mediterranean Basin and other basins in Türkiye.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Diatoms are unicellular microscopic algae that colonize various submerged substrates in aquatic environments and are an important component of phytobenthos (Wetzel 1983; Dixit et al., 1992). Numerous studies were conducted to show the effects of environmental factors on their diversity, abundance, and distribution (Liess et al., 2009; Soininen, 2007; Solak et al., 2020; Tan et al., 2017; Tanabe et al., 2010). They are one of the most often used indicators for assessing the ecological status of rivers due to their shorter life cycles, fast growth rates, quick reactions to environmental changes, greater sensitivity to pollution and eutrophication than other organisms in the water, and ease of sampling (Çetin & Demir, 2019; Delgado et al., 2012; Karaouzas et al., 2019; Poikane et al., 2016; Vilmi et al., 2015; Waite et al., 2020).
Although diatoms have been used in the evaluation of running waters and different indices have been developed for many years, after the implementation of the Water Framework Directive (WFD) (European Union, 2000), biological quality elements (phytobenthos, benthic invertebrates, macrophytes, fish, and phytoplankton) have been favored primarily as the main assessment component, and consequently, diatom studies have gained great importance (Hering et al. 2010). As a result, countries that are part of the European Union have developed their own methods for evaluating the ecological quality of running waters using diatom communities (Žutinić et al., 2020). Diatom indices based on relative abundances of the species and weighted averages of environmental optima or autecological values for the taxa were utilized for this purpose (Smol & Stoermer, 2010). In addition to the indices developed in previous years such as Specific Pollution Sensitivity Index (IPS) (Cemagref, 1982), Trophic Diatom Index (TDI) (Kelly & Whitton, 1995), Eutrophication/Pollution Index (EPI-D) (Dell’Uomo, 2004), and the Biological Diatom Index (IBD) (Coste et al., 2009), new diatom indices have started to be created in European Union member countries (e.g., Álvarez-Blanco et al., 2013; Dell'Uomo & Torrisi, 2011). Important studies have been carried out in other countries in the last decades to develop new diatom indices (e.g., Lavoie et al., 2010; Ponader et al., 2007; Potapova & Charles, 2007). Trophic Index Türkiye (TIT) was developed in Türkiye in order to be used as a region-specific index in studies of ecological quality (Çelekli et al., 2017, 2019a). It has been successfully tested in numerous rivers and basins (Çelekli et al., 2018, 2019b; Sevindik et al., 2021).
The WFD's primary objective is to ensure that all surface waters in member states—including streams and rivers—achieve good ecological status (European Union, 2000). The Ecological Quality Ratio (EQR), which is the ratio between the values expected in a minimally disturbed condition and the observed value of a candidate surface water, must be established in order to assess the ecological status (Kelly et al., 2006, 2012). River types differ due to factors such as altitude, drainage area, geology, stream size, and so on. Furthermore, biological communities differ between habitats and ecoregions. Different river types and ecoregions should be considered while defining reference conditions (O’Driscoll et al., 2012). The Ministry of Agriculture and Forestry, Directorate General of Water Management (DGWM) carried out the "Establishment of Reference Monitoring Network in Türkiye, 2017–2020" project to assess reference conditions in rivers of various types in 25 basins of Türkiye. Candidate reference areas in the Western Mediterranean basin were also revealed as part of this project. TIT and several diatom indices have been used in recent years to assess the ecological quality of rivers in the Western Mediterranean Basin (Çelekli & Lekesiz, 2020; Toudjani et al., 2017). However, no studies on the bio-assessment of minimally disturbed rivers in this basin were performed. As a result, the goals of this study were to: (i) use multivariate approaches to evaluate the interactions between diatoms and environmental stressors in these rivers; (ii) assess the ecological status of minimally disturbed rivers; and (iii) identify reference sites with different types in the Western Mediterranean Basin using different diatom indices.
Materials and methods
The Western Mediterranean Basin and studied sites
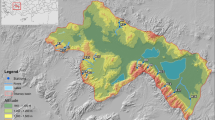
The Western Mediterranean Basin is situated in Türkiye's southwest Anatolian region, between 36° 06′–37° 35′ N and 27° 13′–30° 34′ E. The Büyük Menderes, Burdur, and Antalya basins, as well as the Aegean and Mediterranean Seas, surround it. The provinces of Antalya, Burdur, Denizli, Muğla, and Aydın are all located within the limits of the Western Mediterranean Basin. The surface area is about 21,223 km2, which constitutes 2.7% of Türkiye (DGWM 2016, 2018; Ministry of Agriculture and Forestry-MAF, 2019). The Dalaman, Eşen, Alakır, Akçay, Demre, Namnam, Kargıcak, Kocadere, and Sarıçay rivers, which release their water into the Aegean and the Mediterranean seas, are the principal running waters in the basin. The total length of the designated river water bodies is 1145 km, and the drainage area of these rivers is 14,693 km2. The basin's altitudes range from sea level to 3086 m, with an average altitude of 877 m. The Western Taurus Mountain Range contains the basin's most important mountains, including Beşparmak, Babadağ, Gölgeli, Dumanlı, Akdağlar, Eşeler, Honaz, Bozdağ, and Sandıras (DGWM, 2016, 2018; MAF, 2019).
The typical Mediterranean climate dominates in the basin which is characterized by mild and wet winters, and dry and hot summers. The majority of precipitation and runoff events occur in the winter, although snowfall is rare. However, the characteristics of a continental climate begin to appear in the basin's interior. The basin's annual average precipitation and temperature were 840 mm and 15.8 °C, respectively (DGWM, 2016, 2018; MAF, 2019).
The sampling sites were mostly found in the Western Taurus Mountain Range, and their altitudes ranged from 16 to 1583 m (Fig. 1). A geographic information system and preliminary fieldwork were used to determine the least disturbed river sites. Attention was paid to the absence of agricultural activities and settlements near and above the selected points. Generally, the vegetation type specific to the Mediterranean flora region (maquis and coniferous forests) dominated around the sites. Samples were collected from 44 sampling sites (R1 to R44) three times (in spring, summer, and fall) during 2017. Information on the typology, latitude, longitude, altitude, and mean discharge of the sampling stations is presented in Supplementary Material Table 1. The latitude, longitude, and altitude of the sites were determined using a geographic positioning system (Magellan Sportrak Pro model GPS). River typology was determined according to the EU Water Framework Directive (European Union, 2000), and river typology information was provided by the Ministry of Agriculture and Forestry, Directorate General of Water Management (DGWM). Discharge values from the sampling stations were obtained using a dwarf flow meter (MCM-02 model) or an acoustic Doppler (Isco 4250 model area velocity, Teledyne Isco Inc.).
Analysis of environmental variables
Sampling for chemical analyses and measurement of physical variables were conducted at 44 sampling sites. Water temperature (T) (°C), dissolved oxygen concentration (DO) (mg L−1), electrical conductivity (EC) (µS cm−1), and pH were measured 10 cm below the water surface using a Hach HQ40 model multimeter instrument. Total organic carbon (TOC) (mg L−1), chemical oxygen demand (COD) (mg L−1), alkalinity (ALK) (mg L−1), total phosphorus (TP) (mg L−1), nitrate–nitrogen (NO3–N) (mg L−1), organic nitrogen (ON) (mg L−1), and total nitrogen (TN) (mg L−1) were analyzed spectrophotometrically according to standard methods (APHA 2012) in an accredited laboratory of DGWM.
Analysis of epilithic diatoms
Sampling of epilithic diatoms was conducted at 44 sampling sites. The sampling sites (R3, R6, R10, R15, and R38) found in intermittent rivers were sampled during the flow period (Gallart et al. 2017) to ensure that the study was standardized with permanent rivers. The upper surface of at least five stones was brushed in 100 mL of distilled water, and these samples were fixed with Lugol’s solution (European Committee for Standardization, 2010). Diatom samples were cleaned with hydrochloric acid and hot hydrogen peroxide, and permanent slides were mounted with Naphrax (European Committee for Standardization, 2010). Before counting, it was ensured that all species were identified by navigating randomly selected fields on the slide. More than 400 diatom valves were counted on the selected horizontal transect of the slide for each sample with an Olympus BX51 microscope using 1000 × magnification. The relative abundance of diatom species for each sample was also determined for subsequent analysis. Taxonomic books such as Krammer and Lange-Bertalot (1986, 1991a, 1991b, 1999), Lange-Bertalot (2001), Krammer (2000, 2002, 2003), Bąk et al. (2012), and Lange-Bertalot et al. (2017) were used to identify epilithic diatom species, and the current names of the species were checked according to Guiry and Guiry (2022).
Trophic Index Türkiye (TIT) was calculated according to Toudjani et al. (2017) and Çelekli et al., (2017, 2019a). ‘OMNIDIA’ program 5.2 (Lecointe et al., 1993) was used to calculate different diatom indices (Supplementary Material Table 2). All the indices except TIT were transformed to range from 0 to 20 in Omnidia software. The ecological status assessment of the ROTTs, EPI-D, IDP, and TDI was also done by using class boundaries according to Rott et al., (1997), Guida (2004), Gómez and Licursi (2001), Rakowska and Szczepocka (2011).
Data analysis
The statistical differences in environmental parameters among the sites studied in this study were determined with an analysis of variance (one-way ANOVA) test using SPSS 20.0 software. Also, the statistical differences in common environmental parameters between the sites studied in this study and disturbed sites in other studies (Çelekli & Lekesiz, 2020; Toudjani et al., 2017) were detected with one-way ANOVA. Tukey HSD was used as post hoc multiple comparison. Physical and chemical variables were logarithmically transformed for linear regression analysis. Linear regression analysis between diatom indices and environmental variables was performed with SPSS 20.0 software. To determine the suitability of canonical correspondence analysis (CCA), the gradient length was determined firstly by detrended correspondence analysis (DCA). Redundancy analysis (RDA) was performed in Canoco 5.0 (ter Braak & Smilauer, 2012) to reveal the relations between diatom species and environmental variables since the response data have a gradient 2.2 SD long. RDA was carried out on the log-normal transformed abundance data. The statistical significance of the environmental predictor variables was assessed by 999 restricted Monte Carlo permutations. Diatom species occurring in more than three samples with a relative abundance larger than 1% in at least one sample (33 taxa) were described as abundant species and were used in the RDA (Weilhoefer & Pan, 2006). Moreover, indicator species (9 taxa) were detected with Indicator Species Analysis (IndVal) and were used in the RDA. Before the IndVal, samples were clustered according to environmental variables. Clustering was performed by calculating the Euclidean distances and linking them by the Ward method according to XLSTAT software (Addinsoft, 2014). Indicator diatom species for the clusters were identified by using indicator value (IndVal) analysis (Dufrene & Legendre, 1997). Monte Carlo simulations (999 permutations) were used to assess the significance of each species as an indicator for each cluster. The IndVal analysis of indicator species in clusters was done using the labdsv package (Roberts, 2019) of R software. To analyze the relationship between diatoms and 11 environmental variables (T, EC, pH, DO, TOC, COD, ALK, TP, ON, TN, NO3–N), we performed a RDA using relative abundance values of the 39 taxa (both indicator and abundant species). RDA was performed, initially on the whole environmental and diatom datasets. Forward selection indicated that 7 of the 11 environmental variables made a significant contribution to the variance in the diatom data. The mean values of three seasons were used to determine the relative abundance of diatoms, diatom indices, and environmental variables for RDA and linear regression analysis.
Results
Environmental variables
Table 1 and Supplementary Material Table 3 display the environmental parameters measured at 44 sampling sites. Except for TP, all environmental variables were significantly different between sampling sites (p < 0.01). The mean T values ranged between 11.50 and 23.10 °C, with Kozağaç River (R21) with the highest mean T value (f = 5.72, p < 0.01). The mean pH and ON values were highest in Gürleyik River-1 (R27) (f = 8.69, f = 8.61, respectively, p < 0.01), while the mean TOC and COD values were highest in Karabeyyurdu River (R35) (f = 5.59, f = 7.57, respectively, p < 0.01). The highest mean EC (3090.00 μS cm−1) value was found in Akyaka River (R42), while the lowest (160.03 μS cm−1) was recorded in Suçıkan River (R23) (f = 516.24, p < 0.01). The mean NO3–N and TN values were highest in Gürleyik River-3 (R40) (f = 4.02, f = 5.27, respectively, p < 0.01). Moreover, the mean NO3–N value was also higher in the Akyaka River (R42), while the mean TN values were also measured higher in Gürleyik River-1 (R27), Dalaman River (R37), and Akyaka River (R42). The mean DO values were measured between 3.72 and 11.19 mg L−1, and the highest value was found in Av River (R24) (f = 16.61, p < 0.01). The mean ALK values fluctuated between 103.73 and 395.00 mg L−1, and the highest value was recorded in Araplar River (R29) (f = 24.58, p < 0.01).
The boxplot graphs of common environmental parameters (T, EC, pH, TP, TN, NO3–N) measured in two previous studies on disturbed rivers in the Western Mediterranean Basin (Çelekli & Lekesiz, 2020; Toudjani et al., 2017) and in this study are shown in Supplementary Material Fig. 1. Moreover, the mean and standard deviation (SD) of environmental variables measured in the disturbed rivers of Western Mediterranean Basin in two different studies (Çelekli & Lekesiz, 2020; Toudjani et al., 2017) are given in Supplementary Material Table 4. There was no statistical difference in EC values in all three studies. The highest pH (f = 6.53, p < 0.01) and the lowest T (f = 16.25, p < 0.01), TN (f = 24.31, p < 0.01), NO3–N (f = 9.68, p < 0.01), and TP (f = 40.53, p < 0.01) values were recorded in our study.
Epilithic diatoms and environmental parameters
In the 44 sampling sites of the Western Mediterranean Basin, a total of 140 diatom taxa were recorded. Thirty-three diatom taxa occurring in at least three samples and accounting for more than 1% of total relative abundance are given in Supplementary Material Table 5. Nine diatom taxa were determined as indicator species (Table 2). Amphora ovalis (Kützing) Kützing, Cocconeis lineata Ehrenberg, Cymbella affinis Kützing, Cymbella neocistula Krammer, Fragilaria capucina Desmazières, Gomphonema affine Kützing, Gomphonema angustum Agardh, Gomphonema minutum (Agardh) Agardh, Navicula angusta Grunow, Navicula radiosa Kützing, and Ulnaria ulna (Nitzsch) Compère were commonly recorded in the sites of the basin.
The results of RDA are given in Fig. 2. The diatom–environmental correlations of RDA axes 1 and 2 are high, and the first two axes account for 73.0% of the variance in the diatom–environmental relationships. Most of the indicator species such as A. ovalis, Campylodiscus hibernicus Ehrenberg, Gyrosigma acuminatum (Kützing) Rabenhorst, N. angusta, Nitzschia dissipata (Kützing) Rabenhorst, Nitzschia sigmoidea (Nitzsch) Smith, Surirella amphioxys Smith were strongly correlated with NO3–N, TN and site R40. ALK, COD, and EC were correlated mainly with sites such as R10, R29, R34, R35, R42, and species such as U. ulna, Delicatophycus delicatulus (Kützing) Wynne, Gomphonema commutatum Grunow, and Ulnaria acus (Kützing) Aboal. In the opposite side of the first axis, pH was separated from other parameters with sites such as R14, R17, R19, R23, R25, R27, and species such as C. affinis, G. angustum, G. minutum, Gomphonema parvulum (Kützing) Kützing, Meridion circulare (Greville) Agardh, and N. radiosa.
Ordination diagram of the redundancy analysis (RDA) with the scores of diatoms relative abundance and environmental variables in different sampling sites. Environmental variables: T: water temperature, EC: electrical conductivity, TN: total nitrogen, NO3–N: nitrate–nitrogen, ALK: alkalinity, COD: chemical oxygen demand. Full names and abbreviations of species are given in Table 2 and Supplementary Material Table 5. Indicator species were colored with blue
Diatom indices
Linear regression analysis was performed on the index scores and environmental parameters (Table 3). All the environmental parameters except T, pH, and TP showed significant correlations with 13 index scores (p < 0.01 or p < 0.05). EC, COD, NO3–N, and ALK had 6, 4, 3, and 3 significant correlations with the diatom indices, respectively. The average scores of 18 diatom indices are given in Table 4. Due to the high correlation of indicator species with NO3–N and TN in the RDA, the ecological status of four indices (EPI-D, TDI, IDP, ROTTs) that show high correlation with NO3–N and TN is also given in Table 4. Average percentage information about the species pool which is used by diatom indices is given in Fig. 3. The EPI-D, TDI, IDP, and ROTTs indices used 88%, 99%, 65%, and 77% of the species, respectively.
Discussion
Due to the mountainous geography of the Western Mediterranean Basin, few studies on its rivers have been conducted, and the water quality of lower altitude (lowland) rivers has been investigated in these studies (Çelekli & Lekesiz, 2020; Kazancı & Dügel, 2000; Toudjani et al., 2017). The measured environmental variables, including biological oxygen demand, EC, TOC, TP, TN, and NO3–N, showed that some rivers were seriously nutrient and organically polluted. However, in some areas with less severe anthropogenic activity, good or high water quality was found. Although industrial activity-related pollution has been seen in some rivers near cities like Antalya, Denizli, Aydın, and Muğla, domestic waste and agricultural activity-related pollution are the predominant pollutants in the basin (Directorate General of Environmental Management-DGEM, 2016; Toudjani et al., 2017). Furthermore, intensive tourism and aquaculture activities have impacted the water quality of the basin's rivers (DGEM, 2016). In terms of environmental parameters (TP, TN, NO3–N), it was seen that the values measured in our study are lower than the values measured in two previous studies on disturbed rivers in the Western Mediterranean Basin (Çelekli & Lekesiz, 2020; Toudjani et al., 2017). The most important reason for this is that these 44 sites selected as candidates for minimally disturbed rivers are chosen from the high-altitude points away from human influence as much as possible.
Based on the RDA results, NO3–N, TN, and site R40 were correlated with most of the indicator species such as A. ovalis, C. hibernicus, G. acuminatum, N. angusta, N. dissipata, N. sigmoidea, and S. amphioxys. With the exception of N. angusta, all of these species were classified as pollution-tolerant taxa in European rivers (Kelly et al., 2008; Van Dam et al., 1994). However, N. angusta was described as pollution-sensitive taxa (Carayon et al., 2019; Van Dam et al., 1994). Studies have not been found that give information about the trophic status and ecological preferences of this species in the rivers of Türkiye. It is interesting that this species, which was defined as oligotrophic in rivers of Europe, showed a high correlation with NO3–N in our study. A. ovalis, N. angusta, and N. dissipata were also reported in the disturbed rivers of the Western Mediterranean Basin (Çelekli & Lekesiz, 2020; Toudjani et al., 2017). Site R40 was described as moderate or pool ecological status based on IDP and TDI indices. Similar to the findings of Toudjani et al. (2017), NO3–N was found as the influential environmental variable in the distribution of diatom species in our study. Although the selected sites were chosen from places where human impact is minimal, untreated domestic wastes from small-scale settlements may affect the NO3–N concentration, especially in site R40.
Species such as D. delicatulus, G. commutatum, U. acus, and U. ulna and sites such as R8, R10, R29, R34, R35, and R42 were correlated mainly with ALK, COD, and EC. These species except D. delicatulus were classified as intermediate-pollution-tolerant or pollution-tolerant taxa in different studies (Kelly et al., 2008; Salinas-Camarillo et al., 2021; Van Dam et al., 1994). In particular, U. ulna was highly correlated with EC and COD in the RDA. Bere and Tundisi (2011) have reported this species as tolerant to organic pollution, and it has broad tolerance to EC ranges (Bere & Tundisi, 2009; Venkatachalapathy & Karthikeyan, 2012). On the other hand, D. delicatulus is the indicator species of oligotrophic to mesotrophic waters with rich calcium bicarbonate (CaCO3) content (Lange-Bertalot et al., 2017). Carbonate-rich sedimentary rocks such as calcite (CaCO3) have been described as the most common geogenic source of ALK in stream water (Wedepohl, 1978). As a matter of fact, this species showed higher correlations with ALK and sites R8 (74.2% mean abundance) and R34 (59.1% mean abundance) in the RDA. All these sites (R8, R10, R29, R34, R35, R42) except R35 were high or good ecological status based on EPI-D, TDI, and ROTTs indices. The R35 site indicated medium ecological quality according to the ROTTs, while good ecological quality according to the EPI-D and TDI. In this station, EC and COD values were high and U. ulna was the dominant species with a mean abundance of 75.6%.
In Danish streams, Baattrup-Pedersen et al. (2022) have found that pollution-sensitive and pollution-tolerant taxa coexisted in highly alkaline rivers, and it became difficult to explain the causal relationship between ALK and pollution indicator environmental variables. Kelly et al. (2020) have also stated that ALK stands out as the most important abiotic predictor variable in phytobenthos reference sites, and it became difficult to detect least disturbed conditions in high ALK rivers due to geological conditions which often resulted in situations well-suited to settlement and agriculture. Similarly in our study, high ALK, EC, COD, and pollution-sensitive and pollution-tolerant taxa were observed in streams with different trophic conditions (high, good, medium). Therefore, a new approach should be developed by taking into account the ALK status of the least disturbed rivers during the revision of the ecological quality indices.
Species such as C. affinis, G. angustum, G. minutum, G. parvulum, M. circulare, and N. radiosa were located near sites such as R14, R17, R19, R23, R25, R27 and were associated with high pH in the RDA. All these sites were high or good ecological status based on EPI-D, TDI, and ROTTs indices. These aforementioned species, except C. affinis and N. radiosa, were also found in other studies of this basin (Çelekli & Lekesiz, 2020; Toudjani et al., 2017). Cymbella affinis, G. angustum, M. circulare, and N. radiosa were described as oligo-saprobic species (Bere & Tundisi, 2011; Carayon et al., 2019; Çelekli et al., 2019a, 2019b; Jüttner et al., 1996; Kelly et al., 2008; Van Dam et al., 1994), while G. minutum and G. parvulum were proposed as intermediate-pollution tolerant taxa in different watercourses (Çelekli et al., 2019a; Hausmann et al., 2016; Kelly et al., 2008; Sevindik & Kucuk, 2016).
Four indices (EPI-D, TDI, IDP, and ROTTs) were significantly correlated with NO3–N and TN values. Among these indices, EPI-D and ROTTs indicated high ecological quality at 35 and 37 sites, while they indicated good ecological quality at 9 and 6 sites, respectively. On the other hand, IDP defined the sites as good or moderate ecological quality. TDI determined 15 sites as high, and 26 sites as good ecological status. Site 40 which was highly correlated with TN, NO3–N, and pollution-tolerant species, was determined as poor ecological quality according to TDI. Besides, sites which contain intermediate-pollution-tolerant and pollution-tolerant species such as U. ulna, U. acus, G. parvulum, G. minutum, G. commutatum were described as good ecological status with TDI. For this reason, considering the diatom species and environmental parameters, it can be concluded that the TDI gives better results in determining the ecological quality of the sites. This index was used in studies such as Karasu River (Gürbüz and Kivrak 2002), Upper Porsuk Creek (Solak, 2011), and Acarlar Floodplain (Sevindik & Kucuk, 2016) and found compatible with indicator diatom species and environmental parameters. EPI-D and TIT indices were tested in previous studies of the rivers in the Western Mediterranean Basin (Çelekli & Lekesiz, 2020; Toudjani et al., 2017). TIT index, which was highly correlated with TP, was found effective in evaluating the ecological quality of these rivers. However, in our study, TIT did not give convenient results, since it did not show a significant correlation with TP or other environmental parameters. This index, which gives consistent results in more disturbed rivers of the same basin, did not work in pristine rivers; consequently, it should be revised considering these kinds of pristine rivers.
In conclusion, it was found that 44 pristine rivers which were selected to determine their potential as reference sites in the Western Mediterranean Basin, had better water quality than the disturbed rivers in terms of environmental variables (TP, TN, NO3–N) measured in the basin. According to the RDA results, the distribution of abundant and indicator diatom species in the rivers was highly influenced by the catchment geology and nutrients, as evidenced by the NO3–N, TN, ALK, EC, pH, and COD values. Due to the high correlation between NO3–N, TN, and indicator species, the ecological quality status of 4 indices (EPI-D, TDI, IDP, and ROTTs), which showed high correlations with NO3–N and TN, was determined. However, TDI gave more consistent results with environmental parameters and diatom species. The 15 high-ecological quality sites according to TDI will help set up reference conditions for future studies in the Western Mediterranean Basin and other basins in Türkiye.
Availability of data and materials
Not applicable.
Code availability
Not applicable.
References
Addinsoft. (2014). XLSTAT 5.03. Addinsoft, USA.
Álvarez-Blanco, I., Blanco, S., Cejudo-Figueiras, C., & Bécares, E. (2013). The Duero Diatom Index (DDI) for river water quality assessment in NW Spain: Design and validation. Environmental Monitoring and Assessment, 185(1), 969–981. https://doi.org/10.1007/s10661-012-2607-z
APHA-American Public Health Association. (2012). Standard methods for the examination of water and wastewater (22nd ed.). American Water Works Association, Water Environment Federation.
Baattrup-Pedersen, A., Johnsen, T. J., Larsen, S. E., & Riis, T. (2022). Alkalinity and diatom assemblages in lowland streams: How to separate alkalinity from inorganic phosphorus in ecological assessments? Science of the Total Environment, 823, 153829. https://doi.org/10.1016/j.scitotenv.2022.153829
Bąk, M., Witkowski, A., Żelazna-Wieczorek, J., Wojtal, A. Z., Szczepocka, E., Szulc, K., & Szulc, B. (2012). The key for the determination of diatoms in phytobenthos for the purpose of assessing the ecological status of surface waters in Poland. Environmental Monitoring Library. in Polish.
Bere, T., & Tundisi, J. G. (2009). Weighted average regression and calibration of conductivity and pH of benthic diatom assemblages in streams influenced by urban pollution–São Carlos/SP, Brazil. Acta Limnologica Brasiliensia, 21(3), 317–325.
Bere, T., & Tundisi, J. G. (2011). Diatom-based water quality assessment in streams influence by urban pollution: effects of natural and two selected artificial substrates, São Carlos-SP, Brazil. Brazilian Journal of Aquatic Science and Technology, 15(1), 54–63. https://doi.org/10.14210/bjast.v15n1.p54-63
Carayon, D., Tison-Rosebery, J., & Delmas, F. (2019). Defining a new autoecological trait matrix for French stream benthic diatoms. Ecological Indicators, 103, 650–658. https://doi.org/10.1016/j.ecolind.2019.03.055
Çelekli, A., Kayhan, S., Lekesiz, Ö., Toudjani, A. A., & Çetin, T. (2019b). Limno-ecological assessment of Aras River surface waters in Turkey: Application of diatom indices. Environmental Science and Pollution Research, 26(8), 8028–8038. https://doi.org/10.1007/s11356-019-04295-y
Çelekli, A., & Lekesiz, Ö. (2020). Eco-assessment of West Mediterranean basin’s rivers (Turkey) using diatom metrics and multivariate approaches. Environmental Science and Pollution Research, 27(22), 27796–27806. https://doi.org/10.1007/s11356-020-09140-1
Çelekli, A., Toudjani, A. A., Gümüş, E. Y., Kayhan, S., Lekesiz, H. Ö., & Çetin, T. (2019a). Determination of trophic weight and indicator values of diatoms in Turkish running waters for water quality assessment. Turkish Journal of Botany, 43, 90–101. https://doi.org/10.3906/bot-1704-40
Çelekli, A., Toudjani, A. A., Kayhan, S., Lekesiz, H. O., & Gümüş, E. Y. (2017). Establishment of water quality ecological evaluation system specific to our country (project no: 20011K050400). T.R. Ministry of Forestry and Water Affairs, General Directorate of Water Management. in Turkish.
Çelekli, A., Toudjani, A. A., Lekesiz, H. Ö., & Çetin, T. (2018). Ecological quality assessment of running waters in the North Aegean catchment with diatom metrics and multivariate approach. Limnologica, 73, 20–27. https://doi.org/10.1016/j.limno.2018.09.001
Cemagref. (1982). Study of quantitative biological methods for the assessment of water quality. A.F.B. Rhȏne-Mediterranee-Corse. in French.
Çetin, T., & Demir, N. (2019). The use of phytobenthos for the ecological status assessment in Upper Sakarya Basin, Turkey. Applied Ecology and Environmental Research, 17(4), 10155–10172. https://doi.org/10.15666/aeer/1704_1015510172
Coste, M., Boutry, S., Tison-Rosebery, J., & Delmas, F. (2009). Improvements of the Biological Diatom Index (BDI): Description and efficiency of the new version (BDI-2006). Ecological Indicators, 9(4), 621–650. https://doi.org/10.1016/j.ecolind.2008.06.003
Delgado, C., Pardo, I., & García, L. (2012). Diatom communities as indicators of ecological status in Mediterranean temporary streams (Balearic Islands, Spain). Ecological Indicators, 15(1), 131–139. https://doi.org/10.1016/j.ecolind.2011.09.037
Dell’Uomo, A. (2004). The eutrophication/pollution diatomic index (EPI-D) in the monitoring of running waters. Guide lynx. APAT Agency for Environmental Protection and Technical Services. in Italian.
Dell’Uomo, A., & Torrisi, M. (2011). The Eutrophication/Pollution Index-Diatom based (EPI-D) and three new related indices for monitoring rivers: The case study of the river Potenza (the Marches, Italy). Plant Biosystems-an International Journal Dealing with All Aspects of Plant Biology, 145(2), 331–341. https://doi.org/10.1080/11263504.2011.569347
Descy, J. P. (1979). A new approach to water quality estimation using diatoms. Nowa Hedwigia, 64, 305–323.
Descy, J. P., & Coste, M. (1991). A test of methods for assessing water quality based on diatoms. Verhandlungen Der Internationalen Vereinigung Für Theorestische Und Angewandte Limnologie, 24, 2112–2116. https://doi.org/10.1080/03680770.1989.11899905
DGEM-Directorate General of Environmental Management. (2016). Western Mediterranean Basin Pollution Prevention Action Plan, Ministry of Environment and Urbanization, Ankara, Turkey (in Turkish).
DGWM-Directorate General for Water Management. (2016). Project on the establishment of an ecological assessment system for water quality in Turkey: The West Mediterranean basin, Ankara, Turkey (in Turkish).
DGWM-Directorate General for Water Management. (2018). Western Mediterranean Basin Drought Management Plan, Volume I—General Description of The Basin and Drought Analysis, Ankara, Turkey (in Turkish).
Dixit, S. S., Smol, J. P., Kingston, J. C., & Charles, D. F. (1992). Diatoms: Powerful indicators of environmental change. Environmental Science and Technology, 26(1), 22–33. https://doi.org/10.1021/es00025a002
Dufrene, M., & Legendre, P. (1997). Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecological Monographs, 67, 345–366.
European Committee for Standardization. (2010). Water quality—Guidance standard for the surveying, sampling and laboratory analyses of phytobenthos in shallow running water. European Standard EN, 15708.
European Union. (2000). Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for community action in the field of water policy. Official Journal of the European Communities, L327, 1–73.
Gallart, F., Cid, N., Latron, J., Llorens, P., Bonada, N., Jeuffroy, J., Jiménez-Argudo, S. M., Vega, R. M., Solà, C., Soria, M., & Bardina, M. (2017). TREHS: An open-access software tool for investigating and evaluating temporary river regimes as a first step for their ecological status assessment. Science of the Total Environment, 607, 519–540.
Gómez, N., & Licursi, M. (2001). The Pampean Diatom Index (IDP) for assessment of rivers and streams in Argentina. Aquatic Ecology, 35, 173–181. https://doi.org/10.1023/A:1011415209445
Guida, L. (2004). L’indice Diatomico Di Eutrofizzazione/Polluzione (Epi-D) Nel Monitoraggio Delle Acque Correnti.
Guiry, M.D., & Guiry, G. M. (2022). AlgaeBase. World-Wide Electronic Publication. National University of Ireland, Galway. Retrieved 21 March, 2022, from http://www.algaebase.org.
Gurbuz, H., & Kivrak, E. (2002). Use of epilithic diatoms to evaluate water quality in the Karasu River of Turkey. Journal of Environmental Biology, 23(3), 239–246.
Hering, D., Borja, A., Carstensen, J., Carvalho, L., Elliott, M., Feld, C. K., Heiskanen, A, S., Johnson, R. K., Moe, J., Pont, D., Solheim, A. L., & van de Bund, W. (2010). The European Water Framework Directive at the age of 10: a critical review of the achievements with recommendations for the future. Science of the Total Environment, 408(19), 4007–4019. https://doi.org/10.1016/j.scitotenv.2010.05.031
Hausmann, S., Charles, D. F., Gerritsen, J., & Belton, T. J. (2016). A diatom-based biological condition gradient (BCG) approach for assessing impairment and developing nutrient criteria for streams. Science of the Total Environment, 562, 914–927. https://doi.org/10.1016/j.scitotenv.2016.03.173
Hürlimann, J., & Niederhauser, P. (2002). Method for studying and assessing the state of health of rivers: Diatoms-level R (region). OFEFP. in French.
Jüttner, I., Rothfritz, H., & Ormerod, S. J. (1996). Diatoms as indicators of river quality in the Nepalese Middle Hills with consideration of the effects of habitat-specific sampling. Freshwater Biology, 36(2), 475–486. https://doi.org/10.1046/j.1365-2427.1996.00101.x
Karaouzas, I., Smeti, E., Kalogianni, E., & Skoulikidis, N. T. (2019). Ecological status monitoring and assessment in Greek rivers: Do macroinvertebrate and diatom indices indicate same responses to anthropogenic pressures? Ecological Indicators, 101, 126–132. https://doi.org/10.1016/j.ecolind.2019.01.011
Kazancı, N., & Dügel, M. (2000). An evaluation of the water quality of Yuvarlakçay Stream, in the Köycegiz-Dalyan protected area, SW Turkey. The Turkish Journal of Zoology, 24(1), 69–80.
Kelly, M. G., Gómez-Rodríguez, C., Kahlert, M., Almeida, S. F., Bennett, C., et al. (2012). Establishing expectations for pan-European diatom based ecological status assessments. Ecological Indicators, 20, 177–186. https://doi.org/10.1016/j.ecolind.2012.02.020
Kelly, M. G., Juggins, S., Bennion, H., Burgess, A., Yallop, M., Hirst, H., King, L., Jamieson, J., Guthrie, R., & Rippey, B. (2006). Use of diatoms for evaluating ecological status in UK freshwaters (Vol. 170). Draft final report to Environment Agency, Bristol.
Kelly, M., Juggins, S., Guthrie, R., Pritchard, S., Jamieson, J., et al. (2008). Assessment of ecological status in UK rivers using diatoms. Freshwater Biology, 53(2), 403–422. https://doi.org/10.1111/j.1365-2427.2007.01903.x
Kelly, M. G., Phillips, G., Juggins, S., & Willby, N. J. (2020). Re-evaluating expectations for river phytobenthos assessment and understanding the relationship with macrophytes. Ecological Indicators, 117, 106582. https://doi.org/10.1016/j.ecolind.2020.106582
Kelly, M. G., & Whitton, B. A. (1995). The trophic diatom index: A new diatom index for monitoring eutrophication in rivers. Journal of Applied Phycology, 7, 433–444. https://doi.org/10.1007/BF00003802
Krammer, K. (2000). Diatoms of Europe. In H. Lange-Bertalot (Ed.), The Genus Pinnularia. (Vol. 1). A.R.G. Gantner Verlag.
Krammer, K. (2002). Diatoms of the European Inland Waters and Comparable Habitats, Cymbella (Vol. 3). Gantner Verlag.
Krammer, K. (2003). Diatoms of Europe, Cymbopleura, Delicata, Navicymbula, Gomphocymbellopsis, Afrocymbella (Vol. 4). A.R.G. Gantner Verlag.
Krammer, K., & Lange-Bertalot, H. (1986). Freshwater flora of Central Europe: Bacillariophyceae, I. Naviculaceae. Gustav Fischer Verlag. in German.
Krammer, K., & Lange-Bertalot, H. (1991a). Freshwater flora of Central Europe: Bacillariophyceae. III. Centrales, Fragilariaceae, Eunoticeae. Gustav Fischer Verlag. in German.
Krammer, K., & Lange-Bertalot, H. (1991b). Freshwater flora of Central Europe: Bacillariophyceae. IV. Achnanthaceae, Critical additions to Navicula (Lineolatae) and Gomphonema. Gustav Fischer Verlag. in German.
Krammer, K., & Lange-Bertalot, H. (1999). Freshwater flora of Central Europe: Bacillariophyceae. II. Epithemiaceae, Surirellaceae. Gustav Fischer Verlag. in German.
Lange-Bertalot, H. (2001). Navicula sensu stricto 10 genera separated from Navicula sensu lato Frustulia. In H. Lange-Bertalot (Ed.), Diatoms of Europe—Diatoms of the European Inland waters and comparable habitats. Gantner Verlag.
Lange-Bertalot, H., Hofmann, G., Werum, M., Cantonati, M., & Kelly, M. G. (2017). Freshwater benthic diatoms of Central Europe: Over 800 common species used in ecological assessment. Schmitten-Oberreifenberg, Koeltz Botanical Books.
Lavoie, I., Grenier, M., Campeau, S., & Dillon, P. J. (2010). The Eastern Canadian Diatom Index (IDEC) version 2.0: Including meaningful ecological classes and an expanded coverage area that encompasses additional geological characteristics. Water Quality Research Journal, 45(4), 463–477. https://doi.org/10.2166/wqrj.2010.045
Leclercq, L., & Maquet, B. (1987). Two new chemical and diatomic indexes of common water quality. Application to the Samson and its tributaries (Belgian Meuse Basin). Comparison with other biocenotic and diatomic chemical indices. Royal Belgian Institute of Natural Sciences, Working Document, 38, 1–113. in French.
Lecointe, C., Coste, M., & Prygiel, J. (1993). “Omnidia”: Software for taxonomy, calculation of diatom indices and inventories management. Hydrobiologia, 269(1), 509–513. https://doi.org/10.1007/BF00028048
Liess, A., Lange, K., Schulz, F., Piggott, J. J., Matthaei, C. D., & Townsend, C. R. (2009). Light, nutrients and grazing interact to determine diatom species richness via changes to productivity, nutrient state and grazer activity. Journal of Ecology, 97(2), 326–336.
Lobo, E., Bes, D., Tudesque, L., & Ector, L. (2004). Water quality assessment of the Pardinho River, RS, Brazil, using epilithic diatom assemblages and faecal coliforms as biological indicators. Vie Milieu, 54, 115–125.
MAF-Ministry of Agriculture and Forestry, (2019). Technical assistance project for economic analysis and water efficiency studies within the scope of river basin management plans in 3 pilot basins, strategic environmental assessment. Scoping report. Western Mediterranean River Basin (in Turkish).
O’Driscoll, C., de Eyto, E., Rodgers, M., O’Connor, M., & Xiao, L. (2012). Diatom assemblages and their associated environmental factors in upland peat forest rivers. Ecological Indicators, 18, 443–451. https://doi.org/10.1016/j.ecolind.2011.12.008
Poikane, S., Kelly, M., & Cantonati, M. (2016). Benthic algal assessment of ecological status in European lakes and rivers: Challenges and opportunities. Science of the Total Environment, 568, 603–613. https://doi.org/10.1016/j.scitotenv.2016.02.027
Ponader, K. C., Charles, D. F., & Belton, T. J. (2007). Diatom-based TP and TN inference models and indices for monitoring nutrient enrichment of New Jersey streams. Ecological Indicators, 7(1), 79–93. https://doi.org/10.1016/j.ecolind.2005.10.003
Potapova, M., & Charles, D. F. (2007). Diatom metrics for monitoring eutrophication in rivers of the United States. Ecological Indicators, 7(1), 48–70. https://doi.org/10.1016/j.ecolind.2005.10.001
Prygiel, J., Leveque, L., & Iserentant, R. (1996). IDP: A new practical diatomic index for the evaluation of water quality in a monitoring network. Revue Des Sciences De I’eau, 9, 97–113. in French.
Rakowska, B., & Szczepocka, E. (2011). Demonstration of the Bzura River restoration using diatom indices. Biologia, 66(3), 411–417. https://doi.org/10.2478/s11756-011-0032-3
Roberts, D. W. (2019). labdsv: Ordination and multivariate analysis for ecology. R package version 2.0-1. https://CRAN.R-project.org/package=labdsv.
Rott, E., Hofmann, G., Pall, K., Pfister, P., & Pipp, E. (1997). Indication lists for growth algae in Austrian watercourses, part 1: saprobic indication. Water Management Cadastre, Federal Ministry of Agriculture and Forestry. in German.
Rott, E., Pipp, E., Pfister, P., Van Dam, H., Ortler, K., et al. (1999). Indication lists for growth algae in Austrian watercourses. Part 2: Trophy indication (as well as geochemical preferences, taxonomic and toxicological notes). Water Management Cadastre, Federal Ministry of Agriculture and Forestry. in German.
Rumeau, A., & Coste, M. (1988). Introduction to the systematics of freshwater diatoms. Bulletin Français De La Pèche Et De La Pisciculture, 309, 69. in French.
Salinas-Camarillo, V. H., Carmona-Jiménez, J., & Lobo, E. A. (2021). Development of the Diatom Ecological Quality Index (DEQI) for peri-urban mountain streams in the Basin of Mexico. Environmental Science and Pollution Research, 28, 14555–14575.
Sevindik, T. O., Alemdar, E., Uzun, A., Coşkun, T., & Tunca, H. (2021). Ecological status estimation of eight creeks in the Lake Sapanca Basin (Sakarya, Turkey) using diatom indices. Annales De Limnologie - International Journal of Limnology, 57, 14. https://doi.org/10.1051/limn/2021012
Sevindik, T. O., & Kucuk, F. (2016). Benthic diatoms as indicators of water quality in the Acarlar Floodplain Forest (Northern Turkey). Fresenius Environmental Bulletin, 25(10), 4013–4025.
Sládeček, V. (1986). Diatoms as indicators of organic pollution. Acta Hydrochimica Et Hydrobiologica, 14, 555–566. https://doi.org/10.1002/aheh.19860140519
Smol, J. P., & Stoermer, E. F. (2010). The diatoms: Applications for the environmental and earth sciences. Cambridge University Press.
Soininen, J. (2007). Environmental and spatial control of freshwater diatoms—A review. Diatom Research, 22(2), 473–490. https://doi.org/10.1080/0269249X.2007.9705724
Solak, C. N. (2011). The application of diatom indices in the upper Porsuk Creek Kütahya-Turkey. Turkish Journal of Fisheries and Aquatic Sciences, 11(1), 31–36.
Solak, C. N., Peszek, Ł, Yılmaz, E., Ergül, H. A., Kayal, M., et al. (2020). Use of diatoms in monitoring the Sakarya River Basin, Turkey. Water, 12, 703–723. https://doi.org/10.3390/w12030703
Steinberg, C., & Schiefele, S. (1988). Biological indication of trophy and pollution of running waters. Zeitschrift Für Wasser- Und Abwasser-Forschung, 21(6), 227–234.
Tan, X., Zhang, Q., Burford, M. A., Sheldon, F., & Bunn, S. E. (2017). Benthic diatom based indices for water quality assessment in two subtropical streams. Frontiers in Microbiology, 8, 601. https://doi.org/10.3389/fmicb.2017.00601
Tanabe, Y., Ohtani, S., Kasamatsu, N., Fukuchi, M., & Kudoh, S. (2010). Photophysiological responses of phytobenthic communities to the strong light and UV in Antarctic shallow lakes. Polar Biology, 33(1), 85–100. https://doi.org/10.1007/s00300-009-0687-1
ter Braak, C. J., & Smilauer, P. (2012). Canoco reference manual and user’s guide: software for ordination, (Version 5.0). Microcomputer Power.
Toudjani, A. A., Celekli, A., Gümüş, E. Y., Kayhan, S., Lekesiz, H. Ö., & Çetin, T. (2017). A new diatom index to assess ecological quality of running waters: A case study of water bodies in western Anatolia. Annales De Limnologie - International Journal of Limnology, 53, 333–343. https://doi.org/10.1051/limn/2017012
Van Dam, H., Mertens, A., & Sinkeldam, J. (1994). A coded checklist and ecological indicator values of freshwater diatoms from the Netherlands. Netherlands Journal of Aquatic Ecology, 28(1), 117–133. https://doi.org/10.1007/BF02334251
Venkatachalapathy, R., & Karthikeyan, P. (2012). Environmental impact assessment of Cauvery river with diatoms at Bhavani, Tamil Nadu, India. International Journal of Geology, Earth & Environmental Sciences, 2(3), 36–42.
Vilmi, A., Karjalainen, S. M., Landeiro, V. L., & Heino, J. (2015). Freshwater diatoms as environmental indicators: Evaluating the effects of eutrophication using species morphology and biological indices. Environmental Monitoring and Assessment, 187(5), 1–10. https://doi.org/10.1007/s10661-015-4485-7
Waite, I. R., Pan, Y., & Edwards, P. M. (2020). Assessment of multi-stressors on compositional turnover of diatom, invertebrate and fish assemblages along an urban gradient in Pacific Northwest streams (USA). Ecological Indicators, 112, 106047. https://doi.org/10.1016/j.ecolind.2019.106047
Watanabe, T., Asai, K., & Houki, A. (1990). Numerical simulation of organic pollution in flowing waters. In P. N. Cheremisinoff (Ed.), Encyclopedia of environmental control technology. Vol. 4. Hazardous waste containment and treatment (pp. 251–284). Gulf Publishing Company.
Wedepohl, K. H. (1978). Handbook of geochemistry (Vol. 2). Springer.
Weilhoefer, C. L., & Pan, Y. (2006). Diatom-based bioassessment in wetlands: How many samples do we need to characterize the diatom assemblage in a wetland adequately? Wetlands, 26, 793–802. https://doi.org/10.1672/0277-5212(2006)26[793:DBIWHM]2.0.CO;2
Wetzel, R, G. (1983) Attached algal-substrata interactions: fact or myth, and when and how?. In Periphyton of Freshwater Ecosystems: Proceedings of the First International Workshop on Periphyton of Freshwater Ecosystems held in Växjö (pp. 207–215), Sweden, Springer Netherlands.
Žutinić, P., Kulaš, A., Levkov, Z., Šušnjara, M., Orlić, S., et al. (2020). Ecological status assessment using periphytic diatom communites-case study Krka River. Macedonian Journal of Ecology and Environment, 22(1), 29–44.
Acknowledgements
This study has been prepared by using the data obtained within the scope of “Establishment of Reference Monitoring Network in Türkiye” (2017–2020) (Project Number: 2011K050400) carried out under the responsibility of the Ministry of Agriculture and Forestry, General Directorate of Water Management.
Funding
None.
Author information
Authors and Affiliations
Contributions
TOS designed the experiments, conducted the field sampling, identified and counted the diatoms, analyzed the data, and wrote the text. TÇ participated in project management, designed the experiments, and analyzed the data. UG, HT, and AGT conducted the field sampling and data curation. All authors read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ongun Sevindik, T., Çetin, T., Tunca, H. et al. Ecological status estimation of minimally disturbed rivers of the Western Mediterranean Basin (Türkiye) using diatom indices. COMMUNITY ECOLOGY 24, 243–255 (2023). https://doi.org/10.1007/s42974-023-00147-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42974-023-00147-5