Abstract
Human population is becoming increasingly urbanized, and in this context, private gardens (home gardens) constitute an important component of urban biodiversity and provide access to ecosystem services. This study aims at identifying spatial patterns to understand the socio-ecological processes that influence the urban landscape.
In our study, we analyze private gardens in one of the main urban agglomerations of Argentina to understand whether socio-economic structure or spatial distribution is more strongly influencing the species composition of private gardens.
We selected 50 gardens from the urban area of Gran San Miguel de Tucumán. We surveyed the sociodemographic characteristics of garden owners and we performed vegetation censuses in each of the gardens. In the survey, we also evaluated the main mechanisms of plant acquisition. We used the species composition of each garden to perform a non-metric multidimensional scaling, which reflected the botanical distance between gardens. We used Mantel tests to correlate these botanical distances with the geographic and socio-economic distances between gardens to determine which variable controls the ecological attributes of the garden. To spatially characterize the socio-economic level, we used data from the national population census.
The species composition of the gardens is more strongly associated with socioeconomic conditions than with geographical distance. The exchange of species is the main method of obtaining plants.
Our study permits understanding how socio-economic structure influences the construction of private gardens, which are important components of the landscape and urban ecology. Our results could be explained by the willingness to belong to certain socio-economic groups but also by the interchange of propagules, which may reinforce social ties. Our results highlight the importance of addressing social issues to understand private decisions and design strategies toward a fair distribution of urban vegetation services.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The spatial structure of private gardens, which plays an important role in urban ecosystem biodiversity and the life quality of people, might be associated with socioeconomic conditions. Urban vegetation comprises one of the main scenarios for interactions with wildlife in cities, offering opportunities for the enjoyment of the natural environment (Power 2005; Freeman et al. 2012) in a context in which the human population is undergoing rapid changes toward a dominantly urban way of life (Grimm et al. 2008). Further, projections estimate that by 2050, 68% of the world population will live in urban areas (United Nations 2018). Most of this increase will occur in intermediate-sized urban areas (i.e., between one and three million inhabitants) of developing countries (Crossette 2011). The loss of vegetation is a widespread issue in urban areas sprawling over natural spaces (Paolini et al. 2016). Private gardens are a key component of urban green areas, with an important role in the connection between urban green areas and people’s quality of life (Rudd et al. 2002) and in some cases, they represent a major share of urban green areas (Gaston et al. 2013). However, the governance and management of private gardens, which have a substantial impact on ecosystem services provision and urban biodiversity maintenance, is challenging due to the diversity of actors involved (Loram et al. 2007, 2008; González-García and Sal 2008; Goddard et al. 2010; Peroni et al. 2016). For example, private gardens, whose design is controlled by economic power and the personal choices of owners, may play a relevant role in vegetation and associated animal communities (Avolio et al. 2018).
Most research on urban vegetation focuses on public spaces and the benefits they provide both to the health of people and biodiversity in cities (Chiesura 2004; Boone et al. 2009; Dobbs et al. 2017). Studies addressing private gardens (Loram et al. 2008; González-García and Sal 2008; Peroni et al. 2016) are underrepresented in the research of urban ecology, although in many growing cities the area they occupy, and their biodiversity may be larger than those of other green areas (Thompson et al. 2003; Gaston et al. 2013). Additionally, the social implications of public and private green areas are different and complementary. Public green areas are surrounded by households, they provide experiences with nature and foster social interactions. By contrast, private gardens provide privacy, freedom, and opportunities for gardening (Coolen and Meesters 2011). Regarding species composition, private gardens represent the personal choices of individuals. For example, in many cases, a wide variety of non-native plants are imported to cities, for landscaping and other horticultural objectives (Reichard and White 2001). Thus, the proportion of exotic species in urban environments is generally much larger among plants compared to other taxonomic groups, such as birds, mammals, reptiles, or amphibians (McKinney 2006).
Cities have socio-economic segregation related to the urban form, the provision of infrastructure, and the value based on location (Lima 2001). Several studies have shown that a higher income in households is related to higher vegetation cover, and higher access to ecosystem services (Flocks et al. 2011), and that education level is also positively correlated with vegetation cover (Heynen and Lindsey 2003). The social, economic, and cultural differences are reflected in the urbanized landscape, and explain the access to social and infrastructure services, affecting the spatial pattern of vegetation in urban ecosystems (Pedlowski et al. 2002; Hope et al. 2003; Pickett et al. 2008; Luck et al. 2009; Clarke et al. 2013; Spescha et al. 2020). In Argentina, it has been observed that when green spaces are a scarce commodity, their distribution is mainly controlled by socioeconomic status; in which, sectors with higher income have increased access to urban vegetation (Spescha et al. 2020). Although these authors did not discriminate between public and private vegetation, much of this pattern is likely explained by the contribution of private gardens since in some cases private gardens account for the main share of urban green spaces (Gaston et al. 2013). It is important to highlight the importance of each type of green urban space to biodiversity: public spaces are managed by a single administrative unit, while private gardens have more diverse management (e.g., Gaston et al. 2013). Thus, since socioeconomic factors affect vegetation complexity, social and cultural factors could be expected to influence species composition and abundance of private gardens (Kinzig et al. 2005).
Private gardens are influenced by sociodemographic factors (Philpott et al. 2020). The socioeconomic level is an important factor that influences the characteristics of the gardens and some studies found significant correlations between the diversity of birds and the socioeconomic level, (Melles 2005), and some ecologists (e.g., Gaston et al. 2007) have tried to quantify the scope of wildlife-friendly gardening in the cities. In Vancouver, Canada, it was observed that the vegetation and landscaping of the gardens located in front of the houses, were more similar between neighbors than those of a different street or neighborhood (Zmyslony and Gagnon 1998). It is important to identify whether the contagion process is due to the spatial distance or the socioeconomic distance since it modifies the spatial structure of urban biodiversity and the spatial replacement of species. Therefore, the analysis of these patterns can be difficult due to the effect of “mimicry” or contagious processes in the structure of gardens, which increase spatial autocorrelation of the considered attributes. In some cases, socio-economic characteristics can directly influence garden management, which is reflected in the heterogeneity of urban landscapes (Martin et al. 2004; Grove et al. 2006; Mennis 2006; Troy et al. 2007).
In this study, we analyze private gardens as socio-ecological components to understand the interactions between socio-economic structure and urban vegetation diversity in private gardens. We hypothesize that people with higher income might dedicate more time and economic resources and might have more access to information and resources for gardening, for which we expect a positive association between socioeconomic level and species richness. However, it is likely that other attributes of the garden, such as age and area also influence richness. Another hypothesis is that due to a “mimicry” process and the exchange of genetic material, the socioeconomic level also determines the identity of the species present in gardens, for which we expect to find an association between socio-economic level and garden species composition.
We analyzed the spatial and non-spatial relation of species composition and socio-economic level in a subtropical agglomerate of Argentina: Gran San Miguel de Tucumán. We characterized plant species diversity and composition through plant censuses in 50 private gardens and a short survey of owners. We evaluated the relationship between plant diversity with a socioeconomic index and the different variables obtained in the surveys to explain the pattern of species distribution. We used multivariate analyses to visualize the similarity patterns of species composition among gardens, and we evaluated whether the emerging pattern of these analyses responds to geographic distance or socioeconomic similarity.
Materials and methods
Study area
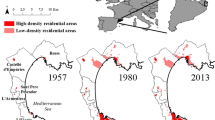
The study area corresponds to the urban agglomerate Gran San Miguel de Tucumán (GSMT) (Fig. 1). GSMT is the main urban center of northwest Argentina, and it is the sixth-largest city in Argentina and the fifth most populated (INDEC 2010). GSMT is located at the foothills of Sierras de San Javier, in a transition area between moist montane forests (Yungas) and dry lowland forests (Chaco), largely modified for agro-industrial activities. The climate is subtropical with a seasonal precipitation regime, leading to warm, rainy summers and cold, dry winters. The mean annual temperature is 19° C and the mean rainfall reaches 1000 annual mm.
The city was founded more than four centuries ago, and it was a pioneer in the sugarcane industry and in the development of railways in Argentina, which led to its constitution as a socioeconomic and urbanization hub. This large urban center concentrates on a population of 800,000 inhabitants (INDEC 2010) and it encompasses five departments: Capital, Cruz Alta, Tafí Viejo, Lules, and Yerba Buena. These departments correspond to administrative units of third order according to the Argentine system. Tucumán has high social inequality compared to other provinces of Argentina (Spescha et al. 2020), which makes it a particularly useful system to evaluate the effects of socio-economic inequities.
Socioeconomic and population data
To spatially characterize the socio-economic level of the GSMT population we used data from the national population census, Instituto Nacional de Estadísticas y Censos de la República Argentina (INDEC). We analyzed the data of the urban agglomerate at the censal radii (CR) level, which is the minimal spatial unit of data aggregation (INDEC, 2010). The size of the CR is defined by the number of households; and each CR has an average of 300 households (INDEC, 2010). Each CR is georeferenced through a polygon that allows working in a Geographic Information System (GIS). To extract information of the censal variables we used the software REDATAM + SP (REcuperación de DATos para Áreas pequeñas por Microcomputador) (INDEC, 2010).
We used the Socioeconomic Status Index (SSI) to characterize the socioeconomic level of each CR (Spescha et al. 2020). This index constitutes a conceptual and quantitative model that summarizes many social and economic aspects to characterize the local population through the material, infrastructure, and household context attributes. The variables used in the index are the maximum educational level obtained by those responsible for households (i.e., primary, secondary, tertiary, and university) and the levels of domestic material quality (i.e., home quality materials and connectivity to basic public services). The educational level achieved by the person in charge of the household is an indicator of occupancy hierarchy, monthly income, and social status, while the characterization of the home reflects its historical value of income and assets. To calculate the SSI, we used the frequency of each category of construction quality and educational level variables and multiplied them by specific factors (Spescha et al. 2020). These factors assigned higher values to the higher construction quality and educational value and ensured that SSI attained a score ranging between 0 and 100. Afterward, the score was categorized into seven groups. Each group (g1, g2, g3, g4, g5, g6, and g7) aggregates RCs units with similar socioeconomic levels, with g1 taking the lowest scores, which indicates the lowest socioeconomic level, and g7 taking the highest scores.
Vegetation Data
We sampled the vegetation of 50 private gardens of GSMT. To select the households, we carried out stratified sampling as a function of the estimated socio-economic level through the SSI. We considered that samples corresponded to gardens of different CR, to encompass all the heterogeneity within the study area. One limitation to performing a strictly stratified sampling was accessibility to households since we considered gardens whose owners had been previously contacted. Thus, although the spatial distribution of gardens encompassed the whole study area, the representativeness of different socioeconomic groups was lower (Fig. 1). After selecting the gardens, we developed a protocol to visit the respective households, including records of the general characteristics of each garden and a short survey to characterize the owners. We filled a spreadsheet with data on the different gardens, with the previous agreement of the owners.
We obtained data on the area of each garden, and we registered the plant species to describe the species composition of each garden. To ensure that all species were recorded we took pictures or parts of the plant in the cases where the species could not be identified in situ, which we later identified through different means (bibliography, flora catalogs, herbaria, etc.). However, certain groups of plants were not considered (e.g., Poaceae), while others were grouped in families (e.g., Cactaceae, Orchidaceae). Species composition allowed us to quantify species richness and diversity considering the diverse life forms: trees, shrubs, herbaceous plants, and climbing plants. The identification of species gave us information about their respective origins, and we distinguished between native (from South America) and exotic species.
We collected data to characterize the owners of the houses through a short survey (Supplementary material), which included questions about the characteristics of the garden and its owners. The variables we used to build the survey were (1) Age of the garden (classified into three categories: less than five years old, between five and fifteen years old, and more than 15 years old), (2) Dedication (time and people) (3) Level of studies reached (4) gender and (5) Age.
As we progressed with sampling in the gardens, we observed that a common behavior among surveyed people was to share opinions about how they obtained certain plants from their gardens, for which we considered it important to include in the survey questions about the origin of the species in their gardens, and how they had acquired them (e.g., through exchange or purchase).
Also, we included questions to evaluate whether people could differentiate between native and non-native species. Both questions were included in the surveys after sampling the fifth garden, for which we obtained answers in 45 out of 50 sampled gardens. These new questions were: What is the way of acquiring the plants of your garden (e.g., Exchange or purchase)? do you identify which species are native and which are not?
Social variables and species richness of gardens
To analyze the relationship between social variables and attributes of private gardens, we carried out linear model analyses between species richness and the following individual variables: age, gender of the person in charge of the garden, maximum level of education reached, time and dedication (hours per week), size of the garden, age of the garden and socioeconomic level (SSI).
Additionally, we analyzed the relation between the size of the garden and socioeconomic level (SSI) through a correlation to observe whether gardens of larger dimensions corresponded to CR with higher levels of SSI.
Analysis of plant species composition in gardens
We performed a multivariate analysis to represent the dissimilarities of species composition among gardens. Multivariate analyses allow summarizing multiple sources of variation in simplified axes. We used the Non-Metric Multidimensional Scaling (NMDS) method, which allows using species presences and absences to calculate dissimilarity using the Bray-Curtis formula. Bray Curtis formula estimates the distance on species composition between each pair of gardens through the use of shared and unshared species. Once the distance matrix among gardens is obtained, the NMDS uses stochastic simulations to find a spatial arrangement in the smaller number of axes, which minimizes the inconsistencies with the observed distances (stress indicator). For that reason, each NMDS run can provide slightly different results. One advantage of the method is that the axes summarize complexity, and their values can be used in subsequent analyses. For this analysis, we considered species that were present in at least five gardens, and we used 96 species (Supplementary material). Once we obtain the best arrangement of the NMDS based on species composition we can plot the species and the garden in this arrangement through the scores they get in each of the axes of the NMDS. These permits comparing the scores of each unit (e.g., species or gardens) through statistical analyses (e.g., ANOVA) or estimating the distance between gardens in the arrangement by calculating their Euclidean distance.
To identify the variable that best explains the distances in garden species composition we compared the adjustment of the obtained distances through the NMDS with the geographic and socioeconomic distances. For this analysis, we used the Mantel correlation (Guillot and Rousset 2013). Mantel tests are adequate for evaluating the correlation between matrices. Their use became popular to evaluate how geographic distances explained structures of other variables considering autocorrelation. In the statistical significance analysis, the Mantel test uses permutations that calculate the probability of obtaining the obtained pattern with a null model. We compared two Mantel tests to determine whether spatial or socio-economic distances among gardens were better predictors of the botanic distances observed in the NMDS. Once each Mantel test was carried out, we evaluated the level of correlation (since they have the same number of observations and parameters) to determine which matrix had higher prediction power. All the analyses of this study were performed using R software (R Core Team 2017).
Results
Garden characteristics
The most variable characteristic of the sampled gardens was their area, with a mean of 407.2 m2 and a range between 50 and 1400 m2. The average age of people in charge of a garden was 53 years; the youngest person was 25 years while the oldest one was 89 years. The time dedicated to gardening varied from one to 13 h per week, with an average of 5.22 hr/per week (Table 1).
Plant composition
We recorded 270 species belonging to 98 families (Table 2). To analyze vegetation structure, we used a simplified life form classification. The most frequent life form corresponded to herbaceous plants, represented by 118 species. Among woody species, composed of trees and shrubs, we registered 92 species. We found 33 climbing species, while other groups of interest such as succulent plants, cacti, and palm trees were less represented. Regarding species origin, exotic species were more frequent, with 165 species, while native species were 105. The plant species that we observed in the sampling of the gardens have mainly ornamental value (aesthetic valuation).
Based on our results we defined older gardens (> 15 years) as the most diverse in plant species. Age and genus (male or female) of the people in charge were not related to species number, and neither was weekly dedication (in hours) to garden maintenance and species richness (Table 2).
Regarding the origin of the species present in gardens, only four of the surveyed people knew whether the species were native or not. Additionally, approximately nine out of ten garden species were obtained through vegetative propagation and seeds, in both cases from relatives or friends.
Representative plant families
Among the 98 identified families, the most representative were Asteraceae, conformed by 16 species: Asparagaceae and Lamiaceae, with 11 species, and Araceae with 10. Other families represented by more than seven species were Bignoniaceae, Oleaceae, Apocynaceae, Arecaceae, Cactaceae, Euphorbiaceae, and Fabaceae. There were also other families represented by less than seven species (e.g., Rutaceae, Myrtaceae, Begoniaceae, Solanaceae), and many families were represented by only one species (e.g., Amaranthaceae, Buxaceae, Portulacaceae, Violaceae).
Results of the correlation analyses between species richness and socioeconomic variables
The correlation analyses (Table 3) show that SSI was significantly related to garden area (r = 0.56, p < 0.001), with larger gardens occurring in CR with higher SSI. (r = 0.41, p < 0.01). A similar result was registered regarding the SSI and species richness (r = 0.41, p < 0.01). The time dedicated to the garden was associated with the SSI (r = 0.32, p < 0.1).
NMDS results
Two axes of the NMDS were enough to summarize the variation of the dissimilarity matrix based on the species composition of the gardens. None of the two axes of the spatial ordination discriminated between native and exotic species or by their life forms. Neither did we observe a spatial pattern responding to these two attributes (Supplementary material).
By contrast, there was an evident spatial pattern of socio-economic groups in the gardens although the socio-economic information was not included in the performance of the NMDS. For example, when gardens were labeled with their socioeconomic level, extreme groups showing a restricted distribution in the bi-dimensional space were observed (e.g., g2, g3 y g7). By contrast, g5, a group of intermediate socio-economic conditions presented a more homogeneous distribution in the bi-dimensional NMDS space and functioned as a link among groups.
The variable that best explained distances in garden species composition was socio-economic distance (r = 0.28) while the influence of spatial distance was weaker (r = 0.08). These are the correlation results obtained from the two Mantel tests aiming at determining whether spatial distance or socio-economic distance among variables had a higher capacity to explain the distance among species observed in the NMDS (Fig. 2). (Acronyms for each species in Supplementary material)
Discussion
Our study explores an aspect that has been scarcely considered in the literature, namely the influence of spatial socio-economic structure on the species composition of private gardens. Our results suggest that species composition plays different roles in structuring social links. On the one hand, species composition may be an indicator of membership in certain socio-economic groups, and, on the other hand, plant sharing (through the interchange of propagules) seems to consolidate social links. It is important to highlight that humans have the unique ability to accumulate culture (Dean et al. 2013), and particularly the use of plants for a wide variety of benefits is passed between groups and from generation to generation (Salali et al. 2016). The patterns found in our study suggest that garden species composition responds to socio-economic conditions, which is probably linked to the exchange of genetic material as a mechanism to strengthen social bonds. Although this type of analysis is not frequent, a similar pattern was found in Burundi (Bigirimana et al. 2012) where garden orientation was structured mostly as a function of socioeconomic level, being utilitarian among lower socio-economic levels, and ornamental among higher socio-economic levels. In contrast, according to our results, in Argentina garden orientation is related to aesthetic appraisal, but the effect of plant species tenure on the socio-economic structure is maintained.
The tradition of exchanging plants with the closest people generates particular spatial patterns of species distribution within urban environments. Therefore, it is necessary to differentiate which are the mechanisms of knowledge exchange and the relationship with the structure of society. (Díaz Reviriego et al. 2016). Our analyses show that exchange between people is the main method to obtain plants. Species composition of gardens arises from such exchange and is defined mainly by belonging to a similar socio-economic rather than to neighborhood or geographic distance. Thus, it is likely that interpersonal relationships in the different socioeconomic sectors significantly affect patterns that structure gardens, thus reinforcing socio-economic segregation within cities. It is also likely that urban plant communities tend to follow the socio-economic polarization process observed in GSMT between 1991 and 2010 (Zamora and Rivas 2017). One alternative in cities with sharp polarities, such as GSMT, could be the reinforcement of existing patches, considering that patches with contrasting communities could increase habitat for a higher diversity of associated species, such as birds (Haedo et al. 2017).
According to our analysis, gardens located in sectors with higher socioeconomic levels are larger, and this implies higher plant species richness. This result is like other studies in which larger gardens presented higher species diversity (e.g., Bernholt et al. 2009). The positive association between time spent in the garden and SSI could be due to the possibility of high-income groups hiring gardeners to take care of the garden.
Our result is consistent with results obtained in other studies showing that higher socio-economic levels have higher access to green spaces and ecosystem services (Flocks et al. 2011; Schwarz et al. 2015; Escobedo et al. 2015; Fernández and Wu 2016). Particularly in Argentina, it has been suggested that when property costs increase in densely populated cities, groups with fewer resources reach higher environmental quality (Spescha et al. 2020). Regarding species richness and garden socioeconomic level, we did find a significant relation, which implies that vegetation in CS with higher SSI was more diverse. These results are consistent with those from a study in New Zealand (van Heezik et al. 2013), in which positive associations between socioeconomic level and plant diversity, and between garden size and plant and bird diversity were found.
Regarding the diversity of life forms registered in our sampling, herbaceous plants were the most representative, possibly because cuttings of herbaceous plants (commonly named “gajos” in Argentine Spanish language) are easily shared and successfully propagated. Additionally, herbaceous species occupy less space, while trees, on the other hand, require a larger area. Thus, lower socio-economic groups might own a smaller number of trees, since based on our results garden area was positively related to socioeconomic level.
The origin of species in urban gardens poses different challenges for the conservation of biodiversity. On the one hand, private gardens contribute to maintaining a considerably high diversity of species. On the other hand, the proliferation of aesthetic-oriented species favors the introduction of exotic species, which can disperse in the urban and surrounding landscapes becoming invasive (Dehnen-Schmutz and Touza 2008; Marco et al. 2008; Powell and Aráoz 2018, Jiménez et al. 2021). The patterns of the richness of exotic species currently observed in the gardens may be more related to human activities of the past than to contemporary actions (Essl et al. 2010). In our study, 62% of the identified species in gardens were nonnative species, and most of the surveyed people were not able to identify native or exotic species, which represents a risk to the environment. Thus, we consider it important to reinforce environmental education and to explain the potential effects of exotic species on the surrounding environment. Further, it should be highlighted that certain exotic plants can become invasive, with drastic consequences for biodiversity and ecosystem functioning (Pimentel et al. 2005). Thus, environmental awareness is a key factor to strike a balance in urban environments.
Urban vegetation is essential to enhance the quality of life of urbanites through the provision of ecosystem services. Environmental justice, the equitation access to the benefits of a healthy environment, might prevent the exacerbation of socioeconomic differences (Spescha et al. 2020). Our results suggest that although private gardens may provide some ecosystem services to the entire society (e.g., temperature regulation, improvement of air quality), others are privative to the most powerful socio-economic groups (e.g., experiences with nature). In the context of diminishing urban vegetation and increasing distance to natural environments (Paolini et al. 2016) private gardens may overcome a historical trend of isolation from natural environments. At this moment, it may be important to involve not only private owners, but the States and the whole society in designing and conserving green spaces open to everyone, which may prevent the proliferation of social silos (Verma et al. 2020). A strategy could imply some incentives for private owners to conserve a proportion of green spaces open to society. In this way, not only the ecosystem services could benefit diverse groups of society, but they would benefit from social interactions between different groups, promoting a more cohesive society.
Using a novel approach, we described the association between socio-economic level and plant species composition in an urban system. However, it is important to mention certain limitations of our study, such as the sample size, which is limited due to the effort involved in sampling each garden. However, studies carried out in countries with more resources had a similar sample size (e.g., van Heezik et al. 2013). On the other hand, most of these studies are carried out in one city, and at least one study addressing similar questions reached similar results (Bigirimana et al. 2012). However, protocols to study this process in other contexts and to replicate our study in other sites should be designed to understand how gardens are structured in different cities. Finally, certain plant groups were more difficult to identify, but in general, they involved particular species that were not very relevant to their owners (e.g., grass/Poaceae). Other groups were identified only at the general level, such as Rosaceae and Orchidaceae.
Conclusions
We can conclude that, regarding factors modeling and structuring plant species composition in gardens of GSMT, garden age conditioned species richness, which might reflect the tradition of exchanging and “accumulating” plant species by their owners. This seems to be associated with belonging to a certain socioeconomic level rather than spatial distance. Thus, urban vegetation seems to provide an additional social service associated with the strengthening of social links. This service, usually neglected in the ecosystem services literature, is key to generating a more cohesive and resilient society. We think that different actors should be involved not only in preventing the loss of green spaces, associated with urban expansion and de-intensification but in increasing environmental justice by ensuring open access to a proportion of private gardens. We consider that this study is an archetype that could be applied and adapted to different spatial scales and sites. This type of project could contribute to ongoing strategies regarding the management of public green spaces through the knowledge of plant patterns in private gardens of a given territory.
Availability of data and material
The authors declare the transparency of the data and material.
Code Availability
Not applicable.
References
Avolio ML, Pataki DE, Trammell TL, Endter-Wada J (2018) Biodiverse cities: the nursery industry, homeowners and neighborhood differences drive urban tree composition. Ecol Monogr 88:259–276. https://doi.org/10.1002/ecm.1290
Bernholt H, Kehlenbeck K, Gebauer J, Buerkert (2009) A plant species richness and diversity in urban and peri-urban gardens of Niamey. Niger Agroforest Syst 77:159. https://doi.org/10.1007/s10457-009-9236-8
Bigirimana J, Bogaert J, Cannière CD, Bigendako M, Parmentier I (2012) Domestic garden plant diversity in Bujumbura, Burundi: role of the socio-economical status of the neighborhood and alien species invasion risk. Landsc Urban Plann 107(2):118–126. https://doi.org/10.1016/j.landurbplan.2012.05.008
Boone CG, Buckley GL, Grove JM, Sister C (2009) Parks and People: an Environmental Justice Inquiry in Baltimore, Maryland. Ann Assoc Am Geogr 99(4):767–787. https://doi.org/10.1080/00045600903102949
Chiesura A (2004) The role of urban parks for the sustainable city. Landsc Urban Plann 68(1):129–138. https://doi.org/10.1016/j.landurbplan.2003.08.003
Clarke LW, Jenerette GD, Davila A (2013) The luxury of vegetation and the legacy of tree biodiversity in Los Angeles, CA. Landscape Urban Plann. 116:48–59. https://doi.org/10.1016/j.landurbplan.2013.04.006
Coolen H, Meesters J (2011) Private and public green spaces: meaningful but different settings. J Hous Built Environ 27(1):49–67. https://doi.org/10.1007/s10901-011-9246-5
Crossette B Estado de la población mundial (2011) 7 mil millones de personas, su mundo, sus posibilidades, Fondo de Población de las Naciones Unidas UNFPA, New York. https://www.unfpa.org/sites/default/files/pub-pdf/SP-SWOP2011_Final.pdf
Dean LG, Vale GL, Laland KN, Flynn E, Kendal RL (2013) Human cumulative culture: a comparative perspective. Biol Rev 89(2):284–301. https://doi.org/10.1111/brv.12053
Díaz-Reviriego I, González-Segura L, Fernández-Llamazares Á, Howard PL, Molina JL, Reyes-García V (2016) Social organization influences the exchange and species richness of medicinal plants in amazonian homegardens. Ecol Soc 21(1). https://doi.org/10.5751/es-07944-210101
Dobbs C, Nitschke C, Kendal D (2017) Assessing the drivers shaping global patterns of urban vegetation landscape structure. Sci Total Environ 592:171–177. https://doi.org/10.1016/j.scitotenv.2017.03.058
Escobedo FJ, Clerici N, Staudhammer CL, Corzo GT (2015) Socio-ecological dynamics and inequality in Bogotá, Colombia’s public urban forests and their ecosystem services. Urban For Urban Greening 14(4):1040–1053. https://doi.org/10.1016/j.ufug.2015.09.011
Essl F, Dullinger S, Rabitsch W, Hulme PE, Hülber K, Jarošík V, Pyšek P (2010) Socioeconomic legacy yields an invasion debt. PNAS 108(1):203–207. https://doi.org/10.1073/pnas.1011728108
Fernández IC, Wu J (2016) Assessing environmental inequalities in the city of Santiago (Chile) with a hierarchical multiscale approach. Appl Geogr 74:160–169. https://doi.org/10.1016/j.apgeog.2016.07.012
Flocks J, Escobedo F, Wade J, Varela S, Wald C (2011) Environmental Justice Implications of Urban Tree Cover in Miami-Dade County. Fla Environ Justice 4(2):125–134. https://doi.org/10.1089/env.2010.0018
Freeman C, Dickinson KJ, Porter S, Heezik YV (2012) My garden is an expression of me”: exploring householders’ relationships with their gardens. J Environ Psychol 32(2):135–143. https://doi.org/10.1016/j.jenvp.2012.01.005
Gaston KJ, Fuller RA, Loram A, Macdonald C, Power S, Dempsey N (2007) Urban domestic gardens (XI): variation in urban wildlife gardening in the United Kingdom. Biodivers Conserv 16(11):3227–3238. https://doi.org/10.1007/s10531-007-9174-6
Gaston KJ, Ávila-Jiménez ML, Edmondson JL (2013) REVIEW: managing urban ecosystems for goods and services. J Appl Eco 50(4):830–840. https://doi.org/10.1111/1365-2664.12087
Goddard MA, Dougill AJ, Benton TG (2010) Scaling up from gardens: Biodiversity conservation in urban environments. Trends Ecol Evol 25(2):90–98. https://doi.org/10.1016/j.tree.2009.07.016
González-García A, Sal AG (2008) Private urban greenspaces or “Patios” as a key element in the Urban Ecology of Tropical Central America. Hum Ecol 36(2):291–300. https://doi.org/10.1007/s10745-007-9155-0
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the Ecology of Cities. Science 319(5864):756–760. https://doi.org/10.1126/science.1150195
Grove JM, Troy AR, O’Neil-Dunne JP, Burch WR, Cadenasso ML, Pickett ST (2006) Characterization of households and its implications for the Vegetation of Urban Ecosystems. Ecosystems 9(4):578–597. https://doi.org/10.1007/s10021-006-0116-z
Guillot G, Rousset F (2013) Dismantling the mantel tests. Methods Ecol Evol 4(4):336–344. https://doi.org/10.1111/2041-210x.12018
Haedo J, Gioia A, Aráoz E, Paolini L, Malizia A (2017) Primary productivity in cities and their influence over subtropical bird assemblages. Urban For Urban Greening 26:57–64. https://doi.org/10.1016/j.ufug.2017.04.017
Heynen NC, Lindsey G (2003) Correlates of Urban Forest Canopy Cover. Public Works Manag Policy 8(1):33–47. https://doi.org/10.1177/1087724x03008001004
Hope D, Gries C, Zhu W, Fagan WF, Redman CL, Grimm NB, Nelson A, Martin C, Kinzig A (2003) Socioeconomics drive urban plant diversity. PNAS 100(15):8788–8792. https://doi.org/10.1073/pnas.1537557100
Jiménez YG, Aráoz E, Grau HR, Paolini L (2021) Linking forest transition, plant invasion and forest succession theories: socioeconomic drivers and composition of new subtropical andean forests Landscape Ecol. 36:1161–11764
Kinzig AP, Warren P, Martin C, Hope D, Katti M (2005) The Effects of Human Socioeconomic Status and Cultural characteristics on urban patterns of Biodiversity. Ecol Soc 10(1). https://doi.org/10.5751/es-01264-100123
Lima JJ (2001) Socio-spatial segregation and urban form: Belem at the end of the 1990s. Geoforum 32(4):493–507. https://doi.org/10.1016/s0016-7185(01)00019-7
Loram A, Tratalos J, Warren PH, Gaston KJ (2007) Urban domestic gardens (X): the extent & structure of the resource in five major cities Landscape Ecol. 22:601–615. https://doi.org/10.1007/s10980-006-9051-9. 4
Loram A, Thompson K, Warren PH, Gaston KJ (2008) Urban domestic gardens (XII): the richness and composition of the flora in five UK cities. J Veg Sci 19(3):321–330. https://doi.org/10.3170/2008-8-18373
Luck GW, Smallbone LT, O’Brien R (2009) Socio-Economics and Vegetation Change in Urban Ecosystems: patterns in space and time. Ecosyst 12(4):604–620. https://doi.org/10.1007/s10021-009-9244-6
Marco A, Dutoit T, Deschamps-Cottin M, Mauffrey J, Vennetier M, Bertaudière-Montes V (2008) Gardens in urbanizing rural areas reveal an unexpected floral diversity related to housing density. C R Biol 331(6):452–465. https://doi.org/10.1016/j.crvi.2008.03.007
Martin CA, Warren PS, Kinzig AP (2004) Neighborhood socioeconomic status is a useful predictor of perennial landscape vegetation in residential neighborhoods and embedded small parks of Phoenix, AZ. Landsc Urban Plann 69(4):355–368. https://doi.org/10.1016/j.landurbplan.2003.10.034
Mckinney ML (2006) Correlated non-native species richness of birds, mammals, herptiles and plants: Scale Effects of Area, Human Population and native plants. Biol Invasions 8(3):415–425. https://doi.org/10.1007/s10530-005-6418-9
Melles S (2005) Urban bird diversity as an indicator of human social diversity and economic inequality in Vancouver, British Columbia, Department of Zoology, University of Toronto, Toronto, Ontario, Canada. https://www.eaglehill.us/urban-habitats/Vol%203/vancouver_pdf.pdf
Mennis J (2006) Socioeconomic-vegetation Relationships in Urban residential land. Photogramm Eng Remote Sens 72(8):911–921. https://doi.org/10.14358/pers.72.8.911
Paolini L, Aráoz E, Gioia A, Powell PA (2016) Vegetation productivity trends in response to urban dynamics. Urban For Urban Greening 17:211–216
Pedlowski MA, Silva VA, Adell JJ, Heynen NC (2002) Urban for environmental inequality in Campos dos Goytacazes, Rio de Janeiro, Brazil. Urban Ecosyst 6(1/2):9–20. https://doi.org/10.1023/a:1025910528583
Peroni N, Hanazaki N, Begossi A, Zuchiwschi E, Lacerda VD, Miranda TM (2016) Homegardens in a micro-regional scale: contributions to agrobiodiversity conservation in an urban-rural context. Ethnobiol Conserv. https://doi.org/10.15451/ec2016-8-5.6-1-17
Philpott SM, Egerer MH, Bichier P, Cohen H, Cohen R, Liere H, Jha S, Li BB (2020) Gardener demographics, experience, and motivations drive differences in plant species richness and composition in urban gardens. Ecol Soc 25(4):8. https://doi.org/10.5751/ES-11666-250408
Pickett ST, Cadenasso ML, Grove JM, Groffman PM, Band LE, Boone CG, Wilson MA (2008) Beyond Urban Legends: an emerging Framework of Urban Ecology, as Illustrated by the Baltimore Ecosystem Study. Bioscience 58(2):139–150. https://doi.org/10.1641/b580208
Pimentel D, Zuniga R, Morrison D (2005) Update on the environmental and economic costs associated with alien-invasive species in the United States. Ecol Econ 52(3):273–288. https://doi.org/10.1016/j.ecolecon.2004.10.002
Powell PA, Aráoz E (2018) Biological and environmental effects on fine-scale seed dispersal of an invasive tree in a secondary subtropical forest. Biol Invasions 20(2):461–473
Power ER (2005) Human–nature relations in Suburban Gardens. Aust Geogr 36(1):39–53. https://doi.org/10.1080/00049180500050847
Reichard SH, White P (2001) Horticulture as a pathway of Invasive Plant Introductions in the United States. Bioscience 51(2):103. https://doi.org/10.1641/0006-3568(2001)051[0103:HAAPOI]2.0.CO;2
Rudd H, Vala J, Schaefer V (2002) Importance of Backyard Habitat in a Comprehensive Biodiversity Conservation Strategy: a connectivity analysis of Urban Green Spaces. Restor Ecol 10(2):368–375. https://doi.org/10.1046/j.1526-100x.200202041.x
Salali G, Chaudhary N, Thompson J, Grace O, Van Der Burgt X, Dyble M, Migliano A (2016) Knowledge-sharing networks in Hunter-Gatherers and the evolution of Cumulative Culture. Curr Biol 26(18):2516–2521. https://doi.org/10.1016/j.cub.2016.07.015
Schwarz K, Fragkias M, Boone CG, Zhou W, Mchale M, Grove JM, Cadenasso ML (2015) Trees grow on money: Urban Tree Canopy Cover and Environmental Justice. PLoS ONE 10(4). https://doi.org/10.1371/journal.pone.0122051
Spescha V, Paolini L, Powell PA, Covaro B, Elías D, Aráoz E (2020) Unequal Appropriation of Urban Vegetation in Argentine Cities. Ecosyst. doi:https://doi.org/10.1007/s10021-019-00476-5
Thompson K, Austin KC, Smith RM, Warren PH, Angold PG, Gaston KJ (2003) Urban domestic gardens (I): Putting small-scale plant diversity in context. J. Veg. Sci, 14(1), 71–78. doi:https://doi.org/10.1111/j.1654-1103. 2003.tb02129.x
Troy AR, Grove JM, O’Neil-Dunne JP, Pickett ST, Cadenasso ML (2007) Predicting Opportunities for Greening and patterns of vegetation on private urban lands. Environ Manage 40(3):394–412. https://doi.org/10.1007/s00267-006-0112-2
Van Heezik YV, Freeman C, Porter S, Dickinson KJ (2013) Garden size, Householder Knowledge, and Socio-Economic Status Influence Plant and Bird Diversity at the scale of Individual Gardens. Ecosyst 16(8):1442–1454. https://doi.org/10.1007/s10021-013-9694-8
Verma P, Singh R, Bryant C, Raghubanshi AS (2020) Green space indicators in a social-ecological system: a case study of Varanasi, India. Sustain Cities Soc. https://doi.org/10.1016/j.scs.2020.102261
Zamora AE, Rivas JJ (2017) La evolución del mapa social del Gran San Miguel de Tucumán. Un estudio del periodo 1991 a 2010 a través del análisis factorial. Geograficando 13(2):027. https://doi.org/10.24215/2346898xe027
Zmyslony J, Gagnon D (1998) Residential management of urban front-yard landscape: a random process? Landsc Urban Plann 40(4):295–307. https://doi.org/10.1016/s0169-2046(97)00090-x
United Nations, Department of Economic and Social Affairs, Population Division. World Urbanization Prospects: The 2018 Revision, Methodology. Report No. ESA/P/WP.252 (2018)
Acknowledgements
We thank Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) Argentina. We gratefully acknowledge the collaboration of Ana Levy Hynes, from “Jardín Botánico de la Fundación Miguel Lillo”, Facultad de Ciencias Naturales, Universidad Nacional de Tucumán (UNT), Tucumán, Argentina; and Dr. Alfredo Grau, professorship ” Diversidad Vegetal III”, Facultad de Ciencias Naturales, Universidad Nacional de Tucumán (UNT), Argentina.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
Verónica Spescha: conceptualization, methodology, formal analysis, investigation, resources, writing original draft, review & editing, visualization, supervision. Ezequiel Aráoz: methodology, formal analysis, writing - review & editing, project administration.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
The authors’ consent to Participate.
Consent for publication
The author’s consent to Publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Spescha, V., Aráoz, E. Socio-economic differences control species composition of urban gardens in a metropolitan area of Argentina. Trop Ecol 65, 142–151 (2024). https://doi.org/10.1007/s42965-023-00293-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42965-023-00293-x