Abstract
Urban and peri-urban agriculture (UPA) significantly contributes to food and nutritional security of urban dwellers in many African countries. Economic and demographic pressures often lead to transformation of subsistence-oriented traditional homegardens into commercial production units. Such transformation is claimed to result in decreasing plant diversity, particularly of local species. A study was therefore undertaken in 51 gardens of Niamey, Niger, to assess the factors determining plant diversity and the suitability of UPA for in situ conservation of plant genetic resources. In each garden, the number and abundance of all human-used plant species were determined, and species density, Shannon index and Shannon evenness were calculated. In the 51 surveyed gardens, a total of 116 plant species were cultivated, most of them for the production of fruits or vegetables. Annual vegetables dominated, particularly exotic species grown for sale. In the cold season, an average of 14 species were cultivated per garden, the Shannon index was 0.96 and evenness was 0.39. Commercial gardens had a species richness similar to that of subsistence gardens, but a lower evenness (P < 0.005), caused by the dominance of a few vegetable species. Gardens of immigrants had a lower Shannon index than those of members of the local Djerma ethnic group. Stepwise multiple regression analysis showed significant influence of various variables on plant species richness and diversity parameters: garden size (richness and Shannon index), ethnicity of the gardener (richness and evenness), gender of the gardener and cash-oriented production (evenness), household size (richness) and garden possession status (Shannon index). Cluster analysis revealed the existence of five garden types. The highest species richness and diversity, particularly of perennial and local species, was found in large, peri-urban, commercial gardens managed by relatively wealthy, elderly gardeners with large families and a regular non-agricultural income.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Niger, whose agricultural area is largely limited to the semi-arid southern Sahelian zone, is the world’s poorest country and recurrently affected by periods of famine (USAID 2002). With its approximately 900,000 inhabitants, the country’s capital Niamey provides an ideal example to study the intensive use of open space by urban and peri-urban agriculture (UPA). Due to the high birthrate and the arrival of migrants from rural areas triggered by low soil fertility, erratic rainfall and poor infrastructure, the city has experienced, over recent years, a high population growth rate (5.3% per year; USAID 2002), leading to a continuous increase in food demand. It is in such a context that, all over Africa, UPA has become an increasingly important activity for improving the quality and quantity of food intake (Maxwell et al. 1998; Bryld 2003). Worldwide, UPA is estimated to produce as much as one-seventh of the total food supply (Drescher 1998). African cities such as Bamako (Mali) or Lubumbashi (Democratic Republic of Congo) are reportedly self-supporting in the supply of vegetables through UPA (Tambwe 2006).
A ‘homegarden’ is generally described as an agroforestry system nearby the gardener’s house, comprising a mixture of annual and perennial crops in several strata and often largely directed towards fulfilling subsistence needs (Nair and Kumar 2006). Nevertheless, some authors refer to mixed species gardens as homegardens even if they are not around the homestead, which is particularly the case in urban areas, due to land scarcity, and in semi-arid regions, where their vicinity to water sources is very important for irrigation (Niñez 1985; Linares 2004; Drescher et al. 2006). In addition, the presence of perennials may be limited under arid and semi-arid conditions (Ceccolini 2002; Gebauer et al. 2007) and urban settings due to land scarcity (Wiersum 2006), water shortages (Thaman et al. 2006) and insecure land tenure (Linares 2004). Thus, urban homegardens in semi-arid regions may not fit the ‘classical’ definition of homegardens, although they could have a similar function as household-based small production units (Drescher et al. 2006). While gardens in an urban setting may also fulfill subsistence needs, they are often heavily market oriented. To avoid confusion in terminology, we use the more general term ‘garden’ instead of ‘homegarden’ in our study. However, given their importance in the literature, we often refer to ‘classical’ homegardens for comparison.
In rural settings, homegardens typically offer a diverse range of products, such as fruits, vegetables, spices, medicine, forage and fuel. The often very high diversity of species in such gardens reportedly reduces the risk of infestation by pests or diseases, offers long-term stability of yields and efficient use of resources and makes year-round availability of crops possible, even under semi-arid conditions (Soemarwoto and Conway 1992; Torquebiau 1992). However, species diversity in homegardens is often very dynamic and largely influenced by different socioeconomic and agroecological factors (Wiersum 2006; Kehlenbeck et al. 2007). Homegardens are often described as sustainable, although quantitative support for this ill-defined statement is rare (Kehlenbeck and Maass 2006). This argument is partly based on the claim that the sustainability of man-made agroecosystems increases with their plant diversity (Soemarwoto and Conway 1992; Torquebiau 1992), which also leads to an improved nutritional status of households managing species-rich systems as compared with species-poor ones (Davis et al. 1994). Homegardens are also regarded as an important land use system for in situ conservation of plant genetic resources (Trinh et al. 2003; Eyzaguirre and Linares 2004), particularly of local species such as indigenous leafy vegetables, which are better adapted to local agroecological conditions (Drescher 1998) and said to have a higher nutritional value than exotic leafy vegetables. Leaves of Gynandropsis gynandra, for example, contain about 80 times more iron than those of spinach (Spinacia oleracea) (Smith et al. 1996). However, in most urban areas, constant economic and demographic pressure as well as high market demand lead to transformation of traditional homegardens towards ornamentalisation or commercialisation (Abdoellah et al. 2006; Wiersum 2006; Kehlenbeck et al. 2007). The related cultural and socioeconomic changes may result in decreasing plant diversity (especially of local species) in the gardens, dominance of a few exotic species/improved varieties for cash crop production, impoverishment of dietary diversity of gardeners’ households or loss of tribal culture and indigenous knowledge (Soemarwoto and Conway 1992; Fundora Mayor et al. 2004; Tesfaye Abebe et al. 2006).
Most available studies of homegardens are from humid areas of tropical Asia and Mesoamerica (Karyono 1990; Soemarwoto and Conway 1992; Trinh et al. 2003; Blanckaert et al. 2004; Kehlenbeck and Maass 2004; Albuquerque et al. 2005; Abdoellah et al. 2006; Peyre et al. 2006), whereas reports from Africa, particularly from urban areas or arid/semi-arid regions, are very limited (Drescher 1998; Linares 2004; Gebauer 2005). Additionally the large majority of the available studies remain rather descriptive, and quantitative methods including multivariate statistics to unravel causes of biodiversity patterns are rarely used (Blanckaert et al. 2004; Kehlenbeck et al. 2007). For urban and rural gardens in the sub-Saharan country of Niger, such information is lacking. The main objectives of this study were therefore to quantify plant diversity in urban and peri-urban gardens of Niamey (Niger) and to analyse the effects of garden and household size, market orientation, ethnic affiliation and gender of the gardener on plant diversity in these gardens. In addition, the study aimed to classify gardens according to species abundance and to assess their suitability for in situ conservation of plant genetic resources.
Materials and methods
Study area
The study was conducted in urban and peri-urban gardens of Niamey (13°31′17” N, 2°6′19” E), the capital of Niger. The typical Sahelian climate at this location can be divided into three distinct periods: the cold season (October to February, no precipitation), the hot season (March to May, no precipitation) and the rainy season (June to September). Mean temperatures are 27°C during the cold, 33°C during the hot and 29°C during the rainy season; the mean annual rainfall amounts to 540 mm (WMO 2007). The rural agriculture in Niger is mainly dominated by fields sown to pearl millet (Pennisetum glaucum) and sorghum (Sorghum bicolor) sparsely relay-cropped with cowpea (Vigna unguiculata), complemented by extensive livestock husbandry. In and around Niamey, intensive horticulture mixed with millet cropping and meat, milk and egg production destined to satisfy the local markets is an important activity (Graefe et al. 2008). The Niger River crosses Niamey and constitutes the most important water source for irrigated horticulture. The native and largest ethnic group in Niamey are the Djerma, but Haussa, Tuareg, Peul and Kanouri are also common (INS 2008).
Data collection
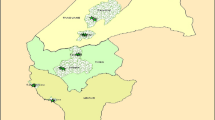
Fieldwork was conducted from January to August 2007 in ten districts of Niamey, including urban and peri-urban ones (Fig. 1), which were chosen based on an earlier study of UPA by Graefe et al. (2008). In these districts, 13 gardens briefly described by Graefe et al. (2008) were selected, to which 38 gardens were added using a ‘snowball’ sampling method for selection. These 51 gardens, comprising 29 urban and 22 peri-urban ones, and covering a total area of 4.4 ha, were surveyed in the three different seasons of the year, except in August (rainy season) where only 45 gardens were available because six gardeners withdrew their participation. Garden sizes were determined using a differential GPS receiver (Global Positioning System; Trimble Pathfinder Pro XR; Westminster, CO, USA). Gardeners were individually interviewed to gather basic socioeconomic data (origin, ethnic affiliation, profession and income sources of the gardener, household size, number and size of land holdings) and garden-related information (plant uses, portion of sold products, use of fertilisers, pesticides and wage laborers, among others). According to Abdoellah et al. (2006), a garden was defined as commercial if more than 50% of its produce was sold and as non-commercial otherwise.
Quickbird satellite image (Google Earth Pro, Google Inc., Mountain View, CA, USA) of Niamey (Niger). The white dots indicate the locations of 29 gardens surveyed in six urban districts in 2007, the white triangles those of 22 gardens in four peri-urban districts. Numbers in brackets show the number of gardens per district
In all gardens, the useful plant species (including ornamentals) and their abundances were determined, whereby weeds were excluded. All species were recorded with their local and scientific names. Species were identified according to Peyre de Fabregues (1979) and Arbonnier (2000) following the nomenclature of Hanelt and the Institute of Plant Genetics and Crop Plant Research (2001) and of Zander et al. (2002) or, if not available there, of the Missouri Botanical Garden (2007). According to the mentioned literature, species originating from sub-Saharan Africa were regarded as ‘local’, and those from other regions as ‘exotic’. As suggested by Kehlenbeck and Maass (2004) and Tesfaye Abebe et al. (2006), all recorded plant species were assigned to the different main use categories fruit, vegetable, stimulant, spice, medicine, staple, wood and multipurpose use (MPU), ornamental or other uses, according to gardeners’ information and the literature used for species identification. For analysis of the vertical vegetation structure, all species were assigned to the ground layer (<1 m), the intermediate layer (1–5 m) or the top layer (>5 m).
Data analysis
Species density was determined as the estimated number of species in a 1,000 m² garden, in order to remove some of the size effects when comparing gardens of different types. Following Evans et al. (1955), we assumed that species number was proportional to log(area + 1). While this species-area curve is only an empirical relationship found in some natural systems, it provides a first approximation to allow for comparisons despite the large differences in garden sizes encountered in this study. In addition, the Shannon index (H′) and the Shannon evenness index (E) were calculated to assess species diversity of the gardens (McCune et al. 2002). To describe and compare the importance of different plant use categories per garden, the summed dominance ratio (SDR) was computed (McCune et al. 2002). First, the relative densities and relative frequencies were used to calculate the SDR for each species, followed by summing up the single SDR values of all species within each of the use categories.
For statistical analysis, SPSS (version 12.0) and MVSP (Multi-Variate Statistical Package, version 13.3p, Kovach Computing Services; Anglesey, Wales, UK) were used. T-tests were used to compare means of two groups; for more than two groups, one-way analysis of variance (ANOVA) followed by post hoc Tukey tests were performed. To analyse temporal changes of diversity between the seasons, paired-samples T-tests were applied. Linear bivariate relations between variables were analysed using Pearson’s correlation coefficients for metric variables and Spearman’s correlation coefficients r S for ordinal variables. Multiple linear regression analysis was performed to identify factors determining plant species richness and diversity using the stepwise method (Backhaus et al. 2006). For these analyses, dependent variables were overall plant species richness, richness of local species, overall diversity expressed by the Shannon index and Shannon evenness index. The independent variables included garden size, characteristics of the gardener and his/her household, and socioeconomic features.
To classify the gardens according to their plant species composition, a hierarchical cluster analysis was carried out based on ln-transformed species abundance data (McCune et al. 2002). For cluster analysis, squared Euclidean distances and the ‘minimum variance’ method in MVSP were applied (Backhaus et al. 2006). To define the correct number of different clusters, the ‘elbow’ criterion was used (Backhaus et al. 2006). Stepwise discriminant analysis was carried out to analyse whether the clusters differed significantly from each other and to determine the species mainly responsible for the separation (McCune et al. 2002).
Results
The 51 surveyed gardens harbored a relatively high but very variable richness and diversity of useful plants. Garden sizes ranged from 37 to 10,355 m² with a mean of 860 m² (Table 1). Almost 70% of the gardeners owned the piece of land they cultivated, but only one lived within his garden. More than 80% of the respondents reported cultivating their gardens mainly for commercial production. Rented gardens were all commercial. Nearly half of the gardens surveyed were managed by the native Djerma ethnic group, whose market orientation was less pronounced than that of other ethnic groups (Table 1). The gardens managed by migrants such as Guarmanché and Mossi from Burkina Faso were highly commercial.
Of all gardeners, 43% reported to have in addition to gardening a supplementary income within their family, such as trade or a retirement pension. One-quarter of those gardeners managing commercial gardens reported to have employees for gardening, often including child labor. The 11 gardens managed by women, all of them Djerma, were smaller than those managed by men, with mean sizes of 312 and 1,010 m², respectively. While 64% of the gardens managed by women were subsistence gardens, 95% of those managed by men were commercial ones.
For irrigation, mainly water from the Niger River, but also from small brooklets and wells, partly equipped with pumps, was used. Nearly all gardeners reported to regularly apply commercial pesticides for plant protection as well as mineral fertilisers, but organic fertilisers were also frequently used. The latter was mostly farmyard manure from their own livestock that was kept by 51% of the respondents.
Species composition, richness and diversity
In total, 116 different plant species from 50 families were cultivated in the 51 surveyed gardens, 71% of which were exotic and 47% were woody perennials (Appendix, Table 7). Most of the cultivated species were used as fruits (33 spp.) and vegetables (31 spp.). For about 30% of the plants with a non-medicinal main use, gardeners mentioned medicine as a secondary use (Appendix, Table 7). However, not all respondents, especially from the younger generations, had retained the traditional knowledge about medicinal plants and their uses. Ornamentals were only rarely planted (Appendix, Table 7).
In the cold season, a total of 115 species were found (Table 2); the most frequently cultivated annual species were the exotic vegetables Lactuca sativa (94% of the gardens) (see Appendix, Table 7 for the common names of all species), Brassica oleracea (80%) and Lycopersicon esculentum (78%), while frequent local annuals were the vegetables Hibiscus sabdariffa (28%) and Abelmoschus esculentus (26%). Among perennials, the most frequently cultivated species were the vegetables Moringa oleifera (55% of gardens) and Adansonia digitata (45%). However, 18% of the gardens had no single perennial species and 43% of the gardeners did not cultivate any fruit trees. In urban locations, perennials were significantly more abundant than in the peri-urban area, with means of 445 and 135 individuals per garden, respectively (P = 0.047). Abundance of fruit trees, however, was slightly higher in peri-urban than in urban areas (34 versus 7 individuals per garden, P = 0.260) and in owned as compared with rented gardens (24 versus 5 tree individuals per garden, P = 0.201). In gardens operated by women, numbers of species and of individuals of perennials were significantly much lower compared with in those operated by men (species number 1.4 versus 7.2; P < 0.001; individual number 33 versus 388; P = 0.001), whereas richness of local species was only slightly lower (1.2 versus 3.2; P = 0.092). The use of market orientation as a categorisation criterion was confirmed by marked differences in species composition and abundances between gardens rated as either commercial or subsistence. In gardens categorised as commercial, a significantly higher proportion of individuals of the most important cash species (Allium porrum, Amaranthus caudatus, Beta vulgaris, B. oleracea, Fragaria × ananassa, H. sabdariffa, L. sativa, Mentha sp., Petroselinum crispum, Pisum sativum, Solanum gilo, Solanum melongena) was found as compared with in subsistence gardens (80% of total individuals versus 37%, P < 0.001).
Forty-four species, including Cola nitida and Theobroma cacao cultivated for experimental reasons, were only recorded from one of the surveyed gardens. Thirty of the 34 species of local origin were cultivated in less than 20% of the gardens (Appendix, Table 7), and 21 were represented by fewer than ten individuals each. One of these rather rare local species, Gossypium arboreum, may be regarded as a relict crop as gardeners mentioned that it was cultivated more frequently in the past for cotton production, while nowadays only its leaves are rarely used as medicine for infants. Several other rare species such as the indigenous fruit tree species Annona senegalensis, Dialium guineense and Grewia tenax were exclusively found in gardens managed by Peul.
In the cold season, mean species richness per garden was 14.06 and mean Shannon index was 0.96, however highly variable (Table 3). The lowest Shannon index was observed in an urban commercial garden, which was largely dominated by L. sativa. The highest Shannon index was found in a very large commercial garden, where many rare species were cultivated.
In the surveyed gardens, the ground layer (<1 m) was dominated by different annual vegetables and spices, particularly L. sativa. In the intermediate layer (1–5 m), small trees such as Citrus spp., Gymnanthemum amygdalinum and M. oleifera were frequent. The dominating species in the top layer (>5 m) were A. digitata, Azadirachta indica and Mangifera indica. In 55% of the gardens, all three strata were observed; 16% of the gardens had the ground and the intermediate layers only; and in 29% of the gardens only the ground layer existed. Several gardeners declared their desire for cultivating more trees, but mentioned the lack of water as a constraint.
Temporal changes of plant species richness and diversity
Total species richness continuously decreased from 115 species in the cold, to 100 in the hot, to 77 in the rainy season (Table 2). This was mainly due to the decrease in the number of annual species such as vegetables, spices and staples. Consequently, species composition was also different from one season to another.
Exotic species of temperate origin such as the vegetables B. oleracea, L. sativa and P. sativum were mainly cultivated in the cold season; other temperate species such as Allium cepa and Anethum graveolens were even exclusively grown in the cold season (Appendix, Table 7). In contrast, in the hot season, vegetables of local origin such as Corchorus olitorius and H. sabdariffa were relatively abundant in the gardens, while the exotic A. caudatus and L. sativa were also widely cultivated. The latter was said to achieve a high price despite its bitter taste during the hot season. Eleven gardeners stopped cultivation of annual species in the hot season. In several gardens, weeds appeared during this season and many plants dried up. Lack of water sources and labor force for irrigating the plots were said to be the limiting resources for gardening during the hot season.
In the rainy season, 21 of the 45 gardeners surveyed halted the cultivation of annual crops. Thirteen of them even stopped all cultivation. Gardeners mentioned that annual crops were difficult to cultivate during the rainy season due to intense rain, temporal flooding or soil silting. In the remaining gardens, mainly A. caudatus and L. sativa were planted, but only in relatively small numbers. Instead staple crops such as S. bicolor and Zea mays were widely cultivated, as well as V. unguiculata that was exclusively cultivated during the rainy season (Appendix, Table 7).
Similarly to the decrease in the total plant species richness from the cold to the hot and the rainy season, a substantial decrease in species diversity parameters was also noted (Table 3). A comparison between the cold and the hot season showed a significant decrease of species richness (P < 0.001), species density (P < 0.001) as well as Shannon index (P = 0.021). From the hot season to the rainy season, species richness (P < 0.001), species density (P < 0.001), Shannon index (P = 0.021) and evenness (P = 0.037) decreased significantly in the 45 surveyed gardens.
Effects of garden and household characteristics on plant species richness and diversity
While total species richness was only slightly correlated with garden size (r = 0.646; P < 0.001; Fig. 2), larger gardens had a higher number of perennial (r = 0.788; P < 0.001) and local plant species (r = 0.797; P < 0.001). The number of vegetation layers increased with garden size (r S = 0.500; P < 0.001). Species richness was also positively correlated with the number of household members per m² of garden area (r = 0.509, P < 0.001).
Compared with commercial gardens non-commercial ones had only a slightly higher Shannon index (1.23 versus 0.90; P = 0.082) and a significantly higher evenness (0.56 versus 0.36; P = 0.005), while species richness was not different (12 versus 15, P = 0.495). Species diversity was also affected by the ethnic affiliation of the gardener. The gardens operated by migrants from Burkina Faso (Gourmanché and Mossi), who mostly rented the land, showed significantly lower Shannon index than gardens managed by the native Djerma, and lower evenness than those of the Peul (Table 4). Species richness was lower in gardens managed by women compared with those managed by men (10 versus 15; P = 0.024), but Shannon index and evenness were higher (H′: 1.24 versus 0.89; P = 0.043; E: 0.56 versus 0.35; P = 0.001).
Most of these results were confirmed by those of the multiple regression analysis. The fit of the model was best for richness of local species (69%) and of overall species (55%; Table 5). Both were positively correlated with garden size and gardener ethnic affiliation (traditionally nomadic groups such as Tuareg or Peul versus sedentary ones). Overall species number was also positively correlated with household size. Only 19% and 32% of the overall Shannon diversity and evenness was explained by the respective regression models (Table 5). Shannon index increased with garden size and property status (rented versus owned). The degree of market orientation had negative effects on evenness that was further determined by gardener ethnic affiliation and gender.
Classification of gardens
Cluster analysis based on crop species abundance data resulted in five distinct clusters (Fig. 3). The discriminant analysis detected the following species (in order of their importance) to be mainly responsible for cluster separation: A. porrum, Talinum triangulare, Tamarindus indica, Dyospyros mespiliformis, S. bicolor, Anethum graveolens, A. caudatus, Musa × paradisiaca, L. esculentum, Borassus aethiopum, Cymbopogon citratus and C. olitorius. The analysis showed that 96% of the original grouped cases were correctly classified. Although the cluster analysis was performed on the basis of species abundances, large differences among clusters were also found with respect to garden size, species richness and diversity and socioeconomic parameters such as ethnic affiliation, gender of gardener or level of market orientation.
Dendrogram resulting from a hierarchical cluster analysis (minimum variance method, squared Euclidean distances) on the basis of ln-transformed abundances of 115 plant species cultivated in 28 urban and 22 peri-urban gardens in Niamey (Niger) in the cold season of 2007. The dotted line marks the cut-off point to define the correct number of clusters according to the ‘elbow’ criterion. The numbers in the clusters refer to the garden number. One urban garden (no. 11) was identified as an outlier before and was, therefore, excluded from this analysis
Cluster 1 comprised rather small subsistence gardens, mostly from peri-urban areas. About 75% of these gardens were managed by native Djerma gardeners and nearly all women-managed gardens were found in this cluster. The Shannon index and evenness of gardens grouped in cluster 1 were relatively high but species richness and density quite low, particularly for local and perennial species (Table 6). No garden of cluster 1 harbored the otherwise common C. olitorius or C. citratus, but L. esculentum and M. × paradisiaca as well as Solanum tuberosum were abundant. Only 35% of these gardens had all three vegetation strata. The three gardens of cluster 2 were large, commercial ones, managed by male, non-Djerma gardeners, who reported to have an additional income besides gardening. These gardens not only showed, together with those of cluster 5, the highest species richness and density as well as number of individuals (especially of fruits and vegetables), but also high Shannon index and high evenness (Table 6). In addition, the number of local and perennial species, as well as fruit tree density, were the highest, resulting in a species-rich top layer. In these gardens, no A. caudatus was cultivated, but Anethum graveolens, B. aethiopum and C. olitorius were abundant. T. indica was found only in gardens of this cluster. Contrary to cluster 2, gardens grouped in cluster 3 were rather small and species poor, including for local and perennial species (Table 6), about 50% of them lacking the top vegetation layer. About half of them were managed by gardeners from Burkina Faso. They were mainly located in the urban area (Fig. 3), rented by the respondents and used for commercial production. Gardens of this cluster showed among the lowest species density, Shannon index and evenness. Only a few A. caudatus were cultivated, but numbers of individuals of S. bicolor was very high in these gardens that also harbored large numbers of F. × ananassa.
Gardens of cluster 4 were all located in the urban area, commercially oriented and owned by gardeners of different ethnic affiliations. This cluster grouped gardens with rather small sizes and intermediate diversity parameters (Table 6). In about 70% of these gardens a complex vegetation stratification existed. No M. × paradisiaca or S. bicolor were grown in gardens of this cluster, but the abundance of A. caudatus was very high. Cluster 5 grouped commercial gardens of intermediate sizes that were mostly rented and managed with the help of several hired workers by retired male, non-Djerma gardeners with rather large families. These gardens had high species richness and density, but relatively low Shannon index and evenness (Table 6) due to the dominance of L. sativa. Gardens of this cluster also showed large quantities of C. citratus and the local vegetable S. gilo and were the only ones where A. porrum and T. triangulare were cultivated. In all these gardens three vegetation strata existed.
Some differences among clusters were also found concerning the summed dominance ratios (SDR) per use category (Fig. 4). Vegetables were the most important use category, particularly in gardens of clusters 1, 3 and 4. In gardens of cluster 1, staple crops were also important. Fruit species as well as wood and MPU species were prevalent only in gardens of cluster 2. Gardens of clusters 3 and 4 lacked stimulant species; ornamental species occurred only in gardens of clusters 2 and 5.
Discussion
The surveyed gardens were highly variable concerning size, plant species composition, richness and diversity, vertical vegetation structure and level of commercialisation. Some gardens resembled species-rich complex agroforestry systems, whereas others were almost managed as a monoculture of annual cash crops in one single vegetation layer. Compared with similar studies, species richness of the 51 surveyed gardens in Niamey (with a total of 116 species and a mean of 14 species per garden in the cold season) (Table 3) seemed relatively high. A recent study of 120 very large commercial urban gardens in Khartoum (Sudan) by Thompson (2007) reported a total species richness of 79 spp. and a mean species richness of only 3 species per garden. In 81 urban homegardens of semi-arid El Obeid (Sudan), the same low mean of 3 species per garden, but an even lower total species richness (32 spp.) were found, probably caused by the lack of irrigation (Gebauer 2005). In Zambia, mean species richness of 31 urban and 21 peri-urban homegardens was 10 and 5 species, respectively (Drescher 1998). Under humid conditions, however, plant species richness in homegardens is often much higher. A total of 602 species and a mean of 7 to 24 species per garden was found in small homegardens on Java, Indonesia (Karyono 1990) and a total of 338 species in homegardens of humid Mexico (Alvarez-Buylla Roces et al. 1989).
The mean Shannon index of H′ = 0.72 to 0.96 observed throughout the year in the surveyed gardens of Niamey (Table 3) was rather low and similar to that of the peri-urban, mostly commercial gardens of Zambia (H′ = 0.81), but lower than in urban gardens of the same country (H′ = 1.35; Drescher 1998). Homegardens in humid Indonesia, on the other hand, had a mean Shannon index of almost 3.0 (Karyono 1990; Kehlenbeck and Maass 2004). It is well known that the Shannon index will decrease if a single species dominates, even if the overall species richness might be high (Drescher 1998). Such a trend was observed in several gardens surveyed in Niamey, where a dominance of a few annual species such as H. sabdariffa or L. sativa, combined with the low abundance of many perennial species (A. digitata or M. oleifera) led to low diversity indices despite the high species richness (Table 6, gardens of cluster 5).
Species seasonality and contribution of gardens to household food supply and income
The large differences of plant species composition, richness and diversity among the three seasons studied were clearly related to the change in climatic conditions throughout the year. The cold season, with its moderate temperature and lower potential evapotranspiration, seemed to be most suitable for horticulture, particularly for the cultivation of annuals. Consequently, the highest species richness and diversity were observed during this period, whereas these parameters were rather low during both the rainy and the hot seasons (Table 3). Thus, the role of sample gardens for income generation and for supplying households with fresh products such as vegetables seemed to decline from the cold to the hot and the rainy season, although some gardens retained a high number of annual vegetable individuals even in the less favourable seasons. The possibility of maintaining a garden during the hot season in Niamey would largely depend on the ability of a household to invest in time (for frequent irrigation), knowledge and cash-demanding infrastructure such as pipes and water pumps, as also reported for homegardens in arid Yemen (Ceccolini 2002). During the rainy season, cultivation of annuals (apart from staple cereals for subsistence) was rare in the studied gardens. Similar findings were also mentioned by Drescher (1998), Linares (2004) and Graefe et al. (2008), and were partly explained by frequent occurrence of fungal diseases on vegetables and high labor demand for cultivating staple crop fields during this season.
Frequent and often indiscriminate use of pesticides was reported to be common in the surveyed gardens and may have consequences for the quality and safety of the food produced. In Niamey, for some short-duration vegetables, residues of pesticides might be high, as well as contamination with pathogens where wastewater was used for irrigation. This, together with the frequent (and sometimes excessive) use of mineral fertilisers, raises concerns about negative externalities of intensive UPA in some of the surveyed gardens.
Contribution of the gardens towards in situ conservation of local plant species
The overall contribution of the surveyed gardens towards in situ conservation of indigenous species may be questioned. More than 70% of all plant species cultivated in the these gardens were of exotic origin; local species were mostly represented by only a few individuals. With increasing market orientation of a garden, local species with low market value are often the first ones to be removed (Soemarwoto and Conway 1992). In Niamey, interviewed gardeners highly appreciated the cultivation of marketable exotic crops, using improved planting material offered by local traders and an outreach program of the International Crops Research Institute for the Semi-Arid Tropics (ICRISAT), Sahelian Centre, located about 25 km south of Niamey. Whether the introduction of exotic, mostly marketable species and improved varieties will indeed enhance the loss of local species and varieties over time as was reported from other case studies in developing countries (Fundora Mayor et al. 2004; Sunwar et al. 2006) can hardly be predicted from a one-time study such as ours. Similarly, no conclusions about the future transformation trends of gardens can be drawn, although a general increase of their degree of market orientation may be assumed (Wiersum 2006).
Some of the surveyed gardens seemed to be more suitable for in situ conservation than others based on differences in plant species richness and diversity, which are reported in the literature to largely depend on a combination of agroecological and socioeconomic factors (Abdoellah et al. 2002; Kehlenbeck and Maass 2004; Sunwar et al. 2006; Kehlenbeck et al. 2007). A positive effect of garden size on species richness such as in our study (Table 5) has been described previously (Abdoellah et al. 2002; Albuquerque et al. 2005; Kehlenbeck et al. 2007). Conversely, ease of access to markets and urbanisation were often reported to have a negative effect (Karyono 1990; Shrestha et al. 2002; Abdoellah et al. 2006; Tesfaye Abebe et al. 2006). The supply of diverse food and the demand for certain crops at the markets seem key forces driving gardeners from subsistence to semi-commercial or commercial production (Peyre et al. 2006). This may lead to a decrease in the number of perennials and the dominance of fast-growing, mostly exotic vegetables, resulting in a simplified vegetation structure in these gardens (Shrestha et al. 2002; Peyre et al. 2006; Thaman et al. 2006). Drescher (1998) reported lower plant species richness and diversity in peri-urban as compared with urban gardens, which he explained by the high level of market orientation observed in the peri-urban gardens. In Niamey, however, even commercial peri-urban gardens such as those classified in clusters 2 and 5 (Table 6) may harbor high plant species richness and diversity, including for local and perennial species. Some other studies reported that garden species diversity may in fact be positively influenced by market proximity and/or ‘semi-commercialisation’ (Trinh et al. 2003; Wezel and Ohl 2005; Kehlenbeck et al. 2007), particularly if there is demand for traditional crops in urban centers (Sunwar et al. 2006).
Gardener’s ethnic affiliation and origin may influence plant species composition and diversity indices (Kusumaningtyas et al. 2006; Kehlenbeck 2007). The higher richness and evenness in gardens of nomads as compared with non-nomads, revealed by the multiple regression analysis (Table 5), may be explained by their rather high mobility and their comprehensive knowledge of the plant species and their uses in different agroecological environments. Regarding gender, only few studies showed effects of this variable on plant species richness and diversity. Women are often reported to play an important role for in situ conservation of plant genetic resources in homegardens by cultivating local species for subsistence, whereas men are often more interested in introduction and cultivation of exotic cash crops (Niñez 1985; Drescher 1998; Del Angel-Pérez and Mendoza 2004; Eyzaguirre and Linares 2004). However, the women gardeners in our study grew fewer species than men, including local ones.
Wealthy households may maintain more perennials (Thaman et al. 2006) and an overall higher plant species richness and diversity in their gardens than poor households because wealthy people in general have a larger compound, greater mobility and better access to new genetic material (Drescher 1998, Shrestha et al. 2002). Although the wealth status of respondents in our study was not assessed in detail, gardeners who rented their plots seemed to be rather poor. The lower Shannon diversity in rented as compared with owned gardens (Table 5) may have been caused by economic pressures on gardeners to generate enough income from their rented plots, thus focussing on cultivation of a very small number of profitable cash crops. Apart from garden size, all factors identified as influencing plant species richness and diversity in our study were also mentioned as important in the summary table published by Wiersum (2006) for Indonesian homegardens.
Cluster analysis may help to better identify those types of gardens generally suitable for in situ conservation of plant genetic resources or containing certain key species targeted for conservation. However, this classification approach is not yet very common (Leiva et al. 2002; Kehlenbeck and Maass 2004; Peyre et al. 2006). Our cluster analysis showed that the garden types most suitable for in situ conservation of plant genetic resources are those managed by wealthier, highly educated people, who were not completely dependent on the generation of cash income through their garden (which is the case for the three gardeners of cluster 2) or those managed by elderly, retired gardeners with large families (the three gardeners of cluster 5). In these gardens not only the highest total species richness, but also the highest amount of local species was found (Table 6), though the latter were often only present in low abundances. These low abundances may limit the general suitability of these urban and peri-urban gardens for in situ conservation, as also mentioned by Alvarez-Buylla Roces et al. (1989) and Kehlenbeck (2007). However, the absolute value of the surveyed gardens for in situ conservation of wild and domesticated indigenous plant species could not be assessed because no list of threatened and endangered species of Niger is available. Sunwar et al. (2006) recommended to prevent the loss of indigenous species by emphasising their traditional prestige, increasing and maintaining the related knowledge, and improving seed supply and the quality of traditional varieties. Diversity studies as such may also promote species conservation because they may raise awareness among consumers and decision-makers and may cause gardeners to be more proud of maintaining a species-rich garden (Kehlenbeck and Maass 2004; Kehlenbeck 2007).
Conclusions
Niamey’s urban and peri-urban gardens seem to play an important role for family nutrition and cash income of the households involved. The relatively high plant species richness and diversity of the studied gardens was mainly dependent on garden size, the gardener’s ethnic affiliation and gender, and on socioeconomic factors. Some gardeners who were not dependent on generating their main cash income from their garden maintained species-rich gardens, including many local species. This type of gardens resembled homegarden-like systems and may be suitable for in situ conservation of plant genetic resources. For an effective promotion of in situ conservation of local plant species in such gardens, suitable threatened key species should be identified and promoted, awareness of gardeners and consumers about the nutritional and cultural value of these species should be increased and their cultivation and marketing be supported.
References
Abdoellah OS, Parikesit, Gunawan B, Hadikusumah HY (2002) Home gardens in the upper Citarum watershed, West Java: a challenge for in situ conservation of plant genetic resources. In: Watson JW, Eyzaguirre PB (eds) Home gardens and in situ conservation of plant genetic resources in farming systems. Proceedings of the second international home gardens workshop, 17–19 July 2001, Witzenhausen, Germany. IPGRI, Rome, p 140–147
Abdoellah OS, Hadikusumah HY, Takeuchi K, Okubo S, Parikesit (2006) Commercialization of homegardens in an Indonesian village: vegetation composition and functional changes. Agrofor Syst 68:1–13. doi:10.1007/s10457-005-7475-x
Albuquerque UP, Andrade LHC, Caballero J (2005) Structure and floristics of homegardens in Northeastern Brazil. J Arid Environ 62:491–506. doi:10.1016/j.jaridenv.2005.01.003
Alvarez-Buylla Roces ME, Lazos Chavero E, García-Barrios JR (1989) Homegardens of a humid tropical region in Southeast Mexico: an example of an agroforestry cropping system in a recently established community. Agrofor Syst 8:133–156. doi:10.1007/BF00123117
Arbonnier M (2000) Arbres, arbustes et lianes des zones sèches d’Afrique de l’Ouest. Centre de Coopération Internationale en Recherche Agronomique pour le Développement, Montpellier
Backhaus K, Erichson B, Plinke W, Weiber R (2006) Multivariate analysemethoden. Eine anwendungsorientierte Einführung. Springer, Berlin
Blanckaert I, Swennen R, Paredes Floresa M, Rosas R, Lopez L, Lira Saade R (2004) Floristic composition, plant uses and management practices in homegardens of San Rafael Coxcatlan, Valley of Tehuacan-Cuicatlan, Mexico. J Arid Environ 57:39–62. doi:10.1016/S0140-1963(03)00100-9
Bryld E (2003) Potentials, problems and policy implications for urban agriculture in developing countries. Agric Human Values 20:79–86. doi:10.1023/A:1022464607153
Ceccolini L (2002) The homegardens of Soqotra island, Yemen: an example of agroforestry approach to multiple land-use in an isolated location. Agrofor Syst 56:107–115. doi:10.1023/A:1021365308193
Davis SD, Heywood VH, Hamilton AC (eds) (1994) Centres of plant diversity. A guide and strategy for their conservation. Volume 1. Europe, Africa, South-West Asia and the Middle East. WWF, IUCN, Cambridge
Del Angel-Pérez AL, Mendoza MA (2004) Totonac homegardens and natural resources in Veracruz, Mexico. Agric Human Values 21:329–346. doi:10.1007/s10460-004-1219-9
Drescher AW (1998) Hausgärten in afrikanischen Räumen. Bewirtschaftung nachhaltiger Produktionssysteme und Strategien der Ernährungssicherung in Zambia und Zimbabwe. Sozioökonomische Prozesse in Asien und Afrika, vol 4. Centaurus, Pfaffenweiler
Drescher AW, Holmer RJ, Iaquinta DL (2006) Urban homegardens and allotment gardens for sustainable livelihoods: management strategies and institutional environments. In: Kumar BM, Nair PKR (eds) Tropical homegardens: a time-tested example of sustainable agroforestry. Advances in agroforestry, vol 3. Springer, Dordrecht, pp 317–338
Evans FC, Clark PJ, Brand RH (1955) Estimation of the number of species present on a given area. Ecology 36:342–343. doi:10.2307/1933244
Eyzaguirre PB, Linares OF (eds) (2004) Home gardens and agrobiodiversity. Smithsonian Books, Washington
Fundora Mayor Z, Shagorodsky T, Castineiras L (2004) Sampling methods for the study of genetic diversity in home gardens of Cuba. In: Eyzaguirre PB, Linares OF (eds) Home gardens and agrobiodiversity. Smithsonian Books, Washington, pp 56–77
Gebauer J (2005) Plant species diversity of home gardens in El Obeid, Central Sudan. J Agric Rural Dev Trop Subtrop 106:97–103
Gebauer J, Luedeling E, Hammer K, Nagieb M, Buerkert A (2007) Mountain oases in northern Oman: an environment for evolution and in situ conservation of plant genetic resources. Genet Resour Crop Evol 54:465–481. doi:10.1007/s10722-006-9205-2
Graefe S, Schlecht E, Buerkert A (2008) Opportunities and challenges of urban and peri-urban agriculture in Niamey, Niger. Outlook Agric 37:47–56
Hanelt P, Institute of Plant Genetics and Crop Plant Research (eds) (2001) Mansfeld’s encyclopedia of agricultural and horticultural crops. Springer, Berlin
INS (Institut National de la Statistique) (2008) Structure de la population résidente de nationalité nigérienne selon l’ethnie et la région au 1er juin 2001. http://www.stat-niger.org. Accessed 18 Nov 2008
Karyono (1990) Homegardens in Java. Their structure and function. In: Landauer K, Brazil M (eds) Tropical home gardens. United Nations University, Tokyo, pp 138–146
Kehlenbeck K (2007) Rural homegardens in Central Sulawesi, Indonesia: an example for a sustainable agro-ecosystem? Dissertation, University of Goettingen, Germany. http://webdoc.sub.gwdg.de/diss/2007/kehlenbeck/kehlenbeck.pdf. Accessed 10 Oct 2007
Kehlenbeck K, Maass BL (2004) Crop diversity and classification of homegardens in Central Sulawesi, Indonesia. Agrofor Syst 63:53–62
Kehlenbeck K, Maass BL (2006) Are tropical homegardens sustainable? some evidence from Central Sulawesi, Indonesia. In: Kumar BM, Nair PKR (eds) Tropical homegardens: a time-tested example of sustainable agroforestry. Advances in agroforestry, vol 3. Springer, Dordrecht, pp 339–354
Kehlenbeck K, Arifin HS, Maass BL (2007) Plant diversity in homegardens in a socio-economic and agro-ecological context. In: Tscharntke T, Leuschner C, Zeller M, Guhardja E, Bidin A (eds) The stability of tropical rainforest margins: linking ecological, economic and social constraints of land use and conservation. Springer, Berlin, pp 297–319
Kusumaningtyas R, Kobayashi S, Takeda S (2006) Mixed species gardens in Java and the transmigration areas of Sumatra, Indonesia: a comparison. J Trop Agr 44:15–22
Leiva JM, Azurdia C, Ovando W, López E, Ayala H (2002) Contributions of home gardens to in situ conservation in traditional farming systems–guatemalan component. In: Watson JW, Eyzaguirre PB (eds) Home gardens and in situ conservation of plant genetic resources in farming systems. Proceedings of the second international home gardens workshop, 17–19 July 2001, Witzenhausen, Germany. IPGRI, Rome, pp 56–72
Linares OF (2004) Casamance, Senegal: home gardens in an urban setting. In: Eyzaguirre PB, Linares OF (eds) Home gardens and agrobiodiversity. Smithsonian Books, Washington, pp 198–212
Maxwell D, Levin C, Csete J (1998) Does urban agriculture help prevent malnutrition? evidence from Kampala. Food Policy 23:411–424. doi:10.1016/S0306-9192(98)00047-5
McCune B, Grace JB, Urban DL (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach
Missouri Botanical Garden (2007) Botanical database TROPICOS. http://www.tropicos.org. Accessed 7 Dec 2007
Nair PKR, Kumar BM (2006) Introduction. In: Kumar BM, Nair PKR (eds) Tropical homegardens: a time-tested example of sustainable agroforestry. advances in agroforestry, vol 3. Springer, Dordrecht, pp 1–10
Niñez V (1985) Working at half-potential: constructive analysis of home garden programmes in the Lima slums with suggestions for an alternative approach. Food Nutr Bull 7:6–14. http://www.unu.edu/unupress/food/8F073e/8F073E02.htm. Accessed 13 Nov 2008
Peyre de Fabregues B (1979) Lexique de noms vernaculaires de plantes du Niger. Institut National de la Recherche Agronomique du Niger, Niamey
Peyre A, Guidal A, Wiersum KF, Bongers F (2006) Dynamics of homegarden structure and function in Kerala, India. Agrofor Syst 66:101–115. doi:10.1007/s10457-005-2919-x
Shrestha P, Gautam R, Rana BR, Sthapit B (2002) Home gardens in Nepal: status and scope for research and development. In: Watson JW, Eyzaguirre PB (eds) Home gardens and in situ conservation of plant genetic resources in farming systems. Proceedings of the second international home gardens workshop, 17–19 July 2001, Witzenhausen, Germany. IPGRI, Rome, pp 105–118
Smith GC, Clegg MS, Keen CL, Grivetti LE (1996) Mineral values of selected plant foods common to southern Burkina Faso and to Niamey, Niger, West Africa. Int J Food Sci Nutr 47:41–53. doi:10.3109/09637489609028560
Soemarwoto O, Conway GR (1992) The Javanese homegarden. J Farming Syst Res-Ext 2:95–118
Sunwar S, Thornström CG, Subedi A, Bystrom M (2006) Home gardens in western Nepal: opportunities and challenges for on-farm management of agrobiodiversity. Biodivers Conserv 15:4211–4238. doi:10.1007/s10531-005-3576-0
Tambwe N (2006) Urban agriculture as a global economic activity with special reference to the city of Lubumbashi in the Democratic Republic of Congo. Afr Asian Stud 5:193–213. doi:10.1163/156920906777906772
Tesfaye Abebe, Wiersum KF, Bongers F, Sterck F (2006) Diversity and dynamics in homegardens of southern Ethiopia. In: Kumar BM, Nair PKR (eds) Tropical homegardens: a time-tested example of sustainable agroforestry. Springer, Dordrecht, pp 123–142
Thaman RR, Elevitch CR, Kennedy J (2006) Urban and homegarden agroforestry in the Pacific Islands: Current status and future prospects. In: Kumar BM, Nair PKR (eds) Tropical homegardens: a time-tested example of sustainable agroforestry. advances in agroforestry, vol 3. Springer, Dordrecht, pp 25–41
Thompson JL (2007) The gardens of urban and peri-urban Khartoum, the Republic of the Sudan. MSc thesis, Faculty of Organic Agricultural Sciences, University of Kassel, Germany
Torquebiau E (1992) Are tropical agroforestry home gardens sustainable? Agric Ecosyst Environ 41:189–207. doi:10.1016/0167-8809(92)90109-O
Trinh LN, Watson JW, Hue NN, De NN, Minh NV, Chu P, Sthapit BR, Eyzaguirre PB (2003) Agrobiodiversity conservation and development in Vietnamese home gardens. Agric Ecosyst Environ 97:317–344. doi:10.1016/S0167-8809(02)00228-1
USAID (United States Agency for International Development) (2002) Niger. Monthly report on food security in Niger. April 26, 2002. http://www.fews.net/docs/Publications/Niger_200203en.pdf. Accessed 03 Feb 2008
Wezel A, Ohl J (2005) Does remoteness from urban centres influence plant diversity in homegardens and swidden fields? A case study from the Matsiguenka in the Amazonian rain forest of Peru. Agrofor Syst 65:241–251. doi:10.1007/s10457-005-3649-9
Wiersum KF (2006) Diversity and change in homegarden cultivation in Indonesia. In: Kumar BM, Nair PKR (eds) Tropical homegardens: a time-tested example of sustainable agroforestry. Advances in agroforestry, vol 3. Springer, Dordrecht, pp 13–24
WMO (World Meteorological Organization) (2007) World Weather Information Service, Niger. http://worldweather.wmo.int/074/c00327.htm. Accessed 08 Apr 2009
Zander R, Erhardt W, Götz E, Bödecker N, Seybold S (2002) Handwörterbuch der Pflanzennamen. Ulmer, Stuttgart
Acknowledgments
We wish to thank the gardeners of Niamey for their friendly cooperation and hospitality, without which it would not have been possible to conduct this research. The support provided by the field assistants Ibrahim Moussa and Moussa Abdourahamane as well as by Rodrigue V.C. Diogo, Martina Predotova, Katja Brinkmann and Dirk Nöding is gratefully acknowledged. This study was supported by the Deutsche Forschungsgemeinschaft (DFG) within the project ‘Plant-animal based matter fluxes and production efficiencies in urban and peri-urban agriculture of a West African city’ (BU 1308).
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Table 7.
Rights and permissions
About this article
Cite this article
Bernholt, H., Kehlenbeck, K., Gebauer, J. et al. Plant species richness and diversity in urban and peri-urban gardens of Niamey, Niger. Agroforest Syst 77, 159–179 (2009). https://doi.org/10.1007/s10457-009-9236-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-009-9236-8