Abstract
Recent years have witnessed remarkable development in the field of nanotechnology and it has been affirmed that carbon-based nanomaterials have wide applications in agriculture, industrial, biomedical and environmental sectors. Due to distinctive physicochemical properties of the carbon nanotubes (CNTs), they have been extensively utilized in plant science as a growth promoter, and thus, could be a boon for biomass production of agricultural products. Studies suggest that CNTs help increase the plant’s ability to absorb water and essential nutrients, thereby increasing growth. Apart from this, CNTs have been scrutinized for their utilization in genetic engineering for the delivery of genes, proteins or drugs. However, the literature discloses mixed effects of CNTs exposure on plants like in inducing oxidative stress by generating reactive oxygen species (ROS). Moreover, studies concerning CNTs interaction with plant system is at a nascent stage and needs further investigations to explore the mechanisms influencing the growth and toxicity in plants. Therefore, this review attempts to highlight the current literature on CNTs (including both single walled and multi walled) exposure on plants. It also explores unresolved challenges, as well as recommendations to ensure sustainable development of CNTs while minimizing any possible adverse health impacts.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Nanotechnology, a new emerging field of science, permits advanced research in several areas. The use of nanoscience for the production of engineered nanomaterials like CNTs is a scientific breakthrough and could open up novel applications in the field of biotechnology, agriculture and others [1]. CNTs are hollow cylinders of carbon atoms that appear like rolled tubes of graphite formed in large bundles of single or multiple sheets of graphene to give single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs), respectively. It is a honeycomb lattice rolled on to itself, with lengths ranging from several hundred nanometers to several micrometers and diameters of a few nanometers (SWCNTs) to dozens of nanometers (MWCNTs) [2]. They are a large group of carbon-based, tube-like nanomaterials, which not only differ in length and the number of layers they consist of but also vary in types of impurity, contents and surface modification. CNTs have become increasingly popular due to unique features such as dimension, structure and topology. They have become one of the most studied and exploited engineered nanomaterials due to their outstanding electronic, mechanical, optical and structural properties. The applications of CNTs include biomedicine, nanoelectronics, bioengineering and mechanical engineering. Currently, the use of CNTs has been further extended to health care and agriculture to improve the quality of life. It can also be used in solving the environmental problems such as air, water and soil pollution where remediation technologies are limited [3, 4]. Some scientists observed that the L‐cysteine moiety in L‐glutathione is responsible for the susceptibility to oxidation by metallic impurities present in the carbon nanotubes. Their results These results assessed the toxicity of carbon‐nanotube materials [5].
Research and development in the field of agriculture is very essential because most of the living beings depend on it. Currently, there is tremendous research interest in nanotechnology-based enhancement of agricultural products [6]. Carbon nanotubes can act as regulators of seed germination and plant growth. Report on MWCNTs has the ability to enhance the growth of tobacco cell culture (55–64%). They found that a correlation between the activation of cells growth exposed to MWCNTs [7]. Major emphasis of these studies is to enhance agricultural outputs, detection of diseases and their remediation processes [8]. Although researchers are keen to develop new techniques that could be suitable for plants to boost their native functions, they are also trying to increase the efficiency of plants to uptake more nutrients from the soil. This will ultimately lead to enhancement in the overall increase of biomass and fruitition of the plants. In the agricultural sector, CNTs might have properties that are effective to crops which aids in, controlling the release of chemical fertilizers and support the nutrients that regulate plant growth which may enhance target activity [6, 9]. In this respect, CNTs have a large potential to provide an opportunity for researchers of plant science and other field, to develop new tools for incorporation into plants that could augment existing functions and include new ones.
In recent years, CNTs have gained interest due to their possible applications in regulating plant growth [10]. Some researchers have investigated the effects of CNTs on plants and achieved significantly higher germination rates for seeds that contain CNTs [11]. It is suggested that CNTs help increase the plant’s ability to uptake water, which abets wholesome growth of the plant, including the roots and shoots, and proliferates branching. Plants have produced two times more flowers and fruits when grown in soil supplemented with CNTs [12]. Studies propose that CNTs can enhance the growth of plants and have an effect on the expression of genes that are essential for cell division and plant development [13]. These studies highlight the potential to use CNTs, and nanomaterials to enhance plant functions.
On the contrary, inhibitory effects of CNTs were also reported in a few literatures [14, 15]. The effect of CNTs on plants is mainly influenced by different plant species, their growth stage and the nature of the CNTs used. Also, their property to penetrate the cell wall and their interactions with intracellular structures owing to their small size and high surface area contribute to potential cellular and genetic toxicity by the induction of oxidative stress. Studies on the cytotoxicity of CNTs include a decrease in cell viability, potential cytotoxicity in Arabidopsis, delayed flowering, reduction in yield and cell death due to apoptosis in rice, reduction in root length in lettuce, wilting and curling of leaves, and pigment loss in red spinach [15]. Cell aggregation, condensation of chromatins, plasma membrane deposition, generation of ROS and DNA damage are some other cellular changes among them.
Several works also highlighted the effects of CNTs on the microbial diversity and their threat to the useful microbial population [16]. Under high concentrations, CNTs significantly affect nutrient retention, decrease enzyme activity and microbial biomass, and degrade enzymes which are responsible for microbial biomass [17]. In a study [18], it was reported that CNTs may exhibit antimicrobial activity when they come in direct contact with bacterial cells, which leads to membrane mutilation and generation of oxidative stress.
Although the major objective of the use of CNTs is to increase agricultural productivity, concerns about possible side effects in ecosystems, human health, and agricultural industries need to be addressed.
2 Classification and characterization of carbon nanotubes
2.1 Classification
On the basis of structural composition, CNTs are mainly classified into three groups: stacked-cup carbon nanotubes (SCCNTs), MWCNTs and SWCNTs [19]. Jackson et al., [20] classified carbon nanotubes into three categories: MWCNTs, double-walled carbon nanotubes (DWCNTs) and SWCNTs. Their functional character changes with the change in structure and symmetry. Structures of carbon nanotubes are unique; they are hollow cylinders of graphite and possess hexagonally arranged carbon rings. Their end contains hexavalent arched structure while they are capped with the pentavalent ring [21]. They possess high tensile strength which resembles the property of graphene. As reported, CNTs remains stable even at very high temperatures and maximize vibrational entropy [22].
SWCNTs have a diameter in the range of 0.4–3 nm and their length ranges in micrometer [23]. They are hexagonally arranged in a bundle and form crystal-like structures [23, 24]. They can be differentiated into different forms in accordance with the type of wrapping, chirality, zigzag, and armchair nature [23]. Properties of SWCNTs are almost the same when compared with MWCNTs except for its high tensile strength [25].
MWCNTs contain many concentric hollow cylinders with an interlayer spacing of 0.34–0.39 nm [23, 26]. The decrease in inner wall diameter depends on wall layers, that is inner wall varies from 0.4 mm to a few nm, while the outer one ranges from 2 to 30 nm [23]. The endings of MWCNTs are closed with dome-shaped half fullerene capping. They are arranged in a way that one carbon nanotube lies another. The diameter of the inner carbon nanotubes is generally smaller than the outer one. This arrangement is also known as a Russian doll arrangement. Yet another arrangement is Parchment, in which one CNT is surrounded or rolled by multiple copies of CNT. Findings also suggest that the outer wall protects the inner wall from chemical reactions [23].
2.2 Characterization
The synthesis procedures available for CNTs are chemical vapour deposition (CVD) [27], arc discharge (AD) [28] and laser ablation (LA) [29] of which CVD is the most popular technique because of well-aligned structure and the desired orientation of the layers [30]. Other synthesis methods for CNTs are also described in the literature [31,32,33]. The produced CNTs are impure due to the presence of different undesired byproducts (fullerenes, carbonaceous residues, amorphous carbon and catalyst impurities). This led to the intensification of research on the purification of the synthesized CNTs [34,35,36,37,38,39]. Conversely, in this modern era of highly innovative technology driven processes, it remains a challenge to synthesize high purity CNTs specific in length and diameter.
Since the available synthesis procedures for CNTs do not ensure the homogeneity of the product in reference to their length and diameter, numerous efforts have been made to characterize CNTs obtained from different synthesis procedures. It should be noted that the toxicity of nanoparticles significantly depends on their morphology [40]. Due to the very small size of the CNTs highly sophisticated techniques are employed to assess their characteristics and morphology. Moreover, their bundle and aggregate structure make their use difficult. For application purposes, it must be dispersed either in water or in any another solvent to enhance its property. This can be achieved through the process of oxidation, sonication, centrifugation and ultrasonication combined with dispersing agents. As the concentration of CNTs in the initial phase is altered during the disaggregation processes and the final concentration in the liquid phase is lower than that initially used for preparation. It is important to study the characterization to determine the concentration of the dispersed CNTs in water. The techniques used to achieve the goal are outlined in Table 1, that can be broadly classified into four categories: microscopic and diffraction, spectroscopic, thermal, and separation techniques (Table 2; Fig. 1).
2.3 Role of CNTs in plant growth
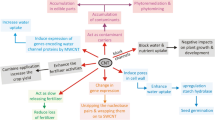
Interaction of CNTs with plants is a complex phenomenon which causes several physiological and morphological changes in the plant species [58]. These changes are influenced by the size, concentration, type of CNTs as well as type of plant species and life stages. Several researchers attempted to study the uptake and translocation of the CNTs in plants and put forward a strategy dependent on the ratio of the size of the CNTs to the pore size of the cell wall. Methods like ultrasonic-assisted chemical oxidative cutting, the introduction of carboxylic groups on CNTs, to make it more soluble, are also used to increase the uptake by plants (Figs. 2, 3).
2.4 Effect of single-walled carbon nanotubes (SWCNTs)
In recent studies, increasing evidences suggest that SWCNTs increase plant growth and development. According to Yan et al. [59] SWCNTs have the ability to accelerate the seminal root growth in maize plants by influencing the gene expression. Similar studies on rice seedlings revealed that SWCNTs promoted growth by upregulating the expression of genes related to root growth [60]. Results also suggested a direct correlation between leaf development and the expression of the related genes in response to the CNTs. The positive effects of CNTs were also observed on the vegetative growth of the plant including increased leaf growth, chlorophyll contents and enhanced photosynthetic rates. Canas et al. [61] studied the effect of exposure of functionalized and non-functionalized SWCNTs on root elongation of six crop species that included cabbage, carrot, cucumber, lettuce, onion and tomato. Enhanced root elongation and increase in root number were reported in onion and cucumber plants.
A study on seed germination and seedling growth of salvia, pepper and tall fescue revealed that SWCNTs increased seed germination rates in the plants and the best SWCNT concentrations for seed germination and seedling growth for salvia and tall fescue were at 30 mg/L of SWCNT and at 10 mg/L for pepper [62]. The outcome of yet another study confirmed, that the CNTs (10–40 mg/L) improved the germination rate of tomato and onion seedlings. Further, there was increase in the dry weight of tomato and radish shoots. This study also revealed that the effectiveness of CNTs may depend on the concentration of CNTs and particular plant species [63].
Similar studies [64] on Rubus adenotrichos, using SWCNTs-COOH and SWCNTs-Fe resulted in elongation of roots and shoots, and enhanced cell metabolism. Khodakovskaya et al., [65] confirmed the increase in biomass in tobacco plants when treated with SWCNTs. In a separate study Mohammad et al., [66] used SWCNTs functionalized with quantum dots (QDs) on tomato seeds and found that addition of CNTs helped in increasing the chlorophyll content, and the total weight and height of the root/shoot system.
The SWCNTs also influences the reproductive growth in plants which are highlighted by the study on tomato plants by significantly increasing the flower and fruit formation. Other effects were increased in plant height, chlorophyll content and the total weight of the root system [12].
A study investigated, the role of SWCNTs under drought stress on Hyoscyamus niger seeds and it was found that SWCNTs helped in providing resistance against drought stress by enhancing water uptake and activating the plant defense system [67]. Another study on SWCNTs treated soybean seeds showed increased tolerance of seeds to drought stress. Apart from this, there was increase in the activity of catalase, superoxide dismutase etc. which may play an important role under stress conditions [68].
Due to the unique properties of SWCNTs, it can easily penetrate the cell wall and deliver chemicals to cells. This aspect was further explored to facilitate the delivery of DNA to the cells. Experiments performed by Corredor et al. [69] in Tobacco and Catharanthus cells evidenced that the SWCNTs translocate through the cell wall and the cell membrane. Wu et al., [70] investigated the cellular delivery of DNA by SWCNTs and observed that they were protected from enzymatic cleavage. They targeted a specific mRNA, which increased the capability of self-delivery and intracellular biostability when compared to free DNA probes.
2.5 Effect of multi-walled carbon nanotubes (MWCNTs)
Research on MWCNTs role in the increase of plant growth, seed germination, biomass etc. has been underway since the last decade. Even the role of CNTs in stress conditions was explored highlighting the fact that MWCNTs can enter into the seeds of broccoli and facilitate water uptake in plants thereby increasing the growth [71]. Further, it was observed that MWCNTs induced changes in lipid composition, rigidity and permeability of the root plasma membranes and ultimately, increased aquaporin transduction under salt-stressed conditions [72]. Furthermore, a study revealed the non-toxic nature of water-soluble CNTs that helped in enhancing the growth of roots, shoots and branching in gram plant (Cicer arietinum) [73]. This property of the CNTs may prove useful in optimum utilization of water in areas face water scarcity.
Research was also carried out on the effects of MWCNTs on seed germination, growth and the development of three important crops (barley, soybean, corn). Reverse transcription polymerase chain reaction (RT-PCR) analysis revealed that the expression of genes encoding water channel proteins increased in soybean, corn, and barley seeds coated with MWCNTs compared with control [74]. A similar study on wheat, maize, peanut and garlic depicted the beneficial effects of functionalized MWCNTs (in a dose-dependent manner) on root and shoot growth, biomass and number of leaves in all the plant species [7]. The ability of MWCNTs to penetrate the seat coat by forming new pores helped in water uptake resulting in the increase of the germination rate. Additionally, it has been also observed that CNTs have the capacity to penetrate plant seed coat [11]. The germination rate and plant growth were found to be significantly higher for seeds that germinated on medium containing CNTs (1040 g/mL) compared to control.
Tiwari et al., [75] investigated the effect of MWCNTs on tomato seedlings and found that at the concentration of 40 μg/mL, absorption of essential nutrient elements increased, which resulted in enhanced growth and biomass of the plant. In another study on tomato, it was found that the production of flowers and fruits were twice as compared to control [12]. Study on maize plant [3], demonstrated the effect of pristine MWCNTs which includes enhanced uptake of water and essential nutrients leading to increase in biomass of the plant. This property of the CNTs may prove to be beneficial for plants growing in nutrient-deficient soil.
Uptake and accumulation of MWCNTs can alter the morphology and biochemical characteristics of Onobrychis arenaria seedlings. Effect of the engineered nanomaterial containing MWCNTs (concentration 100 μg/mL and 1000 μg/mL), on the growth of O. arenaria seedlings suggested that MWCNTs have a capacity to penetrate the cell walls as they accumulate in roots and translocate to the leaves [78].
Larue et al. [79] studied the impact of CNTs on seed germination, root elongation, plant biomass, evapotranspiration, chlorophyll in wheat and rapeseeds. It was observed that less than 0.005% of the applied MWCNT dose is taken up by plant roots and translocated to the leaves. Further, there was no observed effect on photosynthetic activity or oxidative stress in plant.
A study on the effect of MWCNTs on germination and seedling growth of Cichorium intybus L inferred that even though, there was no observed effect on seed germination or plant growth, there was increase in weight of seeds treated with MWCNTs [80].
2.6 Implications in plant system due to use of CNTs
The toxicity of CNTs in plants has been highlighted in several studies. However, few researchers reported that interaction of CNTs with plants manifested toxicity effects, although other studies pointed at the positive correlation or no effect in plants.
Several studies revealed various factors viz. concentration of CNTs, particle size (surface area), plant type, growth stage, growth media etc. that may contribute to toxicity in plants [81, 82]. Further, characterizing CNTs is an essential component of ecotoxicity testing and can include evaluating the starting materials, its characteristics during ecotoxicity tests and the extent to which the CNTs altered during the test. Investigations comparing the toxicity of CNTs to bulk hard carbons should consider potential differences in surface area as a possible variable to explain differences in toxicity. CNT is transpiring novel nano-carbon fertilizer in the agricultural field, but at the same time it can cause deleterious effect on soil microbial density, composition, and population.
A study involving the MWCNT and cotton cellulose nanofibers toxicological effects on freshwater green microalgae Chlorella vulgaris was conducted [83]. It was observed that the uptake of MWCNT and cotton CNF by C. vulgaris led to reductions on algal growth and cell viability [84,85,86].
Wang et al. [87] observed that the carboxylated multi-walled carbon nanotubes cause biochemical and subcellular damages in leaves of broad bean (Vicia faba L.) seedlings under combined stress of lead and cadmium. The study mentioned the deleterious effects i.e. oxidative damage in the leaves due to combined use of MWCNTs-COOH with Pb and Cd while no such effect was observed in the leaves treated with MWCNTs-COOH or Pb and Cd individually. This study concluded that the combination of MWCNTs-COOH with heavy metals may cause phytotoxicity and hence, health risks among individuals.
The application of SWCNTs to rice resulted in the delayed flowering and decreased yield of this essential crop [88] whereas, a study presented a genotoxic potential in Arabidopsis [15] protoplasts relative to their concentration and size. Other cellular changes observed due to SWCNTs application were cell aggregation, condensation of chromatins, plasma membrane deposition and H2O2 accumulation in rice and Arabidopsis. A similar study was also carried out in Arabidopsis using MWCNTs which resulted in reduced cell viabilities and dry weight decreased cell chlorophyll content, and superoxide dismutase activities.
Upon their (MWCNTs) treatment to Amaranthus tricolor, several morphological and physiological changes were observed in the treated leaves like removal in red pigment of leaf, necrosis, curling, and wilting. The effects were not only limited to leaves there was concentration-dependent reduction in root-shoot height, root-shoot weight, and leaf numbers. Also, cell function was impaired due to enhanced electrolytic damage, generation of ROS and increased apoptosis [14]. Researchers reported reduced germination percentage in Cichorium intybus [80], reduced biomass in Zucchini [89] decrease in root lengths in lettuce plants [90] and DNA damage in Allium cepa [91].
Several significant negative effects related to translocation and uptake were reported in different plant species when treated with MWCNTs. Reduction in growth, uptake and translocation was observed in the soybean plants when treated with MWCNTs, 10–50 mg/L [92]. In a study the impact of MWCNTs and accumulation behavior of contaminants in mustard plants were observed. The study found that the permeability and transportability of MWCNTs were intact in mature mustard plants while there was an enhancement in contaminant accumulation [93]. Miralles et al., [94] explained the toxicity and uptake of industrial-grade MWCNTs and their impurities in Alfalfa and Wheat. They found phytotoxicity in both seed germination and seedling growth. Similar studies highlighted the fact that CNTs were acting as contaminant carriers and eventually, translocating the contaminants within edible parts of the crops.
In a study [95], suspension rice cells were used with MWCNTs (Size:10–30 nm and concentration: 20,40 mg/L) that led to several negative effects like condensed chromatin, detachment of cell membrane from cell wall, generation of ROS and decreased cell viability and leading to cell death. In yet, another study the toxic effects of MWCNTs on rice cells were reported [96]. Scanning electron microscope results revealed that cell density decreased with increase in MWCNT concentration. It was concluded that the MWCNTs have direct contact with rice cells and produce an unfavourable effect on rice growth.
Stampoulis et al. [89] focused on the assay-dependent phytotoxicity of nanoparticles on plants. The study investigated the application of nanomaterials viz. MWCNTs, Ag, Cu, ZnO, Si and their response towards seed germination, root elongation, biomass, etc. The hydroponic assay trial showed that after 15 days, the biomass of plants exposed to MWCNTs was reduced to 60–70%.
In addition to this, no observed growth effects on seed germination were reported by few researchers in several plant species like Alfalfa, wheat [94], mustard, black lentil [97], barley, maize, soybean [74] and lettuce [98]. Another, study confirmed that there was no effect of MWCNTs on seed germination on different plant species (rape, radish, ryegrass, lettuce, corn and cucumber) [90]. Others emphasized the accumulation of CNTs in various parts of the plant, changes in gene expression and their involvement in various biochemical changes as discussed earlier.
Therefore, with the development and application of CNTs in the plant system the potential hazards related to this is receiving greater attention [40]. However, this field needs to be explored further prior to arriving at any conclusions about the effects of CNTs. Currently, studies are sparsely related to the mechanisms of CNTs toxicity in plants and hence, more focused approach is needed.
3 Conclusion and future prospects
This review explored recent developments in plant science that focuses on the role of CNTs in plant growth and development and also on plant mechanism. Nanomaterials, like CNTs, due to several advantageous properties i.e. small size, high surface area, ability to penetrate cell wall etc. provide promising possibilities for future studies. On the one hand, they show a great potential to enhance seed germination, plant development and various plant physiological processes. On the other hand, several studies also reported the deleterious effects of CNTs in the plant system. Currently, studies concerning CNTs interaction with plant system is at a nascent stage and needs further investigations to explore the mechanisms influencing the growth and toxicity in plants. There are several gaps in our present knowledge about CNTs than there are certainties and more research is needed in the following proposed areas:
-
Investigations concerning internal mechanisms (toxicity and genotoxicity) in plant system.
-
Outcome of CNTs in the plant system and their role in the food chain.
-
Identification of target plants and detailed analysis of cytotoxicity: toxicity study covering the life cycle of the plant.
-
Toxicity studies related to soil media and microbial population.
-
Need to explore other toxicity indicators in plants for instance biological markers etc.
-
At the genetic level, extensive study related to the expression of genes.
-
Thorough risk assessment of all nanomaterial-integrated products.
-
Phytoremediation.
References
Endo M, Strano MS, Ajayan PM (2007) Potential applications of carbon nanotubes. carbon nanotubes. Springer, Berlin, Heidelberg, pp 13–62
Herrero-Latorre C, Álvarez-Méndez J, Barciela-García J, García-Martín S, Peña-Crecente R (2015) Characterization of carbon nanotubes and analytical methods for their determination in environmental and biological samples: A review. Anal Chim Acta 853:77–94
Gandhi K, Lari S, Tripathi D, Kanade G (2016) Advanced oxidation processes for the treatment of chlorpyrifos, dimethoate and phorate in aqueous solution. J Water Reuse Desalin 6(1):195–203
Ren X, Chen C, Nagatsu M, Wang X (2011) Carbon nanotubes as adsorbents in environmental pollution management: a review. Chem Eng J 170(2–3):395–410
Ambrosi A, Pumera M (2010) Regulatory peptides are susceptible to oxidation by metallic impurities within carbon nanotubes. Chem European J 16(6):1786–1792
Tiwari D, Dasgupta-Schubert N, Cendejas LV, Villegas J, Montoya LC, García SB (2014) Interfacing carbon nanotubes (CNT) with plants: enhancement of growth, water and ionic nutrient uptake in maize (Zea mays) and implications for nanoagriculture. Appl Nanosci 4(5):577–591
Khodakovskaya MV, De Silva K, Biris AS, Dervishi E, Villagarcia H (2012) Carbon nanotubes induce growth enhancement of tobacco cells. ACS nano 6(3):2128–2135
Verma SK, Das AK, Gantait S, Kumar V, Gurel E (2019) Applications of carbon nanomaterials in the plant system: a perspective view on the pros and cons. Sci Total Environ 667:485–499
Tripathi D, Singh K, Mansoori MA (2019) Soil Pollution: Health Implications and Management. In: Kumar M, Tiwari RR (eds) Recent Trends and Advances in Environmental Health, vol 1. Nova Science Publishers. New York, USA, pp 41–64
Khot LR, Sankaran S, Maja JM, Ehsani R, Schuster EW (2012) Applications of nanomaterials in agricultural production and crop protection: a review. Crop Protec 35:64–70
Khodakovskaya M, Dervishi E, Mahmood M, Xu Y, Li Z, Watanabe F, Biris AS (2009) Carbon nanotubes are able to penetrate plant seed coat and dramatically affect seed germination and plant growth. ACS Nano 3(10):3221–3227
Khodakovskaya MV, Kim BS, Kim JN, Alimohammadi M, Dervishi E, Mustafa T, Cernigla CE (2013) Carbon nanotubes as plant growth regulators: effects on tomato growth, reproductive system, and soil microbial community. Small 9(1):115–123
Kwak S-Y, Lew TTS, Sweeney CJ, Koman VB, Wong MH, Bohmert-Tatarev K, Snell KD, Seo JS, Chua N-H, Strano MS (2019) Chloroplast-selective gene delivery and expression in planta using chitosan-complexed single-walled carbon nanotube carriers. Nat Nanotechnol 14(5):447–455
Begum P, Fugetsu B (2012) Phytotoxicity of multi-walled carbon nanotubes on red spinach (Amaranthus tricolor L) and the role of ascorbic acid as an antioxidant. J Hazard Mater 243:212–222
Lin C, Fugetsu B, Su Y, Watari F (2009) Studies on toxicity of multi-walled carbon nanotubes on Arabidopsis T87 suspension cells. J Hazard Mater 170(2–3):578–583. https://doi.org/10.1016/j.jhazmat.2009.05.025
Chen M, Zhou S, Zhu Y, Sun Y, Zeng G, Yang C, Xu P, Yan M, Liu Z, Zhang W (2018) Toxicity of carbon nanomaterials to plants, animals and microbes: Recent progress from 2015-present. Chemosphere 206:255–264
Hao Y, Ma C, Zhang Z, Song Y, Cao W, Guo J, Zhou G, Rui Y, Liu L, Xing B (2018) Carbon nanomaterials alter plant physiology and soil bacterial community composition in a rice-soil-bacterial ecosystem. Environ Pollut 232:123–136
Simon-Deckers A, Loo S, Mayne-L'hermite M, Herlin-Boime N, Menguy N, Reynaud C, Gouget B, Carriere M (2009) Size-, composition- and shape-dependent toxicological impact of metal oxide nanoparticles and carbon nanotubes toward bacteria. Environ Sci Technol 43(21):8423–8429. https://doi.org/10.1021/es9016975
Su W-C, Cheng YS (2014) Carbon nanotubes size classification, characterization and nasal airway deposition. Inhalation Toxicol 26(14):843–852
Jackson P, Jacobsen NR, Baun A, Birkedal R, Kühnel D, Jensen KA, Vogel U, Wallin H (2013) Bioaccumulation and ecotoxicity of carbon nanotubes. Chem Cent J 7(1):154
Ganesh E (2013) Single walled and multi walled carbon nanotube structure, synthesis and applications. Int J Innov Technol Explor Eng 2(4):311–320
Tománek D, Jorio A, Dresselhaus MS, Dresselhaus G (2007) Introduction to the important and exciting aspects of carbon-nanotube science and technology. Carbon Nanotubes. Springer, Berlin, Heidelberg, pp 1–12
Eatemadi A, Daraee H, Karimkhanloo H, Kouhi M, Zarghami N, Akbarzadeh A, Abasi M, Hanifehpour Y, Joo SW (2014) Carbon nanotubes: properties, synthesis, purification, and medical applications. Nanoscale Res Lett 9(1):393
Chico L, Crespi VH, Benedict LX, Louie SG, Cohen ML (1996) Pure carbon nanoscale devices: nanotube heterojunctions. Phys Rev Lett 76(6):971
Vander Wal RL, Berger G, Ticich T (2003) Carbon nanotube synthesis in a flame using laser ablation for in situ catalyst generation. Appl Phys A 77(7):885–889
Ajayan P, Ebbesen T (1997) Nanometre-size tubes of carbon. Rep Prog Phys 60(10):1025
Huczko A (2002) Synthesis of aligned carbon nanotubes. Appl Phys A 74(5):617–638
Iijima S (1991) Helical microtubules of graphitic carbon. Nature 354(6348):56–58
Thess A, Lee R, Nikolaev P, Dai H, Petit P, Robert J, Xu C, Lee YH, Kim SG, Rinzler AG (1996) Crystalline ropes of metallic carbon nanotubes. Science 273(5274):483–487
Cheng H, Li F, Su G, Pan H, He L, Sun X, Dresselhaus M (1998) Large-scale and low-cost synthesis of single-walled carbon nanotubes by the catalytic pyrolysis of hydrocarbons. Appl Phys Lett 72(25):3282–3284
Szabó A, Perri C, Csató A, Giordano G, Vuono D, Nagy JB (2010) Synthesis methods of carbon nanotubes and related materials. Materials 3(5):3092–3140
Prasek J, Drbohlavova J, Chomoucka J, Hubalek J, Jasek O, Adam V, Kizek R (2011) Methods for carbon nanotubes synthesis. J Mater Chem 21(40):15872–15884
Tiwari DK, Valenzuela J, Tripathi D, Mathew S (2020) Fabrication of carbon based nanostructured materials on Si/SiO2substrate and their growth mechanism over different catalysts. Nanomed Nanotechnol Open Access https://doi.org/10.23880/nnoa-16000175
Tendeloo G, Nagy J (1998) Purification of catalytically produced multi-wall nanotubes. J Chem Soc Faraday Trans 94(24):3753–3758
Djordjević V, Djustebek J, Cvetićanin J, Velićknović S, Veljković M, Bokorov M, Stojić BB, Nešković O (2006) Methods of purification and characterization of carbon nanotubes. J Optoelectron Adv Mater 8(4):1631–1634
Shanov V, Yun Y-H, Schulz MJ (2006) Synthesis and characterization of carbon nanotube materials. J University Chem Technol Metall 41(4):377–390
Belin T, Epron F (2005) Characterization methods of carbon nanotubes: a review. Mater Sci Eng, B 119(2):105–118
Mahalingam P, Parasuram B, Maiyalagan T, Sundaram S (2012) Chemical Methods for purification of carbon nanotubes–a review. J Environ Nanotechnol 1(1):53–61
Hou P-X, Liu C, Cheng H-M (2008) Purification of carbon nanotubes. Carbon 46(15):2003–2025
Du J, Wang S, You H, Zhao X (2013) Understanding the toxicity of carbon nanotubes in the environment is crucial to the control of nanomaterials in producing and processing and the assessment of health risk for human: a review. Environ Toxicol Pharmacol 36(2):451–462
Sun Y-P, Fu K, Lin Y, Huang W (2002) Functionalized carbon nanotubes: properties and applications. Acc Chem Res 35(12):1096–1104
Wepasnick KA, Smith BA, Bitter JL, Fairbrother DH (2010) Chemical and structural characterization of carbon nanotube surfaces. Anal Bioanal Chem 396(3):1003–1014
Li J, He Y, Han Y, Liu K, Wang J, Li Q, Fan S, Jiang K (2012) Direct identification of metallic and semiconducting single-walled carbon nanotubes in scanning electron microscopy. Nano Lett 12(8):4095–4101
García-Gutiérrez MC, Nogales A, Hernández JJ, Rueda DR, Ezquerra TA (2007) X-ray scattering applied to the analysis of carbon nanotubes, polymers and nanocomposites. Opt Pura Apl 40(2):195–205
Lopez-Lorente A, Simonet B, Valcárcel M (2014) Raman spectroscopic characterization of single walled carbon nanotubes: influence of the sample aggregation state. Analyst 139(1):290–298
Zang Z, Hu Z, Li Z, He Q, Chang X (2009) Synthesis, characterization and application of ethylenediamine-modified multiwalled carbon nanotubes for selective solid-phase extraction and preconcentration of metal ions. J Hazard Mater 172(2–3):958–963
Attal S, Thiruvengadathan R, Regev O (2006) Determination of the concentration of single-walled carbon nanotubes in aqueous dispersions using UV− Visible absorption spectroscopy. Anal Chem 78(23):8098–8104
Wenseleers W, Vlasov II, Goovaerts E, Obraztsova ED, Lobach AS, Bouwen A (2004) Efficient isolation and solubilization of pristine single-walled nanotubes in bile salt micelles. Adv Func Mater 14(11):1105–1112
Haggenmueller R, Rahatekar SS, Fagan JA, Chun J, Becker ML, Naik RR, Krauss T, Carlson L, Kadla JF, Trulove PC (2008) Comparison of the quality of aqueous dispersions of single wall carbon nanotubes using surfactants and biomolecules. Langmuir 24(9):5070–5078
Grossiord N, Regev O, Loos J, Meuldijk J, Koning CE (2005) Time-dependent study of the exfoliation process of carbon nanotubes in aqueous dispersions by using UV− Visible spectroscopy. Anal Chem 77(16):5135–5139
Schierz A, Parks AN, Washburn KM, Chandler GT, Ferguson PL (2012) Characterization and quantitative analysis of single-walled carbon nanotubes in the aquatic environment using near-infrared fluorescence spectroscopy. Environ Sci Technol 46(22):12262–12271
Pang LS, Saxby JD, Chatfield SP (1993) Thermogravimetric analysis of carbon nanotubes and nanoparticles. J Phys Chem 97(27):6941–6942
Chen C-M, Chen M, Leu F-C, Hsu S-Y, Wang S-C, Shi S-C, Chen C-F (2004) Purification of multi-walled carbon nanotubes by microwave digestion method. Diam Relat Mater 13(4–8):1182–1186
Mansfield E, Kar A, Hooker SA (2010) Applications of TGA in quality control of SWCNTs. Anal Bioanal Chem 396(3):1071–1077
Duesberg G, Blau W, Byrne H, Muster J, Burghard M, Roth S (1999) Chromatography of carbon nanotubes. Synth Met 103(1–3):2484–2485
Flavel BS, Moore KE, Pfohl M, Kappes MM, Hennrich F (2014) Separation of single-walled carbon nanotubes with a gel permeation chromatography system. ACS Nano 8(2):1817–1826
Lopez-Pastor M, Domínguez-Vidal A, Ayora-Canada M, Simonet B, Lendl B, Valcarcel M (2008) Separation of single-walled carbon nanotubes by use of ionic liquid-aided capillary electrophoresis. Anal Chem 80(8):2672–2679
Patel A, Tiwari S, Parihar P, Singh R, Prasad SM (2019) Carbon nanotubes as plant growth regulators: impacts on growth, reproductive system, and soil microbial community. In: Nanomaterials in plants, algae and microorganisms. Academic Press, pp 23–42
Yan S, Zhao L, Li H, Zhang Q, Tan J, Huang M, He S, Li L (2013) Single-walled carbon nanotubes selectively influence maize root tissue development accompanied by the change in the related gene expression. J Hazard Mater 246:110–118
Zhang H, Yue M, Zheng X, Xie C, Zhou H, Li L (2017) Physiological effects of single-and multi-walled carbon nanotubes on rice seedlings. IEEE Trans Nanobiosci 16(7):563–570
Caa JE, Long M, Nations S, Vadan R, Dai L, Luo M, Ambikapathi R, Lee EH, Olszyk D (2008) Effects of functionalized and nonfunctionalized single-walled carbon nanotubes on root elongation of select crop species. Environm Toxicol Chem Int J 27(9):1922–1931
Pourkhaloee A, Haghighi M, Saharkhiz MJ, Jouzi H, Doroodmand MM (2011) Carbon nanotubes can promote seed germination via seed coat penetration. Seed Technol 33(2):155–169
Haghighi M, da Silva JAT (2014) The effect of carbon nanotubes on the seed germination and seedling growth of four vegetable species. J Crop Sci Biotechnol 17(4):201–208
Flores D, Chacón R, Alvarado L, Schmidt A, Alvarado C, Chaves J (2014) Effect of using two different types of carbon nanotubes for blackberry (Rubus adenotrichos) in vitro plant rooting, growth and histology. Am J Plant Sci 5(24):3510
Khodakovskaya MV, De Silva K, Biris AS, Dervishi E, Villagarcia H (2012) Carbon nanotubes induce growth enhancement of tobacco cells. ACS Nano 6(3):2128–2135
Alimohammadi M, Xu Y, Wang D, Biris AS, Khodakovskaya MV (2011) Physiological responses induced in tomato plants by a two-component nanostructural system composed of carbon nanotubes conjugated with quantum dots and its in vivo multimodal detection. Nanotechnology 22(29):295101
Hatami M, Hadian J, Ghorbanpour M (2017) Mechanisms underlying toxicity and stimulatory role of single-walled carbon nanotubes in Hyoscyamus niger during drought stress simulated by polyethylene glycol. J Hazard Mater 324:306–320
Sun W, Shahrajabian MH, Huang Q (2020) Soybean seeds treated with single walled carbon nanotubes (SwCNTs) showed enhanced drought tolerance during germination. Int J Adv Biol Biomed Res (IJABBR) 8:9–16
Corredor E, Testillano PS, Coronado M-J, González-Melendi P, Fernández-Pacheco R, Marquina C, Ibarra MR, de la Fuente JM, Rubiales D, Pérez-de-Luque A (2009) Nanoparticle penetration and transport in living pumpkin plants: in situsubcellular identification. BMC Plant Biol 9(1):45
Wu Y, Phillips JA, Liu H, Yang R, Tan W (2008) Carbon nanotubes protect DNA strands during cellular delivery. ACS Nano 2(10):2023–2028
Martínez-Ballesta MC, Zapata L, Chalbi N, Carvajal M (2016) Multiwalled carbon nanotubes enter broccoli cells enhancing growth and water uptake of plants exposed to salinity. J Nanobiotechnol 14(1):42
Martinez-Ballesta MC, Chelbi N, Lopez-Zaplana A, Carvajal M (2020) Discerning the mechanism of the multiwalled carbon nanotubes effect on root cell water and nutrient transport. Plant Physiol Biochem 146:23–30
Tripathi S, Sonkar SK, Sarkar S (2011) Growth stimulation of gram (Cicer arietinum) plant by water soluble carbon nanotubes. Nanoscale 3(3):1176–1181
Lahiani MH, Dervishi E, Chen J, Nima Z, Gaume A, Biris AS, Khodakovskaya MV (2013) Impact of carbon nanotube exposure to seeds of valuable crops. ACS Appl Mater Interfaces 5(16):7965–7973
Tiwari D, Dasgupta-Schubert N, Villasenor L, Tripathi D, Villegas J (2013) Interaction of carbon nanotubes with mineral nutrients for the promotion of growth of tomato seedlings. Nano Studies 7:87–96
Mondal A, Basu R, Das S, Nandy P (2011) Beneficial role of carbon nanotubes on mustard plant growth: an agricultural prospect. J Nanopart Res 13(10):4519
Taha RA, Hassan MM, Ibrahim EA, Baker NHA, Shaaban EA (2016) Carbon nanotubes impact on date palm in vitro cultures. Plant Cell Tissue Organ Culture (PCTOC) 127(2):525–534
Smirnova E, Gusev A, Zaytseva O, Sheina O, Tkachev A, Kuznetsova E, Lazareva E, Onishchenko G, Feofanov A, Kirpichnikov M (2012) Uptake and accumulation of multiwalled carbon nanotubes change the morphometric and biochemical characteristics of Onobrychis arenaria seedlings. Front Chem Sci Eng 6(2):132–138
Larue C, Pinault M, Czarny B, Georgin D, Jaillard D, Bendiab N, Mayne-L’Hermite M, Taran F, Dive V, Carrière M (2012) Quantitative evaluation of multi-walled carbon nanotube uptake in wheat and rapeseed. J Hazard Mater 227:155–163
Pilevar ZT, Mahmoodzadeh H, Eshaghi A (2015) Impact of multi-walled carbon nanotubes on seed germination and seedling growth of Cichorium intybus L. L J Biol Environ Sci 6(1):438–445
Keller AA, Wang H, Zhou D, Lenihan HS, Cherr G, Cardinale BJ, Miller R, Ji Z (2010) Stability and aggregation of metal oxide nanoparticles in natural aqueous matrices. Environ Sci Technol 44(6):1962–1967
Dasgupta-Schubert N, Tiwari D, Francis ER, Torres PM, Villaseñor L, Mora CV (2017) Plant responses to nano and micro structured carbon allotropes: Water imbibition by maize seeds upon exposure to multiwalled carbon nanotubes and activated carbon. Adv Nano Res 5(3):245
Pereira MM, Mouton L, Yéprémian C, Couté A, Lo J, Marconcini JM, Ladeira LO, Raposo NR, Brandão HM, Brayner R (2014) Ecotoxicological effects of carbon nanotubes and cellulose nanofibers in Chlorella vulgaris. J Nanobiotechnol 12(1):15
Ferguson PL, Chandler GT, Templeton RC, DeMarco A, Scrivens WA, Englehart BA (2008) Influence of sediment− amendment with single-walled carbon nanotubes and diesel soot on bioaccumulation of hydrophobic organic contaminants by benthic invertebrates. Environ Sci Technol 42(10):3879–3885
Petersen EJ, Huang Q, Weber J, Walter J (2008) Bioaccumulation of radio-labeled carbon nanotubes by Eisenia foetida. Environ Sci Technol 42(8):3090–3095
Bullard-Dillard R, Creek KE, Scrivens WA, Tour JM (1996) Tissue Sites of Uptake of14C-Labeled C60. Bioorg Chem 24(4):376–385
Wang C, Liu H, Chen J, Tian Y, Shi J, Li D, Guo C, Ma Q (2014) Carboxylated multi-walled carbon nanotubes aggravated biochemical and subcellular damages in leaves of broad bean (Vicia faba L.) seedlings under combined stress of lead and cadmium. J Hazard Mater 274:404–412
Lin S, Reppert J, Hu Q, Hudson JS, Reid ML, Ratnikova TA, Rao AM, Luo H, Ke PC (2009) Uptake, translocation, and transmission of carbon nanomaterials in rice plants. Small 5(10):1128–1132
Stampoulis D, Sinha SK, White JC (2009) Assay-dependent phytotoxicity of nanoparticles to plants. Environ Sci Technol 43(24):9473–9479
Lin D, Xing B (2007) Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ Pollut 150(2):243–250
Ghosh M, Chakraborty A, Bandyopadhyay M, Mukherjee A (2011) Multi-walled carbon nanotubes (MWCNT): induction of DNA damage in plant and mammalian cells. J Hazard Mater 197:327–336
Zhai G, Gutowski SM, Walters KS, Yan B, Schnoor JL (2015) Charge, size, and cellular selectivity for multiwall carbon nanotubes by maize and soybean. Environ Sci Technol 49(12):7380–7390
Chen G, Qiu J, Liu Y, Jiang R, Cai S, Liu Y, Zhu F, Zeng F, Luan T, Ouyang G (2015) Carbon nanotubes act as contaminant carriers and translocate within plants. Scientific Rep 5:15682
Miralles P, Johnson E, Church TL, Harris AT (2012) Multiwalled carbon nanotubes in alfalfa and wheat: toxicology and uptake. J R Soc Interface 9(77):3514–3527
Tan X-m, Lin C, Fugetsu B (2009) Studies on toxicity of multi-walled carbon nanotubes on suspension rice cells. Carbon 47(15):3479–3487
Tan X-m, Fugetsu B (2007) Multi-walled carbon nanotubes interact with cultured rice cells: evidence of a self-defense response. J Biomed Nanotechnol 3(3):285–288
Ghodake G, Seo YD, Park D, Lee DS (2010) Phytotoxicity of carbon nanotubes assessed by Brassica juncea and Phaseolus mungo. J Nanoelectron Optoelectron 5(2):157–160
Giraldo JP, Landry MP, Faltermeier SM, McNicholas TP, Iverson NM, Boghossian AA, Reuel NF, Hilmer AJ, Sen F, Brew JA (2014) Plant nanobionics approach to augment photosynthesis and biochemical sensing. Nat Mater 13(4):400–408
Acknowledgement
Authors are highly grateful for the CONACYT Basic Science project (A1-S-47641), DST FIST Laboratory, Vimala College (Autonomous), Thrissur and DBT STAR College Scheme, Department of Biotechnology, Govt. of India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mathew, S., Tiwari, D.K. & Tripathi, D. Interaction of carbon nanotubes with plant system: a review. Carbon Lett. 31, 167–176 (2021). https://doi.org/10.1007/s42823-020-00195-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42823-020-00195-1