Abstract
Phosphorus (P) is one of the essential macronutrients for plant metabolism. Regardless of its great quantity in inorganic and organic forms, it is generally inaccessible for plant utility due to bond formation with other ions present in soil. Due to the excessive use of agrochemicals, environmental issues have reached their peak. This has increased the interest of the scientific community in finding a sustainable alternative to chemical fertilisers. Diverse microbes like Rhizobium spp., Serratia spp., Pseudomonas spp., Bacillus spp., Azotobacter spp., Penicillium spp., Rhizopus spp., Fusarium spp., and various actinomycetes have been isolated and screened as phosphorus solubilizing microorganisms (PSMs). The PSMs also act as biological control agents (bioagents) and help to withstand extreme stress circumstances (like heavy metal toxicity) by producing ACC deaminase. With the advent of time, organic farming is gaining attention as this technology is highly eco-friendly, so utilisation of potential microorganisms for solubilisation of phosphorus will improve soil health and crop productivity. PSMs possess significant heavy metal remediation potential; therefore, they can be used in restoration of contaminated soil as well as in enhancing plant health. This review will provide in-depth knowledge about PSMs and their role in sustainable agriculture and bioremediation of toxicants.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
There is a necessity for adequate food requirements to overcome the increasing population demand at global level. To increase the productivity, farmers are using chemical-based fertilizers that are improving crop yield, however it is proven that application of these chemicals have adverse impact on soil productiveness (Yatoo et al. 2021; Fatima et al. 2021). A profitable mode of availing phosphorus to plants is using PSMs (Kirui et al. 2022). Efforts to enhance the soil biological system are essential to address the aforementioned issues. Although phosphorus (P) is a macronutrient, it is crucial for plant health and often required in significant amounts (Kirui et al. 2022; Rawat et al. 2021; Kalayu 2019), contributing to 0.2 percent dry weight of plant (Maharajan et al. 2018). It is one of the main components of phosphoplipids, phosphoproteins, and cofactors and is the constituent of skeletal structure of genetic material of living creatures (Ozane 1980; Bai et al. 2020; Timofeeva et al. 2022; Feng et al. 2024). P is the vital element associated in plant metabolism with many functions like cell division, nitrogen fixation, nucleic acid and protein formation, crop quality, disease resistance (Khan et al. 2014; Nesme et al. 2018; Koch et al. 2018). Though soil holds entire P in organic and inorganic form, maximum of them remains locked thus becomes unavailable to plants (Kalayu 2019; Dong et al. 2023).
Phosphorus deficiency distresses root architecture (Williamson et al. 2001). Phosphorus deficiency leads to pale, dull, blue-green leaves and delayed maturity (Bai et al. 2013). This macronutrient is utilized in an inorganic form either as H2PO44− or HPO42− or both (Hinsinger 2001; Divjot et al. 2021; Li et al. 2021). Numerous microorganisms produce low molecular weight organic acids, such as acetic acid, citric acid, oxalic acid, malic acid, and gluconic acid, which help solubilize P. These acids chelate cationic partners of phosphorus ions, thereby releasing phosphorus directly into the soil solution. Microorganisms, including various species of Aspergillus, Fusarium, Penicillium, Serratia, Bacillus, Azotobacter, Rhizobium and Pseudomonas, play a crucial role in this process. They assimilate phosphorus and prevent its immobilization or fixation, thus enhancing its availability to plants. This microbial activity is vital for maintaining soil fertility and promoting healthy plant growth (Khan et al. 2009a, b, c).
Microorganisms, through various processes such as mineralization, solubilization, and decomposition influences soil fertility. Microbes increase phosphorus availability by solubilizing the locked form of phosphorus (Chen et al. 2006; Kang et al. 2002). Microorganisms acts as a substitute for phosphorus fertilizers (Padmavathi and Usha 2012). From couple of decades numerous PSMs studies were under focus which include various species of Serratia, Micrococcus, Enterobacter, Azotobacter, Rhizobium and Micrococcus (Kirui et al. 2022; Chawngthu et al. 2020; Mohamed et al. 2018; Yadav et al. 2016) Rhizobium, Arthrobacter and Rahnella aquatilis HX2 (Chawngthu et al. 2020; Zhang et al. 2019; Liu et al. 2019), Leclercia adecarboxylata (Teng et al. 2019a), the other fungi function in same way which includes species of Penicillium, Aspergillus, Fusarium, and Hymenella (Zhu et al. 2011; Ichriani et al.2018) are potential PSMs and can be used as an substitute for phosphorus fertilizers (Padmavathi and Usha 2012). Phosphate solubilizing microbes produce some essential phyto hormones like cytokinins, auxins, and gibberellins which have a role in cell division, differentiation, root and shoot development, germination, flowering (Puri et al. 2020).
Moreover, bioremediation has attracted a lot of scientific attention in recent years. Heavy metals like Cu, Pb, Ni, Hg, Cd, and Zn among others, play a role in several environmental problems. Heavy metals pose a significant challenge as pollutants that are not easily broken down. These pollutants can originate from either natural sources or human activities (Tan et al. 2021). Microorganisms like bacteria, fungi, yeast and actinomycetes are known for bioremediation of heavy metals. Microbes have demonstrated remarkable effectiveness in the remediation of environmental pollutants (Ahmed et al. 2022; Hamid et al. 2023a; Hamid et al. 2023b). Various phosphate solubilizing microbial strains which belong to genus Bacillus, Pseudomonas, Paenibacillus, Staphylococcus, Aspergillus, Rhizopus, Trichoderma and Brevibacterium have been reported as bioremediators (Khambhaty et al. 2008; Vala and Sutariya 2012; Yahaghi et al. 2018; Bashir et al. 2018a, b).
2 Phosphorus Solubilizing Microorganisms (PSMs)
PSMs are abundant in the soil and may be readily isolated from a diverse rhizospheric soils (Manoj et al. 2018). The number of P solubilizers present in a soil are based on many factors like organic content and properties of soil (Menezes-Blackburn et al. 2013). Phosphorus solubilizers are observed in rhizosphere where many chemical reactions occur (Mitra et al. 2020; Chawngthu et al. 2020). PSMs play central part in phosphorus cycling in the environment (Khan et al. 2024; Mitra et al. 2020; Saha and Biswas 2009). Since 1903, the PSMs (bacteria, fungi, actinomycetes) that occurs naturally as per reports of Khan et al. (2009a, b, c) play an important role in phosphorus solubilization in soil.
Phosphorus occurs either in organic form or as phosphorite and many solubilizing microorganisms have been obtained and identified from varied habitats (Acevedo et al. 2014; Taktek et al. 2015; Bashir et al. 2018a; Chawngthu et al. 2020; Divjot et al. 2021). PSMs includes many species of Azotobacter, Bacillus, Serratia, Enterobacter, Micrococcus, Rhizobium, Penicillium, Aspergillus, Rhizopus, Fusarium etc. (Kour et al. 2020). These microbial species have been isolated and screened from aquatic and terrestrial habitats (De Souza et al. 2000; Chakdar et al. 2018). These were also isolated and identified from rhizosphere of rice, banana, vegetable, apple, pear, walnut (Chawngthu et al. 2020; Bashir et al. 2019a, b; Bashir et al. 2017, 2018a, b; Kumar et al. 2013; Naik et al. 2008). A part from many species of Bacillus and Pseudomonas other potential P-solubilizers are Rhodococcus, Serratia, Arthrobacter, Chryseobacterium, Xanthomonas, Azotobacter, Klebsiella, Enterobacter, Acinetobacter (Wani et al. 2007; Kumar et al. 2013; Chen and Liu 2019; Chawngthu et al. 2020; Zhao et al. 2023). Here are some examples of PSMs that have been studied (Table 1).
Among all PSMs in soil PSB constitutes 1 to 50%, fungi constitute 0.1 to 0.5% (Chen et al. 2006). Among actinomycetes, 20% of the genera Micromonospora, Actinomyces, and Streptomyces can solubilize P (Aallam et al. 2021; De Zutter et al. 2022). Soil is the hotspot for diverse microflora. Phosphorus solubilizers are the microorganisms that have ability of changing locked form of P in to a plant accessible form (Chawngthu et al. 2020; Silva et al. 2023; Khan et al. 2024). Their potency for solubilization of P can be analysed via both qualitative and quantitative approach (Mehta and Nautiyal 2001). These PSMs are diversified in nature, bacteria from the genera Serratia, Leclercia adecarboxylata, Enterobacter, Pantoea, Bacillus, Pseudomonas, Arthrobacter and fungi like Aspergillus niger, Penicillium spp, Acremonium, Hymenella and Neosartorya are potent PSMs (Kirui et al. 2022; Biswas et al. 2018; Bashir et al. 2018a, b; Zhang et al. 2019; Teng et al. 2019a; Ichriani et al. 2018; Rojas et al. 2018; Sulbaran et al. 2009). Phosphate solubilizers have been reported as N2 fixers and regulate plant hormone levels (Unnikrishnan and Binitha 2024). There are various reports on isolation and characterization of diverse phosphorus solubilizers from different environmental sources. These PSMs by their metabolic activities perform a dynamic role in soil. These microbes act as potent biofertilizers that enhance agriculture produce and maintain soil fitness (Rajwar et al. 2018).
Phosphorus solubilizers produce various hormones like auxins, cytokinins, gibberellins that enhance seed germination, cell differentiation, shoot elongation and flowering (Puri et al. 2020). An innovative choice strain like Pseudomonas plecoglossicida isolated from rhizospheric soil of soybean, also produced growth promoting hormones like Indole-3-Acetic Acid (IAA) (Astriani et al. 2020). Studies suggested that Trichoderma harzianum can also solubilize P, the strain produced IAA, improved root and shoot biomass, leaf number and size observed on Solanum lycopersicum L. (Bader et al. 2020). Various microorganisms, including bacteria and fungi, are known for their ability to produce organic acids. Pseudomonas produces gluconic acid to increase phosphate solubility, which has become an important technology for improving phosphate fertilizer management in modern agriculture (Wang et al. 2022; Rai et al. 2023). Here are some common organic acids and the microorganisms that often produce them (Table 2).
3 Mechanisms of Inorganic Phosphorus Solubilization
The PSMs uses various strategies for phosphorus solubilization. Out of which the principal mechanism is low molecular weight (MW) secretions which include organic acids (Citric acid, Malic acid, Gluconic acid, and Oxalic acid) (Saeid et al. 2018; Marra et al.2015; Buch et al. 2008; Liu et al. 2024).Gluconic acid is the main acid secreted by these PSMs (Zhang et al. 2023a, b).The generation of these low MW secretions `causes acidification of microbial cells as well as its surroundings (Lin et al. 2006; Chen et al. 2006; Silva et al. 2023; Khan et al. 2024). Subsequent ionization of acid occurs, and proton released becomes responsible for expelling phosphorus from phosphate via proton substitution for calcium, aluminium, and iron or chelate cations from carboxylic anions and releasing phosphate anions (Aliyat et al. 2022). The release of organic acids has also been well established (Park et al. 2009; Lin et al. 2006). Studies suggested that there occur some genes which are indirectly or directly involved in secretions of these low molecular weight organic acids (Babu-Khan et al. 1995; Buch et al. 2010; Pang et al. 2024). The -COOH and -OH functional groups of these acids compete with Ca2+, Al3+ and Fe2+, make bonds with metal ions (chelation) and thus transforms phosphorus from insoluble to soluble state (Kpomblekou and Tabatabai 1994). The estimation of these compounds (acids) can be done by HPLC (Wei et al. 2017; Park et al. 2009).Siderophores i.e., Fe2+ chelating agents, phosphatases and extracellular polysaccharides (EPS) synthesized by PSBs results in availability of P in its soluble form from fixed phosphate form (Sharma et al. 2013; Yi et al. 2008; Chen and Arai 2023; Thampi et al. 2023). Thus, the various chelating agents, low molecular weight organic acids and enzymes produced by PSMs are main compounds that cause solubilization of inorganic phosphorus.
4 Mechanism Involved in Organic Phosphorus Mineralization
Organic matter is the key basis of phosphorus availability in the soil, various organic phosphorus compounds are nucleic acids, phospholipids, and phosphodiesters and so on (Rodríguez and Fraga 1999). Besides pesticides, antibiotics, that are released in to the environment also contain organic phosphorus. Phosphorus mineralization means solubilization of organic phosphorus. Microorganisms solubilize P by secretion of various enzymes as among them are those that dephosphorylate the phosphorester or phosphoanhydride bond in organic compounds. PSM, mostly release phosphomonoesterases or phosphatases (Nannipieri et al. 2011), these enzymes i.e., phosphomonoesterases can occur in both acidic as well as in alkaline forms (Jorquera et al. 2011). Other class of enzyme secreted by phosphate solubilizers in the process of mineralization of organic P is phytase (Silva et al. 2023; Khan et al. 2024). This phytase enzyme releases the phosphorus from organic materials like plant seeds and pollen. The phytase during degradation process releases phosphorus in a form which is available for plant uptake (Richardson and Simpson 2011).
5 Phosphate Solubilizers as Plant Growth Enhancers
Microorganisms that solubilize phosphates exert indirect or direct influences on plant health and their yield (Zveushe et al. 2023). Direct process comprises improved solubilization of minerals such as K, P, Si and Zn etc. (Prakash and Arora 2019; Bashir et al. 2017; Hayat et al. 2010). The substances /chemicals released by PSB that enhance plant growth are mentioned in (Table 3).
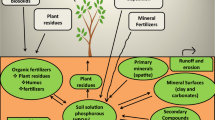
The PSBs have showed good performance for plant development (Prakash and Arora 2019; Wen et al. 2019). These phosphate solubilizing microbial strains increase plant health and yield as they improve the biogeochemical cycling (Fig. 1) (Mitra et al. 2020; Hutchins et al. 2019), prevents from pathogen infestation (Bononi et al. 2020; Saravana et al. 2008), yield some vitamins like riboflavin, biotin, niacin etc., for plant development (Jaiswal et al. 2021; Revillas et al. 2000), phytohormones (Mažylytė et al. 2022; Kalayu 2019; Prakash and Arora 2019), and iron scavenging molecules (Purwaningsih et al. 2021; Prakash and Arora 2019) (Table 4).
6 Phosphate Solubilizing Microbes as Biocontrol Agents
Increased application of chemical pesticides has given rise to environmental contamination, reduced beneficial soil biota, and enhanced vulnerability of crops to disease (Yatoo et al. 2021, 2022). The strategy to combat disease is currently being explored, with a focus on using PSMs as biological control agents. In addition to giving plants vital nutrients, PSMs operate as plant growth-promoters and hinder the growth of a variety of diseases, including nematodes (Thomas et al. 2005; Khan and Kounsar 2000; Oyekanmi et al. 2008; Khan et al. 2009a, b, c). PSMs are efficient biocontrol agent for plant diseases. They affect directly different stages of development in nematodes; eggs, larvae and adults (Pocasangre et al. 2007; Pant and Pandey 2001; Oyekanmi et al. 2008; Sharon et al. 2009).
A PSB, Bacillus amyloliquefaciens can inhibit the growth of F. oxysporum can prevent the growth of F. oxysporum through the production of antifungal components 1-amino-cyclo-propane-carboxylic-aciddeaminase, and enzymes like chitinase and cellulase (Gowtham et al. 2016). Chitinase enzymes can inhibit F. oxysporum attack on chilli seeds (Suryanto et al. 2014), and G. philippii (Widyastuti and Sumardi 1998) causing red roots in acacia plants.
Several bacteria from the genera Azotobacter, Pseudomonas and Bacillus are recognised to be vital in the solubilization of soil minerals and to inhibit plant diseases (Khan et al. 2005; Mardhiansyah 2011; Gill et al. 2016; Bashir et al. 2017; Bashir et al. 2018a; Prihatini et al. 2018; Bashir et al. 2018b; Bashir et al. 2019a, b., Bashir et al.2019b; Gowtham et al. 2016). To suppress parasitic nematodes, different phosphate solubilizing bacteria were used in various plants. For example Azospirillum lipoferum was used against Meloidogyne incognita in green gram (Vigna radiata) (Khan and Kounsar 2000), Bacillus subtilis was used against Rootknot nematode (Meloidogyne spp.) of tomato (Solanum lycopersicum) (Jiménez-Aguirre et al. 2023), Pseudomonas fluorescens against Heterodera schachtii in sugar beet (Beta vulgaris) Oostendrop and Sikora (1989), B. subtilis was used against M. incognita in ornamental plants (Khan et al. 2005b), P. fluorescens was used against Heterodera schachtii in sugar beet, Beijerinkia indica was used against M. incognita in green gram (Khan and Akram 2000; Khan et al. 2009a, b, c). These bacteria suppress nematode pathogenesis, as well as encourage plant development by solubilizing minerals in the soil (Khan et al.2009a, b, c).
Phosphate solubilizing bacteria suppress the growth of plant pathogens by adapting different mechanisms. Bacteria gets attached to roots by means of pili in case of Pseudomonas fluorescens on wheat roots (Patil et al. 2002; Khan et al. 2009a, b, c) or involve development and multiplication on the root surface. Various antifungal components are also produced by bacteria that are active against various pathogens including nematodes, these include iturin, mycosubtilin, surfactin, bacilysin, fengymycin HCN, mycobacilin, ammonia, 2,4-diacetylphloroglucinol, xantobaccin, butyrolactones, oomycin, kanosamine, oligomycin, phenazine-1-carboxylicacid, viscosinamide, pyrrolnitrin, zwittermycin A etc. (Khan et al. 2009a, b, c). They stimulate the systemic resistance (SR) in the host against the pathogenic microbes. After inoculating with the biocontrol bacteria, they bring the following changes in the host; increased the level of enzymes like phenylalanine ammonia lyase, polyphenol oxidase, peroxidase and chitinase (M’Piga et al. 1997; Chen and Dickson 2004; Mansoor et al. 2023a), increased production of phytoalexins, enhanced gene expression related to response to stress, deposition of barriers and strengthening of cell walls outside infection sites including phenolics, lignin and cellulose (Khan et al. 2009a, b, c). Bacteria produce a large number of siderophores or iron chelators that have high affinity for iron. They sequester the iron present in the rhizosphere making it inaccessible to pathogens, thus limiting their growth (Khan et al. 2009a, b, c). P. fluorescens and B. subtilis promote growth of plants directly as well as indirectly. Growth promotion via indirect means is based on the suppression of plant parasites (soil borne) and pathogenic microorganisms in the rhizosphere, whereas direct growth promotion is primarily exerted through the release of growth factors. B. subtilis and P. fluorescence synthesizes plant growth promoting hormones such as gibberellins, indole acetic acid, zeatin and cytokinins that enhances plant growth (Khan et al. 2009a, b, c).
Various P solubilizing fungi inhibits the growth of various microbes including nematodes (Khan and Kounsar 2000; Oyekanmi et al. 2008) and other pathogenic fungi (Padmavathi and Madhumathi 2009). In addition to supplying vital nutrients in plants (Thomas et al. 2005), imparts a vital role on solubilizing insoluble phosphorus (Turan 2006) allowing plants to use it (Mittal et al. 2008; Oyekanmi et al. 2008; Khan and Kounsar 2000). Some fungi Aspergillus niger, Penicillium digitatum, Penicillium anatolicum, Trichoderma viride and Trichoderma harzianum, play a major part in phosphate solubilisation and have been used in biomanagement of different pathogenic fungi and nematodes in different plants like A. niger was used in controlling Meloidogyne incognita in tomato (Tayade et al. 2019), okra (Sharma et al. 2005), Paecilomyceslilacinus was used in controlling Meloidogyne spp. in tomato (Schenek 2004), tobacco, P. anatoticum was used in controlling Globodera rostochinensis in potato (Jatala 1986), Trichoderma harzianum, and P. lilacinus was used in controlling M. incognita in chickpea (Pant and Pandey 2001), Trichoderma harzianum and Pseudomonas fluorescens against M. javanica in tomato (Siddiqi and Shaukat 2004). The many developmental stages of nematodes, including eggs, larvae, and adults, may be directly impacted by phosphorus solubilizing fungi (PSF) (Pocasangre et al. 2007, Pant and Pandey 2001; Oyekanmi et al. 2008; Sharon et al. 2009). Trichoderma, a main biocontrol fungus is found in almost all soils and in a diversity of habitats. They are the most common culturable fungi found in soil and colonize plant roots. Trichoderma sp. invade, parasitize, or feed on other fungi. Because it is usually abundant in healthy roots, they evolved many mechanisms to combat other fungi and encourage the growth of plants and roots (Benítez et al. 2004).
The effect of Trichoderma harzianum on different isolates of Xanthomonas was studied and it was observed that Xanthomonas was completely lysed by T. harzianum and inhibited their growth (Padmavathi and Madhumathi 2009). The phosphate-solubilizing fungi adapt different mechanisms to suppress plant pathogens. According to Lipping et al. (2008), antibiosis gives phosphate-solubilizing fungus species of Trichoderma, Penicillium and Aspergillus, a competitive saprophytic advantage. Different species of Trichoderma and Aspergillus produce low molecular weight compounds both volatile and non-volatile that impedes the growth of harmful microbes including in the root zone. Some of the compounds produced by Aspergillus sp. and Trichoderma sp. include harzianic acid, viridin, tricholin, alamethicins, glisoprenins, peptaibols, massoilactone, heptelidic acid, antibiotics, 6-penthyl-pyrone, oxalic acid, gliovirin, and enzymes (Khan et al. 2009a, b, c).
It is believed that the antagonistic action of phosphorus dissolving fungus, like strains of Trichoderma, A. niger and P. digitatum, on the invading pathogen allows them to defend plants against various pathogens (Khan et al. 2009a, b, c). Trichoderma strains have the potential to cause plants to exhibit hypersensitive reactions systemic generated resistance as well as acquired systemic resistance (Harman et al. 2004). Different species of Trichoderma induces resistance in their host by stimulating the production of genes that stimulate the production of phytoalexins, Pathogenesis-related (PR) proteins and other chemicals that increase plant pathogen resistance. Trichoderma harzianum induced resistance in various plants like cucumber, bean, and cotton (Khan et al. 2009a, b, c). Trichoderma releases growth factors that enhance plant growth, crop yield as well as seed germination rates (Benítez et al. 2004). Trichoderma sp and A. niger also produces phytohormones like zeatin and gibberellic acid, and their production might enhance bio fertilization. Thus, with increased nutrient absorption and the synthesis of these plant hormones directly influence plant development, control of different pathogens and enhancing defence mechanism of plants (Khan et al. 2009a, b, c).
7 Phosphate Solubilizers and Bioremediation
Bioremediation has fascinated a lot of scientific attention in recent years. Its mechanisms are based on redox transformations, absorption, and changes in the reaction of the medium. Presently, the most common methods of microbial removal of heavy metals are bioleaching, biomeneralization, biosorption, bioaccumulation, and production of biosurfactants, oxidation–reduction, and biovolatilization (Rahman and Singh 2020). Some specific genera of microorganisms called extremophiles are involved in such processes, these specific microbial strains, are able to develop protective systems to avoid negative effects of heavy metal based pollution. Therefore, the ability of microorganisms to remain viable under the influence of heavy metals in the restoration of disturbed areas is of pivotal importance (Ayangbenro and Babalola 2017; Hamid et al. 2023a, b). According to literature data, it is possible to use Bacteroidetes and Firmicutes for As-contaminated areas. Their abundance positively correlates with this pollutant in contaminated areas. It is also noted that proteobacteria are resistant to high concentrations of Zn, as well as Pb (Fajardo et al. 2019). Under the heavy metal stress some microbial strains are able to secrete or release extracellular substances such as proteins, polysaccharides, lipids containing various heavy metal binding sites (Tarfeen et al. 2022). Diverse phosphate solubilizing microbial communities have been reported which play important role in heavy metal remediation.
Considerable attention is being given to the microbial breakdown of agricultural chemicals such as pesticides, insecticides, fungicides, chemical fertilizers, and other toxic compounds (Rafa and Chiampo 2021). Besides this microbial community benefit the plants by direct or indirect way like phosphate solubilization, potassium solubilization, zinc solubilization, silicate solubilization, nitrogen fixation, 1-aminocyclopropane-1-carboxylate deaminase (ACCD) production, HCN, siderophore (iron chelating agents) production and phytohormone secretions (Glick et al. 2007; Bashir et al. 2017, 2018a, b; Gouda et al. 2018; Kalam et al. 2020; Park et al. 2021; Saeed et al. 2021; Kaur et al. 2024).
Several research reports have shown that PSMs strains including Bacillus spp, Azospirillum lipoferum, Pseudomonas fluorescence, Rhizobium spp that remediate chemicals and also secrete gibberellic acid (GBA) that induce plant health (Bottini et al. 2004; Gouda et al. 2018). These rhizobacterial phosphate solubilizers also secrete EPS which showed effect on growth and development of plant and drought tolerance (Naeem et al. 2018; Bashir et al. 2021; Kaur et al. 2024). Numerous plant growth-promoting features of phosphate solubilizers such as organic acid production (citric, malic, oxalic, propionic acids), secretion of iron chelating agents (siderophores), indole acetic acid production and ACC deaminase activity enhances the phytoremediation capability of plants (Park et al. 2009; Cui et al. 2022). Plentiful P solubilizing strains belonging to the genera of Pseudomonas, Bacillus, Paenibacillus, Brevibacterium, and Staphylococcus have been reported to solubilize phosphate as well as dissolves the Lead (Pb) mineral. PSM in addition to chelating agent Ethylene diamine tetra acetic acid when added to Arsenic contaminated soil, phytoremediation property of Echinochloa frumentacia is enhanced and it has been reported within 85 days from the seeding of Echinochola frumentacia, arsenic contamination in the soil decreases when compared with initial concentration (Yahaghi et al. 2018). Inoculation with both Brevibacterium frigoripolerans YSP40 and Bacillus paralicheniformis YSP151 and their consortium have been reported to enhance the growth and Pb uptake of Brassicajuncea plants grown in metal contaminated soil. Hence, phosphate solubilizers have an effective role to play in bioremediation (Yahaghi et al. 2018).
8 Conclusion
Phosphorus is an indispensable mineral element amongst the macronutrients needed for different plant metabolic functions. Phosphorus is available in soil as inorganic as well as in organic form, but due to its limited accessibility since it occurs in an insoluble form. Because of its complex formation and meagre soil solubility only few percent of total phosphorus are available to plants. Phosphorus accounts for around 0.2–0.8% of the dry weight of the plant. To fulfil the needs of crop nutrition, phosphorus is generally added to soil in chemical form but the synthesis of phosphorus fertilizer is expensive and effortful process. In addition, plants utilize a small quantity of this phosphorus and 75–90% of added phosphorus forms metal-cation complexes, gets precipitated and is quickly locked in soils. Microbiologists and experts in soil science have setup a way to make phosphorus available to crops, a replacement for crop fertilization that is economically successful. Such ecological concerns result in exploration of crop nutrition for a sustainable way of phosphorus. In this regard, Phosphorus solubilizing microorganism is seen as the best environmentally sustainable means for crop phosphorus nutrition, which farmers can afford. Bioremediation being an interesting and highly applied tool for decontamination of heavy metal polluted soils and sits, number of microbes possess remediation capacity and are utilized at large scale. Phosphorus solubilizing microorganisms has proven to be a potential bio-remediating agent and can be applied in agriculture soils for solubilisation as well as remediation purpose. Future insight about improving bioremediation potential of phosphorus solubilizing microorganisms need to be explored for enhancing quality of soil health and plant health.
Abbreviations
- ACC:
-
1-Aminocyclopropane-1-carboxylic acid
- EDTA:
-
Ethylene diamine tetra acetic acid
- EPS:
-
Exopolysaccharides
- HCN:
-
Hydrogen cyanide
- IAA:
-
Indole-3-acetic acid
- PAL:
-
Phenylalanine ammonia lyase
- PSMs:
-
Phosphorus solubilizing mmicroorganisms
- PSF:
-
Phosphorus solubilizing fungi
- PSB:
-
Phosphorus solubilizing bacteria
- ACCD:
-
1-Aminocyclopropane-1-carboxylate deaminase
- MW:
-
Molecular weight
References
Aallam Y, Dhiba D, Lemriss S, Souiri A, Karray F, Rasafi TE, Hamdali H (2021) Isolation and characterization of phosphate solubilizing streptomyces sp. endemic from sugar beet fields of the BeniMellal region in Morocco. Microorganisms 9(5):914. https://doi.org/10.3390/microorganisms9050914
Acevedo E, Galindo-Castaneda T, Prada F, Navia M, Romero HM (2014) Phosphate-solubilizing microorganisms associated with the rhizosphere of oil palm (Elaeis guineensis Jacq) in Colombia. Appl Soil Ecol 80:26–33. https://doi.org/10.1016/j.apsoil.2014.03.011
Afzal A, Bano A, Fatima M (2010) Higher soybean yield by inoculation with N-fixing and P-solubilizing bacteria. Agron Sustain Dev 30:487–495. https://doi.org/10.1051/agro/2009041
Ahmed NE, Salem SS, Hashem AH (2022) Statistical optimization, partial purifcation, and characterization of phytase produced from Talaromyces purpureogenus NSA20 using potato peel waste and its application in dyes de-colorization. Biointerface Res Appl Chem 12:4417–4431
Akintokun AK, Akande GA, Akintokun PO, Popoola TOS, Babalola AO (2007) Solubilization of insoluble phosphate by organic acid producing fungi isolated from Nigerian soil. Int J Soil Sci 2:301–307. https://doi.org/10.3923/ijss.2007.301.307
Aliyat FZ, Maldani M, El Guilli M, Nassiri L, Ibijbijen J (2022) Phosphate solubilizing bacteria isolated from phosphate solid sludge and their ability to solubilize three inorganic phosphate forms: calcium, iron, and aluminium phosphates. Microorganisms 10:980. https://doi.org/10.3390/microorganisms10050980
de Almeida Leite R, Martins da Costa E, Cabral Michel D, do Amaral Leite A, de Oliveira-Longatti SM, de Lima W, Konstantinidis KT, de Souza Moreira FM (2024) Genomic insights into organic acid production and plant growth promotion by different species of phosphate-solubilizing bacteria. World J Microbiol Biotechnol 40(10):311. https://doi.org/10.1007/s11274-024-04119-3
Altomare C, Norvell WA, Bjorkman T, Harman GE (1999) Solubilization of Phosphates and micronutrients by the plant growth promoting and biocontrol fungus Trichoderma hazarianum Rifai. Appl Environ Microbiol l44:1295–22. https://doi.org/10.1128/AEM.65.7.2926-2933.1999
Arwidsson Z, Johansson E, Kronhelm TV, Allard B, VanHees P (2010) Remediation of metal contaminated soil by organic metabolites from fungi I-production of organic acids. Water Air Soil Pollut 205:215–226. https://doi.org/10.1016/j.jhazmat.2009.08.141
Astriani M, Zubaidah S, Abadi AL, Suarsini E (2020) Pseudomonas plecoglossicida as a novel bacterium for phosphate solubilizing and indole-3-acetic acid-producing from soybean rhizospheric soils of East Java, Indonesia. Biodiversitas 21:578–586. https://doi.org/10.13057/biodiv/d210220
Ayangbenro AS, Babalola OO (2017) A New strategy for heavy metal polluted environments: a review of microbial bio sorbents. Int J Environ Res Public Health 14:94
Babu-Khan S, Yeo TC, Martin WL, Duron MR, Rogers RD, Goldstein AH (1995) Cloning of a mineral phosphate-solubilizing gene from Pseudomonas cepacia. Appl Environ Microbiol 61:972–978
Bader AN, Salerno GL, Covacevich F, Consolo VF (2020) Native Trichoderma harzianum strains from Argentina produce indole-3 acetic acid and phosphorus solubilization, promote growth and control wilt disease on tomato (Solanum lycopersicum L.). J King Saud Univ Sci 32:867–873. https://doi.org/10.1016/j.jksus.2019.04.002
Bai Z, Li H, Yang X, Zhou B, Shi X, Wang B, Zhang F (2013) The critical soil P levels for crop yield, soil fertility and environmental safety in different soil types. Plant Soil 372:27–37. https://doi.org/10.1007/s11104-013-1696-y
Bai YC, Chang YY, Hussain M, Lu B, Zhang JP, Song XB (2020) Soil chemical and microbiological properties are changed by long-term chemical fertilizers that limit ecosystem functioning. Microorganisms 8:694. https://doi.org/10.3390/microorganisms8050694
Bashir Z, Zargar MY, Mohiddin FA, Kousar S, Husain M, Rasool F (2017) Phosphorus solubilizing microorganisms: mechanism and diversity. Int J Chem Stud 5(5):666–673. https://doi.org/10.18782/2320-7051.5446
Bashir Z, Zargar MY, Baba ZA, Mohiddin FA, Peer FA, Mir SA, Najar GR (2018a) Isolation and characterization of phosphorus solubilizing bacteria (PSB) from rhizospheric soils of apple (Malus Domestica). J Pharmacogn Phytochem 7(2):2997–3000
Bashir Z, Zargar MY, Mohiddin FA, Baba ZA, Hamid B (2019b) Isolation and biochemical characterization of phosphorus solubilizing bacteria from Rhizospheric region of Apricot and Peach. J Res Dev Univ Kashmir 19:0972–5407
Bashir SS, Hussain A, Hussain SJ, Wani OA, Zahid Nabi S, Dar NA, Mansoor S (2021) Plant drought stress tolerance: Understanding its physiological, biochemical and molecular mechanisms. Biotechnol Biotechnol Equip 35(1):1912–1925. https://doi.org/10.1080/13102818.2021.2020161
Bashir Z, Zargar MY, Bhat T, Kousar S, Baba ZA, Mohiddin FA (2018b) Solubilization of Phosphorus Containing Mineral by Bacteria from Rhizospheric Region of Walnut (Juglans regia L). Int J Curr Microbiol App Sci 7(8):2391–2398. https://doi.org/10.20546/ijcmas.2018.708.241
Bashir Z, Zargar MY, Vishwakarma DK (2019a) Potassium solubilizing microorganisms for sustainable agriculture. Applied agricultural practices for mitigating climate change, Taylor and Francis- CRC press 2:17-28. https://doi.org/10.1201/9780429326400-2
Baudoin E, Benizri E, Guckert A (2002) Impact of growth stages on bacterial community structure along maize roots by metabolic and genetic fingerprinting. Appl Soil Ecol 19:135–145. https://doi.org/10.1016/S0929-1393(01)00185-8
Benítez T, Rincón AM, Limón MC, Codón AC (2004) Biocontrol mechanisms of Trichoderma strains. Inter Microbiol 7:249–260
Bilal L, Asaf S, Hamayun M, Gul H, Iqbal A, Ullah I, Lee IJ, Hussain A (2018) Plant growth promoting endophytic fungi Aspergillus fumigatus TS1 and Fusarium proliferatum BRL1 produce gibberellins and regulates plant endogenous. Symbiosis 76:117–127. https://doi.org/10.1007/s13199-018-0545-4
Birhanu G, Tsegay Z, Tefera G, Aynalem E, Wassie M (2017) Phosphate solubilizing fungi isolated and characterized from Teff rhizosphere soil collected from North Showa zone, Ethiopia. Afr J Microbiol Res 11(17):687–696. https://doi.org/10.5897/AJMR2017.8525
Biswas JK, Banerjee A, Rai M, Naidu R, Biswas B, Vithanage M, Dash MC, Sarkar SK, Meers E (2018) Potential application of selected metal resistant phosphate solubilizing bacteria isolated from the gut of earthworm (Metaphire posthuma) in plant growth promotion. Geoderma 330:117–124. https://doi.org/10.1016/j.geoderma.2018.05.034
Boiero L, Perrig D, Masciarelli O, Penna C, Cassa´n F, Luna V (2007) Phytohormone production by three strains of Bradyrhizobium japonicum and possible physiological and technological implications. Appl Microbiol Biotechnol 74:874–880. https://doi.org/10.1007/s00253-006-0731-9
Bononi L, Chiaramonte JB, Pansa CC, Moitinho MA, Melo IS (2020) Phosphorus-solubilizing Trichoderma spp. from Amazon soils improve soybean plant growth. Sci Rep 10:1–13. https://doi.org/10.1038/s41598-020-59793-8
Bottini R, Cassan F, Piccoli P (2004) Gibberellin production by bacteria and its involvement in plant growth promotion and yield increase. Appl Microbiol Biotechnol 65:497–503
Buch A, Archana G, Kumar GN (2008) Metabolic Channeling of glucose towards gluconate in phosphate-solubilizing Pseudomonas aeruginosa P4 under phosphorus deficiency. Res Microbiol 159:635–642. https://doi.org/10.1016/j.resmic.2008.09.012
Buch A, Archana G, Kumar GN (2010) Heterologous expression of phosphoenolpyruvate carboxylase enhances the phosphate solubilizing ability of fluorescent pseudomonads by altering the glucose catabolism to improve biomass yield. Biores Technol 101:679–687. https://doi.org/10.1016/j.biortech.2009.08.075
Chakdar H, Dastager SG, Khire JM, Rane D, Dharne MS (2018) Characterization of mineral phosphate solubilizing and plant growth promoting bacteria from termite soil of arid region. Biotech 8:1–11. https://doi.org/10.1007/s13205-018-1488-4
Chandra S, Choure K, Dubey RC, Maheshwari DK (2007) Rhizosphere competent Mesorhizobium loti MP6 induces root hair curling, inhibits Sclerotinia Sclerotium and enhances growth of Indian mustard (Brassica campestris). Braz J Microbiol 38:124–130. https://doi.org/10.1590/S1517-83822007000100026
Chatli AS, Beri V, Sidhu BS (2008) Isolation and characterisation of phosphate solubilising microorganisms from the cold desert habitat of salix alba Linn. In Trans Himalayan region of Himachal Pradesh. Ind J Microbiol 48:267–273. https://doi.org/10.1007/s12088-008-0037-y
Chawngthu L, Hnamte R, Lalfakzuala R (2020) Isolation and characterization of rhizospheric phosphate solubilizing bacteria from wetland paddy field of Mizoram, India. Geomicrobiol 37:366–375. https://doi.org/10.1080/01490451.2019.1709108
Chen A, Arai Y (2023) A review of the reactivity of phosphatase controlled by clays and clay minerals: implications for understanding phosphorus mineralization in soils. Clay Clay Miner 71:119–142. https://doi.org/10.1007/s42860-023-00243-7
Chen Q, Liu S (2019) Identification and characterization of the phosphate-solubilizing bacterium Pantoea sp. S32 in reclamation soil in Shanxi, China. Front Microbiol 10:2171. https://doi.org/10.3389/fmicb.2019.02171
Chen YP, Rekha PD, Arun AB, Shen FT, Lai WA, Young CC (2006) Phosphate solubilizing bacteria from subtropical soil and their Tricalcium phosphate solubilizing abilities. Appl Soil Ecol 34:33–41. https://doi.org/10.1016/j.apsoil.2005.12.002
Chen S, Dickson DW (2004) Biological Control of Nematodes by Fungal Antagonists. In: Nematology: Advances and Perspectives. Nematode Management and Utilization, Z.X. Chen, S.Y. Chen, and D.W. Dickson [eds.] Cambridge, MA: Tsinghua. University Press and CABI 2:656. https://doi.org/10.1079/9780851996462.0979
Cui K, Xu T, Chen J, Yang H, Liu X, Zhuo R, Peng Y, Tang W, Wang R, Chen L, Zhang X (2022) Siderophores, a potential phosphate solubilizer from the endophyte Streptomyces sp. CoT10, improved phosphorus mobilization for host plant growth and rhizosphere modulation. J Clean Prod 367:133110. https://doi.org/10.1016/j.jclepro.2022.133110
De Souza MJ, Nair S, Chandramohan D (2000) Phosphate solubilizing bacteria around Indian peninsula. Ind J Mar Sci 29:48–51
De Zutter N, Ameye M, Vermeir P, Verwaeren J, De Gelder L, Audenaert K (2022) Innovative rhizosphere-based enrichment under P-limitation selects for bacterial isolates with high-performance P-solubilizing traits. Microbiol Spectrum 10(6):e0205222. https://doi.org/10.1128/spectrum.02052-22
Dimkpa C, Ales S, Dirk M, Georg B, Erika K (2008) Hydroxamate siderophores produced by Streptomyces Acidiscabies E13 bind nickel and promote growth in cowpea (Vignaunguiculata L.) under nickel stress. Can J Microb 54:163–172. https://doi.org/10.1139/w07-130
Divjot K, Rana KL, Tanvir K, Yadav N, Yadav AN, Kumar M (2021) Biodiversity, current developments and potential biotechnological applications of phosphorus-solubilizing and-mobilizing microbes: a review. Pedosphere 31:43–75. https://doi.org/10.1016/S1002-016020:60057-1
Do Carmo TS, Moreira FS, Cabral BV, Dantas RC, de Resende MM, Cardoso VL, Ribeiro EJ (2019) Phosphorus recovery from phosphate rocks using phosphate-solubilizing bacteria. Geomicrobiol 36:195–203. https://doi.org/10.1080/01490451.2018.1534901
Dong Z, Liu Y, Li M, Ci B, Lu X, Feng X (2023) Effect of different NPK fertilization timing sequences management on soil-petiole system nutrient uptake and fertilizer utilization efficiency of drip irrigation cotton. Sci Rep 13:14287. https://doi.org/10.1038/s41598-023-40620-9
Duan S, Declerck S, Feng G, Zhang L (2023) Hyphosphere interactions between Rhizophagus irregularis and Rahnella aquatilis promote carbon-phosphorus exchange at the peri-arbuscular space in Medicago truncatula. Environ Microbiol 25:867–879. https://doi.org/10.1111/1462-2920.16333
Fajardo C, Costa G, Nande M, Botías P, García-Cantalejo J, Martin M (2019) Pb, Cd, and Zn soil contamination: Monitoring functional and structural impacts on the microbiome. Appl Soil Ecol 135:56–64. https://doi.org/10.1016/j.apsoil.2018.10.022
Farajzadeh D, Yakhchali B, Aliasgharzad N, Bashir NS, Farajzadeh M (2012) Plant growth Fenice (2000) Application of encapsulated Penicillium variabilis P16 in dissolution of Tricalcium phosphate. World J Microbiol Biotechnol 24:1059–1065. https://doi.org/10.1007/s00284-012-0083-x
Fatima F, Ahmad MM, Verma SR, Pathak N (2021) Relevance of phosphate solubilizing microbes in sustainable crop production: a review. Inter Environ Sci Technol 1–14. https://doi.org/10.1007/s13762-021-03425-9
Feng Y, He J, Zhang H, Jia X, Hu Y, Ye J, Gu X, Zhang X, Chen H (2024) Phosphate solubilizing microorganisms: a sustainability strategy to improve urban ecosystems. Front Microbiol 14:1320853. https://doi.org/10.3389/fmicb.2023.1320853
Fernandes GC, Rosa PA, Jalal A, Oliveira CES, Galindo FS, Viana RS (2023) Technological quality of sugarcane inoculated with plant-growth promoting Bacteria and residual effect of phosphorus rates. Plan Theory 12:2699. https://doi.org/10.3390/plants12142699
Gill W, Eyles A, Glen M, Mohammed C (2016) Structural host responses of Acacia mangium and Eucalyptus pellita to artificial infection with the root rot pathogen, Ganoderma philippii. Forest Pathol 46:369–375. https://doi.org/10.1111/efp.12286
Glick BR, Cheng Z, Czarny J, Duan J (2007) Promotion of plant growth by ACC deaminase producing soil bacteria. Eur J Plant Pathol 119:329–339. https://doi.org/10.1080/07352680701572966
Gong A, Wang G, Sun Y et al (2022) Dual activity of Serratia marcescens Pt-3 in phosphate-solubilizing and production of antifungal volatiles. BMC Microbiol 22:26. https://doi.org/10.1186/s12866-021-02434-5
Gouda S, Kerry RG, Das G, Paramithiotis S, Shin HS, Patra JK (2018) Revitalization of plant growth promoting rhizobacteria for sustainable development in agriculture. Microbiol Res 206:131–140. https://doi.org/10.1016/j.micres.2017.08.016
Gowtham HG, Pariprasad P, Nayak PS, Niranjana SR (2016) Application of rhizobacteria antagonistic to Fusarium oxysporum f. sp. lycopersici for the management of Fusarium wilt in tomato. Rhizosphere 2:72–74. https://doi.org/10.1016/j.rhisph.2016.07.008
Guimarães SL, Bonfim-Silva EM, Polizel de Souza AC, Simeon BG (2019) Efficiency of Inoculation with Rhizobium in Peanuts (Arachis hypogaea L.) Grown in Brazilian Cerrado Soil. Agric Sci 10:7. https://doi.org/10.4236/as.2019.107072
Gulati A, Sharma N, Vyas P, Sood S, Rahi P, Pathania V, Prasad R (2010) Organic acid production and plant growth promotion as a function of phosphate solubilization by Acinetobacter rhizosphaerae strain BIHB 723 isolated from the cold deserts of the trans-Himalayas. Arch Microbiol 192:975–983. https://doi.org/10.1007/s00203-010-0615-3
Gupta A, Rai V, Bagdwal N, Goel R (2005) In situ characterization of mercury-resistant growth promoting fluorescent pseudomonads. Microbiol Res 160:385–388. https://doi.org/10.1016/j.micres.2005.03.002
Hamid B, Majeed N, Ganai BA, Hassan S, Bashir Z, Wani PA, Perveen K, Sayyed RZ (2023a) Heavy-metal tolerant bacterial strains isolated from industrial sites and scrap yards in Kashmir, India. J Basic Microbiol. https://doi.org/10.1002/jobm.202300349
Hamid B, Yatoo AM, Sayyed RZ, Dineshkumar R, Al-Khayri JM, Bashir Z, Sillanpää M, Majeed N (2023b) Microbial-based conversion of food waste for sustainable bioremediation and utilization as compost. Biomass Convers Biorefin 1–15. https://doi.org/10.1007/s13399-023-04794-6
Harman GE, Howell CR, Viterbo A, Chet I, Lorito M (2004) Trichoderma species opportunistic, a virulent plant symbionts. Nat Rev 2:43–56. https://doi.org/10.1038/nrmicro797
Hassan TU, Bano A (2016) Biofertilizer: A novel formulation for improving wheat growth, physiology and yield. Pak J Bot 48:2233–2241
Hayat R, Ali S, Amara U, Khalid R, Ahmed I (2010) Soil beneficial bacteria and their role in plant growth promotion: a review. Annals Microbiol 60(4):579–598. https://doi.org/10.1007/s13213-010-0117-1
He T, Xu ZJ, Wang JF, Wang FP, Zhou XF, Wang LL, Li QS (2022) Improving cadmium accumulation by Solanum nigrum L. via regulating rhizobacterial community and metabolic function with phosphate solubilizing bacteria colonization. Chemosphere 287(2):132209. https://doi.org/10.1016/j.chemosphere.2021.132209
Hii YS, San Chan Y, Lau SW, Michael D (2020) Isolation and characterisation of phosphate solubilizing microorganisms from peat. Biocatal Agric Biotechnol 26:101463. https://doi.org/10.1016/j.bcab.2020.101643
Hinsinger P (2001) Bioavailability of trace elements as related to root-induced chemical changes in the rhizosphere. In: Gobran GR, Wenzel WW, Lombi E (eds) Trace elements in the rhizosphere. CRC press, Boca Raton, Fl, USA. https://doi.org/10.1201/9781420039993.ch2
Hutchins DA, Qu P, Fu FX, Kling J, Huh M, Wang X (2019) Distinct responses of the nitrogen-fixing marine cyanobacterium Trichodesmium to a thermally-variable environment as a function of phosphorus availability. Front Microbiol 10:1282. https://doi.org/10.3389/fmicb.2019.01282
Ichriani GI, Nuraini Y, Handayanto (2018) Formulation of biochar compost and phosphate solubilizing fungi from oil palm empty fruit bunch to improve growth of maize in an ultisol of Central Kalimantan. J Ecol Eng 19:45–55. https://doi.org/10.12911/22998993/92891
Islam MK, Sano A, Majumder MSI, Hossain MA, Sakagami J (2019) Isolation and molecular characterization of phosphate solubilizing filamentous fungi from subtropical soils in Okinawa. Appl Ecol Environ Res 17(4):9145–9157. https://doi.org/10.15666/aeer/1704_91459157
Jain R, Saxena J, Sharma V (2012) Solubilization of inorganic phosphates by Aspergillus awamori S19 isolated from rhizosphere soil of a semi-arid region. Ann Microbiol 62:725–773
Jaiswal SK, Mohammed M, Ibny FY, Dakora FD (2021) Rhizobia as a source of plant growth-promoting molecules: Potential applications and possible operational mechanisms. Front Sustain Food Sys 4:619676
Jamshidi R, Jalili B, Bahmanyar MA, Salek-Gilani S (2016) Isolation and identification of a phosphate solubilising fungus from soil of a phosphate mine in Chaluse, Iran. Int J Fungal Biol 7:3. https://doi.org/10.1080/21501203.2016.1221863
Jatala P (1986) Biological control of plant parasitic nematodes. Annu Rev Phytopathol 24:453–491. https://doi.org/10.1146/annurev.py.24.090186.002321
Jesús A, Ibarra-Galeana, Castro-Martínez C, Fierro-Coronado RA, Armenta-Bojórquez AD, Ignacio E, Maldonado-Mendoza (2017) Characterization of phosphate-solubilizing bacteria exhibiting the potential for growth promotion and phosphorus nutrition improvement in maize (Zea mays L.) in calcareous soils of Sinaloa, Mexico. Ann Microbiol 67:801–811
Jha Y, Mohamed HI (2023) Inoculation with Lysinibacillus fusiformis strain YJ4 and Lysinibacillus sphaericus strain YJ5 alleviates the effects of cold stress in maize plants. Gesunde Pflanzen 75:77–95. https://doi.org/10.1007/s42161-023-01338-9
Jiménez-Aguirre MA, Padilla-Hurtado BE, Gonzalez-Cardona C, Ceballos-Aguirre N, Cardona-Agudelo LD, Montoya-Estrada CN (2023) Antagonist effect of native bacteria of the genus Bacillus on the root-knot nematode (Meloidogyne spp.) in tomato germplasm. Chil J Agric Res 83(5). https://doi.org/10.4067/s0718-58392023000500565
Jorquera MA, Crowley DE, Marschner P, Greiner R, Fernandez MT, Romero D (2011) Identification of b-propeller phytase-encoding genes in culturable Paenibacillus and Bacillus sp. from the rhizosphere of pasture plants on volcanic soils. FEMS Microbiol Ecol 75:163–172. https://doi.org/10.1111/j.1574-6941.2010.00995.x
Kadmiri IM, Chaouqui L, Azaroual SE, Sijilmassi B, Yaakoubi K, Wahby I (2018) Phosphate-solubilizing and auxin-producing rhizobacteria promote plant growth under saline conditions. Arab J Sci Eng 43:3403–3415. https://doi.org/10.1007/s13369-017-3042-9
Kalam S, Basu A, Podile AR (2020) Functional and molecular characterization of plant growth promoting Bacillus isolates from tomato rhizosphere. Heliyon 6:e04734. https://doi.org/10.1016/j.heliyon.2020.e04734
Kalayu G (2019) Phosphate solubilizing microorganisms: promising approach as biofertilizers. Inter J Agron 2019(1):4917256. https://doi.org/10.1155/2019/4917256
Kang SM, Khan MA, Hamayun M, Kim LR, Kwon EH, Kang YS (2021) Phosphate-solubilizing Enterobacter ludwigii AFFR02 and Bacillus megaterium Mj1212 rescues alfalfa’s growth under post-drought stress. Agriculture 11:485. https://doi.org/10.3390/agriculture11060485
Kang SC, Ha CG, Lee TG, Maheshwari DK (2002) Solubilization of insoluble inorganic phosphates by a soil-inhabiting fungus Fomitopsis sp. PS 102. Curr Sci 82(4):439–442. https://www.jstor.org/stable/24106657
Kaur H, Mir RA, Hussain SJ, Prasad B, Kumar P, Aloo BN, Sharma CM, Dubey RC (2024) Prospects of phosphate solubilizing microorganisms in sustainable agriculture. World J Microbiol Biotechnol 40:291. https://doi.org/10.1007/s11274-024-04086-9
Khambhaty Y, Mody K, Basha S, Jha B (2008) Hg(II) Removal from aqueous solution by dead fungal biomass of marine Aspergillus niger: kinetic studies. Sep Sci Technol 43:1221–1238. https://doi.org/10.1080/01496390801888235
Khan MR, Akram M (2000) Effect of certain antagonistic fungi and rhizobacteria on wilt disease complex caused by Meloidogyne incognita and Fusarium oxysporium sp. lycopersici on tomato. Nematol Mediter 28:139–144
Khan MR, Kounsar K (2000) Effects of certain bacteria and fungi on the growth of mung bean and reproduction of Meloidogyne incognita. Nematol Mediter 28:221–226
Khan MR, Tarannum Z (1999) Effects of field application of various microorganisms on Meloidogyne incognita on tomato. Nematol Mediter 27:233–238
Khan MR, Khan SM, Mohiddin FA (2005) Root-knot problem of some winter ornamental plants and its bio management. J Nematol 37:198–206
Khan AA, Jilani G, Akhtar MS, Naqvi SMS, Rasheed M (2009a) Phosphorus solubilizing bacteria: occurrence, mechanisms and their role in crop production. J Agric Biol Sc 1:4858
Khan MS, Zaidi A, Wani PA, Ahemad M (2009c) Functional diversity among plant growth-promoting rhizobacteria. In: Khan MS, Zaidi A, Musarrat J (eds) Microbial strategies for crop improvement. Springer, Berlin, pp 105–132. https://doi.org/10.1007/978-3-642-01979-1_6
Khan N, Siddiqui MH, Ahmad S, Ahmad MM, Siddiqui S (2024) New insights in enhancing the phosphorus use efficiency using phosphate-solubilizing microorganisms and their role in cropping system. Geomicrobiol J 41(5):485–495. https://doi.org/10.1080/01490451.2024.2331111
Khan MR, Altaf S, Mohidin FA, Khan U, Anwer A (2009b) Biological control of plant nematodes with phosphate-solubilizing microorganisms. pp 395–426
Khan M, Zaidi SA, Ahmad E (2014) Mechanism of phosphate solubilization and physiological functions of phosphate solubilizing microorganisms. In: MS Khan. Eds. https://doi.org/10.1007/978-3-319-08216-5_2
Kirui CK, Njeru EM, Runo S (2022) Diversity and phosphate solubilization efficiency of phosphate solubilizing bacteria isolated from semi-arid agroecosystems of eastern Kenya. MicrobiologyInsights 15:11786361221088992. https://doi.org/10.1177/11786361221088991
Koch M, Kruse J, Eichler-Löbermann B, Zimmer D, Willbold S, Leinweber P, Siebers N (2018) Phosphorus stocks and speciation in soil profiles of a long-term fertilizer experiment: evidence from sequential fractionation, P K-edge XANES, and 31P NMR spectroscopy. Geoderma 316:115–126. https://doi.org/10.1016/j.geoderma.2017.12.003
Kour D, Yadav AN (2023) Alleviation of cold stress in wheat with psychrotrophic phosphorus solubilizing Acinetobacter rhizosphaerae EU-KL44. Braz J Microbiol 54:371–383. https://doi.org/10.1007/s42770-023-00913-7
Kour D, Rana KL, Sheikh I, Kumar V, Yadav AN, Dhaliwal HS, Saxena AK (2020) Alleviation of drought stress and plant growth promotion by Pseudomonas libanensis EU-LWNA-33, a drought-adaptive phosphorus-solubilizing bacterium. Proc Natl Acad Sci India Sect B: Biol Sci 90:785–795. https://doi.org/10.1007/s40011-019-01151-4
Kpomblekou AK, Tabatabai MA (1994) Effect of organic acids on release of phosphorus from phosphate rock. Soil Sci 158:442–453
Krishnaswamy U, Muthuswamy M, Perumalsamy M (2009) Studies on the efficiency of the removal of phosphate using bacterial consortium for the biotreatment of phosphate wastewater. Eur J Appl Sci 1:6–15
Kumar V, Singh P, Jorquera MA, Sangwan P, Kumar P, Verma AK, Agrawal S (2013) Isolation of phytase-producing bacteria from Himalayan soils and their effect on growth and phosphorus uptake of Indian mustard (Brassica juncea). World J Microbiol Biotechnol 29:1361–1369. https://doi.org/10.1007/s11274-013-1299-z
Kusale SP, Attar YC, Sayyed RZ (2021) Production of plant beneficial and antioxidants metabolites by Klebsiella variicola under salinity stress. Molecules 26:1894. https://doi.org/10.3390/molecules26071894
Lallawmkima I, Singh SK, Sharma M (2018) Application of Azotobacter, Vesicular Arbuscular Mycorrhiza and Phosphate Solubilizing Bacteria for potato cultivation in Central Plain Zone (Pb-3) of Punjab. J Environ Biol 985–989. https://doi.org/10.22438/jeb/39/6/MRN-463
Li Z, Bai T, Dai L, Wang F, Tao J, Meng S, Hu Y, Wang S, Hu S (2016) A study of organic acid production in contrasts between two phosphate solubilizing fungi: Penicillium oxalicum and Aspergillus niger. Sci Rep 6:1–8. https://doi.org/10.1038/srep25313
Li H, Song C, Yang L, Qin H, Cao X, Zhou Y (2021) Nutrients regeneration pathway, release potential, transformation pattern and algal utilization strategies jointly drove cyanobacterial growth and their succession. J Environ Sci 103:255–267. https://doi.org/10.1016/j.jes.2020.11.010
Lin TF, Huang HI, Shen FT, Young CC (2006) The protons of gluconic acid are the major factor responsible for the dissolution of tricalcium phosphate by Burkholderia cepacia CC-A174. BioresourTechnol 97:957–960. https://doi.org/10.1016/j.biortech.2005.02.017
Lipping Y, Jiatao X, Daohong J, Yanping F, Guoqing L, Fangcan L (2008) Antifungal substances produced by Penicillium oxalicum strain PY-1-potential antibiotics against plant pathogenic fungi. World J Microbiol Biotechnol 24:909–915. https://doi.org/10.1007/s11274-007-9626-x
Liu C, Mou L, Yi J, Wang J, Liu A, Yu J (2019) The eno gene of Burkholderia cenocepacia strain 71–2 is involved in phosphate solubilization. Curr Microbiol 76:495–502. https://doi.org/10.1007/s00284-019-01642-7
Liu J, Liu X, Zhang Q, Li S, Sun Y, Lu W (2020) Response of alfalfa growth to arbuscular mycorrhizal fungi and phosphate-solubilizing bacteria under different phosphorus application levels. AMB Express 10:200. https://doi.org/10.1186/s13568-020-01137-w
Liu H-y, Jin H, Yang X-y, Xu Z-x, Cui Z-t (2022) Molecular identification, diversity and functional characterization of fungal communities isolated from Stipa purpurea. S Afr J Bot 149:117–123. https://doi.org/10.1016/j.sajb.2022.05.050
Liu F, Qian J, Zhu Y, Wang P, Hu J, Lu B (2024) Phosphate solubilizing microorganisms increase soil phosphorus availability: a review. Geomicrobiol J 41:1–16. https://doi.org/10.1080/01490451.2023.2272620
Lucero CT, Lorda GS, Anzuay MS, Ludueña ML, Taurian T (2021) Peanut Endophytic Phosphate Solubilizing Bacteria Increase Growth and P Content of Soybean and Maize Plants. Curr Microbiol 78:1961–1972. https://doi.org/10.1007/s00284-021-02469-x
Maharajan T, Ceasar SA, Ajeesh Krishna TP, Ramakrishnan M, Duraipandiyan V, Naif Abdulla AD (2018) Utilization of molecular markers for improving the P efficiency in crop plants. Plant Breed 137:10–26. https://doi.org/10.1111/pbr.12537
Maliha R, Samina K, Najma A, Sadia A, Farooq L (2004) Organic acid production and phosphate solubilization by phosphate solubilizing microorganisms under in vitro conditions. Pak J Biol Sci 7:187–196
Manoj S, Srivastava PC, Douza SF (2018) Phosphate-Solubilizing Microbes: Diversity and Phosphates Solubilization Mechanism. Springer Nature Singapore Pte Ltd. 13: 978-981. https://doi.org/10.1007/978-981-13-0044-8_5
Mansoor S, Ali A, Kour N, Bornhorst J, AlHarbi K, Rinklebe J, Chung YS (2023a) Heavy metal induced oxidative stress mitigation and ROS scavenging in plants. Plants 12(16):3003. https://doi.org/10.3390/plants12163003
Mansoor S, Sakina A, Mir MA, Mir JI, Wani AA, Masoodi KZ (2023b) Elucidating the role of reactive oxygen species metabolism and phenylpropanoid pathway during an incompatible interaction between apple-Venturia inaequalis host-pathosystem. S Afr J Bot 160:428–436. https://doi.org/10.1016/j.sajb.2023.07.020
Mardhiansyah (2011) Potensipengendalian Trichoderma spp. terhadap Ganoderma sp. di pertanaman Acacia mangiumum urenambulan. SAGU 10:29–34
Marra LM, Oliveira-Longatti SM, Soares CRFS, Lima ML, Olivares FL, Moreira FMS (2015) Initial pH of medium affects organic acids production but do not affect phosphate solubilization. Braz J Microbiol 46:367–375. https://doi.org/10.1590/S1517-838246246220131102
Mažylytė R, Kaziūnienė J, Orola L, Valkovska V, Lastauskienė E, Gegeckas A (2022) Phosphate Solubilizing Microorganism Bacillus sp. MVY-004 and Its Significance for Biomineral Fertilizers’ Development in Agrobiotechnology. Biology 11:254. https://doi.org/10.3390/biology11020254
Mehta S, Nautiyal CS (2001) An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr Microbiol 43:51–56. https://doi.org/10.1007/s002840010259
Mendes R, Garbeva P, Raaijmakers JM (2013) The rhizosphere microbiome: significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms affiliations expand. FEMS Microbiol Rev 37(5):634–663. https://doi.org/10.1111/1574-6976.12028
Menezes-Blackburn D, Jorquera MA, Greiner R, Gianfreda L, de la Luz MM (2013) Phytases and phytase-labile organic phosphorus in manures and soils. Crit Rev Environ Sci Technol 43(9):916–954. https://doi.org/10.1080/10643389.2011.627019
Mingkwan D, Phookamsak R, Yan K, Suwannarach N (2020) Screening of phosphate solubilizing fungi from Air and Soil in Yunnan. Front Microbiol 5:11–18. https://doi.org/10.3389/fmicb.2020.585215
Mitra D, Anđelković S, Panneerselvam P, Senapati A, Vasić T, Ganeshamurthy AN, Radha TK (2020) Phosphate-solubilizing microbes and biocontrol agent for plant nutrition and protection: current perspective. Commun Soil Sci Plant Anal 51:645–657. https://doi.org/10.1080/00103624.2020.1729379
Mittal V, Singh O, Nayyar H, Kaur J, Tewari R (2008) Stimulatory effect of phosphate solubilizing fungal strains (Aspergillus awamori) and Penicillium citrinum) on the yield of chickpea (Cicerarietitinum L. cv.GPF2). Soil Biol Biochem 40:718–727. https://doi.org/10.1016/j.soilbio.2007.10.008
Mohamed EAH, Farag AG, Youssef SA (2018) Phosphate solubilization by Bacillus subtilis and Serratia marcescens isolated from tomato plant rhizosphere. J Environ Prot 09:266–277. https://doi.org/10.4236/jep.2018.93018
Munir I, Bano A, Faisal M (2019) Impact of phosphate solubilizing bacteria on wheat (Triticum aestivum) in the presence of pesticides. Braz J Biol 79:29–37. https://doi.org/10.1590/1519-6984.172213
Nacoon S, Jogloy S, Riddech N, Mongkolthanaruk W, Kuyper TW, Boonlue S (2020) Interaction between phosphate solubilizing bacteria and arbuscular mycorrhizal fungi on growth promotion and tuber inulin content of Helianthus tuberosus L. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-61846-x
Nacoon S, Seemakram W, Ekprasert J, Jogloy S, Kuyper TW, Mongkolthanaruk W (2022) Promoting growth and production of sunchoke (Helianthus tuberosus) by co-inoculation with phosphate solubilizing bacteria and arbuscular mycorrhizal fungi under drought. Front Plant Sci 13:1022319. https://doi.org/10.3389/fpls.2022.1022319
Naeem M, Aslam Z, Khaliq A, Ahmed JN, Nawaz A, Hussain M (2018) Plant growth promoting rhizobacteria reduce aphid population and enhance the productivity of bread wheat. Braz J Microbiol 49:9–14. https://doi.org/10.1016/j.bjm.2017.10.005
Naik PR, Sahoo N, Goswami D, Ayyadurai N, Sakthivel N (2008) Genetic and functional diversity among fluorescent pseudomonads isolated from the rhizosphere of banana. Micro Ecol 56:492–504. https://doi.org/10.1007/s00248-008-9368-9
Nannipieri P, Giagnoni L, Landi L, Renella G (2011) Role of phosphatase enzymes in soil,” in Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling. Soil Biology, Eds E. Bunemann, A. Oberson, and E. Frossard (Berlin: Springer) 215-243. https://doi.org/10.1007/978-3-642-15271-9_9
Nesme T, Metson GS, Bennett EM (2018) Global P flows through agricultural trade. Glob Environ Change 50:133–141. https://doi.org/10.1016/j.gloenvcha.2018.04.004
Oostendrop M, Sikora RA (1989) Seed treatment with antagonistic rhizobacteria for the suppression of Heterodera schachtii early root infection of sugar beet. Rev Nematol 12:77–83
Oyekanmi ZDL, Coyne, Fawole LB (2008) Utilization of the potentials of selected microorganisms as biocontrol and biofertilizer for enhanced crop improvement. J Biol Sci 8:746–752
Ozane PG (1980) Phosphate nutrition of plants-general treatise. In: Khasawneh FE, Sample EC, Kamprath EJ (eds) The role of phosphorus in agriculture. American Soc Agron, Crop Sci Soc America, Soil Sci Soc America, Madison, WI, USA, pp 559–589. https://doi.org/10.2134/1980.roleofphosphorus.c21
Padmavathi T, Madhumathi G (2009) Phosphate solubility and biocontrol activity of Trichoderma harzianum. Turk J Biol 35(2011):593–600
Padmavathi T, Usha S (2012) Phosphate-solubilizing microbes and their occurrence in the rhizospheres of Piper betel in Karnataka, India. Turk J Biol 36:25–35
Pang F, Li Q, Solanki MK, Wang Z, Xing YX, Dong DF (2024) Soil phosphorus transformation and plant uptake driven by phosphate solubilizing microorganisms. Front Microbiol 15:1383813. https://doi.org/10.3389/fmicb.2024.1383813
Pant H, Pandey G (2001) Efficacy of biocontrol agents for the management of root-knot nematode on chickpea. Ann Plant Protect Sci 9:117–170
Park KH, Lee CY, Son HJ (2009) Mechanism of insoluble phosphate solubilization by Pseudomonas fluorescens RAF15 isolated from ginseng rhizosphere and its plant growth-promoting activities. Lett Appl Microbiol 49(2):222–228. https://doi.org/10.1111/j.1472-765X.2009.02642.x
Park JH, Bolan N, Megharaj M, Naidu R (2011) Isolation of phosphate solubilizing bacteria and their potential for lead immobilization in soil. J Hazard Mater 185:829–836. https://doi.org/10.1016/j.jhazmat.2010.09.095
Park S, Kim AL, Hong YK, Shin JH, Joo SH (2021) A highly efficient auxin-producing bacterial strain and its effect on plant growth. J Genet Eng Biotechnol 19(1):179. https://doi.org/10.1186/s43141-021-00252-w
Patil MG, Sayyed RZ, Chaudhari AB, Chincholkar SB (2002) Phosphate Solubilizing Microbes: A Potential Bioinoculant for Efficient Use of Phosphate Fertilizers. In: Reddy SM, Reddy SR, Grisham S (eds) Bioinoculants for Sustainable Agriculture and Forestry. Scientific Publisher, Jodhpur, pp 107–118
Paulraj P, Gunaseelan R, Samrot AV, Ismail AA, Iyappan P, Chandramohan M, Fung HZ, Selvarani A J (2020) Isolation and characterization of phosphate solubilizing bacteria from rhizosphere soils for potential use in agricultural practices of Solanum melongena. Eco Env & Cons. pp S53–S61
Peix A, Lang E, Verbarg S, Spröer C, Rivas RIS, Mateos PF, Molina EM, Barrueco CR, Velázquez E (2009) Acinetobacter strains IH9 and OCI1, two rhizospheric phosphate solubilizing isolates able to promote plant growth, constitute a new genomovar of Acinetobacter calcoaceticus. Sys Appl Microbiol 32:334–341. https://doi.org/10.1016/j.syapm.2009.03.004
Piga M, Belager RR, Paulitz TC, Benhamou N (1997) Increased resistance to Fusarium oxysporumf.sp. radices-lycopersici in tomato plants treated with the endophytic bacterium P. fluoresecns strain 63–28. Physiol Mole Plant Pathol 50:301–320. https://doi.org/10.1006/pmpp.1997.0088
Pocasangre E, zum Felde A, Cañizares C, Muñoz J, Suarez P, Jimenez C, Riveros AS (2007) Field evaluation of the antagonistic activity of endophytic fungi towards the burrowing nematode, Radopholussimilis, in plantain. In: Latin America Pro Musa Symposium, Greenway Woods Resort. White River
Prakash J, Arora NK (2019) Phosphate-solubilizing Bacillus sp. enhances growth, phosphorus uptake and oil yield of Mentha arvensis L. 3 Biotech 9(4):1–9. https://doi.org/10.1007/s13205-019-1660-5
Prihatini I, Rimbawanto A, Puspitasari D, Fauzi D (2018) Pengujian penandajenisspesifik pada jamur yang berpotensise bagaia genspengendalihayatipenyakitbusukakar pada akasia. Jurnal Pemuliaan 12(1):1–11. https://doi.org/10.20886/jpth.2018.12.1.1-11
Puri A, Padda KP, Chanway CP (2020) In vitro and in vivo analyses of plant-growth-promoting potential of bacteria naturally associated with spruce trees growing on nutrient-poor soils. Appl Soil Ecol 149:103538. https://doi.org/10.1016/j.apsoil.2020.103538
Purwaningsih S, Agustiyani D, Antonius S (2021) Diversity, activity, and effectiveness of Rhizobium bacteria as plant growth promoting rhizobacteria (PGPR) isolated from Dieng, central Java. Iran J of Microbiol 13:130. https://doi.org/10.18502/ijm.v13i1.5504
Qarni A, Billah M, Hussain K, Shah SH (2021) Isolation and characterization of phosphate solubilizing microbes from rock phosphate mines and their potential effect for sustainable agriculture. Sustainability 13(4):2151. https://doi.org/10.3390/su13042151
Rafa CM, Chiampo F (2021) Bioremediation of agricultural soils polluted with pesticides: a review. Bioeng 8:92. https://doi.org/10.3390/bioengineering8070092
Rahman Z, Singh VP (2020) Bioremediation of toxic heavy metals (THMs) contaminated sites: Concepts, applications and challenges. Environ Sci Pollut Res Int 27:27563–27581. https://doi.org/10.1007/s11356-020-08903-0
Rai A, Sharma NK, Singh VK, Dwivedi BS, Singh JS, Rai PK (2023) Study of phosphate solubilizing fluorescent Pseudomonas recovered from rhizosphere and endorhizosphere of Aloe barbadensis (L.). Geomicrobiol J 40:347–359. https://doi.org/10.1080/01490451.2023.2171165
Rajwar J, Chandra R, Suyal DC, Tomer S, Kumar S, Goel R (2018) Comparative phosphate efficiency of psychrotolerant Pseudomonas jesenii MP1 and Acinetobacter sp. ST02 against chickpea for sustainable hill agriculture. Biologia 73:793–802. https://doi.org/10.2478/s11756-018-0089-3
Rawat P, Das S, Shankhdhar D, Shankhdhar SC (2021) Phosphate-solubilizing microorganisms: mechanism and their role in phosphate solubilization and uptake. J Soil Sci Plant Nutr 21:49–68. https://doi.org/10.1007/s42729-020-00342-7
Revillas JJ, Rodelas B, Pozo C, Martinez-Toledo MV, Gonzalez LJ (2000) Production of B-Group vitamins by two Azotobacter strains with phenolic compounds as sole carbon source under diazotrophic and adiazotrophic conditions. J Appl Microbiol 89:486–493. https://doi.org/10.1046/j.1365-2672.2000.01139.x
Reyes I, Baziramakenga R, Bernier L, Antoun H (2001) Solubilization of phosphate rocks and minerals by a wild-type strain and two UV induced mutants of Penicillium regulosum. Soil Biol Biochem 33:1741–1747. https://doi.org/10.1016/S0038-0717(01)00099-2
Rezakhani L, Motesharezadeh B, Tehrani MM, Etesami H, Hosseini HM (2019) Phosphate solubilizing bacteria and silicon synergistically augment phosphorus (P) uptake by wheat (Triticum aestivum L.) plant fertilized with soluble or insoluble P source. Ecotoxicol Environ Safety 173:504–513. https://doi.org/10.1016/j.ecoenv.2019.02.060
Rfaki A, Zennouhi O, Aliyat NL, Ibijbijen J (2020) Isolation, selection and Characterization of root-associated rock phosphate solubilizing bacteria in Moroccan wheat (Triticum aestivum L.). Geomicrobiol J 37:230–241. https://doi.org/10.1080/01490451.2019.1694106
Richa G, Khosla B, Reddy MS (2007) Improvement of maize plant growth by phosphate solubilising fungi in rock phosphate amended soils. World J Agricul Sci 3:481–484
Richardson AE, Simpson RJ (2011) Soil microorganisms mediating phosphorus availability. Plant Physiol 156:989–996. https://doi.org/10.1104/pp.111.175448
Rodríguez H, Fraga R (1999) Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol Adv 17:319–339. https://doi.org/10.1016/S0734-9750(99)00014-2
Rojas YD, Arias RM, Ortiz RM, Aguilar DT, Heredia G, Yon YR (2018) Effects of native arbuscular mycorrhizal and phosphate-solubilizing fungi on coffee plants. Agrofor Syst 93:961–972. https://doi.org/10.1007/s10457-018-0190-1
Saeed Q, Xiukang W, Haider FU, Kučerik J, Mumtaz MZ, Holatko J, Naseem M, Kintl A, Ejaz M, Naveed M, Brtnicky M, Mustafa A (2021) Rhizosphere bacteria in plant growth promotion, biocontrol, and bioremediation of contaminated sites: a comprehensive review of effects and mechanisms. Int J Mol Sci 22(19):10529. https://doi.org/10.3390/ijms221910529
Saeid A, Prochownik E, Dobrowolska-Iwanek J (2018) Phosphorus solubilization by Bacillus species. Mol 23:2897. https://doi.org/10.3390/molecules23112897
Saha N, Biswas S (2009) Mineral phosphate solubilizing bacterial communities in agro-ecosystem. Afr J Biotechnol 8:6863–6870
Saravana KD, Lavanya N, Muthumeena B, Raguchander T, Suresh S, Samiyappan R (2008) Pseudomonas fluorescens enhances resistance and natural enemy population in rice plants against leaf folder pest. J Appl Entomol 132(6):469–479. https://doi.org/10.1111/j.1439-0418.2008.01278.x
Schenek C (2004) Control of nematodes in tomato with Paecilomyceslilacinus. Hawaii Agriculture Research Center Vegetable Report 5
Shahid M, Hameed S, Imran A, Ali S, Elsas J (2012) Root colonization and growth promotion of sunflower (Helianthus annuus L.) by phosphate solubilizing Enterobacter sp. Fs-11. World J Microbiol Biotechnol 28:2749–2758. https://doi.org/10.1007/s11274-012-1086-2
Sharma SB, Sayyed RZ, Trivedi MH, Gobi TA (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. Springer plus 2:587. https://doi.org/10.1186/2193-1801-2-587
Sharma HK, Prasad D, Sharma P (2005) Compatibility of fungal bioagents as seed dressers with carbofuran in okra against Meloidogyne incognita. Natl Symp Rec Adv Res Priorities in Indian Nematol IARI New 72:9–11
Sharon E, Chet I, Spiegel Y (2009) Improved attachment and parasitism of Trichoderma on Meloidogyne javanica in vitro. Eur J Plant Pathol 123:291–299. https://doi.org/10.1007/s10658-008-9366-2
Sheng XF, Xia JJ (2006) Improvement of rape (Brassica napus) plant growth and cadmium uptake by cadmium-resistant bacteria. Chemosphere 64:1036–1042. https://doi.org/10.1016/j.chemosphere.2006.01.051
Shin W, Ryu J, Kim Y, Yang J, Madhaiyan M, Sa T (2006) Phosphate solubilization and growth promotion of maize (Zea mays L.) by the rhizosphere soil fungus Penicillium oxalicum. In: 18th World congress of soil science. Philadelphia, PA
Siddiqi IA, Shaukat SS (2004) Trichoderma harzianum enhances the production of nematicidal compounds in vitro and improve biocontrol of M. javanica by Pseudomonas fluorescens in tomato. Lett Appl Microbiol 38:169–175. https://doi.org/10.1111/j.1472-765X.2003.01481.x
Sierra S, Rodelas B, Martinez-Toledo MV, Pozo C, Gonzalez-Lopez J (1999) Production of B-group vitamins by two Rhizobium strains in chemically defined media. J Appl Microbiol 86:851–858. https://doi.org/10.1046/j.1365-2672.1999.00765.x
Silva LID, Pereira MC, Carvalho AMXD, Buttrós VH, Pasqual M, Dória J (2023) Phosphorus-solubilizing microorganisms: a key to sustainable agriculture. Agriculture 13(2):462. https://doi.org/10.3390/agriculture13020462
Srinivasan R, Yandigeri SM, Kashyap S, Alagawadi RA (2012) Effect of salt on survival and P-solubilization potential of phosphate solubilizing microorganisms from salt affected soils. Saudi J Biol Sci 19(4):427–434. https://doi.org/10.1016/j.sjbs.2012.05.004
Stajkovic O, Delic D, Josˇic D, Kuzmanovic D, Rasulic N, Knezevic´-Vukcevic J (2011) Improvement of common bean growth by co-inoculation with Rhizobium and plant growth-promoting bacteria. Rom Biotechnol Lett 16:5919–5926
Sulbaran M, Perez E, Ball MM, Bahsas A, Yarzabal LA (2009) Characterization of the mineral phosphate-solubilizing activity of Pantoeaaglomerans MMB051 isolated from an iron-rich soil in south eastern Venezuela (Bolivar state). Curr Microbiol 58:378–383. https://doi.org/10.1007/s00284-008-9327-1
Suryanto D, Asril M, Munir E, Kardhinata EH (2014) Assay of antagonistic bacteria of single isolate and combination to control seedling-off in chili seed caused by Fusarium oxysporum. J Pure Appl Microbiol 8(2):645–650
Taktek S, Trépanier M, Servin PM, St-Arnaud M, Piché Y, Fortin JA, Antoun H (2015) Trapping of phosphate solubilizing bacteria on hyphae of the arbuscular mycorrhizal fungus Rhizophagsirregularis DAOM 197198. Soil Biol Biochem 90:1–9. https://doi.org/10.1016/j.soilbio.2015.07.016
Tan B, Wang H, Wang X, Ma C, Zhou J, Dai X (2021) Health risks and source analysis of heavy metal pollution from Dust in Tianshui, China. Minerals 11:502. https://doi.org/10.3390/min11050502
Tarfeen N, Nisa KU, Hamid B, Bashir Z, Yatoo AM, Dar MA, Mohiddin FA, Amin Z, Ahmad RA, Sayyed RZ (2022) Microbial remediation: a promising tool for reclamation of contaminated sites with special emphasis on heavy metal and pesticide pollution: a review. Processes 10(7):1358. https://doi.org/10.3390/pr10071358. MDPI
Tayade A, Geetha P, Anusha S, Dhanapal R, Hari K (2019) Bio-intensive modulation of sugarcane ratoon rhizosphere for enhanced soil health and sugarcane productivity under tropical Indian condition. Sugar Tech 21:278–288. https://doi.org/10.1007/s12355-018-0669-0
Teng Z, Chen Z, Zhang Q, Yao Y, Song M, Li M (2019a) Isolation and characterization of phosphate solubilizing bacteria from rhizosphere soils of the Yeyahu Wetland in Beijing, China. Environ Sci Pollut Res 26:33976–33987. https://doi.org/10.1007/s11356-018-2955-5
Teng Z, Shao W, Zhang K, Huo Y, Li M (2019b) Characterization of phosphate solubilizing bacteria isolated from heavy metal contaminated soils and their potential for lead immobilization. J Environ Manag 231:89–197. https://doi.org/10.1016/j.jenvman.2018.10.012
Thampi M, Dhanraj N, Prasad A, Ganga G, Jisha M (2023) Phosphorus solubilizing microbes (PSM): biological tool to combat salinity stress in crops. Symbiosis 91:15–32. https://doi.org/10.1007/s13199-023-00947-3
Thomas GV, Shantaram MV, Saraswathy N (2005) Occurrence and activity of phosphate solubilizing fungi from coconut plantation soils. J Plant Sci 87:357–364. https://doi.org/10.1007/BF02181903
Timofeeva A, Galyamova M, Sedykh S (2022) Prospects for using phosphate solubilizing microorganisms as natural fertilizers in agriculture. Plants 11:2119. https://doi.org/10.3390/plants11162119
Tittabutr P, Awaya JD, Li QX, Borthakur D (2008) The cloned 1-aminocyclopropane-1-carboxylate (ACC) deaminase gene from Sinorhizobium sp. strain BL3 in Rhizobium sp. Strain TAL1145 promotes nodulation and growth of Leucaena leucocephala. Syst Appl Microbiol 31(141):150. https://doi.org/10.1016/j.syapm.2008.03.001
Tripathi M, Munot HP, Shouche Y, Meyer JM, Goel R (2005) Isolation and functional characterization of siderophore producing lead and cadmium resistant Pseudomonas putida KNP9. Curr Microbiol 50:233–237. https://doi.org/10.1007/s00284-004-4459-4
Turan M (2006) Evaluation of the capacity of phosphate solubilizing bacteria and fungi on different forms of phosphorus in liquid culture. J Sustain Agric 28:99–108. https://doi.org/10.1300/J064v28n03_08
Unnikrishnan BV, Binitha NK (2024) Positive effect of inoculation with an Aspergillus strain on phosphorus and iron nutrition plus volatile organic compounds in rice. Folia Microbiol 1–10. https://doi.org/10.1007/s12223-024-01129-4
Uzah GA, Ire FS, Ogugbue CJ (2024) Isolation and molecular characterization of microorganisms with biofertilizer potential. Scientia Afr 23(1):173–188. https://doi.org/10.4314/sa.v23i1.15
Vala AK, Sutariya V (2012) Trivalent arsenic tolerance and accumulation in two facultative marine fungi. Jundishapur J Microbiol 5(4):542–545. https://doi.org/10.5812/jjm.3383
Vazquez P, Holguin G, Puente ME, Lopez-Cortes A, Bashan Y (2000) Phosphate-solubilizing microorganisms associated with the rhizosphere of mangroves in a semiarid coastal lagoon. Biol Fertil Soils 30:460–468. https://doi.org/10.1007/s003740050024
Vivas A, Biro B, Ruiz-Lozano JM, Barea JM, Azcon R (2006) Two bacterial strains isolated from a Zn-polluted soil enhance plant growth and mycorrhizal efficiency under Zn toxicity. Chemosphere 52:1523–1533. https://doi.org/10.1016/j.chemosphere.2005.06.053
Vyas, Gulati A (2009) Organic acid production in vitro and plant growth promotion in maize under controlled environment by phosphate-solubilizing fluorescent Pseudomonas. BMC Microbiol 9:174. https://doi.org/10.1186/1471-2180-9-174
Wang L, Zhou F, Zhou J, Harvey PR, Yu H, Zhang G (2022) Genomic analysis of Pseudomonas asiatica JP233: An efficient phosphate-solubilizing bacterium. Genes 13:2290. https://doi.org/10.3390/genes13122290
Wani PA, Khan MS, Zaidi A (2007) Synergistic effects of the inoculation with nitrogen fixing and phosphate-solubilizing rhizobacteria on the performance of field grown chickpea. J Plant Nutr Soil Sci 170:283–287. https://doi.org/10.1002/jpln.200620602
Wei Y, Zhao Y, Shi M, Cao Z, Lu Q, Yang T, Fan Y, Wei Z (2017) Effect of organic acids production and bacterial community on the possible mechanism of phosphorus solubilization during composting with enriched phosphate-solubilizing bacteria inoculation. Biores Technol 26:30921–30929. https://doi.org/10.1016/j.biortech.2017.09.092
Wen ZL, Yang MK, Du MH, Zhong ZZ, Lu YT, Hua XM, Fazal A, Mu CH, Yan SF, Zhen Y, Qi YRW (2019) Enrichments/derichments of root-associated bacteria related to plant growth and nutrition caused by the growth of an EPSPS-transgenic maize line in the field. Front Microbiol 10:1335. https://doi.org/10.3389/fmicb.2019.01335
Widyastuti SM, Sumardi AS (1998) Pengendalian Hayati Penyakit Akar Merah Pada Akasia Dengan Trichoderma= Biological control of red root-rot disease of acacia using Trichoderma. J Plant Prot 4:65–72
Williamson LC, Ribrioux SP, Fitter AH, Leyser HM (2001) Phosphate availability regulates root system architecture in arabidopsis. Plant Physiol 126:875–882. https://doi.org/10.1104/pp.126.2.875
Yadav A, Yadav K, Vashistha A (2016) Phosphate solubilizing activity of Pseudomonas fluorescens PSM1 isolated from wheat rhizosphere. J Appl Nat Sci 8:93–96. https://doi.org/10.31018/jans.v8i1.754
Yahaghi Z, Shirvani M, Nourbakhsh F, de la Peña TC, Pueyo JJ, Talebi M (2018) Isolation and characterization of Pb-Solubilizing bacteria and their effects on Pb uptake by Brassica juncea: implications for Microbe-Assisted phytoremediation. J Microbiol Biotechnol 28(7):1156–1167. https://doi.org/10.4014/jmb.1712.12038
Yatoo AM, Ali MN, Baba ZA, Hassan B (2021) Sustainable management of diseases and pests in crops by vermicompost and vermicompost tea. A review. Agron Sustain Develop 41:1–26. https://doi.org/10.1007/s13593-020-00657-w
Yatoo AM, Ali MN, Zaheen Z, Baba ZA, Ali S, Rasool S, Hamid B (2022) Assessment of pesticide toxicity on earthworms using multiple biomarkers: a review. Environ Chem Lett 20:2573–2596. https://doi.org/10.1007/s10311-022-01386-0
Yi Y, Huang W, Ge Y (2008) Exopolysaccharide: a novel important factor in the microbial dissolution of Tricalcium phosphate. World J Microbiol Biotechnol 24:1059–1065. https://doi.org/10.1007/s11274-007-9575-4
Zaheer A, Mirza BS, Mclean JE, Yasmin S, Shah TM, Malik KA, Mirza MS (2016) Association of plant growth-promoting Serratia spp. with the root nodules of chickpea. Res Microbiol 167:510–520. https://doi.org/10.1016/j.resmic.2016.04.001
Zai XM, Fan JJ, Hao ZP, Liu XM, Zhang WX (2021) Effect of coinoculation with arbuscular mycorrhizal fungi and phosphate solubilizing fungi on nutrient uptake and photosynthesis of beach palm under salt stress environment. Sci Rep 11:5761. https://doi.org/10.1038/s41598-021-84284-9
Zhan X, Rajendran A, Grimm S, Sun X, Lin H, He R, Bo Hu (2023) Screening of calcium- and iron-targeted phosphorus solubilizing fungi for agriculture production. Rhizosphere 26:100689. https://doi.org/10.1016/j.rhisph.2023.100689
Zhang L, Fan J, Feng G, Declerck S (2019) The arbuscular mycorrhizal fungus Rhizophagus irregularis MUCL 43194 induces the gene expression of citrate synthase in the tricarboxylic acid cycle of the phosphate-solubilizing bacterium Rahnella aquatilis HX2. Mycorrhiza 29:69–75. https://doi.org/10.1007/s00572-018-0871-7
Zhang C, Chen H, Dai Y, Chen Y, Tian Y, Huo Z (2023a) Isolation and screening of phosphorus solubilizing bacteria from saline alkali soil and their potential for Pb pollution remediation. Front Bioeng Biotechnol 11:1134310. https://doi.org/10.3389/fbioe.2023.1134310
Zhang J, Han X, Su Y, Staehelin C, Xu C (2023b) T-DNA insertion mutagenesis in Penicillium brocae results in identification of an enolase gene mutant impaired in secretion of organic acids and phosphate solubilization. Microbiology 169:001325. https://doi.org/10.1099/mic.0.001325
Zhao R, Huang L, Peng X, Fan L, Chen S, Qin P, Zhang J, Chen A, Huang H (2023) Effect of different amounts of fruit peel-based activator combined with phosphate-solubilizing bacteria on enhancing phytoextraction of cd from farmland soil by ryegrass. Environ Pollut 316:1206. https://doi.org/10.1016/j.envpol.2022.120602
Zheng BX, Ding K, Yang XR, Wadaan MA, Hozzein WN, Penuelas J, Zhu YG (2019) Straw biochar increases the abundance of inorganic phosphate solubilizing bacterial community for better rape (Brassica napus) growth and phosphate uptake. Sci Total Environ 647:1113–1120. https://doi.org/10.1016/j.scitotenv.2018.07.454
Zhu F, Qu L, Hong X, Sun X (2011) Isolation and characterization of a Phosphate-Solubilizing Halophilic Bacterium Kushneria sp. YCWA 18 from Daqiao Saltern on the Coast of Yellow Sea of China. Evid Based Complement Alternat Med 1–6
Zveushe OK, De Dios VR, Zhang H, Zeng F, Liu S, Shen S (2023) Effects of co-inoculating Saccharomyces spp. with Bradyrhizobium japonicum on atmospheric nitrogen fixation in soybeans (Glycine max (L.). Plants 12:681
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interests
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bashir, Z., Hamid, B., Yatoo, A.M. et al. Phosphorus Solubilizing Microorganisms: An Eco-Friendly Approach for Sustainable Plant Health and Bioremediation. J Soil Sci Plant Nutr (2024). https://doi.org/10.1007/s42729-024-02007-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42729-024-02007-1