Abstract
Economically feasible bioethanol production from lignocellulosic biomass requires solid loadings ≥ 15% dry matter (DM, w/w). However, increased solid loadings can lead to process difficulties, which are characterized by high apparent slurry viscosity, insufficient substrate mixing and limited water availability, resulting in reduced final glucose yields. To overcome these limitations, this study focused on enzymatic hydrolysis of 10–35% DM solid loadings with steam-exploded wheat straw in two different particle sizes. At solid loadings of 20 and 25% DM small particle size of ≤ 2.5 mm yielded 16.9 ± 1.1% and 10.2 ± 1.4% increased final glucose concentrations compared to large particle size of 30 ± 20 mm. Small particle size also positively influenced slurry viscosity and, therefore, miscibility. As a key finding of this investigation, high gravity enzymatic hydrolysis with solid loadings of 30–35% DM was indeed successfully employed when wheat straw was applied in small particle size. Here, the highest final glucose yield was achieved with 127.9 ± 4.9 g L−1 at 35% DM solid loading. An increase in the solid loading from 10 to 35% DM in small particle size experiments resulted in a 460% increase in the final glucose concentration.
Article Highlights
-
Fiber size reduction benefits enzymatic high gravity hydrolysis of steam-exploded wheat straw.
-
Small fiber size of ≤ 2.5 mm enables saccharification at 35% w/w solid loadings.
-
Water-retention capacity of cellulosic fibers influence slurry viscosity and yielded glucose concentration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Lignocellulose is the most abundant source of renewable biomass and an attractive material for bioethanol production [1]. Over the last decades, a number of demonstration plants were introduced for the production of lignocellulosic bioethanol [2]. Some countries now produce bioethanol from cellulosic feedstock at different development stages [3]. Important factors for processing economics and energy balance are pretreatment efficiency, saccharification of the lignocellulosic biomass and fermentation of the slurry phase [4, 5].
Over the last years, enzymatic hydrolysis has been the preferred method to recover fermentable monosaccharides from lignocellulosic biomass [5]. One approach to improve monosaccharide yields and, in-terms, economic feasibility is to apply high gravity enzymatic hydrolysis [6,7,8]. High gravity enzymatic hydrolysis is defined as a saccharification step at solid loadings ≥ 15% dry matter (DM, w/w). It offers advantages over hydrolysis at low solid loadings [3]. For instance, it decreases operational costs due to lower energy demand for cooling and heating steps [7, 9] and reduces required equipment volumes, water consumption, wastewater generation and costs for additional separation steps [10, 11]. A major advantage of high gravity enzymatic hydrolysis is that it can result in a slurry with a bioethanol concentration of ≥ 4% w/w, which is required for an economically feasible and efficient distillation step [10, 12].
However, high gravity enzymatic hydrolysis poses technical difficulties. With an increase in solid loading the apparent viscosity of the slurry increases, resulting in poor mixing properties of liquid and solid fractions such as limited heat and mass transfer rates. Mixing is required for sufficient contact between enzymes and substrate solids. Poor mixing results in areas with high concentrations of hydrolysis products (e.g., cellobiose, glucose and xylose), which hinders enzyme catalysis reactions due to limited substrate diffusion into the active site, or product diffusion out [13].
It was shown that an increase in solid loading can result in a decrease in the final glucose yield [14]. This effect is known as the high-solids effect and has mostly been attributed to the non-productive binding of enzymes to lignin [15, 16] or the inhibition of enzymes resulting from high concentrations of hydrolysis products [14, 17]. Several studies have linked the ability of lignocellulosic polymers to constrain water with the inhibition of cellulase [18, 19]. Water acts as medium through which enzymes diffuse to and degradation products away from the enzymatic binding sites and operates as a reactant in the cleavage of the glycosidic bonds within the cellulose polymers [20]. They suggested that there is a strong correlation between the degree to which the polymers constrain the surrounding water and the inhibition of cellulase. However, it is still not fully understood if this correlation is dominated by the non-productive binding of enzymes to lignin or poor mixing quality of the slurry.
Plant fibers consist of a hydrophilic polymer matrix with micro-pores in the cell wall and macro-pores within the plant tissue, which are known to absorb the surrounding fluid. The literature implies that the extent of fluid absorption depends mainly on chemical and physical characteristics such as substrate porosity and particle size [21]. With an increase in solid loading, more water gets constrained within the cellulosic fibers, making it unavailable as a reactant and dispersant agent [17]. If the slurry reaches a certain solid loading, the average particle separation distance is less than the particle radius and the slurry is considered “dense” [22]. The ability of biological particles to absorb water increases this effect substantially. A further increase in the solid loading of a “dense” slurry causes all available water to be absorbed or entrained within the plant fibers [23]. A lack of freely available water decreases the lubricating fluid layer between particles and causes the slurry to become unsaturated. In an unsaturated slurry, the fluid layer gets replaced by air, which causes the fibrous particles to become further entangled and agglomerate to larger flocs or fiber networks [24]. The agglomerated fibers behave as a wet granular material with a high apparent viscosity that causes shearing difficulties and poor mixing.
Altering the physical characteristics, such as the particle size, of the biomass could help to further understand the mechanism behind the cellulase inhibition. It is known that the particle size in correlation with several substrate properties such as water-retention capacity, chemical composition of fibers and inter-fiber interactions influence the rheological behavior of slurries and the amount of fermentable monosaccharides released during enzymatic hydrolysis (sugar yield) [14, 25,26,27]. Thus, this study investigated the reduction in particle size as a way to alter the water-polymer interaction and reduce cellulase inhibition at a high solid loading. This included the investigation of preferential steam-explosion conditions, glucose yields, substrate conversion and rheological behavior of the slurry.
2 Experimental
2.1 Biomass feedstock
Wheat straw (Triticum aestivum L.) was grown, harvested, dried and coarsely chopped by Ihinger Hof (University of Hohenheim, Germany) in 2017. The straw consisted of 44.0% cellulose, 19.8% hemicellulose, 32.3% lignin and 4.5% ash, determined according to Sluiter et al. [28]. Wheat straw was initially cut to a length of 30 ± 20 mm to investigate large fiber size (LFS). A milling pretreatment (type SR2. Retsch GmbH, Germany) with a 2.5 mm sieve was applied to investigate small fiber size (SFS). The milling pretreatment did not impose any significant changes in substrate composition.
2.2 Experimental setup
The experimental setup investigated (1) preferential steam explosion conditions and subsequently (2) enzymatic hydrolysis with different particle sizes and solid loadings. Steam explosion is known to partly solubilize the biomass, resulting in the release of sugar and the formation of sugar derived degradation products (inhibitors) that can negatively impact the enzymatic hydrolysis [29, 30].
To be able to differentiate between the inhibiting effects caused by the high-solids effect during enzymatic hydrolysis and accumulated inhibitors, formed during steam explosion, it is necessary to remove the latter prior to enzymatic hydrolysis. Additionally, the goal was to find pretreatment conditions that would increase the accessibility of the cellulosic fibers to hydrolytic enzymes, without significantly altering the physiochemical characteristics of the substrate. Therefore, steam explosion experiments with subsequent washing steps were conducted to investigate the influence of the pretreatment severity on formation of inhibitors and changes of material composition.
2.3 Preferential steam explosion conditions
The pretreatment experiments were performed with water-soaked substrate in a double-walled 20 L steam explosion reactor (H & K GmbH Behälter und Edelstahltechnik, Kehl, Germany), heated by indirect steam injection. The severity parameter (S0) was used to adjust the degradation, and depolymerization of the material and was calculated using the following equation [31]:
where T is the temperature (°C) and t the time (min). A reactor temperature of 170 °C and pressure of 600–700 kPa were kept for 45 and 120 min to apply a low (S0 = 3.75) and high (S0 = 4,18) severity pretreatment, accordingly. At the end of the reaction time, a reactor valve was opened to abruptly reduce the reactor pressure to ambient pressure. Steam explosion conditions were adapted according to the optimization studies by Fleischer [32].
The pretreated material was water-washed in a ratio of 1:5 g g−1 in a stirred beaker (10 min), centrifuged at 10,000 × g and the liquid phase removed. The washing step (WS) was repeated three times and the materials subsequently dried at 65 °C for 96 h. Liquids from the pretreatment and washing steps were analyzed for soluble contents. The amount of soluble content removed with each washing step was calculated by accounting for their content in the solid and liquid phase following pretreatment and washing.
2.4 Enzymatic hydrolysis
Enzymatic hydrolysis was carried out in 0.5 L stainless steel vessels equipped with a crossbar stirrer and closeable plastic lids at 50 °C for 96 h at a volume of 0.2 L. All experiments were set up in triplicates with the pretreated substrates, 50 mM citrate buffer at pH 4.8 and a cellulolytic enzyme (Cellic® CTec2. Novozymes, Denmark) at enzyme loading of 15 FPU g−1 DM. In a first experimental setup, both particle sizes were investigated in solid loadings of 10, 15, 20 and 25% DM. Additionally, a second experiment investigated very high solid loadings of 30 and 35% DM. Enzymatic hydrolysis was also performed with SFS, when the substrate was milled after steam explosion, to investigate the effect of particle size on the severity of the pretreatment.
2.5 Rheological measurement
The slurry viscosity was measured using a modular compact rheometer (MCR 92. Anton Paar GmbH, Germany). The rheometer was operated in rotational mode at 20 °C with logarithmically increasing shear rates between 0.1 and 10 s−1 and equipped with a plate/plate geometry with a 1.5 mm gap. Each measurement was repeated five times with fresh samples. Results are given in mean values and standard deviation.
2.6 Conversion analysis
The enzymatic hydrolysis efficiency was evaluated by glucan conversion analysis. Vessel liquids were sampled every 24 h and analyzed for cellobiose, glucose, xylose and arabinose concentrations with high-performance liquid chromatograph (HPLC) refractive index detector (RID, Shodex RI-101, Thermo Fisher, USA). All samples were centrifuged at 4000 × g for 10 min and the supernatant water-diluted in a ratio of 1:3. The supernatant was subsequently 0.45 µm filtered and analyzed via HPLC equipped with a Phenomenex Rezex RHM column at 80 °C with 1 mM H2SO4 as eluent at a flow rate of 0.6 mL min−1. The calculation of the converted glucose was adapted from Zhu et al. [33]. The formula was developed to investigate the true lignocellulosic conversion to glucose during enzymatic hydrolysis performed at high solid loadings and considers varying liquid densities and volumes resulting from the constant solubilization of solid matter (Eq. 2).
Yg is the conversion of biomass to glucose (glucan conversion), cg and cg0 the glucose concentration, Vh and Vh0 the volume of the hydrolysate, Xis0 the initial mass fraction of insoluble solids in total solids, φG the molecular weight ratio of glucose to glucan monomer (φG = 1.11), ρh0 the initial density of the hydrolysate, xg0 the initial mass fraction of glucan in insoluble solids and ƒtS0 the initial mass fraction of total solids in the slurry. All terms with subscript 0 correspond to initial values at the start of the experiment. Results are given in mean values and standard deviation.
2.7 Degradation product determination
Liquid samples from the pretreatment and washing steps were analyzed for potentially inhibiting compounds. Furfural, 5-Hydroxymethylfurfural (5-HMF), formic and acetic acid were quantified using the previously described HPLC method. The total phenolic content was determined by means of Folin–Ciocalteu reagent according to Singleton et al. [34]. Units of all degradation products, including sugars are presented in mg g−1 based on initial biomass loading at the start of the experiment.
3 Results and discussion
3.1 Preferential steam explosion conditions
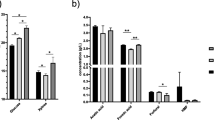
To define preferential steam explosion conditions, the soluble contents were analyzed after steam explosion at low and high severity (Table 1). The initial amount of solubilized sugars changed with the severity of the pretreatment. The content of glucan monomers decreased from 34.8 ± 0.4 mg g−1 to 26.0 ± 0.4 mg g−1 and xylan monomers increased from 90.4 ± 1.0 mg g−1 to 113.3 ± 6.8 mg g−1 when altering from low to high severity conditions. Additionally, the concentration of inhibitors increased significantly. The concentration of organic acids showed the highest increase from 6.9 ± 0.2 mg g−1 formic acid and 16.3 ± 0.0 mg g−1 acetic acid at low severity to 21.6 ± 0.2 mg g−1 formic acid and 28.6 ± 0.3 mg g−1 acetic acid at high severity, respectively. The concentration of total phenolic contents remained below 0.2 mg g−1 for both severity factors. Approximately 50% of the soluble contents were removed in the first washing step. Further washing steps removed most of the remaining contents (≥ 78%). The content reduction was calculated based on the relation of washing step three (WS3) and the initial content of the liquid phase after steam explosion.
The solubilization of biomass also caused a change in the composition of the remaining insoluble solids (Table 2). By accounting for glucan and xylan content in the liquid and solid phases, their conversion ratios can be calculated. While the glucan content is relatively unaffected by steam explosion and remains in the solid state, a large amount of xylan becomes solubilized. Based on low and high severity steam explosion, at least 7.4% and 5.9% of glucan as well as 50.5% and 57.3% of xylan were solubilized. It is likely that they were removed during the subsequent washing steps. Material composition analysis showed that there were no significant differences in material composition between steam-exploded SFS and LFS.
Pretreating the material at a higher severity, in theory, increases the accessibility of the cellulosic fibers to hydrolytic enzymes and therefore increases the glucan conversion [35, 36]. However, the literature implies that high severity steam explosion most likely resulted in significant changes of the material composition. For instance, Kellock et al. [37] showed that a high severity pretreatment causes structural changes in lignin that are associated with increased binding and inactivation of cellulase. In addition, Ehrhardt et al. [38] reported that an increase in severity can affect the rheological behavior of the slurry and can cause particle size reduction. This suggests that pretreatments at high severity could negatively alter the physiochemical characteristics of the substrate and could interfere with the effects of the particle size reduction. Therefore, subsequent enzymatic hydrolysis experiments were performed on triple-washed material, pretreated at S0 = 3.75.
3.2 Glucan conversion in LFS and SFS experiments
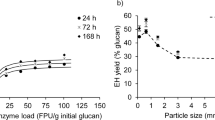
The enzymatically hydrolyzed glucose concentrations of small fiber size (SFS) and large fiber size (LFS) wheat straw in experimental setups with solid loadings of 10–25% DM are shown in Fig. 1. After 96 h, the highest glucose concentrations were 74.1 ± 2.7 g L−1 for LFS and 82.5 ± 0.8 g L−1 for SFS at a solid loading of 25% DM. The glucan conversion was determined with values of 58.3 ± 0.8%, 60.2 ± 2.9%, 52.6 ± 1.4% and 52.3 ± 1.5% for LFS and 57.1 ± 0.0%, 59.0 ± 1.6%, 64.9 ± 0.9% and 59.7 ± 0.5% for SFS in reactors with solid loadings of 10, 15, 20 and 25% DM, respectively. Furthermore, similar glucose concentrations were determined, when the milling process for SFS was applied after steam explosion. This indicated that the milling process had no impact on the severity of the steam explosion (Figure A1 = attachment).
The results of the determined glucose yields showed that the glucan conversion was lower when enzymatic hydrolysis was performed with LFS at solid loadings ≥ 20% DM. This reduction in glucose yield with increasing solid loading is in accordance with the results from other researchers performing high gravity enzymatic hydrolysis [2, 14, 39, 40]. Significantly higher final glucose yields and glucan conversions were present in SFS experiments at 20 and 25% DM solid loadings. In SFS experiments, the glucan conversion was always ≥ 57%, even at a solid loading of 25% DM.
In direct comparison, the amount of released glucose was increased by 16.9 ± 1.1% and 10.2 ± 1.4% in experiments with SFS compared to LFS, when performed at solid loadings of 20 and 25% DM (Table 3). The results indicated that a reduced fiber size is possibly more resilient to high-solids effects and leads to improved slurry mixing properties and beneficial enzyme accessibility to the fibers.
3.3 Slurry viscosity
Enzymatic hydrolysis with LFS showed increased inhibition at ≥ 20% DM solid loading compared to SFS experiments (Table 3). At 20% DM solid loading, it is suspected that convective mixing and freely available water are negligible, likely indicating the point as mass transfer limitations, known as high-solids effect. Therefore, the slurry viscosity was investigated at 20% DM solid loading for SFS and LFS (Fig. 2).
Both slurries exhibit shear-thinning behavior in the range of 0.1–10 s−1 shear rates. However, LFS showed an increased viscosity by factor 7–14 from high to low shear rates. A possible explanation could be that the milling process caused a breakdown of plant tissue resulting in particles that contained fewer or smaller macro-pores, such that less liquid is entrained within these particles. Further, interactions among the fibers due to friction or entanglement are possibly less likely to impact the rheological behavior with SFS. These effects allow the mixture to remain a saturated slurry with a comparably low viscosity, even at solid loadings ≥ 20% DM, while LFS showed the typical rheological behavior of wet granular (Fig. 3).
The lower viscosity of SFS indicated that the slurry does possibly contain enough free water to keep a fluid layer between the fibers, causing the contact between them to remain lubricated and not frictional. The combination of improved glucose yield and significantly lower viscosity shows that a reduction in particle size potentially provides an efficient method to reduce the ability of polymers to constrain water. The effect of different particle size dimensions on enzymatic hydrolysis has often been studied. Many studies impose that particle size reduction increases the accessibility between enzymes and cellulose, which results in improved glucan conversion [41]. For example, a recent modeling and simulation study from Gaikwad [42] investigated the effect of particle size dimensions of 0.78–25.52 µm on enzymatic hydrolysis of cotton cellulose. He stated that particle size reduction also increases the available reactive surface area for mass transfer actions, which favors enzyme adsorption and also reduces the probability of inhibition by lignin or hydrolysis products.
However, the fact that the improved glucose yield in our experiments is only present at solid loadings ≥ 20% w/w supports the hypothesis that the inhibitory potential of lignocellulosic polymers is mainly caused by the degree to which they absorb the surrounding liquid. If the increased glucose yield was caused by improved accessibility between enzymes and cellulose, an improvement would also be present at solid loadings ≤ 20% w/w. Gatt et al. [43] studied the effects of a bioextrusion pretreatment on high gravity enzymatic hydrolysis with solid loadings of up to 40% w/w and came to a similar conclusion. Their pretreatment enhanced the subsequent enzymatic hydrolysis of raw corn crop residues by reducing particle size and improving mixing conditions. However, surface cellulose accessibility analysis showed that the negative impact of a high solid loading was not associated with the lack of exposed cellulose. This suggests that a particle size reduction most likely had no impact on the hydrolysis efficiency itself, but helped reduce the high solids effect caused by poor mixing of the slurry. DeMartini et al. [44] investigated a milling pretreatment on steam-exploded aspen wood chips. Similar to our results, their experiments indicated that an increased surface area did not improve glucan conversion. To address these results, they performed a Simons’ stain test based on competitive adsorption of two different-sized molecular dyes. The test results suggested that the milling pretreatment did not improve the ratio of adsorbed dye and therefore most likely did not improve enzyme accessibility. They concluded that steam explosion treatment sufficiently expanded pore size in the material and an additional milling pretreatment was not necessary.
3.4 Very high gravity enzymatic hydrolysis
The second experimental setup investigated the enzymatically hydrolyzed glucose yields at very high solid loadings of 30 and 35% DM (Fig. 4).
While solid loadings of 30 and 35% DM could be applied to SFS, it was not possible to conduct the experiment with LFS, due to apparent high viscosity and technical mixing problems.
The final glucose concentrations after 96 h enzymatic hydrolysis were 116.1 ± 0.9 g L−1 and 127.9 ± 4.9 g L−1 for 30 and 35% DM, respectively. The glucan conversions were determined with 66.4 ± 0.4% and 58.6 ± 1.9%.
In previous experiments at lower solid loadings, most of the glucose was released within 24–48 h. During enzymatic hydrolysis performed at solid loadings of 30 and 35% DM, at least 72 h were required to reach comparable glucose concentrations. While the process seemed to release glucose slower at an early stage, it is likely that with enhancing hydrolysis time, the liquefaction of the slurry increased, leading to beneficial rheological properties.
While the beneficial effects of a particle size reduction have often been studied, scientific studies which consider both aspects of particle size reduction and solid loadings at ≥ 30% w/w simultaneously are scarce. Weiss et al. [19] showed that a milling pretreatment increased the cellulose conversion yield of steam pretreated wheat straw at solid loadings of 5–30%. However, they observed a decrease in glucan yields with increased solid loadings, but missed to provide more specifics on particle size dimensions. In contrast to their results, the key finding of our manuscript was that an efficient enzymatic hydrolysis with no reduction in glucan conversion was indeed possible at solid loadings of 30 and 35% w/w with SFS. It is likely that a reduction in particle size to ≤ 2.5 mm is necessary to achieve these results.
A study from Chundawat et al. [45] investigated the impact of a particle size reduction in corn stover on ammonia fiber expansion (AFEX) treatment and enzymatic hydrolysis. Their results showed that a particle size reduction from 0.85–0.5 to < 0.15 mm enhanced glucan conversion by 15–20% at 1% w/w glucan loading. While our experiments focused specifically on a particle size reduction from large (30 ± 20 mm) to small (≤ 2.5 mm) particles, the results from Chundawat et al. indicated that a further reduction in particle size could potentially lead to an even higher glucan conversion. On the contrary, Liu et al. [46] reported a decrease in glucan conversion with decreasing particle size (25–5 mm) during enzymatic hydrolysis of steam-exploded corn stover. They showed that the specific surface area increased and the crystallinity index decreased for larger particles. Our study showed that a reduction in particle size is clearly beneficial for improved slurry mixing at high solid loadings. However, differences in feedstock type and applied pretreatment method seem to play a very important role in enzyme-cellulose interaction. The beneficial effects of particle size reduction could possibly be improved by application of optimized pretreatment methods.
SFS offers great potentials for very high gravity hydrolysis ≥ 35% w/w. It has the potential to achieve highly concentrated hydrolysate streams, which could be a crucial factor for significant economic benefits due to operational and capital cost savings. However, the energy consumption required for the milling pretreatment should also be considered. Adapa et al. [47] investigated the energy consumption for milling wheat straw to smaller particle sizes using a ball mill. Their results showed that milling wheat straw to fit through a 1.6 mm screen required 42.57 ± 2.04 kWh t−1, while using a 6.4 mm screen only required 12.26 ± 0.70 kWh t−1. Additionally, they found that performing substrate milling after steam explosion reduced the energy consumption by approximately 60%. This shows that it is necessary to include energy consumption studies in order to investigate economically sound high gravity hydrolysis with reduced particle sizes.
4 Conclusion
The experiments showed the importance of particle size on the yield of high gravity enzymatic hydrolysis. Reducing the particle size of wheat straw prior to enzymatic hydrolysis was found to be an effective method to increase the final glucose yield. A reduced particle size has the potential to enable enzymatic hydrolysis at a solid loading of up to 35% DM. It is likely that these improvements are mainly caused by destruction of the porous structure of the large fibers and thus decreased water-retention capacities.
Sugar concentrations up to 127.9 ± 4.9 g L−1 glucose can be achieved with enzymatic hydrolysis of SFS wheat straw. Implementing enzymatic hydrolysis with a solid loading of 35% DM has the potential to increase final glucose concentration by up to 460%, compared to 10% solid loading. The investigation of small particle sizes provides important information on process optimization potentials for lignocellulosic bioethanol production. For a successful implementation of high gravity enzymatic hydrolysis on a larger scale, we recommend specific investigations on optimized milling pretreatments. Combining SFS with novel reactor designs for high gravity hydrolysis could potentially enable a feasible enzymatic hydrolysis process at solid loadings ≥ 35% DM.
Availability of data and material
The data that support the findings of this study are available from the corresponding author, Luis Hoppert, upon reasonable request.
References
Soccol CR, Faraco V, Karp SG, Vandenberghe LPS, Thomaz-Soccol V, Woiciechowski AL, Pandey A (2019) Lignocellulosic bioethanol: current status and future perspectives. Biofuels: alternative feedstocks and conversion processes for the production of liquid and gaseous biofuels. Elsevier, pp 331–354
Jørgensen H, Vibe-Pedersen J, Larsen J, Felby C (2007) Liquefaction of lignocellulose at high-solids concentrations. Biotechnol Bioeng 96(5):862–870. https://doi.org/10.1002/bit.21115
da Silva ASA, Espinheira RP, Teixeira RSS, de Souza MF, Ferreira-Leitão V, Bon EPS (2020) Constraints and advances in high-solids enzymatic hydrolysis of lignocellulosic biomass: a critical review. Biotechnol Biofuels. https://doi.org/10.1186/s13068-020-01697-w
Heller D, Einfalt D (2020) Wildflower mixtures for bioethanol production - Pretreatment and enzymatic hydrolysis. Biomass Bioenerg 141:105727. https://doi.org/10.1016/j.biombioe.2020.105727
Aditiya HB, Mahlia TMI, Chong WT, Nur H, Sebayang AH (2016) Second generation bioethanol production: a critical review. Renew Sustain Energy Rev 66:631–653. https://doi.org/10.1016/j.rser.2016.07.015
Koppram R, Tomás-Pejó E, Xiros C, Olsson L (2014) Lignocellulosic ethanol production at high-gravity: challenges and perspectives. Trends Biotechnol 32(1):46–53. https://doi.org/10.1016/j.tibtech.2013.10.003
Humbird D, Mohagheghi A, Dowe N, Schell DJ (2010) Economic impact of total solids loading on enzymatic hydrolysis of dilute acid pretreated corn stover. Biotechnol Prog 26(5):1245–1251. https://doi.org/10.1002/btpr.441
Cheng MH, Wang Z, Dien B, Slininger PJ, Singh V (2019) Economic analysis of cellulosic ethanol production from sugarcane bagasse using a sequential deacetylation, hot water and disk-refining pretreatment. Processes 7(10):642. https://doi.org/10.3390/pr7100642
Modenbach AA, Nokes SE (2013) Enzymatic hydrolysis of biomass at high-solids loadings – A review. Biomass Bioenerg 56:526–544. https://doi.org/10.1016/j.biombioe.2013.05.031
Wingren A, Galbe M, Zacchi G (2003) Techno-economic evaluation of producing ethanol from softwood: comparison of SSF and SHF and identification of bottlenecks. Biotechnol Prog 19(4):1109–1117. https://doi.org/10.1021/bp0340180
Quintero JA, Moncada J, Cardona CA (2013) Techno-economic analysis of bioethanol production from lignocellulosic residues in Colombia: a process simulation approach. Biores Technol 139:300–307. https://doi.org/10.1016/j.biortech.2013.04.048
Fan Z, South C, Lyford K, Munsie J, van Walsum P, Lynd LR (2003) Conversion of paper sludge to ethanol in a semicontinuous solids-fed reactor. Bioprocess Biosyst Eng 26(2):93–101. https://doi.org/10.1007/s00449-003-0337-x
Zhang ZC, Li JH, Wang F (2017) Kinetics of cellulase saccharification of corn stover after pretreatment by lignin peroxidase and H2O2. BioResources. https://doi.org/10.15376/biores.12.3.5462-5486
Kristensen JB, Felby C, Jørgensen H (2009) Yield-determining factors in high-solids enzymatic hydrolysis of lignocellulose. Biotechnol Biofuels 2(1):11. https://doi.org/10.1186/1754-6834-2-11
Lu X, Feng X, Li X, Zhao J (2018) The adsorption properties of endoglucanase to lignin and their impact on hydrolysis. Biores Technol 267:110–116. https://doi.org/10.1016/j.biortech.2018.06.031
Saini JK, Patel AK, Adsul M, Singhania RR (2016) Cellulase adsorption on lignin: a roadblock for economic hydrolysis of biomass. Renew Energy 98:29–42. https://doi.org/10.1016/j.renene.2016.03.089
Roberts KM, Lavenson DM, Tozzi EJ, McCarthy MJ, Jeoh T (2011) The effects of water interactions in cellulose suspensions on mass transfer and saccharification efficiency at high solids loadings. Cellulose 18(3):759–773. https://doi.org/10.1007/s10570-011-9509-z
Selig MJ, Thygesen LG, Felby C (2014) Correlating the ability of lignocellulosic polymers to constrain water with the potential to inhibit cellulose saccharification. Biotechnol Biofuels 7(1):159. https://doi.org/10.1186/s13068-014-0159-x
Weiss ND, Felby C, Thygesen LG (2019) Enzymatic hydrolysis is limited by biomass-water interactions at high-solids: improved performance through substrate modifications. Biotechnol Biofuels 12:3. https://doi.org/10.1186/s13068-018-1339-x
Liu Z-H, Chen H-Z (2016) Biomass-water interaction and its correlations with enzymatic hydrolysis of steam-exploded corn stover. ACS Sustain Chem Eng 4(3):1274–1285. https://doi.org/10.1021/acssuschemeng.5b01303
Nguyen TC, Anne-Archard D, Fillaudeau L (2015) Rheology of lignocellulose suspensions and impact of hydrolysis: a review. Adv Biochem Eng Biotechnol 149:325–357. https://doi.org/10.1007/10_2015_323
Stickel JJ, Powell RL (2005) Fluid mechanics and rheology of dense suspensionS. Annu Rev Fluid Mech 37(1):129–149. https://doi.org/10.1146/annurev.fluid.36.050802.122132
Coussot P (2005) Rheometry of pastes, suspensions, and granular materials. Wiley, Hoboken, NJ, USA
Hämäläinen J, Eskola R, Erkkilä A-L, Leppänen T (2011) Rheology in papermaking — from fibre suspension flows to mechanics of solid paper. Korea-Aust Rheol J 23(4):211–217. https://doi.org/10.1007/s13367-011-0026-2
Quiroga AG, Costa A, Maciel Filho R (2010) Analysis of conversion and operation strategies for enzymatic hydrolysis of lignocellulosic biomass in a series of CSTRs with distributed feeding. Bioprocess Biosyst Eng 33(8):901–910. https://doi.org/10.1007/s00449-010-0413-y
Knutsen JS, Liberatore MW (2010) Rheology modification and enzyme kinetics of high solids cellulosic slurries. Energy Fuels 24(5):3267–3274. https://doi.org/10.1021/ef100140g
Rajesh Dasari, Berson Eric (2007) The effect of particle size on hydrolysis reaction rates and rheological properties in cellulosic slurries. Appl biochem Biotechnol. https://doi.org/10.1007/978-1-60327-181-3_26
Sluiter H, Ruiz S, Templeton C (2012) Determination of Structural Carbohydrates and Lignin in Biomass: Laboratory Analytical Procedure (LAP). Golden, CO: National Renewable Energy Laboratory. NREL/TP510-42618
Kim Y, Ximenes E, Mosier NS, Ladisch MR (2011) Soluble inhibitors/deactivators of cellulase enzymes from lignocellulosic biomass. Enzyme Microb Technol 48(4–5):408–415. https://doi.org/10.1016/j.enzmictec.2011.01.007
Kim D (2018) Physico-chemical conversion of lignocellulose: inhibitor effects and detoxification strategies: a mini review. Molecules. https://doi.org/10.3390/molecules23020309
Abatzoglou N, Chornet E, Belkacemi K, Overend RP (1992) Phenomenological kinetics of complex systems: the development of a generalized severity parameter and its application to lignocellulosics fractionation. Chem Eng Sci 47(5):1109–1122. https://doi.org/10.1016/0009-2509(92)80235-5
Fleischer S (2010) Verbesserung der Energie-, Stoff- und Emissionsbilanzen bei der Bioethanolproduktion aus nachwachsenden Rohstoffen. Universität Hohenheim. http://opus.uni-hohenheim.de/volltexte/2011/579
Zhu Y, Malten M, Torry-Smith M, McMillan JD, Stickel JJ (2011) Calculating sugar yields in high solids hydrolysis of biomass. Biores Technol 102(3):2897–2903. https://doi.org/10.1016/j.biortech.2010.10.134
Singleton VL, Orthofer R, Lamuela-Raventós RM (1974) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Acad Press (Methods in Enzymology, 299), S. 152–178. https://doi.org/10.1016/S0076-6879(99)99017-1
Auxenfans T, Crônier D, Chabbert B, Paës G (2017) Understanding the structural and chemical changes of plant biomass following steam explosion pretreatment. Biotechnol Biofuels 10:36. https://doi.org/10.1186/s13068-017-0718-z
Kabel MA, Bos G, Zeevalking J, Voragen AGJ, Schols HA (2007) Effect of pretreatment severity on xylan solubility and enzymatic breakdown of the remaining cellulose from wheat straw. Biores Technol 98(10):2034–2042. https://doi.org/10.1016/j.biortech.2006.08.006
Kellock M, Maaheimo H, Marjamaa K, Rahikainen J, Zhang H, Holopainen-Mantila U, Ralph J, Tamminen T, Felby C, Kruus K (2019) Effect of hydrothermal pretreatment severity on lignin inhibition in enzymatic hydrolysis. Biores Technol 280:303–312. https://doi.org/10.1016/j.biortech.2019.02.051
Ehrhardt MR, Monz TO, Root TW, Connelly RK, Scott CT, Klingenberg DJ (2010) Rheology of dilute acid hydrolyzed corn stover at high solids concentration. Appl Biochem Biotechnol 160(4):1102–1115. https://doi.org/10.1007/s12010-009-8606-z
Lu Y, Wang Y, Xu G, Chu J, Zhuang Y, Zhang S (2010) Influence of high solid concentration on enzymatic hydrolysis and fermentation of steam-exploded corn stover biomass. Appl Biochem Biotechnol 160(2):360–369. https://doi.org/10.1007/s12010-008-8306-0
Ludwig D, Michael B, Hirth T, Rupp S, Zibek S (2014) High solids enzymatic hydrolysis of pretreated lignocellulosic materials with a powerful stirrer concept. Appl Biochem Biotechnol 172(3):1699–1713. https://doi.org/10.1007/s12010-013-0607-2
Sun S, Sun S, Cao X, Sun R (2016) The role of pretreatment in improving the enzymatic hydrolysis of lignocellulosic materials. Biores Technol 199:49–58. https://doi.org/10.1016/j.biortech.2015.08.061
Gaikwad A (2019) Effect of particle size on the kinetics of enzymatic hydrolysis of microcrystalline cotton cellulose: a modeling and simulation study. Appl Biochem Biotechnol 187(3):800–816. https://doi.org/10.1007/s12010-018-2856-6
Gatt E, Khatri V, Bley J, Barnabé S, Vandenbossche V, Beauregard M (2019) Enzymatic hydrolysis of corn crop residues with high solid loadings: new insights into the impact of bioextrusion on biomass deconstruction using carbohydrate-binding modules. Biores Technol 282:398–406. https://doi.org/10.1016/j.biortech.2019.03.045
DeMartini JD, Foston M, Meng X, Jung S, Kumar R, Ragauskas AJ, Wyman CE (2015) How chip size impacts steam pretreatment effectiveness for biological conversion of poplar wood into fermentable sugars. Biotechnol Biofuels 8(1):209. https://doi.org/10.1186/s13068-015-0373-1
Chundawat SPS, Venkatesh B, Dale BE (2007) Effect of particle size based separation of milled corn stover on AFEX pretreatment and enzymatic digestibility. Biotechnol Bioeng 96(2):219–231. https://doi.org/10.1002/bit.21132
Liu Z-H, Qin L, Pang F, Jin M-J, Li B-Z, Kang Y, Dale BE, Yuan Y-J (2013) Effects of biomass particle size on steam explosion pretreatment performance for improving the enzyme digestibility of corn stover. Ind Crops Prod 44:176–184. https://doi.org/10.1016/j.indcrop.2012.11.009
Adapa P, Tabil L, Schoenau G (2011) Grinding performance and physical properties of non-treated and steam exploded barley, canola, oat and wheat straw. Biomass Bioenerg 35(1):549–561. https://doi.org/10.1016/j.biombioe.2010.10.004
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hoppert, L., Einfalt, D. Impact of particle size reduction on high gravity enzymatic hydrolysis of steam-exploded wheat straw. SN Appl. Sci. 3, 878 (2021). https://doi.org/10.1007/s42452-021-04870-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42452-021-04870-4