Abstract
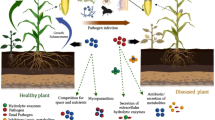
Plants grow associated with complex communities of organisms, and their development depends largely on management practices, available nutrients in the soil, environmental conditions, and the balanced equilibrium established among all these factors. In unbalanced environments, diseases, insects, and weeds can decrease agricultural productivity or even destroy crops. There is great interest in developing effective and complementary strategies and approaches to include in the integrated management of phytopathogens and pests, such as the application of formulations containing microorganisms with biological control capabilities. In Uruguay, several groups have been studying microbes that can be used as biological control agents, and there has been a clear interest in the implementation of biocontrol strategies. In this regard, Uruguay has taken several steps towards more sustainable agricultural practices, such as the implementation of a registration procedure for biopesticides and biofertilizers, and the creation of the National Plan for Fostering Agroecological Production. A positive outcome of these actions is the increase from only 2 to 13 registered products in the last 10 years. Registered formulations are based on bacteria, fungi, and viruses, including antagonists of plant diseases and entomopathogens for the control of insect pests. More than half of the registered products contain organisms isolated in Uruguay. Additionally, Uruguay occupies the eighth position in the world ranking of land destined for organic production. This review presents a summary of the history and outcomes of biological control research in Uruguay on beneficial microorganisms (bacteria and fungi) able to antagonise and control phytopathogenic fungi, oomycetes, and insect pests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Microbial biological control agents and sustainable agriculture
Throughout their life cycle, plants interact with a wide variety of macro- and micro-organisms, and plant health depends significantly on the harmonious equilibrium established by those interactions. In agricultural crops, unbalanced environments, diseases, insects, and weeds can decrease productivity or even cause severe damage to plants. The control of plant diseases has depended mainly on the extensive use of chemically synthesised pesticides. Many of these chemicals have been shown to be toxic to non-targeted micro and macro-organisms, including humans (Spurgeon et al. 2020). Therefore, there is a great interest in finding environmentally friendly ways to prevent, mitigate or control plant diseases. The most promising approaches include the use of biological agents able to suppress plant diseases, either by boosting the plant immune system or by directly inhibiting the pathogen.
The term “biological control” and its abbreviated synonym “biocontrol” were defined in 1987 by the National Academy of Sciences of the United States as “the use of natural or modified organisms, genes or their products to reduce the effects of undesirable organisms and favour the development of desirable organisms. Organisms with the ability to control the development of pathogens belong to the group of Biological Control Agents (BCAs) (Legein et al. 2020). The use of BCAs may have several advantages over chemically synthesised pesticides: (1) usually, they cause less environmental and human health damage; (2) they can multiply at the site of action exerting their effect directly on the colonised tissue and their growth is further controlled by local microflora; (3) and the risk of emergence of resistant pathogens is low because of the mechanisms of action involved and the metabolites produced by BCAs. The mechanisms involved in the biocontrol of phytopathogens include parasitism and predation, induction of host-plant resistance, secretion of antibiotics, generation of organic and inorganic volatile compounds, and production of lytic enzymes (such as chitinases, cellulases, glucanases, proteases and lipases) that can disrupt the cell wall of many phytopathogenic fungi/oomycetes (Narayanasamy 2013). Successful BCAs generally express multiple biological traits that act additively and synergistically to efficiently suppress the pathogen (Tilocca et al. 2020). BCAs are ubiquitous constituents of soils and plant microbiota. They can be found on the surface or inside roots, stems, leaves and flowers of diverse plant genera, and in legume nodules (Eljounaidi et al. 2016; Hong and Park 2016; Martínez-Hidalgo and Hirsch 2017).
The utilisation of biological control strategies has been acknowledged for the capacity to promote the conservation and enrichment of biodiversity (Hulot and Hiller 2021). The reduction in the reliance on chemical pesticides has been shown to have positive impacts on biodiversity. By mitigating the risks associated with non-target effects on beneficial organisms and minimizing the presence of toxic residues in the environment, biological control methods create favourable conditions for microorganisms, plants, and animals to thrive in their natural habitats. This underscores the importance of implementing biological control strategies as a means to support biodiversity preservation and ecosystem health. (Bajsa et al., 2013; Hulot and Hiller 2021). Additionally, the use of biocontrol alternatives lowers negative impacts on human and animal health. Chemical pesticides pose a multitude of health issues including hazards to exposed workers in the field and to consumers due to residues in food products (Junaid et al. 2013; Baker et al. 2020; Hulot and Hiller 2021). In 2001, the Uruguayan government created the Uruguay Natural brand with the aim of promoting and improving the image of the country. In this framework, “natural” goes hand in hand with sustainability and is a positive value per se (https://marcapaisuruguay.gub.uy/marca-pais-uruguay/). This brand identifies national products worldwide and certifies the quality of products obtained with sustainable practices. More than a thousand companies and institutes, both public and private, belonging to different sectors such as tourism, services, agriculture, and research, as well as a wide variety of products in Uruguay, have joined this brand and obtained benefits from its use (https://marcapaisuruguay.gub.uy/socios/).
Regulatory issues for registering microbial biological control agents in Uruguay
A further step in Uruguay towards the promotion of sustainable agriculture was the governmental declaration in 2007 of its interest in the use of microbial biological control agents (MBCA) in agriculture. Subsequently, in 2013, the regulatory authorities approved a registration procedure for MBCAs. This aims to ensure and guarantee the agronomic efficacy of MBCAs while being safe to humans, animals, and plants, and having minimal environmental impact. Briefly, the procedure for registering MBCAs involves the elaboration of a dossier with the following information: MBCA characteristics (molecular identification, biology, mechanisms of action, effects on target organisms), characteristics and composition of the formulation that contains the MBCA, toxicological and ecotoxicological information, MBCA agronomic efficacy and safety, as well as proposed containers, packaging, and proper labelling.
Recently, in December 2018, Law No. 19.717 was passed in Uruguay, stating an interest in the creation of a “National Honorary Commission” and the “National Plan for Fostering Agroecological Production” (MGAP, 2021). This was a significant step towards a continuous governmental commitment towards natural resources, environmental sustainability, and consumer health. This commitment is also mirrored in the position that Uruguay occupies in the world ranking of organic production. Uruguay destines 15.3% of its agricultural production to organic production (2.1 million ha), which places the country 8th in the world ranking of organic producers (Helga and Lernoud 2019).
Strong evidence of the beneficial impacts of the adoption of more sustainable practices in Uruguay is reflected in the reduction of imported pesticides for agricultural uses (Table 1). In 2021, the cost of pesticides imported into Uruguay represented USD 110.4 million less than in 2014 (DIEA – MGAP, 2022). This significant decrease is split among the different types of pesticides: herbicides (27%), fungicides (45.8%), insecticides (78%), and others (81.1%). Since the surface for agricultural use has been relatively stable in Uruguay and the prices of agrochemicals have increased in recent years, the cost reduction in imports is likely to be the result of two interconnected reasons. One of them is the reduced incidence of pests and diseases combined with the availability of alternative products to chemical pesticides. Under an Integrated Pest Management, there is still the use of agrochemicals in conjunction with biological products. Agroecological production, which is growing in Uruguay, also implies the incorporation of microbial inoculants and the lack of use of chemical pesticides. These managements result in reduced amounts of applied pesticides, so fewer quantities of chemical pesticides are required (Table 1). The decrease in the use of chemical pesticides combined with the adoption of biocontrol strategies is also observed worldwide. The global market for microbial pesticides was valued at USD 1.94 billion in 2018 and is forecasted to reach a value of USD 4.75 billion in 2024 (Modor Intelligence, 2019). Among the continents, South America is the fastest-growing market demanding biopesticides, Argentina and Brazil being the leading countries in the region. On the other hand, North America represents approximately 43% of the global market for microbial pesticides. The raising demand in the region is driven by several factors, including the increased interest in sustainable agricultural practices and the restrictions imposed on many conventional chemical products or the loss of performance issues due to emerging resistant pests.
Research on microbial biological control agents in Uruguay
In Uruguay, research programs in biocontrol date back more than 25 years ago, ranging from the isolation and selection of microorganisms with antagonistic activity against fungi, oomycetes, or insects to the analysis of the possible mechanisms of action involved in biological control, the evaluation of different formulations, and the assessment of plants in the field. The steady increase of registered biopesticides in Uruguay in the last ten years partially reflects the interest in the development of new strategies for biological control. While in 2012 there were only two commercial biopesticides, in 2023 there are 13 registered ones, and another 20 products are being currently evaluated for registration (Table 2). They comprise viruses (Granulovirus), bacteria (Bacillus spp.), fungi (Bauveria bassiana, Isaria javanica, Lecanicillum lecanii Metarhizium spp. and Trichoderma spp.), mites (Amblyseius swirskii), and beneficial insects (Aphidoletes aphidimyza, Crysoperla externa and Orius insidiosus).
Current key market constraints for biopesticides are the same as those for biofertilizers: low product efficacy under unfavourable conditions and the low shelf life of microorganisms (Modor Intelligence, 2019; O’Callaghan et al., 2022). The potential opportunities for the development of biopesticides need to reinforce better microbial characterisation and product formulation in order to overcome the limitations and constraints associated with the use of microbes under variable and challenging field conditions.
In this review, we will present a summary of the results obtained in Uruguay on microorganisms (bacteria and fungi) able to antagonise phytopathogenic fungi/oomycetes and on entomopathogenic fungi able to control insect pests, with their potential use in agriculture.
Microbial antagonists of phytopathogens
Bacillus, Burkholderia, Paenibacillus, Pseudomonas and Streptomyces
Fusarium circinatum, known as the pitch canker fungus, was identified as a phytopathogen of Pinus taeda seedling in Uruguay (Alonso and Bettucci 2009). The work performed by Soria et al. (2012) showed that four Bacillus sp. strains and one Burkholderia sp. strain, isolated from healthy tissues of P. taeda, presented in vitro antifungal activity against strains of F. circinatum (isolated from symptomatic P. taeda seedlings). Moreover, the authors observed that thermostable metabolites produced by these bacterial strains were able to reduce over 50% of mycelial growth. Overall, results suggest that these bacterial isolates are candidates to be tested in planta for their ability to protect pines from the pitch canker fungus (Soria et al. 2012).
Other Bacillus sp. strains, isolated from wheat seeds, also showed the ability to antagonise pathogenic fungi (Pan et al. 2015). In that study, researchers found that the four Bacillus spp. strains analysed were able to significantly reduce fungal growth and spore germination of a Fusarium graminearum (previously also known as Gibberella zeae) strain that infects wheat plants producing Fusarium head blight. Losses caused by this disease can reach up to 30% under severe epidemic conditions. An additional concern is the production of mycotoxins that are potentially toxic for humans. Interestingly, the inoculation of wheat with the bacterial isolates reduced the disease incidence and severity of Fusarium head blight in a field assay and caused a reduction in mycotoxin production (Pan et al. 2015).
In Uruguay, Botrytis cinerea has proven to be responsible for the “gray mold” in vineyards, causing significant losses in wine production, interfering with the fermentation process of grapes, and affecting the colour and flavour of the wine obtained. Mondino et al. (2012) demonstrated in field trials that Zimevit® a commercial product composed of a strain of Bacillus subtilis and the yeast Metschnikowia pulcherrima, had the capability to control the “gray mold” caused by B. cinerea in grapes from the Cabernet Franc and Gewurztraminer varieties.
Recently, a bacterial strain isolated from an Arachis villosa root nodule was shown to have in vitro antagonistic effect toward a wide spectrum of phytopathogenic fungi and oomycetes (Costa et al. 2022). The strain was further identified as a new species belonging to the Paenibacillus genus and was named Paenibacillus farraposensis (Roldán et al. 2022). The analysis of some traits putatively involved in biological control demonstrated that the strain produces diverse diffusible and volatile compounds putatively involved in antibiosis (Fig. 1a). Further investigation should be done to evaluate its field performance as a BCA.
Dual plates assay. (a) Antagonistic activity of Paenibacillus farraposensis UY79 (bottom) against Fusarium graminearum Schwabe S127 (top). (b) Trichoderma atroviride T3 (bottom) against Fusarium nygamai Burgess & Trimboli (top). P. farraposensis strain UY79 was isolated from a root-nodule of Arachis villosa (wild peanut); T. atroviride T3 was isolated from a soil sample of a sorghum crop in Uruguay. F. graminearum S127 and F. nygamai are phytopathogenic fungi isolated from infected sorghum grains. Dual plate assay was performed on PDA medium. Photos: (a) Andrés Costa, (b): Belén Corallo
Fluorescent Pseudomonas are traditionally regarded as promising candidates for the development of biopesticides and biofertilizers. These bacteria are well known for their capability to produce secondary metabolites such as antibiotics, induction of systemic resistance in the host plant, and/or competition with phytopathogens for niches and nutrients. In addition, they are suitable for developing market inoculants because they can use a variety of carbon sources, have a high growth rate, and can be introduced into the rhizosphere by seed bacterization. These bacteria have received much attention as BCAs for the suppression of root pathogenic microorganisms, which improves plant growth and crop yields (Weller et al. 2002; Whipps 2001; Yanes and Bajsa 2016).
In Uruguay, forage legumes play an important role in agricultural production as the main high-quality feed source for livestock. Their ability to establish a symbiotic association with nitrogen-fixing rhizobia contributes to the sustainability of crop rotations with pastures (Rebuffo et al. 2006). However, considerable losses in the establishment of legumes are due to soil pathogens, mainly fungi and oomycetes, responsible for low persistence and reduced crop performance. Pre- and post-emergence damping-off is one of the major diseases caused by members of the genera Pythium, Rhizoctonia and Fusarium genera (Altier and Thies 1995). Generally, this disease cannot be prevented by crop rotations or the development of resistant plant varieties, so biological control represents a promising alternative to chemical control. The selection of native fluorescent Pseudomonas strains for the control of forage legume seedling diseases took several stages: first, the isolation, selection, and characterisation of the strains, and then the formulation of an inoculant with the objective of improving the establishment of pastures using a non-aggressive technology for the environment (Bajsa et al. 2005; Yanes and Bajsa 2016). Some native strains were characterised in terms of antagonistic mechanisms, colonisation, and field performance. For instance, Pseudomonas protegens strain UP61, isolated from the rhizosphere of Lotus corniculatus, produced hydrogen cyanide, 2,4-diacetylphloroglucinol, pyrrolnitrin and pyoluteorin, was able to densely colonise the roots of L. corniculatus and alfalfa (Medicago sativa), and was capable of controlling damping-off caused by Pythium spp. on both forage legumes (de la Fuente et al. 2004; Quagliotto et al. 2009).
Among the strains isolated from the alfalfa rhizosphere, P. fluorescens C119, which produces a cyclic lipopeptide with antimicrobial activity, notably promoted the growth of alfalfa. Alfalfa plants that were co-inoculated with the strains P. fluorescens C119 and Sinorhizobium meliloti using a peat-based formulation showed a significant increase in plant yield compared to non-inoculated plants (Yanes et al. 2004, 2012). In addition, in vitro and in-field experiments carried out in lotus and alfalfa plants showed that plants co-inoculated with both bacterial strains were healthier than plants inoculated with only one strain (de Fuente et al. 2002; Quagliotto et al. 2009). Sterile peat is a carrier that has been extensively used for rhizobial formulations (de Fuente et al. 2002; Quagliotto et al. 2009) because it allows bacterial survival during the storage and commercialisation of the final product. It was demonstrated that this carrier is also suitable for fluorescent Pseudomonas, maintaining constant populations of E10 bacteria per gram of peat at 4 °C for 6 months (Bagnasco et al. 1998).
However, compared to the volume of research that has been carried out with these bacteria, few strains have been successfully developed into commercial products for plant biocontrol and biostimulation. Although suitable formulations for bacterial delivery have been developed, some drawbacks in field applications still limit their use. Some variations in field performance have been observed, mainly due to changes in climatic conditions. Moreover, there are some reports of the opportunistic human pathogen Pseudomonas sp. Therefore, the requirements for the registration of new products must consider the use of formulations that provide stability of the MBC in different climatic situations (e.g., dry or excessively humid) while also including strict biosafety control (Höfte and Altier 2010; Yanes and Bajsa 2016).
Another strategy that requires further investigation is the optimisation of management practices to favour the development of antagonistic populations already present in the soil. In that sense, disease-suppressive soils are a natural source of BCAs. Supporting this strategy, Vaz et al. (2018) demonstrated that longer rotations harboured a higher amount of Streptomyces spp. and an increased number of plant-pathogen antagonists than shorter rotations.
Bacteria belonging to the Bacillus, Burkholderia, Paenibacillus, Pseudomonas and Streptomyces genera are commonly present in soils and frequently associated with different parts of plants. Interestingly, it has been observed that some strains can modulate the plant microbiota either by controlling or promoting the presence of certain microbial species, establishing a complex network of interaction and response between the endophytic microbiota, the plant, and the soil (Han et al. 2020). Additionally, as presented in this review, diverse strains have been shown to be effective BCAs. Results of research carried out in Uruguay indicate that these strains are an excellent starting point to assess their use for the development of new biofungicides or as BCAs.
Clonostachys and Trichoderma
Fungi are also well known for their biological control capability against phytopathogens. Mycoparasitism, antibiosis, competition for nutrients and space, and induction of plant defence systems are some of the mechanisms involved in their biological control activity (Latz et al., 2018). In Uruguay, two genera of fungi have been investigated for their potential as BCAs: Clonostachys and Trichoderma.
In Uruguay, onion is produced mainly by family farmers. It has been observed that, during the nursery stage, the disease caused by Botrytis squamosa severely limits its production. In order to evaluate different control strategies to reduce the disease, Silvera-Pérez et al. (1997) performed a study searching for microbial strains able to antagonise the fungal growth. The strategy used in that case was the isolation of antagonists from visibly parasitised B. squamosa sclerotia. The authors selected a fungal strain of Clonostachys rosea (also known as Gliocladium rosea) (Silvera-Pérez et al. 1997), as the best isolate with antagonistic ability against B. squamosa. After the evaluation of different substrates for inoculum production (wheat grains, middling, whole corn grains, ground corn grains), results indicated that the highest yield in C. rosea conidia production was obtained with wheat grains previously sterilised in an autoclave. With the application of this formulation, the authors observed in a field assay a reduced incidence of onion disease in comparison to the untreated control (Silvera-Pérez et al. 1997). Currently, a formulation containing this strain is being analysed for its registration for commercial purposes.
Different groups have worked in Uruguay in the search and selection of Trichoderma strains with biological control activity (Corallo et al. 2021a, b; Pereyra et al. 2005). Although there are different species of Trichoderma with biological control activity, most of the strains studied belong to the species T. asperellum, T. harzianum and T. viride (Harman 2006). It is worth mentioning that the first biological control product for agriculture registered in Uruguay was Trichosoil®, a fungicide whose active ingredient is a strain of Trichoderma harzianum of national origin (Table 1). Trichosoil® was registered in 2009 by Lage and Cia, and was a result of research aimed at reducing the disease caused by Sclerotium rolfsii in garlic crops. As a result of that work, a collection of isolates with demonstrated antagonistic activity against S. rolfsii was achieved, and an isolate of T. harzianum with good characteristics as an antagonist for industrial production was selected from the collection. According to an assay carried out with plants grown in nurseries, the selected T. harzianum isolate not only controlled S. rolfsii, but was also effective against other soil pathogens such as Sclerotinia, Rhizoctonia, Pythium, Fusarium, and some foliar pathogens such as Botrytis (Mondino et al. 2014). Another bioproduct based on Trichoderma asperellum obtained from national research was tested in commercial field conditions and later developed to provide ecological tools for the management of diseases in intensive crops such as Botrytis, Phytium, Peronospora and Sclerotinia (Rodríguez et al. 2022).
For the purpose of controlling Fusarium head blight in wheat, Pereyra et al. (2005) and Cabrera et al. (2020) analysed the effect of different Trichoderma sp. strains. When the commercial strains of T. harzianum KRL-AG2 from the USA (T-22®) and T. harzianum L1 (Trichosoil®) were assayed, significantly lower production of perithecia per mm2 of straw was observed three months after applying these strains (Pereyra et al. 2005). Sixteen strains of Trichoderma sp. with antagonistic activity against different strains of F. graminearum were further isolated from wheat and barley stubble (Pereyra et al. 2005). The strains were identified as Trichoderma koningiopsis, T. harzianum, Trichoderma atroviride and Trichoderma longibrachiatum (Cabrera et al. 2020). To estimate the potential of these fungi as BCAs, the strains were characterised by their ability to produce different enzymes such as cellulases, chitinases and b-1,3-glucanases, as well as their ability to inhibit the growth of the pathogen under in vitro conditions. Four strains of Trichoderma koningiopsis and one of T. atroviride, were further selected and evaluated in vitro for their ability to inhibit the production of F. graminearum perithecia on wheat straw and it was observed that the five strains were able to significantly reduce the production of these fungal structures (Cabrera et al. 2020).
Fusarium nygamai is a pathogen that causes significant losses in sorghum (Sorghum bicolor) crops in Uruguay. This pathogen also produces various contaminating mycotoxins in sorghum grains, such as fumonicin, moniliformin, and beauvericin (Corallo et al. 2021a, b). Aiming to obtain Trichoderma spp. strains able to control this phytopathogen, different fungi were isolated from soils of sorghum commercial plantations (Corallo et al. 2021a, b). The strains were identified as Trichoderma asperellum (two strains), Trichoderma atroviride (five strains), Trichoderma virens (two strains), Trichoderma longibrachiatum (one strain) and Trichoderma sp. (two strains). Two strains with the highest antagonistic activity against F. nygamai (T. asperellum T6 and T. atroviride T21) were further selected to evaluate their ability to protect sorghum plants from the disease (Fig. 1b). Results showed that, while all sorghum seedlings inoculated with F. nygamai evidenced some symptoms of disease, 50% of seedlings inoculated with F. nygamai and treated with T. asperellum T6 were healthy. Additionally, this beneficial fungal strain was also recovered from the internal tissues of roots, stems, and leaves (Corallo et al. 2021a, b). Altogether, these results suggest that T. asperellum strain T6 is a promising candidate to be used to control the disease caused by F. nygamai in sorghum crops.
Biological control of insect pests and diseases with entomopathogenic fungi in Uruguay
Conventional use of entomopathogenic fungi against insect pests
Among the microorganisms used as biocontrol agents, entomopathogenic fungi (EF) have a considerable role in decreasing insect populations (Fig. 2). Beauveria bassiana and Metarhizium anisopliae are the most used EF around the world for the control of insects, and several biopesticides have been developed from them (Brunner-Mendoza et al. 2019; Glare et al. 2012; Leng et al. 2011). Additionally, conidia from these fungal genera are easily mass-produced using solid-state fermentation on grains and formulated as dry or wettable powder for aerial applications for insects dwelling on the plant phyllosphere (Jaronski and Mascarin 2017). In Uruguay, research in this field has focused on tackling insect pests affecting crops where the use of pesticides compromises either the economy, the environment, or the health of humans and animals.
Entomopathogenic fungi. (a) Trialeurodes vaporariorum nymph (white fly) showing hyphae from Lecanicillium lecanii after an application of a formulated conidial suspension on tomato plants. (b) Diloboderus abderus larva (grass grub) naturally infected with Metarhizium spp. found in grassland fields. Photos: F. Rivas-Franco. Grass grub collected by Ximena Cibils
Some of the leaf-cutting ants found in Uruguay are Acromyrmex crassispinus, Acromyrmex heyeri and Acromyrmex lundi, which cause severe defoliation in young plants, representing an economically important pest for agriculture and forestry (Corallo et al. 2017). Leaf-cutting ants (Acromyrmex spp.) were efficiently controlled with M. anisopliae conidia formulations under field conditions after weekly applications for 3 to 4 weeks. The effectiveness of using the strain of M. anisopliae to induce ant mortality and reduce ant nest activity could offer a viable alternative for reducing reliance on pesticides or even replacing them. M. anisopliae, the entomopathogenic fungus, has shown promise as a potential biopesticide for managing ants, with the potential to decrease the use of chemical pesticides or provide an alternative approach altogether (Tiscornia et al. 2014). In this regard, other field studies considered an Integrated Pest Management to reduce ant populations using a combined application of pesticides and EF (Corallo et al. 2017). The main results showed that M. anisopliae combined with fipronil or sulfuramid can inactivate all A. heyeri nests one week before than when used with B. bassiana. Rodríguez et al. (2022), in another field experiment aimed at contributing to local agroecological practices, studied beneficial soil organisms to obtain native fungi for the control of cutting ants (genera Atta spp. and Acromyrmex spp.). The outcome of this research was the registration of a new biopesticide to control ants, named Crebio 1 (Table 3).
Another opportunistic pest is Thaumastocoris peregrinus Carpintero & Dellapé (Heteroptera: Thaumastocoridae), a sap-sucking insect that feeds on leaves of Eucalyptus with a significant impact on the plant’s growth and health. Detected in Uruguay in 2008, it was causing considerable economic losses in Eucalyptus plantations (Corallo et al. 2019). Currently, there is no efficient control of this insect and specific restrictions are imposed on the use of chemicals that can leave residues. The search for promissory fungal isolates was a priority, and promising EF were obtained belonging to several genera, including Beauveria sp., Fusarium sp., Isaria sp., Lecanicillium sp., Paecilomyces sp., Pochonia sp., Purpureocillium sp. and Simplicillium sp. Among the fungal isolates, B. bassiana (FI2403) showed the highest virulence (Corallo et al. 2019).
When developing a biopesticide, the selected strains should not only be able to effectively kill the highest number of target insects in the shortest time interval but also resist environmental conditions that can be detrimental to fungal development (Corallo et al. 2019; Rivas-Franco et al. 2019). Particularly, fungi are affected by UV radiation, high temperatures and low humidity. Several studies have focused on the selection of fungal strains that can tolerate and survive in low-water activities and high temperatures (Corallo et al. 2019; Rivas et al. 2014). Tomato plants in Uruguay are generally produced in greenhouses where temperatures can be extremely high. The whitefly Trialeurodes vaporariorum (Westwood) (Hemiptera: Aleyrodidae) is an important pest of protected horticultural crops worldwide, and tomatoes are significantly affected by it (Rivas et al. 2014). The entomopathogenic fungus Lecanicillium spp. is a natural pathogen for whiteflies, developed in many countries as a biopesticide for crops vulnerable to this insect pest. The development of a biopesticide based on Lecanicillium against T. vaporariorum on tomatoes considered the selection of fungal isolates tolerant to low water activities and high-temperature conditions (Fig. 2a). This research allowed the characterisation of Lecanicillium spp. isolates tolerant to temperatures above 28 °C and low humid conditions, where fungal growth, conidiation and conidia germination were unaffected (Rivas et al. 2014). Research carried out in conjunction with an agricultural cooperative to reduce the incidence of whiteflies Trialeurodes vaporariorum and Bemisia tabacci led to the development of a bioinsecticide with Isaria javanica as a microbial antagonist (Crebio 5, Table 3), which is already being applied in the ecological management of horticultural crops (Rodríguez et al. 2022).
Another insect pest responsible for damaging trees’ health and fruit quality is the Asian citrus psyllid Diaphorina citri Kuwayama (Hemiptera: Liviidae). This insect is a vector of the bacteria Candidatus Liberibacter spp., a putative causal agent of citrus greening disease known as “Huanglongbing”, considered one of the most destructive diseases of citrus orchards worldwide. Corallo et al. (2021a, b) determined in laboratory studies as well as in semi-field conditions that satisfactory levels of pathogenicity on this psyllid were achieved with strains belonging to M. anisopliae 2411 and B. bassiana 2067. Under controlled conditions, the percentage of insect mortality produced by the evaluated fungi varied between 82.8% and 85.9%, while under semi-field conditions, the infection and mortality rates of D. citri caused by M. anisopliae 2411 and B. bassiana 2067 were 78.9% and 51%, respectively. While the mortality rates achieved with the strains are promising, additional studies on mass production, formulation, and shelf-life survival are necessary to further develop these strains into effective biopesticides. In Uruguay, Thaumastocoris peregrinus and Piezodorus guildinii are important hemipteran pests in eucalyptus and soybean plantations, respectively. Initial specific strain characterisation studies on potential fungal biocontrol agents were performed for their development into biopesticides against these insects. In natural conditions, only P. guildinii is extremely resistant to fungal epizootics. Insect cuticular hydrocarbons (CHC) are known to be important determinants of the resistance of these insects to EF (Sessa et al. 2022). Five Uruguayan B. bassiana strains were phenotypically and genetically characterised regarding their response to CHC (n-pentadecane) as the sole carbon source, as well as their pathogenicity and virulence against hemipteran insects (Sessa et al. 2022). The results showed that virulence towards P. guildinii was enhanced in some B. bassiana strains after growth on CHC, whereas virulence enhancement towards T. peregrinus was only observed in one of the hypervirulent strains. The authors conclude that the enhancement of virulence could be attributed to a priming effect in the strains induced by the n-pentadecane, which may upregulate genes related to hydrocarbon assimilation, such as cytochrome P450 genes, as well as genes associated with adhesion and stress tolerance.
Seed coatings with endophytic entomopathogenic fungi for plant protection against soil-dwelling phytopathogens and insect pests
Recently, new insights into the ecology of EF have envisaged new innovative uses for these fungi in addition to their traditional application as biopesticides. Several studies reported that EF not only have the ability to antagonise other fungal phytopathogens, but they can also be found growing on the surface of plant tissues or even as endophytes (Jaber and Ownley 2017; Rivas-Franco et al. 2020b; Vega 2018). The ability of EF to grow associated with the rhizosphere or colonizing roots represents an opportunity to control root-feeding insects and phytopathogens that inhabit the soil (Keyser et al. 2014). Insect larvae and phytopathogens dwelling in the soil are difficult to reach once the crop is installed, and when their presence is noticed, plants are often considerably damaged. Additional advantages of this colonisation capability are that the fungus persists in the root system as a bodyguard, making the re-application of the biocontrol agent unnecessary, and that root colonisation triggers plant-induced resistance (IR) as other biotrophs do (Liao et al. 2014; Rivas-Franco et al. 2020b).
Traditionally, soil-dwelling insects are controlled with EF formulated as granules applied with the seeds when these are sown or just applying the granules onto the soil if the crop is already installed (Jackson and Jaronski 2009). Since the discovery that species belonging to the genus Metarhizum are able to associate with roots, a new application strategy through seed coating has been conceived (Kabaluk and Ericsson 2007; Rivas-Franco et al. 2019, 2020a, b). Growth chamber studies performed with maize-rhizosphere-competent isolates of Metarhizium spp. coated to seeds showed that the used fungi were able to associate with maize root plants colonizing the rhizosphere and also growing as endophytes. This root colonisation reduced Fusarium root rot symptoms by 40% and infected larvae of Costelytra giveni by 60% (Rivas-Franco et al. 2019). However, the positive effects on plant growth promotion were only detected in the presence of the insect pest or the phytopathogen fungus, but not in their absence (Kabaluk and Ericsson 2007; Rivas-Franco et al. 2019). The reason behind this observation could be that the plant-fungus interaction demands photosynthates in addition to triggering the IR in the plant, both facts demanding additional energy and consequently reducing plant growth. On the other hand, in the presence of the biotic challengers (the pest or the phytopathogen), growth is also affected, but the presence of biocontrol EF overcomes those effects and plant growth is improved (Partida-Martínez & Heil 2011; Rivas-Franco et al. 2019). Other studies have shown that EF can also work as biofertilizers because fungi associated with roots are able to transfer nitrogen from infected insects, improving plant nutrition and therefore promoting plant growth (Behie et al. 2012).
Another interesting capability of EF, when cultured under liquid conditions, is the production of resting structures known as microsclerotia. The presence of microsclerotia was first reported in M. anisopliae by Jackson and Jaronski (2009), which represented a milestone in fungal formulations. These resistant structures can tolerate detrimental conditions and survive storage for long periods. Once microsclerotia are returned to appropriate conditions for fungal growth, like the ones found in the soil, microsclerotia germinate, producing hyphae and then high quantities of conidia, which in turn are able to infect insects or associate with plant roots (Jackson and Jaronski 2009; Rivas-Franco et al. 2020a). Several works are now focused on the formulation of EF as microsclerotia contained in diatomaceous earth granules or coated to seeds for its use in plant protection (Lira et al. 2020; Rivas-Franco et al. 2020a).
The existence in Uruguay of large agricultural areas destined for cattle grazing (natural pastures and forages) or the production of grains demands chemical seed treatments to control soil-dwelling insects and phytopathogens causing damping-off. Previous studies of seed coating with EF (Rivas-Franco et al. 2020 and 2020a) were financed by Uruguay’s National Agricultural Research Institute (INIA), seeking alternatives to chemical seed treatments, with the collaboration of New Zealand’s Bio-protection Research Centre and AgResearch institutes. Nowadays, this research line about the application of BCAs to seeds focuses on the characterisation of national isolates of Metarhizium spp., their compatibility with species of rhizobia, and their formulation as seed coatings with natural biopolymers (INIA, 2022). EF are envisaged with wider potential for agricultural use because of their capabilities for plant nutrition and plant protection against insect pests and phytopathogens. The new research lines in Uruguay are aimed at the development of EF as multifunctional bio-inputs for agriculture that can help plant nutrition, control insect pests, and reduce damping off when incorporated in seed coatings for plant protection.
Future prospects
Research investment for improved microbial characterisations, formulations, bioproduction standardisation procedures and field efficacy has shown the benefits obtained from plant protection and plant nutrition from microbes like bacteria and fungi. The positive results obtained in biocontrol efficacy have boosted the availability of BCAs products. Over the last ten years, the range of products and agents available to Uruguayan farmers for biocontrol has expanded significantly. This trend has also been observed in other areas of the world such as Europe, where the market for BCAs is growing and expected to expand by 15% per year for the next decade (Hulot and Hiller 2021). In only 10 years, the conditions set in Uruguay led to an 87% increase in the commercially available biocontrol products, from only 2 to 13, and biocontrol products currently in the process of registration increased by 95% (DIEA, 2022). Among the different categories of biocontrol, those related to microorganisms are the most dynamic segment. Globally, microbial products already occupy two-thirds of the market, and most of the research projects focus on microorganisms (Hulot and Hiller 2021).
In Uruguay, the areas that need a reduction in the use of chemicals are those destined for the production of crops (soy, wheat, barley, and rice), pastures and forages, vegetables and fruits. Crops and pastures occupy an extended surface in Uruguay, and the application of chemicals has a deep impact on the environment. Furthermore, the consumption of fresh fruits and vegetables can pose a risk to consumers’ health due to the presence of chemical residues. In addition, the excessive use of synthetic pesticides in higher quantities than necessary to enhance crop performance is a growing concern, resulting in increased levels of pesticide residues (Scarlato et al., 2022). This has led to a global emphasis on implementing Maximum Residue Limits for most chemical pesticides in agrifood derivatives (Helga and Lernoud 2019). In contrast, microbial pesticides typically have lower residue levels and can be a viable strategy for sustainable agriculture, promoting the health of workers and consumers.
The impact of the indiscriminate use of pesticides on the environment is clearly evidenced in beehive losses, affecting the natural pollination of crops and honey production, and finally causing the rejection of honey consignment because of the presence of pesticide residues (Castelli et al. 2021). The increasing concern from the One Health point of view and the demand of consumers for agricultural products obtained without chemicals requires the use of alternatives to the chemical control of insect pests and microbial diseases. Additionally, in recent years, the detection of insect pests resistant to some transgenic soy and maize crops has promoted the search for alternatives, where biological control represents a potential solution.
Soil-dwelling insect pests are notoriously difficult to control, often requiring the application of persistent synthetic pesticides. With the ongoing de-registration of these categories of pesticides, there is an increased need for BCAs to fulfil this function. To date, there has been very limited research in this area, with most biopesticides used as foliar-application products. Soil-borne fungal diseases causing damping-off and affecting the emergence of seedlings is another area where biocontrol offers a possible solution.
In this regard, the registration of all BCAs to be commercialised guarantees their quality and efficacy, but for small companies, the cost associated with the registration can be high. Among the costs are field trials, toxicological and molecular analyses, and professional advice. Additionally, some toxicological labtests and clinical analyses are done in Argentina and Brazil, elevating costs even further.
From the beginning, the BCAs registration procedure differed from the original procedure for chemical pesticides. Many of the analyses required for phytosanitary products do not apply to microbial BCAs. However, some analyses still require meeting agronomic efficacy values similar to those of chemical pesticides, which requires additional field studies that ultimately limit the availability of biopesticides. In an attempt to alleviate the cost, the government provided an incentive by eliminating the registration fees of approximately USD 1000.
The National Plan for Agroecology defines several axes of action, some of them promoting the use of biocontrol strategies, the conservation of microbial genetic resources, scientific research, human resources training and postgraduate studies in related disciplines (https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/comunicacion/publicaciones/plan-nacional-para-fomento-produccion-bases-agroecologicas/plan-nacional). The rise in the demand for organic products is booming, not only due to an increase in conscientious consumers, but also due to increasing incomes, along with improved farming practices that make organic yields more robust. As a result, the increasing worldwide demand for organic food has increased the area for organic farming. The Common Agricultural Policy limits and restricts the use of synthetic pesticides while promoting the use of biopesticides and organic farming. According to Helga and Lernoud (2019), countries with the most organic agricultural land in 2020 were Australia (35.7 million hectares), Argentina (4.5 million hectares), and Uruguay (2.7 million hectares). The main organic products of Uruguay are meat, honey, fruits and vegetables, cereals, oilseeds, aromatic and medicinal herbs, wines, milk, and preserves. Certified organic production in Uruguay involves around 748 farmers (Helga and Lernoud 2019).
A major constraint in Uruguay for the improvement of biopesticides is the scarcity of financial resources allocated to research, both in public and private institutes. Despite being considered a developing country, Uruguay still grapples with acute social inequalities, which have been further exacerbated by the recent pandemic. Additionally, Uruguay has one of the lowest percentages of GDP (Gross Domestic Product) assigned to research, representing only 0.48% in 2019. Among public research institutions in Uruguay, those advocating for research in Biocontrol are the Biological Research Institute Clemente Estable (IIBCE), the Pasteur Institute of Montevideo (IPM), the National Agricultural Research Institute (INIA), and the Faculties of Agriculture, Chemistry, Engineering, Sciences and Veterinary of the University of the Republic (UdelaR).
Conclusions
The emergence of insects and pathogens resistant to commonly used chemicals, the restrictions imposed by importing countries to certain agrochemicals and toxic compounds of microbial origin, and the growing concerns from a One Health point of view for the environment and people’s health have prompted us to look for alternatives to the chemical control of pests and diseases. Agriculture is Uruguay’s main economic activity, so biological control is a relevant strategy for reducing the incidence of pests and diseases with a negative impact on crop production. The steady increase of registered biopesticides in Uruguay in the last 10 years clearly reflects the advance of biocontrol and the development of sustainable agriculture.
Some additional factors that have influenced the higher availability of biopesticides are public awareness regarding the harmful effects of chemical plant protection products on the environment and human health, the increasing demand for organic food, the known advantages of BCAs over chemical pesticides, and the adoption of this technology by farmers already familiarised with microbial inoculants for nitrogen fixation. Promising antagonistic microorganisms have not yet been developed into commercial products, and further research on bioproduction, formulation and agronomic efficacy is being carried on.
Limitations for a broader biocontrol success are identified in terms of the farmers’ knowledge about the proper application, environmental conditions in the field, and restrictions in commercial or regulatory aspects. In terms of research, some of the aspects that would help consolidate biopesticide development are the survival of biocontrol agent in the formulation, its persistence in the environment, the control efficacy in different crops and environmental conditions, and the ecotoxicity for its safe release. Another valid alternative is to take advantage of the naturally occurring antagonist microbes in soils and promote management practices that increase its occurrence and activity, as a complementary approach to promote sustainable food production systems.
Acknowledgments.
Data Availability
Not applicable.
References
Alonso R, Bettucci L (2009) First report of the pitch canker fungus fusarium circinatum affecting Pinus taeda seedlings in Uruguay. Australasian Plant Disease Notes 4:91–92
Altier N, Thies JA (1995) Identification of resistance to Pythium seedling diseases in alfalfa using a culture plate method. Plant Dis 79(4):341–346
Bagnasco P, De La Fuente L, Gualtieri G, Noya F, Arias A (1998) Fluorescent Pseudomonas spp. As biocontrol agents against forage legume root pathogenic fungi. Soil Biol Biochem 30:1317–1322
Bajsa N, Quagliotto L, Yanes ML, Vaz P, Azziz G, de la Fuente L et al (2005) Selección de Pseudomonas fluorescentes nativas para controlar enfermedades de implantación en praderas. Agrociencia 9:321–325
Bajsa N, Morel MA, Brana V, Castro-Sowinski S (2013) The Effect of Agricultural Practices on Resident Soil Microbial Communities: Focus on Biocontrol and Biofertilization. Molecular Microbial Ecology of the Rhizosphere Vo. 2. Bruijn, F. (ed) https://doi.org/10.1002/9781118297674.ch65
Baker BP, Green TA, Loker AJ (2020) Biological control and integrated pest management in organic and conventional systems. Biol Control 140. https://doi.org/10.1016/j.biocontrol.2019.104095
Behie SW, Zelisko PM, Bidochka MJ (2012) Endophytic insect-parasitic fungi translocate nitrogen directly from insects to plants. Sci (New York NY) 336:1576–1577
Brunner-Mendoza C, Reyes-Montes MdR, Moonjely S, Bidochka MJ, Toriello C (2019) A review on the genus Metarhizium as an entomopathogenic microbial biocontrol agent with emphasis on its use and utility in Mexico. Biocontrol Sci Technol 29:83–102
Cabrera M, Garmendia G, Rufo C, Pereyra S, Vero S (2020) Trichoderma atroviride como controlador biológico de fusariosis de espiga de trigo mediante la reducción del inóculo primario en rastrojo. Terra Latinoam 38:629–651
Castelli L, Balbuena S, Branchiccela B, Zunino P, Liberti J, Engel P, Antúnez K (2021) Impact of chronic exposure to sublethal doses of glyphosate on Honeybee immunity, gut microbiota and infection by pathogens. Microorganisms 15(4):845. https://doi.org/10.3390/microorganisms9040845
Corallo B, Tiscornia S, Galvalisi U, Lupo S, Bettucci L (2017) Combined biological and chemical control of neotropical leaf-cutting ants (Acromyrmex spp.) under field conditions. Trends in Entomology 13:103–108
Corallo B, Simeto S, Martínez G, Gómez D, Abreo E, Altier N et al (2019) Entomopathogenic fungi naturally infecting the eucalypt bronze bug, Thaumastocoris peregrinus (heteroptera: Thaumastocoridae), in Uruguay. J Appl Entomol 143:542–555
Corallo AB, Pechi E, Bettucci L, Tiscornia S (2021a) Biological control of the asian citrus psyllid, Diaphorina citri kuwayama (hemiptera: Liviidae) by entomopathogenic fungi and their side effects on natural enemies. Egypt J Biol Pest Control 31:15
Corallo B, Bettucci L, Tiscornia S (2021b) Selection of Trichoderma strains for biological control of Fusarium nygamai in sorghum (sorghum bicolor L. Moench). Revista Colombiana de Investigaciones Agroindustriales 8:11–22
Costa A, Corallo B, Amarelle V, Stewart S, Pan D, Tiscornia S et al (2022) Paenibacillus sp. Strain uy79, isolated from a root nodule of Arachis villosa, displays a broad spectrum of antifungal activity. Appl Environ Microbiol 88:e01645–e01621
de la Fuente L, Quagliotto L, Bajsa N, Fabiano E, Altier N, Arias A (2002) Inoculation with Pseudomonas fluorescens biocontrol strains does not affect the symbiosis between rhizobia and forage legumes. Soil Biol Biochem 34:545–548
de la Fuente L, Thomashow L, Weller D, Bajsa N, Quagliotto L, Chernin L et al (2004) Pseudomonas fluorescens up61 isolated from birds foot trefoil rhizosphere produces multiple antibiotics and exerts a broad spectrum of biocontrol activity. Eur J Plant Pathol 110:671–681
DIEA – MGAP (2022) Anuario Estadístico Agropecuario. Dirección de Estadística Agropecuaria del Ministerio de Ganadería Agricultura y Pesca, in: https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/comunicacion/publicaciones/anuario-estadistico-agropecuario-2022
Eljounaidi K, Lee SK, Bae H (2016) Bacterial endophytes as potential biocontrol agents of vascular wilt diseases – review and future prospects. Biol Control 103:62–68
Glare T, Caradus J, Gelernter W, Jackson T, Keyhani N, Köhl J et al (2012) Have biopesticides come of age? Trends Biotechnol 30:250–258
Han Q, Ma Q, Chen Y, Tian B, Xu L, Bai Y et al (2020) Variation in rhizosphere microbial communities and its association with the symbiotic efficiency of rhizobia in soybean. ISME J 14:1915–1928
Harman GE (2006) Overview of mechanisms and uses of Trichoderma spp. Phytopathology 96:190–194
Helga W, Lernoud J (2019) The World of Organic Agriculture. Statistics and emerging Trends 2019. Research Institute of Organic Agriculture (FiBL), Frick, and IFOAM – Organics International, Bonn
Höfte M, Altier N (2010) Fluorescent pseudomonads as biocontrol agents for sustainable agricultural systems. Res Microbiol 161:464–471
Hong CE, Park JM (2016) Endophytic bacteria as biocontrol agents against plant pathogens: current state-of-the-art. Plant Biotechnol Rep 10:353–357
Hulot JF, Hiller N (2021) Exploring the benefits of biocontrol for sustainable agriculture. A literature review on biocontrol in light of the European Green Deal, Institute for European Environmental Policy. In: https://ieep.eu/publications/exploring-the-benefits-of-biocontrol-for-sustainable-agriculture/#:~:text=explore%20sustainable%20alternatives.-,As%20a%20non%2Dchemical%20and%20targeted%20input%2 C%20biocontrol%20can%20offer,agronomic%20practices%20on%20plant%20health
INIA (2022) Bioinsumos: agricultura biológica y sustentable en su dimensión ambiental, social y productiva. Revista INIA, Informe Especial 71:61–75.
Jaber RL, Ownley B (2017) Can we use entomopathogenic fungi as endophytes for dual biological control of insect pests and plant pathogens? Biol Control 116:36–45
Jackson MA, Jaronski ST (2009) Production of microsclerotia of the fungal entomopathogen Metarhizium anisopliae and their potential for use as a biocontrol agent for soil-inhabiting insects. Mycol Res 113:842–850
Jaronski ST, Mascarin GM (2017) Chapter 9 - mass production of fungal entomopathogens. In: Lacey LA (ed) Microbial control of insect and mite pests. Academic Press, pp 141–155
Junaid JM, Dar NA, Bhat TA, Bhat AH, Bhat MA (2013) Commercial Biocontrol Agents and their mechanism of action in the management of Plant Pathogens. Int J Mod Plant Anim Sci 1(2):39–57
Kabaluk JT, Ericsson JD (2007) Metarhizium anisopliae seed treatment increases yield of field corn when applied for wireworm control. Agronomy J 99:1377–1381
Keyser CA, Thorup-Kristensen K, Meyling NV (2014) Metarhizium seed treatment mediates fungal dispersal via roots and induces infections in insects. Fungal Ecol 11:122–131
Latz MCA, Jensen B, Collinge DB, Jørgensen HJL (2018) Endophytic fungi as biocontrol agents: elucidating mechanisms in disease suppression. Plant Ecol Divers 11:5–6. https://doi.org/10.1080/17550874.2018.1534146
Legein M, Smets W, Vandenheuvel D, Eilers T, Muyshondt B, Prinsen E et al (2020) Modes of action of microbial biocontrol in the phyllosphere. Front Microbiol 11:1619
Leng P, Zhang Z, Pan G, Zhao M (2011) Applications and development trends in biopesticides. Afr J Biotechnol 10:19864–19873
Liao X, O’Brien TR, Fang W, St Leger RJ (2014) The plant beneficial effects of Metarhizium species correlate with their association with roots. Appl Microbiol Biotechnol 98:7089–7096
Lira ACd, Mascarin GM, Delalibera Júnior Í (2020) Microsclerotia production of Metarhizium spp. for dual role as plant biostimulant and control of Spodoptera frugiperda through corn seed coating. Fungal Biology 124:689–699
Martínez-Hidalgo P, Hirsch AM (2017) The nodule microbiome: N2-fixing rhizobia do not live alone. Phytobiomes J 1:70–82
MGAP (2021) Official communications from the Ministry of Livestock, Agriculture and Fisheries (MGAP) found in: https://www.gub.uy/ministerio-ganaderia-agricultura-pesca/comunicacion/publicaciones/plan-nacional-para-fomento-produccion-bases-agroecologicas/plan-0
Modor Intelligence (2019) https://www.mordorintelligence.com/industry-reports/microbial-pesticides-market
Mondino P, Casanova L, Calero G, Betancur O, Alaniz S (2012) Zimevit: a biofungicide that combines the action of one bacteria and one yeast for the control of gray mold of grape caused by Botrytis cinerea. Revista Brasileira de Agroecologia 7:127–134
Mondino P, Altier N, Vero S, Pereyra S, Folch C (2014) Control biológico de enfermedades de plantas en Uruguay. In: Bettiol W, Wagner M, Mondino P, Montealegre J, Colmenarez Y (eds) Control biológico de enfermedades de plantas en América Latina y el Caribe. Facultad de Agronomía, Universidad de la República, Uruguay
Narayanasamy P (2013) Mechanisms of action of fungal biological control agents. Biological management of diseases of crops progress in biological control, vol 15. Springer, Dordrecht
O’Callaghan M, Ballard RA, Wright D (2022) Soil microbial inoculants for sustainable agriculture: Limitations and opportunities. Soil Use Manag 38:1340–1369. https://doi.org/10.1111/sum.12811
Pan D, Mionetto A, Tiscornia S, Bettucci L (2015) Endophytic bacteria from wheat grain as biocontrol agents of Fusarium graminearum and deoxynivalenol production in wheat. Mycotoxin Res 31:137–143
Partida-Martínez LP, Heil M (2011) The microbe-free plant: fact or artifact? Front Plant Sci 2:1–16
Pereyra S, Garmendia G, Cabrera M, Vero S, Pianzzola M, Dill-Macky R (2005) Control biológico de la fusariosis de la espiga de trigo y cebada. Agrociencia 9:337–343
Quagliotto L, Azziz G, Bajsa N, Vaz P, Pérez C, Ducamp F et al (2009) Three native Pseudomonas fluorescens strains tested under growth chamber and field conditions as biocontrol agents against damping-off in alfalfa. Biol Control 51:42–50
Rebuffo M, Bemhaja M, Risso D (2006) Utilization of forage legumes in pastoral systems: state of art in Uruguay. Lotus Newsl 36:22–23
Rivas F, Nuñez P, Jackson T, Altier N (2014) Effect of temperature and water activity on mycelia radial growth, conidial production and germination of Lecanicillium spp. Isolates and their virulence against Trialeurodes vaporariorum on tomato plants. Biocontrol 59:99–109
Rivas-Franco F, Hampton JG, Morán-Diez ME, Narciso J, Rostás M, Wessman P et al (2019) Effect of coating maize seed with entomopathogenic fungi on plant growth and resistance against Fusarium graminearum and Costelytra giveni. Biocontrol Sci Technol 29:877–900
Rivas-Franco F, Hampton JG, Altier NA, Swaminathan J, Rostás M, Wessman P et al (2020a) Production of microsclerotia from entomopathogenic fungi and use in maize seed coating as delivery for biocontrol against Fusarium graminearum. Front Sustainable Food Syst 4. https://doi.org/10.3389/fsufs.2020.606828
Rivas-Franco F, Hampton JG, Narciso J, Rostás M, Wessman P, Saville DJ et al (2020b) Effects of a maize root pest and fungal pathogen on entomopathogenic fungal rhizosphere colonization, endophytism and induction of plant hormones. Biol Control 150:104347
Rodríguez A, Chia E, Rossi V (2022) Biological control: lessons learned for agroecological transition in Uruguay.Agrociencia Uruguay26(NE3)
Roldán DM, Costa A, Králová S, Busse HJ, Amarelle V, Fabiano E et al (2022) Paenibacillus farraposensis sp. nov., isolated from a root nodule of Arachis villosa. Int J Syst Evol Microbiol 72(3)
Scarlato M, Dogliotti S, Bianchi FJJA, Rossing WAH (2022) Ample room for reducing agrochemical inputs without productivity loss: The case of vegetable production in Uruguay. Sci Total Environ 810:152248.
Sessa L, Pedrini N, Altier N, Abreo E (2022) Alkane-priming of Beauveria bassiana strains to improve biocontrol of the redbanded stink bug Piezodorus guildinii and the bronze bug Thaumastocoirs peregrinus. J Invertebr Pathol 187:107700. https://doi.org/10.1016/j.jip.2021.107700
Silvera-Pérez E, Gonzalez P, Di Candia M, Galván G, Mondino P, Scattolini A et al (1997) Control biológico de la mancha foliar y punta seca de la cebolla (Allium cepa) ocasionada por Botrytis squamosa en almácigo. In: Proceedings of the Congreso Latinoamericano de Fitopatologia, 1997. Montevideo, Uruguay, (Fitopatologia SUd ed)
Soria S, Alonso R, Bettucci L (2012) Endophytic bacteria from Pinus taeda L. as biocontrol agent of Fusarium circinatum Nirenberg & O’Donnell. Chil J Agricultural Res 72:281–284
Spurgeon D, Lahive E, Robinson A, Short S, Kille P (2020) Species sensitivity to toxic substances: evolution, ecology and applications. Front Environ Sci 8:588380
Tilocca B, Cao A, Migheli Q (2020) Scent of a killer: microbial volatilome and its role in the biological control of plant pathogens. Front Microbiol 11:41
Tiscornia S, Lupo S, Corallo B, Sanchez A, Bettucci L (2014) Neotropical leaf-cutting ants (Acromyrmex spp.): biological control under laboratory and field conditions. Trends Entomology 10:55–62
Vaz Jauri PV, Altier N, Pérez CA, Kinkel L (2018) Cropping history effects on pathogen suppressive and signalling dynamics in Streptomyces communities. Phytobiomes 2:14–23
Vega FE (2018) The use of fungal entomopathogens as endophytes in biological control: a review. Mycologia 110:4–30
Weller DM, Raaijmakers JM, Gardener BBM, Thomashow LSJArop (2002) Microbial populations responsible for specific soil suppressiveness to plant pathogens. Ann Rev Phytopathol 40:309–348
Whipps JM (2001) Microbial interactions and biocontrol in the rhizosphere. J Exp Bot 52:487–511
Yanes ML, Bajsa N (2016) Fluorescent Pseudomonas: A Natural Resource from Soil to Enhance Crop Growth and Health. In: Castro-Sowinski, S. (eds) Microbial Models: From Environmental to Industrial Sustainability. Microorganisms for Sustainability, vol 1. Springer, Singapore. 323–349
Yanes ML, Fernández A, Arias A, Altier N (2004) Método para evaluar protección contra Pythium debaryanum y promoción del crecimiento de alfalfa por Pseudomonas fluorescentes. Agrociencia 8:23–32
Yanes ML, De La Fuente L, Altier N, Arias A (2012) Characterization of native fluorescent Pseudomonas isolates associated with alfalfa roots in uruguayan agroecosystems. Biol Control 63:287–295
Acknowledgements
We acknowledge the support of the following research organisations, financial agencies and research institutes: Programa Nacional de Desarrollo de las Ciencias Básicas (PEDECIBA), Agencia Nacional de Investigación e Innovación (ANII), Sistema Nacional de Investigadores (SNI), Instituto de Investigaciones Biológicas Clemente Estable – Ministerio de Educación y Cultura (IIBCE-MEC), Universidad de la República (UdelaR), Instituto Nacional de Investigación Agropecuaria (INIA), Ministerio de Ganadería, Agricultura y Pesca (MGAP), AgResearch (AgR), and Bioprotection Research Centre (BPRC), as well as the support of Karina Punschke (DGSA, MGAP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
On behalf of all authors, the corresponding author states that there are no conflicts of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Financial interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bajsa, N., Fabiano, E. & Rivas-Franco, F. Biological control of phytopathogens and insect pests in agriculture: an overview of 25 years of research in Uruguay. Environmental Sustainability 6, 121–133 (2023). https://doi.org/10.1007/s42398-023-00275-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42398-023-00275-8